Abstract
Cells derived from the placenta have become the focus of extensive research concerning their ability to be used for regenerative medicine or cellular therapies. In a previous study, we characterized amnion-derived multipotent progenitor cells, or AMP cells, by in vitro methods and showed they were able to inhibit antigen-specific T-cell proliferation in a cell-to-cell contact-dependent fashion. Here we examine specific mechanisms involved in immunomodulation by AMP cells. We found that AMP cells significantly inhibited monocyte-derived myeloid dendritic cell (DC) maturation when placed in coculture. Cocultured monocytes retained the nondifferentiated macrophage marker CD14 while exhibiting significant reduction in DC maturation markers CD83 and CD1a, indicating an immature DC maturation state that is pivotal in determining its immune stimulatory or regulatory status. This effect was again dependent on cell-to-cell contact interaction. We also found a significant shift in cytokines present in the microenvironment of cocultures, which indicated a regulatory DC function rather than a stimulatory cell type. Here supernatants taken from AMP cell/monocyte cocultures yielded significant levels of regulatory cytokines, such as PGE2, IL-6, IL-10, and MIC-1. The soluble form of HLA-G was also found at higher levels in cocultures. In contrast, supernatants contained significantly less amounts of the T-cell-stimulating factor IL-12, which is normally produced by activated DCs. Interestingly, cocultured monocytes acquired significant expression of HLA-G on their cell surface over time. HLA-G has multifaceted immunological implications and may be a key molecule in influencing these cells to behave as regulatory DCs. Together, the influence of AMP cells on maturing DCs may favor a regulatory pathway that can be useful for therapeutic applications for immune-mediated disorders or transplantation therapies.
Keywords
Introduction
The ability of the semiallograph fetus to successfully evade the maternal immune response has elicited extensive research over the past decade. How the maternal immune system tolerates rather than rejects the genetically disparate fetus is still not fully understood. It is the responsibility of the cells from the trophoblast, which arises from the trophectoderm layer of the blastocyst, that interact with the maternal environment and protect the fetus from maternal immune attack (19). Other tissues, such as the amnion, are believed to be involved in this protective effect. Fetal-derived amniotic tissue is in close proximity to the trophoblast tissue and is directly descended from the epiblast. Epithelial cells isolated from the amnion, or amniotic epithelial cells (AECs), have also been reported to exhibit stem cell-like properties with the potential to differentiate into various tissues (31,32). AECs have been shown to be nonimmunogenic and exhibit immunomodulatory capabilities (1,13,37). We have previously characterized a subpopulation of AECs, termed amnion-derived multipotent progenitor cells (AMP cells), which are amniotic cells propagated under proprietary serum-free conditions (5). Phenotypically, AMP cells are identified by the absence of major histocompatibility complex (MHC) II, cluster of differentiation 45 (CD45), CD34, CD49d, CD117, CD140b, CD80, and CD86 while positive for CD104, CD90, CD29, CD49b, CD49c, CD49f, CD9, CD10, and stage-specific embryonic antigen 4 (SSEA-4) (5). AMP cells lack the ability to stimulate an immune response by traditional methods such as in a mixed lymphocyte reaction; however, they possess significant immune-modulatory characteristics. AMP cells were able to downregulate the peripheral blood mononuclear cell (PBMC) proliferative responses to several types of stimuli, including nonspecific mitogen, alloantigen, and specific recall antigen, in a dose-dependent manner (5).
Although the immune-modulatory properties of AMP cells have been documented, the mechanism of immune regulation remains unknown. Here we test the possibility that AMP cells modulate the immune response by direct regulation of dendritic cells (DCs), the most effective and potent antigen-presenting cells (APCs). DCs are the major inducers of the immune response, yet they are also the most effective cells of immune regulation. Indeed, immature DCs, although very capable of acquisition and processing of antigen, are naturally tolerogenic and elicit anergistic effects on T lymphocytes (41).
We have previously found that the in vitro immunemodulatory effects of AMP cells are dependent on cell-to-cell contact and primarily do not involve soluble mediators (5). A key membrane-bound molecule that may play a role in the ability of AMP cells to modulate immune responses is the tissue-restricted, nonclassical MHC class I molecule human leukocyte antigen (HLA)-G (2). The regulatory properties of HLA-G are well described, as the molecule is one of the factors allowing fetal escape from maternal immune surveillance (18,38). Further, HLA-G has been shown to have immune-modulatory capabilities on lymphoid, myeloid, and natural killer (NK) cells through interaction with various immunoglobulin-like transcript receptors on the surfaces of these cells (7).
Although previous work had demonstrated that AMP cells were indeed immunomodulatory, the actual immune effector cells involved were not characterized. It was necessary to examine the effects AMP cells have on these cells, specifically APCs, to determine the potential consideration AMP cells may have as a cellular therapeutic agent. In the present study, we investigated the possible mechanisms of how AMP cells modulate the maturation of monocytes to myeloid DCs. This was accomplished by determining both the phenotype and function of monocytes cocultured with AMP cells. The impact of AMP cells on monocytes was through blocking their maturation to functional DCs, and this effect was amplified when AMP cells were preactivated with interferon (IFN)-γ. DCs were maintained in an immature, macrophage-like state during coculture with AMP cells, as indicated by 1) the retention of the macrophage marker CD14; 2) downregulation of DC maturation markers CD83 and CD1a; 3) inhibition of T helper 1 (Th1) cytokine interleukin (IL)-12; and 4) increased production of immune-modulatory cytokines such as IL-10, IL-6, macrophage inhibitory cytokine 1 (MIC-1), and prostaglandin E2 (PGE2). Monocytes cultured with IFN-γ-activated AMP cells also expressed HLA-G on their membrane surface, and soluble HLA-G was detected at significant levels in cocultured supernatants.
Materials and Methods
Isolation of Amnion-Derived Multipotent Progenitor Cells
Twenty-six anonymized human placentas were obtained after uncomplicated elective cesarean deliveries from consenting healthy mothers and with institutional review board approval. AMP cells were isolated from human full-term placentas as previously described (5). AMP cells were banked by cryopreservation in CryoStor™ CS-5 (BioLife Solutions, Bothell, WA, USA) at a concentration of 20 × 106/ml and stored in the vapor phase of liquid nitrogen. The cells were subsequently thawed and cultured in proprietary serum-free culture media at 37°C, 5% carbon dioxide, and 95% air humidity and were grown to confluency in 24-well tissue culture plates (BD Falcon, Franklin Lakes, NJ, USA) for coculture experiments and further analysis.
Isolation of Monocytes and T Lymphocytes From Peripheral Blood Mononuclear Cells
Normal PBMCs were isolated from 27 buffy coats supplied by the Central Blood Bank of Pittsburgh (Product Code E5318; anonymized with minimum age of 17 years old) by way of Ficoll-Hypaque (Life Technologies, Grand Island, NY, USA) density gradient centrifugation at 1,500 rpm (Sorvall Heraeus Legend RT Centrifuge with swinging bucket rotor 7500-6445 and rectangular buckets 7500 6446; radius 19 cm; Thermo Scientific, Waltham, MA, USA) for 30 min. The buffy coat containing the PBMCs was aspirated and washed three times with Roswell Park Memorial Institute (RPMI) media (Life Technologies) at speeds of 1,500 rpm, 1,250 rpm, and 950 rpm for 12 min each. After the final wash, cells were resuspended in two 50-ml conical tubes (Fisher Scientific, Fairlawn, NJ, USA) with 35 ml of RPMI + 5% human AB serum (Gemini Bioproducts, West Sacramento, CA, USA) and kept at room temperature overnight. The next day, cells were washed three times in RPMI at speeds of 1,500 rpm, 1,250 rpm, and 950 rpm. Cells were then counted, and viabilities were assessed using the Guava EasyCyte (Millipore, Billerica, MA, USA). Mononuclear cells were isolated following a Percoll (Life Technologies) density gradient consisting of isotonic Percoll diluted with Iscove's modified Dulbecco's medium (IMDM; Life Technologies) containing 10% fetal calf serum (FCS, Hyclone, Logan, UT, USA). Cells were counted, and between 10 and 20 × 106 viable lymphocytes and monocytes were separately cryogenically frozen and stored in liquid nitrogen using CryoStore™ CS-5. One vial of lymphocytes was used to determine MHC class I and class II antigens by molecularly based HLA typing methods. For cellular assays, monocytes were further purified using Miltenyi microbeads and Midi-Macs separation columns (Miltenyi Biotec, Bergisch Gladbach, Germany). Monocytes were isolated by negative selection using the Miltenyi Monocyte Isolation Kit II according to manufacturer's instructions. The percentage of CD14-positive cells isolated was equal or greater than 95% by flow cytometry.
Cell Morphology
Cell morphology was evaluated by phase contrast microscopy and Wright—Giemsa staining (Sigma-Aldrich, St. Louis, MO, USA). Briefly, cells were air dried on cytospin slides and fixed in cold methanol (Fisher Scientific) for 5 min. Slides were then stained for 20 min in a 1:20 dilution of Giemsa in deionized water at room temperature. Slide images were visualized using a Nikon™ TE2000-U (Nikon, Melville, NY, USA) inverted microscope, 20× objective, and captured with a Photometrics® Coolsnapes2 camera (Roper Scientific, Sarasota, FL, USA). Images were then processed with Adobe Photoshop 8.0 software (Adobe, San Francisco, CA, USA).
AMP Cell and Monocyte Coculture
AMP cells were plated at a concentration of 250,000 cells/ml in tissue culture-treated 24-well plates in proprietary serum-free media containing 20 ng/ml epidermal growth factor (EGF; Life Technologies) in the presence or absence of 10 ng/ml IFN-γ (R & D Systems, Minneapolis, MN, USA). Cultures were started 4 days prior to coculture setup, and factor-supplemented media was changed 2 days later. On the fourth day of culture, media was aspirated from the adherent AMP cell monolayer, and the cells were washed three times with phosphate-buffered saline (PBS, Life Technologies). One hundred fifty thousand purified monocytes were added to each well in 1 ml RPMI + 5% FCS complete media [RPMI containing 5% FCS, 1% Penn-Strep, 1% Glutamax, and 8 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer; all Life Technologies] containing 500 IU premium-grade human granulocyte-macrophage colony-stimulating factor (GM-CSF; Miltenyi Biotec) and 500 IU premium-grade human IL-4 (Miltenyi Biotec). This was considered day 0 of the AMP cell—monocyte coculture. Five hundred international units GM-CSF and 500 IU IL-4 were resup-plemented to the cultures 3 days later (day 3). On day 5, 500 ng/ml lipopolysaccharide (LPS; Sigma Aldrich) with, or without, 10 ng/ml IFN-γ was added to the coculture plates for 2 additional days. Control monocyte-derived immature DCs (imDCs) were generated by culturing 150,000 monocytes alone in 1 ml complete media containing 500 IU human GM-CSF and 500 IU human IL-4 in 24-well tissue culture plates for 5 days with a resupplement of GM-CSF and IL-4 at day 3 at 37°C 5% CO2. Mature DCs (mDCs) were induced by adding 500 ng/ml LPS to each well with, or without, 10 ng/ml IFN-γ and culturing for an additional 2 days. Both imDCs and mDCs were also cultured separately in the presence of 10 ng/ml IFN-γ as a control. Cells were analyzed by flow cytometry to confirm DC phenotype. AMP cells and monocytes were analyzed for surface cell expression of various markers on days 0, 2, 3, 5, 6, and 7.
Flow Cytometric Analysis of AMP Cells, Monocytes, and Maturating Dendritic Cells
All cells were analyzed for cell surface marker expression using a Becton Dickinson FACSVantage SE Cytometer with a digital acquisition and analysis (FACSDiVa) option (Becton Dickinson, Franklin Lakes, NJ, USA). All conjugated and streptavidin secondary conjugated antibodies were obtained from BD Pharmingen (San Diego, CA, USA) unless otherwise stated (see Table 1). AMP cell—monocyte mixed coculture cell populations were distinguished by gating on allophycocyanin-conjugated antibodies for CD45-positive (monocytes) and CD45-negative (AMP cells) populations. Coculture populations were harvested for analysis on days 0, 2, 3, 5, 6, and 7. Relevant monocyte and DC markers included CD14, CD1b, CD80, CD83, CD1a, MHC I, MHC II, and CD86. Nonspecific fluorescence was determined using isotype-matched monoclonal antibody controls. Relevant AMP cell markers analyzed from the CD45 negatively gated cells included HLA-G and the programmed death ligand 2 (PD-L2). Finally, an immunological panel was analyzed on cocultured cells on day 4 only. This panel consisted of PD-L1, PD-L2, HLA-G, immunoglobulin (Ig)-like receptor 2 (ILT-2), ILT-3, ILT-4, CD200, CD200R1, CD47, and CD172a, glucocorticoid-induced tumor necrosis factor receptor family related (GITR), and GITR ligand (GITRL) for the CD45-positive population (imDCs) to further characterize the immunological synapse between imDCs and AMP cells.
Antibodies Used by Flow Cytometry for Cell Surface Marker Expression Analysis

CD45, cluster of differentiation 45; PE, phycoerythrin; FITC, fluorescein isothiocyanate; MHC, major histocompatibility complex; HLA, human leukocyte antigen; PD-L1, programmed death ligand 1; ILT-2, immunoglobulin (Ig)-like receptor 2; GITR, glucocorticoid-induced tumor necrosis factor receptor family related.
Ancell (Bayport, MN, USA).
Abcam (Cambridge, MA, USA).
BD Bioscience (San Jose, CA, USA).
BioLegend (San Diego, CA, USA).
R&D Systems (Minneapolis, MN, USA).
Cytokine Analysis in Cell Culture Supernatants
Culture supernatants were collected from cultures of monocytes alone, monocytes supplemented with 10 ng/ml IFN-γ, and from cocultures consisting of monocytes with AMP cells and monocytes with activated AMP cells. Supernatants were analyzed on days 5 and 7 following addition of maturation factors consisting of 500 ng/ml LPS alone or 500 ng/ml LPS plus 10 ng/ml IFN-γ. A 1× Serine Protease Inhibitor Cocktail Set I (EMD-Calbiochem, Torrey Pines, CA, USA) was added to each sample before freezing at −80°C. At the time of assay, culture supernatants were diluted 1:10 in culture media except for PGE2 samples. Culture supernatants were analyzed by enzyme-linked immune-sorbant assay (ELISA) for the cytokines IL-12 p40 subunit (IL-12p40), IL-12p70, transforming growth factor (TGF)-β1, IL-10, IL-6, macrophage-colony stimulating factor (M-CSF), MIC-1 (all from Raybiotech, Norcross, GA, USA), PGE2 Parameter Competitive ELISA (R&D Systems), human indoleamine 2,3-dioxygenase (IDO; NovaTeinBio, Woburn, MA, USA), and soluble HLA-G (Ex-Bio, Vestec, Czech Republic) according to manufacturer's instructions.
Statistical Analysis
Data are represented as mean ± standard deviation. Means were compared using analysis of variance between multiple comparison groups (AMP cells alone, AMP cells + IFN-γ, monocytes alone, monocytes + IFN-γ, AMP cells + monocytes, activated AMP cells + monocytes) followed by Holm—Bonferroni adjustment to correct for multiple comparison between groups. Values of p < 0.05 were considered statistically significant. Statistical analyses were performed using R software 3.0 (www.r-project.org/).
Results
Morphological Appearance of Monocytes Cocultured with AMP Cells
Maturation of monocytes into DCs is indicated by well-characterized changes in morphology. During activation, the morphological appearance of cell cytoplasm and membranes changes from rounded, large nucleated cells with very few membrane extensions to that of larger, dense, granulated cells with multiple membrane protrusions, resembling neural dendrites (23). Morphological changes of monocytes cocultured with AMP cells or with IFN-γ preactivated AMP cells are summarized in Figure 1. AMP cells were easily distinguished from monocytes by having epithelial cell morphology, while monocytes were much smaller and more rounded in appearance. Overall gross coculture observations showed that monocytes cultured with AMP cells formed very few aggregates and remained mostly as adherent single cells or small clusters (Fig. 1A1). On the contrary, monocytes cultured with activated AMP cells formed tight adherent clusters throughout the well (Fig. 1A2). We next examined changes in monocyte morphology during the DC maturation process in the absence or presence of AMP cells (Fig. 1B). By day 4, monocytes cultured alone in the presence of IL-4 and GM-CSF proceeded to an immature DC state and appeared rounded with several pseudopodia and cytoplasmic membrane extensions (“dendrites”; Fig. 1B1). On day 5, cultures were treated with LPS for an additional 2 days to promote maturation of DCs. These matured DCs exhibited numerous membrane-associated dendritic protrusions completely enveloping the cells and had the characteristics of mature, activated DCs (Fig. 1B2). At day 4, monocytes cultured with AMP cells had no apparent morphological differences compared to monocytes cultured alone (Fig. 1B3). There was, however, a remarkable difference in day 7 LPS-stimulated monocytes cocultured with AMP cells (Fig. 1B4). In these cultures, monocytes were significantly smaller and had only a few dendritic extensions and appeared more veil-like overall. These data suggest that monocytes cultured with AMP cells were maintained in a more undifferentiated, immature state and did not exhibit the morphology of activated cells.

AMP cell—monocyte cocultures. (A) Images were taken by light microscopy using a 20× objective (Nikon instruments) at day 4. (1) Amnion-derived multipotent progenitor (AMP) cell monolayer with attached monocytes. (2) Monocytes attached to activated AMP cell monolayer. Scale bar: 100 μm. (B) Giemsa staining of monocytes differentiated into immature dendritic cells (DCs) in the presence of 500 IU interleukin 4 (IL-4), and 500 IU granulocyte-macrophage colony-stimulating factor (GM-CSF) (imDCs; day 4) and mature DCs in the presence of 500 ng/ml lipopolysaccharide (LPS) (mDCs; day 7). (1 and 2) Monocytes differentiated alone. (3 and 4) Monocytes differentiated in the presence of AMP cells. Scale bar: 10 μm.
Impact of AMP Cells on Monocyte Surface Marker Expression
Dendritic Cell Maturation Markers
Cell surface marker expression by flow cytometry was used to quantify the effect of AMPs on monocyte differentiation. In Table 2, we summarize the expression of the costimulatory molecules CD80 and CD86, the macrophage marker CD14, and mature DC markers CD83, CD1a, and CD1b on monocytes cocultured with AMP cells and activated AMP cells. Monocytes in the presence of maturation factors GM-CSF and IL-4 lost their CD14 expression as early as day 3 (day 3: 4.9%; day 5: 0.5%), while monocytes cocultured with activated AMP cells significantly retained CD14 expression (day 3: 63.1%; day 5: 33.7%; p < 0.01). This process was less evident in the presence of nonactivated AMP cells, whereby monocytes lost their CD14 by day 5. The hallmark of DC maturation is the increased expression of CD1a and CD83, as shown on the monocytes cultured with GM-CSF and IL-4 and stimulated with LPS. However, in the presence of activated AMP cells, the maturation markers CD1a and CD83 were significantly inhibited on monocytes (CD83 day 7: 20.2% vs. 77.8%, p < 0.01 and CD1a day 5: 5.4% vs. 69.6%, p < 0.05). AMP cells that were not preactivated had no effect on CD83 expression but did reduce CD1a levels at days 5 and 7; however, these differences did not reach statistical significance. A typical representative example of the impact of AMP cells and activated AMP cells on expression of the monocyte marker CD14 and DC maturation markers CD1a and CD83 over the coculture period (days 0, 3, 5, and 7) is depicted in Figure 2. Overall, there was no significant change in the expression of costimulatory molecules CD80 and CD86, as well as CD1b on monocytes regardless of culture conditions (Table 2). These findings are supportive of the coculture morphological observations indicating that monocytes in the presence of activated AMPs maintain an immature phenotype.

A representative example of surface marker expression of CD1a, CD14, and CD83 on monocytes cultured alone, in the presence of AMP cells, or in the presence of activated AMP cells. Here monocytes were analyzed by flow cytometry on days 0, 3, 5, and 7. Percent positive cells for each surface marker are expressed by histograms. Actual percent positive values are shown within each histogram analysis box. CD1a, cluster of differentiation 1a.
Effects of Culturing Monocytes With AMP Cells Alone and IFN-γ Preactivated AMP Cells as Measured by Pertinent Surface Marker Expression

Monocytes were cultured alone, in the presence of nonactivated amnion-derived multipotent progenitor (AMP) cells, or in the presence of AMP cells preactivated with interferon (IFN)-γ. Cells were cultured for a maximum of 7 days in monocyte differentiation media containing 500 U/ml granulocyte-macrophage colony-stimulating factor (GM-CSF) and 500 U/ml interleukin 4 (IL-4). On day 5, the cells were supplemented with 500 ng/ml lipopolysaccharide (LPS) for an additional 2 days. Monocytes were positively gated by CD45 and examined for cell surface expression of markers involved in cell maturation. Data are represented as mean percent positive compared to isotype control ± standard deviation.
p < 0.05 versus monocytes alone.
p < 0.01 versus monocytes alone.
Dendritic Cell Immune-Modulatory Markers
In order to further characterize the effect of AMPs on monocytes, we examined a panel of ligands and receptors that are involved in immune-modulatory pathways of myeloid antigen-presenting cells and downstream effects on T lymphocytes. Differential expression induced on monocytes or DCs may provide evidence to the behavioral aspects of DCs modulated by AMP cells (6,28,34,35). These receptors and ligands included HLA-G and the HLA-G binding receptors immunoglobulin-like transcripts ILT-2, ILT-3, and ILT-4; the programmed death ligands PD-L1 and PD-L2; the glucocorticoid-induced TNFR-related protein GITR and its ligand GITRL; the myeloid membrane protein CD200 (OX2) and its receptor CD200R; and the myeloid membrane protein CD47 and its receptor CD172a (signal-regulatory protein α; SIRPα). The summary of five separate experiments is shown in Table 3. There were no significant differences in monocyte expression of ILT-2, ILT-3, ILT-4, CD200, CD200R, CD47, and CD172a in all five culture conditions tested (data not shown). HLA-G expression on monocytes was significantly elevated in the presence of activated AMP cells, reaching up to 51.9% of monocytes with positive expression (p < 0.01 vs. monocytes alone). PD-L1 was elevated on monocytes in both AMP cell cocultured groups, but only in the presence of activated AMP cells did expression reach over 90% (p < 0.001). Furthermore, monocytes stimulated with IFN-γ had a similar increase in PD-L1 expression. In contrast, PD-L2 and GITR levels were not upregulated on monocytes in any group tested and were below isotype control (negative). Although GITRL was significantly increased on monocytes treated with IFN-γ versus monocytes alone (p < 0.05), the addition of AMP cells and activated AMP cells induced its highest expression on these cells (p < 0.05 with AMP cells and p < 0.001 with activated AMP cells). A representative histogram example for surface marker expression of HLA-G on monocytes is shown in Figure 3. In this typical sample, there was a significant increase in surface expression of HLA-G on monocytes cocultured for 4 days with activated AMP cells (45.4%) compared with monocytes alone or monocytes cultured with IFN-γ (2.4% and 1.9%, respectively). These data indicate that AMP cells effected monocytes by inducing immunomodulatory ligands on their cell surface, which may facilitate regulatory downstream activities.
Surface Cell Marker Expression of Pertinent Immunological Molecules on Monocytes

Surface cell marker expression for PD-L1, PD-L2, HLA-G, GITR, and GITRL were determined for monocytes cultured alone, with 10 ng/ml IFN-γ, in the presence of nonactivated AMP cells, or in the presence of AMP cells preactivated with IFN-γ for 4 days (n = 5). Values are expressed as mean percent positive cells compared to isotype controls ± standard deviation.
p < 0.001 versus monocytes alone,
p < 0.001 versus all groups,
p < 0.05 versus monocytes alone and monocytes + activated AMPs,
p < 0.05 versus monocytes alone,
p < 0.001 versus monocyte alone.

HLA-G expression on monocytes cocultured with AMP cells: a representative experiment. Flow cytometric histogram of fluorescence intensity of monocytes cultured alone (red histogram), with 10 ng/ml interferon (IFN)-γ (cyan histogram), with AMP cells (orange histogram), and with activated AMP cells (green histogram). Histogram analysis shows nearly 50% of the monocytes cultured with activated AMP cells expressed human leukocyte antigen (HLA)-G as indicated by a several log shift in fluorescence intensity (x-axis). *p < 0.001 versus monocytes alone, monocytes + IFN-γ, and monocytes + AMP cells.
The Impact of Coculture on AMPs Surface Marker Expression
The impact of monocytes on AMP cells was also determined by analyzing the surface marker expression of the immune-modulatory ligands HLA-G and PD-L2 on AMP cells following coculture. The mean percent expression of HLA-G and PD-L2 on AMP cells (n = 5) over the coculture period (0 to 7 days) is summarized in Figure 4A. Resting AMP cells expressed low levels of these markers over the entire culture period. Stimulation with IFN-γ (10 ng/ml) promoted HLA-G expression on the activated AMP cells over the first 3 days (55-60%); however, it gradually decreased to 24% by day 7 (Fig. 4A, activated AMPs). Post-IFN-γ exposure, PD-L2 expression on activated AMP cells also rose immediately to 65% but dropped rapidly over time to near undetect-able levels by day 7 of culture. Addition of monocytes to AMPs did not affect their expression of HLA-G and PD-L2. However, following stimulation of these cocultures with LPS on day 5, there was a significant increase in HLA-G and PD-L2 on AMP cells (50% and 72.9%, respectively; Fig. 4A, AMPs + Mos). AMP cells treated with LPS without monocytes (control) did not upregulate HLA-G or PD-L2 and looked similar to the resting AMP cells (data not shown). Coculturing of activated AMP cells with purified monocytes had a synergistic effect on HLA-G and PD-L2 expression (Fig. 4A, activated AMPs + Mos). The expression of HLA-G increased throughout the coculture period, reaching nearly 90% positive cell expression by day 7. The PD-L2 pattern on activated AMP cells mirrored expression to that of AMP cells cultured with monocytes but at significantly higher levels. Here activated AMP cells had 82.5% positive expression at day 0, which declined rapidly to nearly 25% by day 5 before it increased again to nearly 62% on day 7, post-LPS stimulation. Representative histograms of flow cytometric fluorescence intensity at days 3 and 7 are shown in Figure 4B.
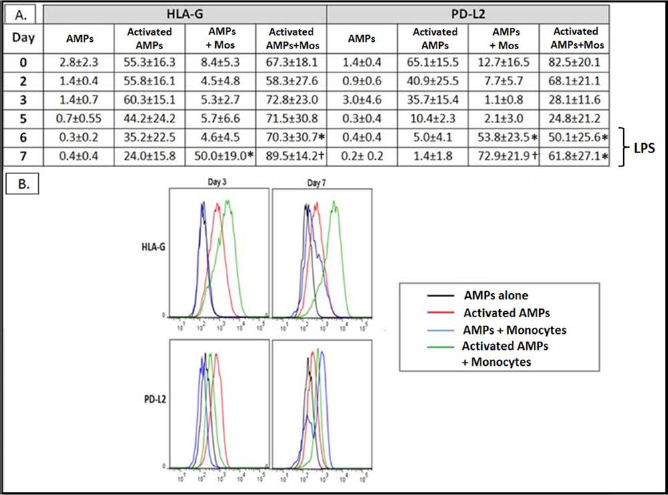
Expression of HLA-G and PD-L2 on cocultured AMP cells. (A) Expression of HLA-G and programmed death ligand 2 (PD-L2) over time on AMP cells and activated AMP cells in the presence or absence of monocytes (n = 5). AMP cells were gated on the CD45-negative population by flow cytometry. Data are represented as percent positive versus isotype controls ± standard deviation. Mean percent positive values are represented numerically. *p < 0.05 versus activated AMPs. †p < 0.01 versus activated AMPs. (B) Individual histogram example of HLA-G and PD-L2 expression by AMP cells on days 3 and 7 of culture. Histogram designations are as follows: AMPs (black histogram), activated AMPs (red histogram), AMPs + monocytes (blue histogram), and activated AMPs + monocytes (green histogram).
Cytokine Quantification and Analysis in the Coculture
Cytokines were measured in cultures containing monocytes with and without AMP cells to evaluate the cytokine environment produced by the different culturing conditions. The panel consisted of both proinflammatory and regulatory cytokines. Cytokine profiles were performed using ELISA on culture supernatants from day 5 and day 7 postaddition of maturation factors LPS alone or LPS + IFN-γ (n = 5) (Fig. 5). There was no detection of IDO, M-CSF, or TGF-β1 in any of the samples tested at any time during the study (data not shown). On day 5, monocyte cocultures with both AMPs and activated AMPs contained significantly higher levels of IL-6, MIC-1, and PGE2 in comparison to monocytes alone (*p < 0.05, **p < 0.01, ***p < 0.001) (Fig. 5). The addition of IFN-γ induced significant levels of MIC-1 in cultures of monocytes alone but was less than that seen in supernatants collected from monocytes and AMP cell cocultures (**p < 0.01). IL-12p40, IL-12p70, and IL-10 were not detected at significant levels through the first 5 days of all five culture conditions (see Fig. 5). On day 7, elevations in IL-6, PGE2, IL-10, and MIC-1 were observed in supernatants derived from monocyte cocultures with both AMP cell groups, with significantly higher levels found in the monocyte/activated AMP cell coculture supernatants. The maturation of monocytes to DCs, in the presence of LPS and IFN-γ, resulted in elevated levels of both IL-12 isoforms compared to LPS alone (data not shown). Addition of AMP cells significantly decreased these levels of IL-12, especially when monocytes were cocultured with activated AMPs (**p < 0.01, ***p < 0.001) (see Fig. 5). In contrast to the other mediators described in Figure 5, sHLA-G levels were significantly increased only in the activated AMP cell—monocyte cocultures (**p < 0.01, ***p < 0.001) (Fig. 6). These data indicate that the coculturing of monocytes with AMP cells, especially activated AMP cells, induced a cytokine microenvironment characteristic of tolerogenic DCs.

Cytokine levels measured by ELISA in the supernatants of the various culture conditions (gray day 5 and black day 7). Supernatants were collected from cultures on days 5 and 7 and assayed for IL-10, IL-6, macrophage inhibitory cytokine 1 (MIC-1), IL-12 p40 subunit (IL-12p40), IL-12p70, indoleamine 2,3-dioxygenase (IDO), macrophage-colony-stimulating factor (M-CSF), and transforming growth factor (TGF)-β1 by ELISA. Prostglandin E2 (PGE2) was assayed by competitive ELISA. Data are expressed as mean ± standard deviation (n = 5). IL-12p40, IL-12p70, IL-6, PGE2, IL-10, and MIC-1 are expressed in pg/ml. There was no detection of M-CSF, IDO, or TGF-β1 at any time of assay (data not shown). No cytokines were found in cultures of monocytes (MØ) alone. AMP cells and IFN-γ-activated AMP cells alone did produce some PGE2 and MIC-1 compared to monocytes alone. Significant increases in cytokines were detected in supernatants of cocultured monocytes compared to monocytes alone or monocytes treated with IFN-γ, as shown by *p < 0.05, **p < 0.01, and ***p < 0.001 for groups indicated by the horizontal line. Cell culture media was negative for all cytokines analyzed.

Soluble (s)HLA-G measurements by ELISA in supernatants of various culture conditions (gray day 5 and black day 7). sHLA-G is expressed as units/ml. Data are expressed as mean ± standard deviation (n = 5). AMP cells alone had a slight (not significant) increase in sHLA-G versus monocytes alone with or without IFN-γ. Cocultures of monocytes with AMP cells and IFN-γ-activated AMP cells had significant increases in sHLA-G versus monocytes alone or with IFN-γ, with activated AMP cell cocultures producing the highest amounts detected. **p < 0.01 and ***p < 0.001 for groups indicated by the horizontal line. Cell culture media was negative for all cytokines analyzed.
Discussion
In this study, we demonstrate for the first time that AMP cells, significantly impair the maturation and differentiation process of peripheral blood monocytes to mature myeloid DCs. Activated AMP cells were able to significantly inhibit the expression of the DC maturation markers CD83 and CD1a during monocyte maturation to mature DCs. As well as maintaining an immature phenotype of monocytes, activated AMP cells also preserved the retention of the monocyte marker CD14 for a significantly longer period of time compared to monocytes differentiated under normal conditions. Supernatants taken from cocultures showed significantly lower levels of proinflammatory cytokines IL-12p40 and IL-12p70, and increased levels of immune-regulatory cytokines, such as IL-6, IL-10, MIC-1, and PGE2, in comparison to monocytes cultured alone. Also, the presence of immune-modulating soluble HLA-G was detected at high levels in these coculture conditions. Furthermore, expression of HLA-G was demonstrated on monocytes maturing to DCs in the presence of activated AMP cells. These phenomena represent strong evidence of a regulatory DC phenotype induced by both nonactivated and activated AMP cells.
We have previously shown that AMP cells lack the necessary molecules to be immunogenic in vitro, such as MHC class II and costimulatory molecules. We also showed that these cells are potent immunomodulators and have the capacity to downregulate immune response in an antigen-dependent manner (5). Others have extensively studied AECs and found similar findings, but the mechanism of the immunomodulatory capabilities has yet to be elucidated (4,17,20). Recent attention has been steered toward the effects of immunomodulatory cells and molecules on DCs.
DCs are the central directors of the immune response. They play a key role in the initiation of primary immune responses and tolerance induction, depending on their developmental maturity. Immature DCs residing in tissue display a high ability for antigen uptake and processing and a moderate ability for T-cell stimulation. Maturation is induced by locally produced cytokines, initiating a change in monocyte/macrophage phenotype from a processing cell to a presenting cell, characterized by upregulation of MHC and costimulatory molecules, production of IL-12, and migration to lymphoid tissues. DC maturation is a prerequisite for inducing immunogenic T-cell responses, whereas tolerance is observed when antigens are presented by immature or semimature DCs (10,22,29,36,43,50). Two populations of APCs, macrophages and DCs, reside in the human decidualized endometrium throughout pregnancy. These powerful, multifunctional leukocytes are located in close proximity to invasive cytotrophoblast cells, uterine glandular epithelium, and uterine blood vessels and are proposed to play central roles in uterine and placental homeostasis as well as immune modulation (19). In vivo, amniotic-derived cells may provide fetal immune privilege from macrophages and DCs by directly influencing cell phenotype and function. Cellular immunomodulation by AMP cells may involve alteration in DC function by affecting the maturation state of initially naive monocytes. Other cell types have been extensively studied involving immune modulation by altering maturation of DCs. Megatti et al. have recently demonstrated how amnion-derived mesenchymal cells may block the maturation of monocytes into DCs both in terms of phenotype and ability to stimulate T-cell responses (30). These mesenchymal amniotic cells caused an arrest in DC cell cycle at G0 phase and abolished proinflammatory cytokines such as TNF-α, IFN-γ-induced protein 10 (IP-10), monokine induced by IFN-γ (MIG) and regulated upon activation, normally T-expressed, and presumably secreted (RANTES), but an upregulation of Th2 cytokines was observed in coculture supernatants (30). Mesenchymal stem cells (MSCs) have also been shown to immunomodulate immune responses by interfering with DC differentiation, maturation, and function (14,21,23,25,29,33,40,47). In contrast with the above studies, a prerequisite for AMP cells to induce immune modulation is the cell-to-cell contact during the exposure of the monocytes to the cytokines and stimuli required for DC maturation. We previously have shown that when AMP cells were separated from PBMCs in a mixed lymphocyte culture by Transwell apparatus, they failed to inhibit allogeneic PBMC proliferation (5). There was no effect of AMP cells on DC markers when AMP cells and monocytes were cultured in suspension, aiding the belief that cell-to-cell contact was a prerequisite for the AMP cell modulatory effect (data not shown). Inhibition of DC maturation markers CD83 and CD1a concomitant with the persistence of the macrophage marker CD14 are the hallmark of activated AMP cell effect on monocytes using current adherent culture methods (see Table 2). Although LPS stimulation did promote substantial loss of CD14 from monocytes cocultured with AMP cells, these cells never gained adequate expression of the DC maturation markers.
Other immune-modulating molecules were induced on immature DCs by AMP cells. GITR and its ligand (GITRL) were examined on monocytes cocultured with nonactivated AMP cells and activated AMP cells. GITR is a member of the tumor necrosis factor receptor superfamily (TNFRSF) and is a strong regulator of T-cell activation (42). Ronchetti et al., using GITR-/- knockout mice models, found that DCs devoid of GITR when cocultured with CD4+CD25- T-cells produced higher amounts of IL-10 compared to GITR+/+ DCs (42). These cells also originated forkhead box P3-positive (FoxP3+) T lymphocytes that secreted IL-10 and TGF-β during coculture. Monocytes cultured with either AMP cells or activated AMP cells expressed very low levels of GITR by surface marker expression (see Table 2). Hence, nonexpression of GITR on AMP cell-modulated DCs may in effect promote regulatory DC function when interacting with T lymphocytes. On the contrary, GITRL was significantly induced on monocytes cocultured with both types of AMP cells. GITRL is expressed in macrophages, immature and mature DCs, and B-cells, but not resting or activated T-cells (34,35). GITRL itself, when expressed in DCs, has been shown to cause induction of T-regulatory cells (8). Therefore, elevation of this ligand on AMP cell-modulated monocytes may also play a role in a regulatory phenotype. The programmed cell death ligand, PD-L1, was also induced on immature DCs but mainly by activated AMP cells. Interestingly, IFN-γ alone may also induce expression of this ligand on immature DCs. Programmed death ligands play key roles in T-cell-mediated tolerance of the semiallogeneic fetus, as binding of these ligands to the PD-1 receptor inhibits antigen-stimulated T-cell activation and cytokine production in vitro. Indeed, these ligands are highly expressed on AMP cells when induced with proinflammatory cytokines such as IFN-γ and are a major mechanism of immune modulation of trophoblastic cells in vivo (38).
Of particular interest was the impact of monocytes on the expression of immune-modulatory ligands on AMP cells. Expression of HLA-G on AMP cells rose significantly in the presence of monocytes and was maintained at much higher levels for a longer period than when AMP cells were cultured with only recombinant IFN-γ (Fig. 4). This may represent a two-way interaction during cell-to-cell contact. Monocytes cocultured with activated AMP cells, but not AMP cells, also acquired significant amounts of HLA-G on their cell surface. This finding cannot be explained as a result of IFN-γ since monocytes cultured in the presence of recombinant IFN-γ failed to express HLA-G. One potential explanation that may account for this phenomenon is trogocytosis. Trogocytosis is a transfer of membrane fragments and not individual molecules. During such, all molecules contained within a certain membrane area are transferred, including some that do not participate in cell-to-cell crosstalk and thus transfer nonspecifically (16). Trogocytosis of membrane-bound HLA-G is not a new phenomenon and has been studied in various cellular systems (9,16,26,44,48). Caumartin et al. have shown that HLA-G1 can indeed be acquired from tumor cells by activated but not resting NK cells (9). When this occurred, NK cells stopped proliferating, were no longer cytotoxic, and behaved as suppressor cells capable of inhibiting cytotoxic functions of other NK cells. Lamaoult et al. were able to show HLA-G1 transfer from activated APCs to effector T-cells demonstrating highly efficient regulatory T-cells can be generated immediately by reversing the function of effector immune cells and without the requirement of a maturation process (26). In another study, resting and activated monocytes were able to acquire membrane-bound HLA-G from HLA-G tumor cells by trogocytosis (16). The acquisition of HLA-G APCs may be a key mechanism of tolerance-inducing trophoblast-derived cells protecting the semiallogeneic fetus. It has been accepted that tolerogenic DCs demonstrate low expression of costimulatory molecules with high inhibitory receptor expression (48). Monocytes maturing to DCs in the presence of activated AMP cells may acquire an immunosuppressive DC phenotype by having low expression of DC maturation markers with high inhibitory receptor expression as well as acquisition of membrane-bound HLA-G.
The importance of HLA-G expression in immune modulation cannot be trivialized. HLA-G exerts its inhibitory function against NK cells, T lymphocytes, and APCs through direct binding to the inhibitory receptors ILT-2 (expressed on all immune cell subsets), ILT-4 (expressed on myeloid cells), and killer cell immunoglobulin-like receptor, two domains, long cytoplasmic tail, 4 (KIR2DL4; expressed on NK cells) (12,41). We found relatively high expression of the inhibitory receptors ILT-2, ILT-3, and ILT-4 within all monocyte/DC treatment groups studied, with some variability in ILT-4 expression. All three of these receptors are implicated in tolerogenic DC phenotypes when expressed on the cell surface, but only ILT-2 and ILT-4 have been shown so far to be receptors for HLA-G (24,46). Compared to other HLA class I molecules, HLA-G has enhanced direct inhibitor functions and inhibits NK cell and cytotoxic T lymphocyte cytolytic functions, CD4+ alloproliferation, and DC maturation (27,28). Indeed, cells that express HLA-G are protected from NK cell cytolysis through interaction with the killing-inhibitory receptors found on NK cell surface. Hunt has shown that antigen-presenting cells transfected with HLA-G1 not only prevent T helper cell proliferation but also directs them toward an immunosuppressive/Th2 phenotype (19).
Suppressive APC types produce anti-inflammatory cytokines such as PGE2, IL-10, and TGF-β1 and have increased expression of PD-L2, ILT-3, and DC-specific intracellular adhesion molecule (ICAM-3)-grabbing nonintegrin (DC-SIGN), all of which are markers associated with immune evasion and activation of macrophages into a suppressive profile. These cells also secrete low levels of IL-12 and may induce Th2 cells when cocultured with naive CD4 T-cells (19). Similarly, supernatants derived from cocultured AMP cells with monocytes contained elevated levels of immune modulatory cytokines IL-6, PGE2, MIC-1, and IL-10 and lower levels of proinflammatory IL-12 cytokine compared to monocyte cultures alone (Fig. 5). IL-10 has been observed to promote tolerogenic DCs with enhanced endocytic activity and poor antigen-presenting function (43). Tolerogenic DCs have been shown to produce IL-10 and IL-6, along with PGE2 and TGF-β1 (22,41,43). Also, MIC-1 secreted by decidual cells in early pregnancy may promote an increase of a tolerogenic DC subtype in decidua (45). MIC-1 was associated with the downregulation of costimulatory molecules CD25, CD83, and CD86 on peripheral blood monocyte-derived DCs, thus generating a phenotypically immature subtype of DCs (45). Our findings with surface markers and cytokine profiles support the notion that in the presence of AMP cells, the maturation of monocytes is skewed toward regulatory-type DCs. The presence of immune-modulating cytokines IL-6, PGE2, M-CSF, TGF-β1, and IL-10 in cocultures of MSCs with differentiating DCs were also implicated in playing a role in interfering with DC maturation (14,15,21,30,43,48). In our system, TGF-β1 and M-CSF were not detected in monocyte—AMP cell cocultures at any point analyzed. Increased levels of soluble HLA-G that were found in culture conditions consisting of monocytes together with either type of AMP cells may also promote immune modulation. Soluble HLA-G has been shown to inhibit T-cell proliferation and cell cycle progression, but not apoptosis (3). Clinically, soluble HLA-G has been found circulating in maternal peripheral blood during pregnancy and may have a role in maternal/fetal tolerance (18). Furthermore, increased soluble HLA-G plasma levels were detected in patients with a reduced incidence of acute and chronic rejection after several types of solid organ transplantation, such as liver, renal, and combined liver—kidney transplantation (11,39,49).
In conclusion, our findings may suggest that the immunomodulatory pathway mediated by AMP cells is through their impact on maturation of monocyte-derived DCs. These DCs have distinct phenotypic and functional characteristics of regulatory DCs that can dampen T-cell activation and may promote regulatory/suppressor T-cells. The maturation of monocyte-derived DCs in the presence of AMP cells is shifted toward regulatory-type DCs as illustrated by phenotypical and functional analysis. Furthermore, the unique characteristic of AMP cells to express high levels of HLA-G and to promote the expression of HLA-G on the surface of immature DCs may also provide the immunological basis for the use of these cells for therapy in immune-related phenomena such as allograft transplantation. Indeed, upregulation of HLA-G on allograft cells has been shown to be associated with significantly better graft acceptance (7). These findings also suggest AMP cells may have a significant effect as an immunomodulatory application in several immunemediated scenarios such as graft-versus-host disease, autoimmune syndromes, and facilitation of allogeneic cellular grafts such as bone marrow and hematopoietic stem cells. Thus, the unique properties of AMP cells may qualify them as a cellular therapeutic agent for a variety of immune-mediated disorders and circumstances involving regulated immune responses. Further studies with specific animal models of such immune-mediated disorders would be necessary to confirm the full potential of these cells.
Footnotes
Acknowledgments
This work was funded by the Advanced Medical Development Program, US Navy Bureau of Medicine and Surgery. The authors declare no conflicts of interest.
