Abstract
We previously demonstrated that mesenchymal cells from human amniotic membrane (hAMTCs) inhibit the generation and maturation of monocyte-derived dendritic cells (DCs) in vitro. Considering the crucial role of DCs in the immune response and that epithelial cells of the human amniotic membrane (hAECs) share some of the immunoregulatory properties of hAMTCs, we investigated whether hAECs also modulate monocyte-derived DCs. We compared hAECs with hAMTCs in a cell-to-cell contact setting and their secreted factors in modulating DC differentiation and function. First, we demonstrated that primary and expanded hAMTCs strongly inhibited the differentiation of DCs and induced a shift toward M2-like macrophages. This was observed when hAMTCs were cultured in contact (hAMTC-DCcont) or in Transwells (hAMTC-DCtw) with monocytes and even when medium conditioned by hAMTCs was used instead of hAMTCs. hAECs also prevented DC development, but to a lesser extent than hAMTCs. hAECs were more effective when cultured in contact with monocytes (hAEC-DCcont) rather than in Transwells (hAEC-DCtw). The modulatory capacity of hAECs changed during passaging unlike the hAMSCs. The ability to stimulate CD4+ and CD8+ T-cell proliferation was almost completely abolished by hAMTC-DCcont, whereas hAMTC-DCtw and hAEC-DCcont displayed only a reduced ability to stimulate CD8+ T cells. Furthermore, monocytes cocultured with hAMTCs and hAECs showed some similarities, but also differences in cytokine/chemokine secretion. Similarities were observed in the inhibition of IL-12p70 and TNF-α and the increase in IL-10 in supernatants taken from monocyte-DCs cocultured with hAMTCs and hAECs in contact and Transwell settings. The inflammatory factors IL-8, CXCL9, and MIP-1α were significantly lower in hAMTC-DCcont, hAMTC-DCtw, and hAEC-DCcont conditions. In contrast, only hAMTCs (in both contact and Transwell conditions) were able to significantly increase IL-1β and CCL2. Altogether, we demonstrated that hAMTCs and hAECs affect DC differentiation, but that hAMTCs exerted a stronger inhibitory effect, abolished T-cell proliferation, and also induced more changes in cytokine/ chemokine production.
Keywords
Introduction
The human amniotic membrane is a valuable biomaterial that is widely utilized in ophthalmic and dermatological applications (13). It is also an easily accessible and rich source of two distinct stem/progenitor cell populations. Human amniotic epithelial cells (hAECs) form a monolayer lying above a collagen-rich area that harbors monocyte-macrophages, fibroblasts, and mesenchymal stromal cells, referred to as human amniotic mesenchymal tissue cells (hAMTCs) (46). Both hAMTCs and hAECs display multiple properties that may aid regeneration and/or repair of damaged or diseased tissues and organs (52,61,62,72). Indeed, therapeutic effects have been observed following the application of these cells in preclinical settings, especially in disease models involving inflammation and fibrosis (10,52,53,58,63,72). Interestingly, their therapeutic effects were observed despite their absence or rare presence in host tissues after cell transplantation. Such findings have shifted the focus away from their ability to differentiate into tissue-specific cells (28,56,58,78,79) toward their ability to release paracrine-acting factors with anti-inflammatory and anti-fibrotic properties (12,42,65) and possible regulatory effects on immune cells (11,51,63).
Elucidating the effects of amniotic membrane-derived cells on immune cells in vivo is complex, as several distinct cell populations act together in initiating, maintaining, or suppressing immune reactions, whereas in vitro studies have yielded valuable data on effects on innate and adaptive immune cell populations. In this context, it is well known that hAMTCs and hAECs inhibit T-cell proliferation induced by alloantigens, T-cell receptor cross-linking, or mitogens. Cell-to-cell contact and/ or soluble bioactive molecules secreted by these cells appear to play a role in inhibiting T-cell proliferation (3,5,40,46,80). Further, we have reported that the medium conditioned by cultured hAMTCs inhibits the proliferation of lymphocytes in vitro (46,68). Studies investigating other possible cellular targets reported that soluble factors from hAECs inhibit the migration of murine neutrophils and macrophages, and significantly reduce the proliferation of B cells after mitogenic stimulation (40). Furthermore, we have previously shown that hAMTCs strongly inhibit the generation, maturation, and function of monocyte-derived dendritic cells (DCs) in vitro (47).
DCs are “professional” antigen-presenting cells crucial to initiating and modulating immune responses (7,74). Immature DCs are strategically positioned throughout the body to capture antigens from invading pathogens or other foreign bodies. Once DCs process the antigen, they migrate to areas rich in T cells, such as the lymphoid tissues, where they undergo complete maturation, associated with upregulation of chemotactic signals (e.g., CCR7), T-cell costimulatory molecules (CD80, CD86), and activation molecules (CD83), loss of phagocytic receptors, and changes and upregulation of MHC class II compartments (6). Mature DCs may induce CD4+ T-cell expansion and polarization toward T-helper (Th)-1, Th-2, or Th-17 cells (39,59,66,71), stimulate proliferation of CD8+ T cells to bring about cytotoxic activities (1,27,31), or induce differentiation toward T-regulatory or FOXP3+ cells that perform suppressive activities (30,43). In addition, in lymphoid tissues, DCs may interact with B cells and natural killer cells (21–23). Besides their role in initiating immunity to foreign antigens, DCs play an important role as inducers of tolerance to self-antigens, by deleting self-reactive thymocytes and inducing anergy or deleting mature T cells (16). The type, maturation, and activation state of DCs, in combination with a spectrum of environmental and endogenous stimuli (e.g., microbial products, cytokines, endogenous ligands) present in the microenvironment, together determine whether stimulatory versus tolerogenic lymphocyte functions will be induced (44).
Considering therefore the pivotal role of DCs in the immune response, stem/progenitor cell-based strategies that could control DC maturation and function and/or the cytokine microenvironment would be a valuable tool for counteracting many clinical situations involving T cells, including, for example, graft rejection after transplantation. In this scenario, elucidating whether DC differentiation and function is a target of the immunosuppressive activity of amniotic cells may provide a basis for their use in transplantation settings. We previously demonstrated that hAMTCs affect the major stages of the DC life cycle, including maturation, differentiation, and function in vitro (47). Since hAECs have been reported to share some of the immunoregulatory roles of hAMTCs, we investigated whether hAECs can also inhibit monocyte differentiation toward DCs in a manner similar to hAMTCs and if these effects could be exerted by primary as well as expanded cells. To better understand the mechanisms involved, we studied the influence of cell-to-cell contact and secreted factors on DC maturation. Furthermore, we examined the factors produced by the DCs and the effects of these DCs on T-cell proliferation.
Materials and Methods
Ethics Statement
Human term placentae (n = 30) were collected from healthy women after vaginal delivery or cesarean section. All placentae were from female babies. Peripheral blood was collected from healthy male adult donors (n = 12). Samples were collected after obtaining informed written consent according to the guidelines set by the Ethics Committee for the Institution of Catholic Hospitals (CEIOC) and after their authorization to use placenta for experimental research (Document “Parere 16/2012”).
Culture Media
The culture media used were: Roswell Park Memorial Institute (RPMI complete medium): RPMI-1640 medium (Lonza, Basel, Switzerland), supplemented with 10% heat-inactivated fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA), 2 mM l-glutamine (Lonza), and 100 U/ml penicillin plus 100 mg/ml streptomycin (herein referred to as P/S; both from Euroclone, Whetherby, UK); DMEM complete medium: DMEM (Lonza) containing 10% FBS, 2 mM l-glutamine, and P/S; DMEM/ F12 complete medium: DMEM/F12 medium (Invitrogen, Carlsbad, CA, USA) containing 10 ng/ml epithelial growth factor (Sigma-Aldrich), 10% FBS, 2 mM l-glutamine, and P/S.
Isolation and Culture of hAMTCs and hAECs
Immediately after placenta collection, the amniotic membrane was manually separated from the chorion, washed extensively, and treated with antibiotic and anti-fungal solutions as previously described (68).
hAMTCs were isolated by digesting amnion tissue fragments (≈3 × 3 cm2) as previously described (68).
hAECs were prepared as described by Miki et al. (57), with the following modifications. Amniotic membrane fragments (≈15 × 15 cm2) were initially incubated for 10 min at 37°C in phosphate-buffered saline (PBS) containing 0.5 mM EDTA and P/S, and then in 1× trypsin/ EDTA solution (10 ml for each fragment) for 5 min at 37°C. The reagents were purchased from Sigma-Aldrich. After discarding debris, the fragments were incubated in fresh trypsin/EDTA solution for 10 min at 37°C, tissue shaken briefly and gently, and the trypsin inactivated by adding three to four volumes of RPMI complete medium. The fragments were then washed twice with PBS and again digested in trypsin/EDTA. The cells from the second and third digests were pooled and centrifuged at 300 × g for 10 min. Cell suspensions were then filtered through a 100-μm cell strainer (BD Biosciences, San Jose, CA, USA), centrifuged, and counted.
To obtain cells at different passages, freshly isolated hAMTCs and hAECs designated as passage 0 (P0) cells were plated at a density of 50 × 103 and 120 × 103/cm2, respectively. hAMTCs were cultured in DMEM complete medium and hAECs in DMEM/F12 complete medium at 37°C and 5% CO2. Upon reaching confluence, adherent cells were trypsinized and then subcultured at a density of 25 × 103/cm2 for P1 and 12 × 103/cm2 for subsequent passages in the case of hAMTCs and 40 × 103/cm2 at P1 and 20 × 103/cm2 at subsequent passages for hAECs.
Preparation of Conditioned Medium (CM) From Amniotic Cells
To obtain CM from hAMTCs (CM-hAMTC) and hAECs (CM-hAEC), P0 cells were cultured for 6 days in 24-well plates (Corning, Corning, NY, USA) at a density of 2 × 105 cells/well in 0.5 ml of their respective complete medium. At the end of this culture period, CM was collected, centrifuged at 300 × g, filtered through a 0.8-μm sterile filter (Sartorius Stedim, Florence, Italy), and kept frozen at −80°C until use. To minimize donor variability and obtain a better representation of the factors released by the cells, we tested three CM-hAMTC or CM-hAEC pools with each pool containing CM from five to six different cell preparations.
Isolation of Peripheral Blood Mononuclear Cells (PBMCs), Monocytes, and T Cells
Human PBMCs were obtained through density gradient centrifugation (Histopaque; Sigma-Aldrich) of heparinized whole blood samples or buffy coats (n = 10).
Nine different preparations of monocytes were obtained from PBMCs through positive selection using anti-CD14- coated microbeads and MACS® separation columns (Miltenyi Biotec, Bergisch Gladbach, Germany), according to the manufacturer's instructions. Monocytes were stained with phycoerythrin (PE)-conjugated CD14 (clone MϕP9; BD Biosciences), as described below in Flow Cytometric Analysis of Cell Phenotype. The percentage of CD14+ cells was demonstrated by flow cytometry analysis to be greater than 95% of the total cells recovered.
Three different preparations of T-lymphocytes were purified from PBMCs through a magnetic labeling system using the Pan T-cell Isolation Kit II (Miltenyi Biotec), according to the manufacturer's instructions. T cells were stained with allophycocyanin (APC)-conjugated CD3 (clone UCHT1; BD Biosciences), as described below in Flow Cytometric Analysis of Cell Phenotype. The percentage of CD3+ cells was demonstrated by flow cytometry to be more than 95% of the total cells recovered.
Generation of Monocyte-Derived DCs
Peripheral blood monocytes were differentiated into DCs following protocols based on the use of interleukin-4 (IL-4) and granulocyte-macrophage colony-stimulating factor (GM-CSF) (70), followed by maturation with lipopolysaccharide (LPS) (36). Specifically, 1 × 106 monocytes were cultured in 1 ml of RPMI complete medium, containing 50 ng/ml recombinant human GM-CSF and 50 ng/ml IL-4 (both kindly provided by Dr. Schweighoffer, Novartis, Vienna, Austria), in 24-well plates at 37°C for 4–5 days. Cells were then stimulated by adding 100 ng/ml LPS (Sigma-Aldrich) for 2 days.
Coculture of Amniotic Membrane Cells with Monocytes and Treatment with CM
Cocultures of monocytes and amniotic membrane-derived cells were established either in direct cell-to-cell contact or with physical separation using Transwell chambers. On the basis of preliminary titration experiments performed by maintaining a constant number of monocytes (1 × 106) and decreasing the number of hAMTCs or hAECs added (to obtain ratios of monocytes/amniotic membrane cells of 1:0.4, 1:0.2, 1:0.1, 1:0.05, and 1:0), unless otherwise stated, we selected the following for all experiments reported: for contact experiments, 5 × 104 P0 hAMTCs or hAECs (monocytes/amniotic membrane cells ratio of 1:0.05) were seeded in 24-well plates in 1 ml of complete DMEM and DMEM/F12 medium, respectively; for Transwell experiments, 2 × 105 P0 hAMTCs or hAECs (monocytes/amniotic membrane cells ratio of 1:0.2) were seeded in Transwells (0.4 μm pore, polycarbonate membrane; Corning) placed in 24-well plates in 1 ml of the corresponding culture medium. For both coculture conditions, P0 hAMTCs or hAECs were initially plated alone and cultured for 4 days at 37°C and 5% CO2 to allow the cells to adhere to the plastic and grow as a monolayer.
Then, for cell contact experiments, the amniotic membrane cells were irradiated (30 Gy) to block proliferation, and the supernatant was removed to discard nonadherent dead cells. Afterward, 1 × 106 monocytes in RPMI complete medium were added into the same well (for contact experiments) or into the lower compartment of 24-well plates (for Transwell experiments). Monocyte differentiation toward DCs was carried out as described above.
Experiments performed with hAMTCs and hAECs at P2 were as described for P0 cells, except that only 1 day of culture at 37°C was necessary to allow the cells to adhere. Different monocytes/amniotic membrane-derived cells ratios were applied and are as follows: 1:0.05 for the hAMTCs and 1:0.05, 1:0.1, 1:0.2 for hAECs in the cell-to-cell contact settings; 1:0.2, 1:0.4, 1:0.8 for the hAMTCs and hAECs in the Transwell settings.
Experiments performed with hAMTCs and hAECs at P4 were as described for P2, with the exception that only one cell ratio (monocytes/amniotic membrane-derived cells) was used for each condition (1:0.05 for cell-to-cell contact and 1:0.4 for Transwell).
To study the influence of CM, monocyte differentiation was induced as described above in 0.5 ml of complete RPMI containing GM-CSF, IL-4, and LPS together with 0.5 ml of CM-hAMTC or CM-hAEC from P0 cells. Controls were treated with 0.5 ml DC differentiation medium plus 0.5 ml of DMEM or DMEM/F12.
T-Cell Proliferation Assays
Monocytes that had been differentiated in the presence or absence of hAMTCs and hAECs were collected after culture, washed in RPMI complete medium, and used as stimulators to induce proliferation of alloreactive T cells (responders). To avoid contamination by amniotic cells (collected when plated in contact with monocytes), the monocyte-derived cells were isolated through positive selection using anti-CD45-coated microbeads and MACS® separation columns (Miltenyi Biotec), according to the manufacturer's instructions.
Irradiated (30 Gy) stimulator cells (5 × 103) were seeded with 1.5 × 105 responder T cells in round-bottom 96-well tissue culture plates (Corning), in a volume of 150 μl of RPMI complete medium. All cultures were carried out in triplicate.
T-cell proliferation was assessed after 5 days by adding [3H]thymidine (0.67 mCi/well; PerkinElmer Life & Analytical Sciences, Zaventem, Belgium) for 16–18 h. Cells were then harvested with a Filtermate Harvester (PerkinElmer), and thymidine incorporation was measured using a microplate scintillation and luminescence counter (Top Count NXT; PerkinElmer).
T-cell proliferation was also measured by the analysis of carboxyfluorescein diacetate, succinimidyl ester (CFSE) fluorescence. T-lymphocytes were labeled with 5 mM CFSE (Invitrogen) in PBS/0.5% FBS for 4 min. T cells were washed three times in RPMI complete medium, counted, and 1.5 × 105 cells were plated with 5 × 103 stimulators as described above. After 6 days of coculture, cells were harvested and stained with APC-conjugated CD4 (dilution 1:250, clone SK3; BD Biosciences) and CD8 (1:500, clone SK1; BD Biosciences) as described below in flow cytometric analysis of cell phenotype. The dilution of the CFSE-label, which correlates with division of T cells, was analyzed by flow cytometry on a FACS Calibur machine using CellQuest Software (BD Biosciences). T-cell proliferation was calculated as percentage of CD4+ and CD8+ proliferating cells. Where indicated, T-cell proliferation was also reported as proliferation index (PI), which is the sum of the cells in all generations divided by the calculated number of original parent cells. PI was calculated by using FCS express v4.07 (DeNovo Software, Los Angeles, CA, USA).
Flow Cytometric Analysis of Cell Phenotype
For evaluation of phenotype, amniotic cells and monocyte-derived cells were washed with FACS buffer [0.1% sodium azide (Sigma-Aldrich) and 0.1% bovine serum albumin (BSA; Sigma-Aldrich) in PBS]. Cells were then incubated for 20 min at 4°C with anti-human fluorescein isothiocyanate (FITC), PE, APC, Alexa-488 or Alexa-647-conjugated monoclonal antibodies, or isotype-matched controls specified below, together with 20 mg/ml polyglobin (gammagard®; Baxter, Illinois, USA), which was prepared in PBS with 1% BSA and added to block nonspecific binding. To detect intracellular staining of the macrophage marker (see below), cells were fixed and permeabilized with Cytofix/cytoperm solution (BD Biosciences) for 20 min at 4°C. After washing with 1× perm/wash buffer solution (BD Biosciences), cells were stained with the macrophage marker antibody for 30 min at room temperature and washed with 1× perm/wash buffer solution. Dead cells were gated out by propidium iodide (PI, 0.1 μg/ml; Sigma-Aldrich) staining (for cell surface staining) or by side scatter and forward scatter gating (for intracellular staining). Cells were acquired and analyzed on a FACS Calibur machine using CellQuest Software (BD Biosciences).
The dilution, clones, and suppliers of the antibodies used in this study were as follows.
Phenotype of Amniotic Membrane Cells
Monoclonal antibodies against CD13 (dilution 1:500, clone L138), CD44 (1:50, L178), CD73 (1:50, AD2), CD90 (1:500, 5E10), CD117 (1:200, 104D2), CD146 (1:50, P1H12), CD166 (1:100, 3A6), CD326 (1:50, EBA-1), SSEA-4 (1:50, MC813-70), CD49b (1:500, AK-7), CD49c (1:10, C3 II.1), CD49d (1:100, 9F10), CD14 (1:250, MϕP9), CD45 (1:250, 2D1), HLA-ABC (1:100, G46-2.6), and HLA-DR (1:50, TU36) were all purchased from BD Bio-sciences and those against CD105 (1:50, SN6), CD49a (1:50, TS2/7), and CD49f (1:50, NKI-GoH3) from Serotec (Oxford, UK).
Phenotype of Monocytes and Monocyte-Derived Cells
Monoclonal antibodies against CD1a (1:150, clone HI149), CD14 (1:250, MϕP9), CD80 (1:100, L307.4), CD83 (1:25, HB15e), CD86 (1:200, 2331, FUN-1), CD197 (1:100, 3D12), CD23 (1:50, M-L233), and CD163 (1:50, GHI/61) were all purchased from BD Biosciences; those against CD32 (1:50, FUN-2) from Biolegend (San Diego, CA, USA); and the macrophage marker (1:1,000, PM-2K) from ACRIS Antibodies (Herford, Germany).
FITC/PE/APC/Alexa-488 or Alexa-647-conjugated mouse IgG1, IgG2b, IgG3, and rat IgG2a served as isotype controls (1:250); all obtained from BD Biosciences, except for FITC-, PE-, and APC-conjugated mouse IgG2b, which were purchased from Biolegend and Alexa-647-conjugated rat IgG2a isotype control from Serotec.
Cytokine Assays
Cell-free supernatants from monocytes differentiated toward DCs±amniotic membrane cells were collected and frozen at −80°C. As controls, supernatants from amniotic cells cultured alone and in the presence of stimulants needed for DC differentiation (GM-CSF, IL-4, and LPS) were also collected and frozen.
A multiplex bead-based immunoassay (BD CBA Flex Set System from BD Biosciences) was used to simultaneously quantify the levels of human IL-1α, IL-1β, IL-6, IL-8, IL-10, IL-12p70, monocyte chemoattractant protein-1 (MCP-1/CCL2), monokine induced by interferon-γ (MIG/ CXCL9), macrophage inflammatory protein (MIP)-1α, MIP-1β, and tumor necrosis factor-α (TNF-α). The bead arrays were analyzed on a FACSAria machine and data evaluated with FCAP Array software (BD Biosciences).
Statistical Analysis
Data are expressed as mean±standard deviation (SD). Comparison between two samples was performed using the paired sample t-test. Analysis of variance was used to assess differences between groups. The t-test and analysis of variance were performed using 2010 Microsoft Excel (Microsoft Corporation, Redmond, WA, USA). Raw p values were adjusted by Holm-Bonferroni's procedure for multiple comparisons. A value of p < 0.05 was considered statistically significant.
Results
Characterization of hAMTCs and hAECs
The morphology and immunophenotype of P0 and cells expanded up to P4 were examined.
At P0, hAMTCs formed a monolayer of fibroblast-like cells, with the typical mesenchymal stromal morphology, whereas the hAECs were cuboidal in shape and displayed the typical epithelial cobblestone appearance. These adherent cells could be expanded without visible morphological changes at P4 under our culture conditions (data not shown).
To ensure that the different cell preparations were comparable with each other in terms of their immunophenotype, batches of each cell preparation were cryo-preserved after isolation and serial passaging, and then simultaneously thawed and analyzed by flow cytometry. Specifically, cells were analyzed for the expression of: i) a panel of well-described mesenchymal markers [i.e., CD13, CD44, CD73, CD90 (Thy-1), CD105 (endoglin), CD117 (c-kit), CD146 (the melanoma cell adhesion molecule described for pericytes, as well as for MSCs with colony-forming unit-fibroblast capacity) (17), and CD166 (ALCAM)] (20,45); ii) the epithelial marker CD326 (EpCAM); iii) the stage-specific embryonic antigen SSEA-4; iv) different types of integrins (CD49-a, CD49-b, CD49-c, CD49-d, CD49-f); v) the hematopoietic markers CD14 and CD45; and iv) HLA-A, -B, -C, and HLA-DR major histocompatibility antigens.
Consistent with our previous findings (48), the hAMTCs expressed CD13, CD44, CD73, and CD90 from P0 to P4. The number of CD73+, CD90+, and CD105+ cells increased with passaging (Fig. 1A). CD117, CD146, and CD166, as well as EpCAM and SSEA-4 were expressed by very few cells (Fig. 1A). All of the examined integrins were expressed, although at different levels, and the number of integrin+ cells increased with expansion except for CD49a (Fig. 1A). The hAMTCs also expressed HLA-A, -B, and -C antigens, and the percentage of positive cells decreased during culture passages. Interestingly, as we previously reported (46), hAMTCs also included a subpopulation of cells positive for CD14, CD45, and HLA-DR, and this subpopulation decreased markedly during culture passages (Fig. 1A).
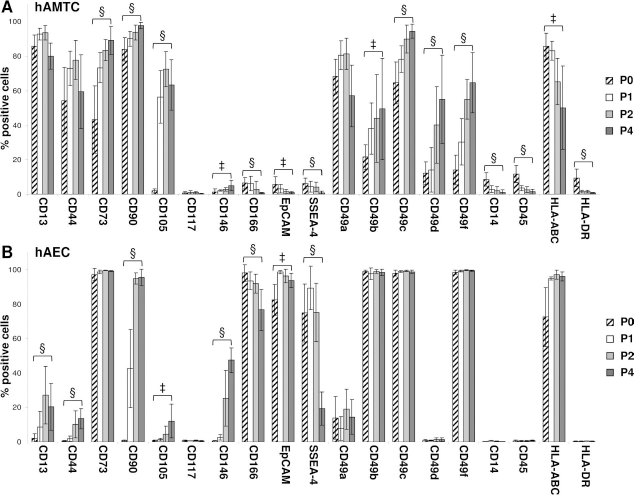
Immunophenotype of human amniotic mesenchymal tissue cells (hAMTCs) and human amniotic epithelial cells (hAECs) at different culture passages. hAMTCs (A) and hAECs (B) at passage 0 (P0), P1, P2, and P4 were analyzed for markers shown in the figure using flow cytometry. The figure shows the percentages of cells staining positive for each marker. The data are expressed as mean ± SD of at least five (hAMTCs) and six (hAECs) different cell preparations. Statistically significant differences of cells at P0 versus cells at P4: ‡p < 0.01; §p < 0.001.
The hAECs expressed the epithelial marker EpCAM and SSEA-4 (Fig. 1B), as reported previously (9,18,28, 65,73). Among the mesenchymal markers tested, CD73 and CD166 were expressed at high levels both at P0 and subsequent passages, while CD90 was absent at P0, but the numbers of positive cells rapidly increased with passaging (Fig. 1B). CD13, CD44, CD105, and CD146 were present in few P0 cells and increased moderately following passaging; similarly to hAMTCs, CD117 was present in a few hAECs (Fig. 1B). In terms of the integrins, P0 and passaged hAECs expressed CD49b, CD49c, and CD49f, while some cells expressed CD49a, and almost none expressed CD49d (Fig. 1B). As previously reported (9,18,73), we confirmed that hAECs do not express CD14 and CD45, and are negative for HLA-DR, but are positive for HLA-A, -B, and -C and the percentage of positive cells, in contrast with hAMTCs, remained fairly unchanged during culture (Fig. 1B).
Effects of P0 Cells on the Differentiation of Monocytes Toward DCs
To study the effects on DC development/function, CD14+ monocytes were differentiated toward DCs with GM-CSF and IL-4 followed by exposure to LPS. This was performed in the absence or presence of hAMTCs or hAECs, either in direct contact with monocytes or with physical separation using a Transwell system. Cells resulting from these cultures are referred to as mDCs (mature DCs) for monocytes differentiated in the absence of amniotic membrane-derived cells; hAMTC-DCcont and hAMTC-DCtw for monocytes differentiated toward DCs in the presence of hAMTCs, in cell-to-cell contact (cont), and Transwell (tw) settings, respectively; and hAEC-DCcont and hAEC-DCtw for monocytes differentiated toward DCs in the presence of hAECs, in cell-to-cell contact (cont), and Transwell (tw) settings, respectively. Using FACS analyses, a combination of the presence or absence of various surface markers was used to assess the differentiation state of these cells.
As expected, mDCs were CD1a+ and did not express the monocytic marker CD14 (Fig. 2, Table 1). The mDCs were positive for the activation marker CD197 (CCR7), the costimulatory molecules CD80 (B7.1) and CD86 (B7.2), and the DC maturation marker CD83 (Fig. 2, Table 1).

Effect of PO hAMTCs and hAECs on differentiation of monocytes to dendritic cells (DCs). Phenotypic analysis of CD14+ monocytes and cells resulting from their culture under DC differentiation conditions, in the absence (mDC) or presence of PO hAMTCs or hAECs, in cell-to-cell contact (hAMTC-DCcont and hAEC-DCcont) and Transwell (hAMTC-DCtw and hAEC-DCtw) coculture settings. The markers reported in the figure were evaluated using flow cytometry by incubation with corresponding monoclonal antibodies (open histograms) or isotype-matched IgGs (control, filled histograms). The percentage of positive cells is indicated in each plot. The results are representative of at least four individual experiments.
Effect of Passage 0 (PO) Human Amniotic Mesenchymal Tissue Cells (hAMTCs) and Human Amniotic Epithelial Cells (hAECs) on Differentiation of Monocytes Toward Dendritic Cells (DCs)

Percentage of positive monocyte DCs to indicated markers. Mean±SD of four (hAMTC-DCcont and hAMTC-DCtw) or five (hAEC-DCcont and hAEC-DCtw) experiments. Experiments were performed using three, four, and five different preparations of monocytes, hAMTCs, and hAECs, respectively. Mo, monocyte; mDC, mature dendritic cells; hAMTC-DCcont and hAMTC-DCtw, monocytes differentiated toward DCs in the presence of hAMTCs in contact (cont) and Trans well (tw) conditions; hAEC-DCcont and hAEC-DCtw, monocytes differentiated toward DCs in the presence of hAECs in contact (cont) and Transwell (tw) conditions. The monocyte/amniotic cell ratios are shown. Statistically significant differences to control mDC:
p<0.001,
p<0.05,
p<0.01.
In contrast, both hAMTC-DCcont and hAMTC-DCtw were CD14+, and the presence of CD1a+ cells was completely abolished (hAMTC-DCcont) or greatly reduced (hAMTC-DCtw) (Fig. 2, Table 1). The percentage of cells expressing CD80 and CD83 was greatly reduced compared to that of mDCs. Of note, the percentage of CD86+ cells did not change (the entire population expressed CD86, as well as the control mDCs) (Fig. 2, Table 1), but the level of expression was significantly reduced. In fact, the median fluorescence intensity (MFI) of CD86 was 39.3 ± 28.0 for hAMTC-DCcont and 57.2 ± 35.2 for hAMTC-DCtw compared to a MFI of 223 ± 133 for mDCs (p < 0.05). A reduced percentage of cells with the DC activation/maturation marker CD197 was also observed (Fig. 2, Table 1). Moreover, both hAMTC-DCcont and hAMTC-DCtw expressed CD32, CD163, and CD23 and were positive for the macrophagespecific antibody (clone PM-2K) (Fig. 2, Table 1). This antibody has been reported to be negative for DCs and monocytes and can be used to differentiate between monocytes, macrophages, and DCs (75).
These findings confirm that hAMTCs strongly inhibit the generation of cells with a DC phenotype, when cultured in contact and in Transwell conditions with stimulated monocytes. They also demonstrate that the monocytes that are unable to differentiate toward a dendritic lineage shift toward a macrophage phenotype (clone PM-2K+) and specifically a M2-like macrophage due to the presence of M2 markers CD14, CD163, and CD23, and the decreased expression or absence of M1 markers CD80 and CD197.
The presence of hAECs resulted in a significant reduction in cells positive for CD1a and CD197 and a slight increase in the percentage of cells expressing CD14 compared to mDCs (for hAEC-DCcont and hAEC-DCtw). The reduction of CD1a was less evident in Transwell cultures, even though no statistically significant difference was seen between hAEC-DCcont and hAEC-DCtw (Fig. 2, Table 1). hAEC-DCcont and hAEC-DCtw also expressed CD32 and low levels of CD23, but not CD163 and the macrophage marker PM-K2 (Fig. 2, Table 1). In addition, the costimulatory molecules CD80, CD83, and CD86 did not change (both in terms of percentage and MFI) compared with the control mDCs (Fig. 2, Table 1). These data indicate that hAECs alter monocyte differentiation toward DCs, inducing myeloid cells at an intermediate state of differentiation/maturation. Taken together, these results show that hAECs and hAMTCs are both able to inhibit the initial differentiation of DCs from monocytes, but that the suppressive effect of hAECs was not as strong as hAMTCs. Moreover, the resulting phenotype of the cells exposed to hAECs or hAMTCs is dissimilar.
Effects of Passaged Cells on Differentiation of Monocytes Toward DCs
We then analyzed the effect of hAMTC and hAEC at P2 and P4 on the differentiation of monocytes toward DC.
Specifically, in the cell contact system, the inhibitory capacity of P2 hAMTCs was similar to that observed at P0 using the same ratio of cells (monocyte/hAMTC ratio of 1:0.05). In the Transwells, using the same ratio of P0 cells (monocyte/hAMTC ratio of 1:0.2), again we observed a modulation of the phenotype profile similar to that found with P0 cells. However, in order to obtain the same statistical significance observed with P0 cells, it was necessary to increase the number of P2 hAMTC (monocyte/hAMTC ratios of 1:0.4 and 1:0.8). In these coculture conditions, modulation occurred in a dose-dependent manner, and changes in phenotypic profile reached statistical significance (Table 2).
Effect of P2 hAMTC on Differentiation of Monocytes Toward DCs

Percentage of positive monocyte DCs to indicated markers. Mean±SD of at least three experiments. Experiments were performed using three and four different preparations of monocytes and hAMTCs, respectively. Mo, monocyte; hAMTC, human amniotic mesenchymal tissue cells; mDC, mature dendritic cells; hAMTC-DCcont and hAMTC-DCtw, monocytes differentiated toward DCs in the presence of hAMTCs in contact (cont) and Transwell (tw) conditions. The monocyte/hAMTC ratios are shown. Statistically significant differences to control mDC:
p<0.001
p<0.01
p<0.05.
The inhibitory capacity of P0 hAECs was strongly reduced when P2 hAECs were tested in cell contact and Transwells. In contact cultures the only statistically significant effect was a slight reduction in CD1a and induction of CD32 compared with mDCs, even when the hAEC numbers were increased (monocyte/hAEC ratios of 1:0.1 and 1:0.2). In Transwells, the only statistically significant effect was a slight induction of CD14 and CD32 at all ratios tested (hAEC/monocyte ratios of 1:0.2, 1:0.4, and 1:0.8). When hAEC numbers were increased (hAEC/ monocyte ratio of 1:0.8) we also observed an increase in the expression of CD23 (Table 3).
Effect of P2 hAECs on Differentiation of Monocytes Toward DCs

Percentage of positive monocyte DCs to indicated markers. Mean±SD of two experiments. Experiments were performed using two different preparations of monocytes and hAECs. Mo, monocyte; hAEC, human amniotic epithelial cells; mDC, mature DCs; hAEC-DCcont and hAEC-DCtw, monocytes differentiated toward DCs in the presence of hAECs in contact (cont) and Transwell (tw) conditions. Monocyte/hAEC ratios are shown. Statistically significant differences to control mDC:
p<0.05,
p<0.01.
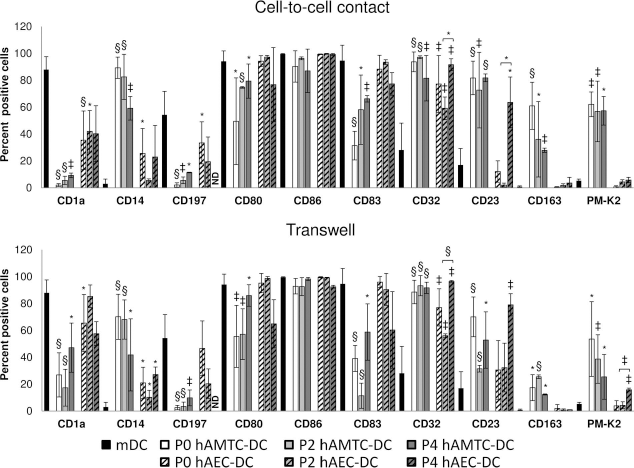
We also performed a study whereby we compared hAMTCs and hAECs at different passages up to P4 at the monocytes/amniotic membrane-derived cell ratio of 1:0.05 for cell-to-cell contact and 1:0.2 for Transwell conditions at P0 and 1:0.4 for Transwell conditions at P2 and P4 (Fig. 3). Interestingly, we observed that the inhibitory capacity of hAMTCs was consistent during cell passages. Specifically, significant changes in phenotypic profile found at P2 were retained up to P4, and no additional changes were observed due to passages (Fig. 3). Moreover, when compared to hAMTCs, hAECs possessed a reduced capacity of modulating DC differentiation at different passages. However, the ability of hAECs to modulate CD32 and CD23 observed at P2 was confirmed and even more evident at P4, as well as a slight increase of PM-K2 only in the Transwell condition (Fig. 3).

Effect of passaged hAMTCs and hAECs on differentiation of monocytes to DCs. Phenotypic analysis of monocytes differentiated to DC in the absence (mDC) or presence of P0, P2, and P4 hAMTCs or hAECs (P0 hAMTC-DC, P2 hAMTC-DC, P4 hAMTC-DC and P0 hAEC-DC, P2 hAEC-DC, P4 hAEC-DC), in cell-to-cell contact (upper panel) and Transwell (lower panel) coculture settings. The percentage of the different markers analyzed is reported as mean ± SD of at least four individual experiments. Statistically significant differences of P0 hAMTC-DC, P2 hAMTC-DC, P4 hAMTC-DC, and P0 hAEC-DC, P2 hAEC-DC, P4 hAEC-DC versus control mDCs: *p < 0.05; ‡p < 0.01; §p < 0.001. The brackets show where statistically significant differences were observed between P2 and P4. ND, value not determined.
Effects of CM-hAMTC and CM-hAEC on Monocyte Differentiation Toward DCs
An inhibitory effect of hAMTCs on DC differentiation was observed even when the hAMTCs were in Transwells, suggesting the involvement of hAMTC-secreted factors. To test this hypothesis, we treated monocytes with CM obtained from P0 hAMTCs and found that the effects were similar to that of hAMTC-DCtw, with the exception of CD80 (Fig. 4, Table 4). Indeed, as previously observed for CD86, the percentage of CD80+ and CD86+ cells did not change (compared to the control mDCs), but changes were seen in the intensity of expression (MFI CD80 10.3 ± 1.3, CD86 72.2 ± 35.2 for CM-hAMTC-DC; MFI CD80 20.1 ± 2.13, CD86 162 ± 48.2 for mDC; p < 0.001 for CD80 and p < 0.05 for CD86). These data indicate that the immunomodulatory effect exerted by hAMTCs is predominantly due to the constitutive secretion of inhibitory factors. We also tested the effect of CM-hAEC and obtained similar results as those from hAEC-DCtw. Specifically, we observed a slight reduction of CD1a and CD197, accompanied by a small retention of CD14, and the increased expression of CD32. Moreover, in the presence of CM-hAEC, we observed the upregulation of CD23, compared to the control mDCs (Fig. 4, Table 4).
Effect of conditioned medium (CM)-hAMTC and CM-hAEC on differentiation of monocytes to DCs. Phenotypic analysis of CD14+ monocytes and cells resulting from their culture under DC differentiation conditions, in the absence (mDC) or presence of CM-hAMTC or CM-hAEC. Markers reported in the figure were evaluated using flow cytometry after incubation with corresponding monoclonal antibodies (open histograms) or isotype-matched IgGs (control, filled histograms). The percentage of positive cells is indicated in each plot. The results are representative of at least four individual experiments.
Effect of Conditioned Medium (CM) on Differentiation of Monocytes Toward DCs

Percentage of positive monocyte DCs to indicated markers. Mean±SD of at least four experiments. Experiments were performed using three different preparations of monocytes and CM-hAMTC or CM-hAEC pools. mDC, mature DCs; CM-hAMTC-DC and CM-hAEC-DC, monocytes differentiated toward DCs in the presence of 50% CM obtained by hAMTCs and hAECs, respectively. Statistically significant differences to control mDC: *p < 0.001, †p < 0.01, ‡p < 0.05.
Effect of hAMTC- or hAEC-Treated Monocytes on T-Cell Proliferation
Next, it was important to assess whether the altered immunophenotype corresponded to an alteration in their functions. Given that mature DCs stimulate T cells, we tested whether P0 hAMTC-DCcont, hAMTC-DCtw, hAEC-DCcont, and hAEC-DCtw could act as stimulators in T-cell proliferation assays. We found that exposure to amniotic cells reduced the ability to stimulate the proliferation of allogeneic T-lymphocytes, although at different levels. Indeed, hAMTC-DCtw and especially hAMTC-DCcont significantly impaired T-cell proliferation compared to mDCs. hAEC-DCcont also had a significantly reduced stimulatory ability with respect to mDCs and similar to that of hAMTC-DCtw. In contrast, hAEC-DCtw stimulated T-cell proliferation as seen for mDCs (Fig. 5A).
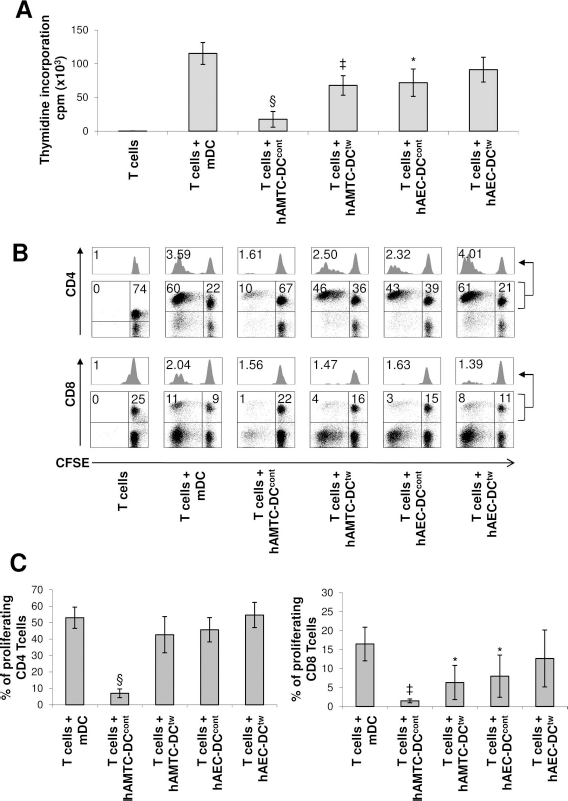
Effect of P0 hAMTC- and hAEC-treated monocyte-derived DCs on T-cell proliferation. (A) Monocyte-derived cells that had been differentiated in the absence (mDC) or presence of hAMTCs (hAMTC-DCcont and hAMTC-DCtw) and hAECs (hAEC-DCcont and hAEC-DCtw) were collected after culture and used as stimulators to induce proliferation of alloreactive T cells (responders). T-cell proliferation was assessed by [3H]thymidine incorporation after 5 days of culture and expressed in counts per minute (cpm). The results shown are the mean ± SD of five individual experiments. (B) Representative flow plots of CD4+ (upper panel) and CD8+ (lower panel) CFSE-labeled T cells cocultured for 6 days with mDCs, hAMTC-DCcont, hAMTC-DCtw, hAEC-DCcont, and hAEC-DCtw. Above each dot plot, the proliferation histogram of CD4- or CD8-gated T cells is reported. Within each dot plot, the percentage of proliferating (upper-left quadrant) and nonproliferating (upper-right quadrant) T cells is indicated. Within histograms, the proliferation index of CD4 or CD8 T cells is indicated. C) CD4+ and CD8+ proliferating CFSE-labeled T cells under different coculture conditions. The results shown are the mean ± SD of four individual experiments. Data shown in (A) and (C) are generated from two preparations of monocytes, five (A) or four (B) preparations of amniotic cells. Statistically significant differences of hAMTC-DCcont, hAMTC-DCtw, hAEC-DCcont, and hAEC-DCtw versus control mDCs: *p < 0.05; ‡p < 0.01; §p < 0.001.
Since mDCs may differ in their ability to induce CD4+ and CD8+ T-cell proliferation (6), we also investigated the effects of hAMTC-DCcont, hAMTC-DCtw, hAEC-DCcont, and hAEC-DCtw on the proliferation of these T-cell subtypes. hAMTC-DCcont almost completely impaired CD4+ and CD8+ T-cell proliferation. hAMTC-DCtw and hAEC-DCcont displayed a reduced ability only toward CD8+ T-cell proliferation, and hAEC-DCtw did not significantly affect the T-cell stimulatory capability of CD4+ and CD8+ T cells (Fig. 5B, C).
Impact of hAMTCs and hAECs on DC Cytokine Production
To investigate whether the presence of amniotic membrane-derived cells could interfere with cytokine production by DCs, we studied the output of several factors. These included proinflammatory cytokines involved in T-cell priming and maturation (IL-1β, IL-12p70, TNF-α, IL-6, and IL-8); anti-inflammatory cytokine (IL-10), chemokines of the CC subfamily (MCP-1/CCL2, MIP-1α, and MIP-1β), and the CXC subfamily (and MIG/ CXCL9), which are involved in monocyte/macrophage and lymphocyte chemoattraction. These factors were measured in supernatants collected from monocyte cultures treated with P0 amniotic cells in Transwell and cell-to-cell contact settings. The supernatants from P0 hAMTCs or hAECs treated with ±GM-CSF, IL-4, and LPS served as additional controls. Data from control cultures are as follows. hAMTCs cultured in the presence of GM-CSF, IL-4, and LPS did not produce IL-1β, IL-12p70, TNF-α, IL-10, and MIG/CXCL9. Instead, they secreted IL-6 (contact: 7.738+1.837 ng/ ml; Transwell 8.989 + 0.224 ng/ml), IL-8 (contact: 9.994±5.298 ng/ml; Transwell: 8.679±6.737 ng/ml), MCP-1/CCL2 (contact: 7.812±2.079 ng/ml; Transwell: 10.264±0.831 ng/ml), MIP-1β (contact: 2.595±1.489 ng/ml; Transwell: 3.314±2.256 ng/ml), and MIP-1α (contact: 410 ± 205 pg/ml; Transwell: 497 ± 282 pg/ml). hAECs cultured in the presence of GM-CSF, IL-4, and LPS did not produce IL-1β, IL-12p70, TNF-α, IL-10, MIG/CXCL9, MCP-1/CCL2, MIP-1α, and MIP-1β. On the other hand, they produced low levels of IL-6 (contact: 100±10 pg/ml; Transwell: 6.51±2.24 pg/ml) and IL-8 (contact: 68.9 ± 43.8 pg/ml; Transwell: 68.2 ± 27.5 pg/ml).
We found that the supernatants from hAMTC-DCcont and hAMTC-DCtw had significantly reduced levels of MIG/ CXCL9, IL-12p70, TNF-α, IL-8, and MIP-1-α, compared with the samples from mDCs (Fig. 6). The levels of IL-10 were significantly higher in hAMTC-DCcont and hAMTC-DCtw compared to mDCs (Fig. 6). We also detected a significant increase in the amount of MCP-1/CCL2 and IL-1-β production when monocytes were exposed to hAMTCs or their supernatants (Fig. 6). However, it is of note that the observed increase in MCP-1/CCL2 may be influenced by the fact that hAMTCs secrete this cytokine. No significant differences were observed in the supernatants from hAMTC-DCcont and hAMTC-DCtw for the other cytokines investigated (IL-6, IL-1-α, and MIP-1β) when compared with the samples from mDCs (Fig. 6).
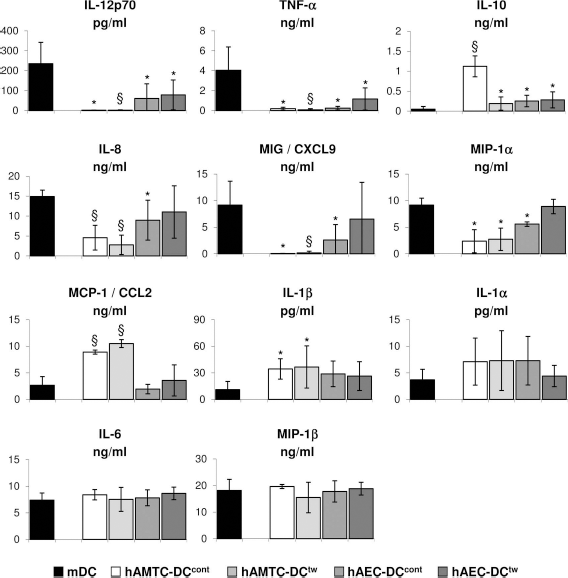
Effect of P0 hAMTCs and hAECs on DC cytokine production. The cytokines/chemokines were quantified from supernatants from monocyte cultures differentiated toward DCs in the absence (mDC) or presence of P0 hAMTCs or P0 hAECs, both in cell-to-cell contact and Transwell conditions. The results are the mean ± SD of three individual supernatants for each cell population. Statistically significant differences of hAMTC-DCcont, hAMTC-DCtw, hAEC-DCcont, and hAEC-DCtw versus control mDCs: *p < 0.05; §p < 0.001.
In the supernatants of the cocultures established using hAECs, similar to the results obtained with hAMTCs, we found significantly reduced production of IL-12p70 and TNF-α by hAEC-DCcont and hAEC-DCtw compared to mDCs (Fig. 6), whereas the reduction in MIG/CXCL9, IL-8, and MIP-1α reached a statistical significance only for hAEC-DCcont (Fig. 6). hAEC-DCcont and hAEC-DCtw also produced a significantly higher amount of IL-10 with respect to mDCs (Fig. 6). The levels of MCP-1, IL-1β, IL-1α, and MIP-1β were not significantly affected by the exposure of stimulated monocytes to hAECs, either in contact or in Transwell settings (Fig. 6).
Discussion
We have previously demonstrated that hAMTCs strongly inhibit the differentiation and function of monocyte-derived DCs in vitro when cultured in Transwell settings (44). Here we extended the study to include primary and expanded hAMTCs and hAECs in cell-to-cell contact-dependent and independent coculture systems. We also evaluated the effects of secreted factors from amniotic cells on DC differentiation, the influence of amniotic cells on cytokine production by the differentiated monocytes, and the effects of these monocytes on T-cell proliferation.
We found that hAMTCs affect DC generation from monocytes when cultured in contact or in a Transwell setting. The Transwell system demonstrated the involvement of inhibitory secreted factor(s), consistent with our previous findings where we reported the inhibitory action of hAMTCs on lymphocytes (46) and on the proliferation of different cancer cell lines (48), which occurs independently of cell contact. This is in contrast to studies from mesenchymal stem cells (MSCs) obtained from bone marrow where cell-to-cell contact (34,64), or the addition of other cells (monocytes, dendritic cells) (8,25,41), was a prerequisite for their immunomodulatory effects. Moreover, the CM-hAMTC inhibited DC differentiation as effectively as the Transwell cultures. This suggests that the secretion of inhibitory factors by hAMTCs can occur independently of DC-activating stimuli and is in agreement with a previous study showing the inhibitory action of CM-hAMTC on T-cell proliferation in vitro (33,68). However, we found that cell-to-cell contact plays an important role; fewer numbers of hAMTCs were needed to inhibit DC differentiation compared with the Transwell cultures (1:0.05 vs. 1:0.2). Since the set of molecules secreted by the cells, referred to as the secretome, could vary depending on the passage and culture conditions, it is tempting to speculate that partially diverging secretomes of culture-expanded hAMTCs may underlie the differences in suppressing DC generation observed with passaged cells. However, this remains to be determined.
Although in a different manner compared to hAMTCs, we found that hAECs could also affect the differentiation of monocytes toward DCs in cell-to-cell contact and Transwell settings. Contrasting data regarding the involvement of inhibitory secreted factors in hAEC immunomodulatory actions have been reported. Some reports demonstrate that the CM from hAECs inhibits T- and B-cell proliferation in vitro (40) and possesses anti-inflammatory properties able to inhibit corneal inflammation (32). On the other hand, other studies demonstrate that the immunomodulatory effect on T cells is dependent on cell-to-cell contact (5,80). Moreover, Banas and colleagues (4) underline the importance of cell-to-cell contact as a prerequisite for a subpopulation of hAECs (termed amnion progenitor cells) to modulate monocyte-derived DC phenotype. In our experimental setting, even the use of CM-hAEC affected the phenotype of monocytes differentiating toward DCs, confirming the production of inhibitory secreted factor(s). However, effects on T cells and the cytokine profile of the resulting monocytes were more pronounced in the cell-to-cell contact conditions compared to the Transwell setting. Hence, it is conceivable that direct contact between monocytes and hAECs would play a more important role during the differentiation and maturation of DCs than indirect action of hAEC-secreted factors. However, in contrast to what we observed with hAMTCs, the inhibitory capacity of hAECs was lower even when the number of hAECs was increased. In terms of surface markers, passaged hAECs acquired mesenchymal markers (including CD13, CD44, CD90, CD105, and CD146) while retaining the epithelial marker EpCAM, and CD166 and SSEA-4 markers that were present in low levels or absent in hAMTCs. It has been reported that expanded hAECs undergo phenotypic changes consistent with an epithelial–mesenchymal transition (2,9,18,65), along with changes in immunosuppressive properties and secretion of immunomodulatory factors (65). Indeed the immunomodulatory capacity of cells derived from the amniotic membrane (76), or other perinatal sources such as Wharton's jelly (37,38), after culture passages is an important matter of study given the relevancy for clinical application of these cells.
Interestingly, we observed that hAMTCs had a consistent capacity to inhibit monocyte differentiation to DCs, and to induce M2 differentiation, both in contact and in Transwell conditions, up to passage 4. On the other hand, hAECs had a decreased potential to inhibit differentiation toward DCs, and we observed that their modulatory capacity changed during passaging.
Irrespective of the differences, hAMTCs and hAECs impacted on the phenotype of monocytes. In our previous study (47) we found that monocytes differentiated toward DCs in the presence of hAMTCs and showed an expression pattern similar to that observed for monocytes before induction of differentiation. In this study, we demonstrated that hAMTCs do not simply block the differentiation at the monocytic level, but skewed differentiation to cells with macrophage features. Monocytes are precursors of both DCs and macrophages, therefore a switch/shift induced by hAMTCs toward one direction rather than another could influence the outcome of the immune response. The ability to induce this shift in monocyte differentiation has been demonstrated through in vitro studies for MSCs from bone marrow (14,29,60,67,81), umbilical cord Wharton's jelly (69), as well as for fibroblasts (15,19), and human renal tubular epithelial cells (35). Monocyte-macrophages are a heterogeneous group of cells, and the term M1 was proposed for proinflammatory macrophages, and M2 for macrophage activation other than M1, involved in type II responses, immunoregulation, and tissue remodeling (50). Although the characterization of human M1 and M2 profiles is far from complete, some surface markers suggested for M1 macrophages include the chemokine receptor CCR7 (CD197) and high levels of the costimulatory molecules CD80 and CD86, while for M2 macrophages CD14, CD23 (Fc∊-RII), the scavenger receptors CD163 and CD204, the mannose receptor CD206, and CD209 (DC-SIGN) were suggested (50,54,77). In accordance with these markers, there appears to be a shift toward M2-like macrophages. Indeed, the resulting monocytic cells expressed the macrophage-specific antibody clone PM-2K, and are positive for the M2 markers CD14, CD163, and CD23, while they lacked or expressed low levels of the M1 markers CD197 and CD80.
In terms of hAECs, we are unable to describe the nature of cells resulting from the cocultures. Indeed, these cells had a phenotype that did not resemble the initial monocytes, macrophages, or mature DCs, but seem to be myeloid cells at an intermediate state of differentiation/maturation. Fewer cells had CD1a and CD197, indicating an immature maturation state, but also had high levels of costimulatory molecules CD80, CD86, and CD83. Moreover the cells had CD32, which is usually expressed on macrophages, but neither the macrophage marker CD163 nor PM-K2, except for this last marker only at P4 and only in the Transwell condition. Interestingly, not only with hAMTCs, but also hAECs (in some experimental conditions, when we used CM-hAECs or high number of passaged cells), we observed a high percentage of Fc∊RII/CD23+ cells in the resulting cocultured monocytes. CD23 has been identified as low-affinity receptor for IgE and has been described to a play a critical role during the immune response, including IgE synthesis, the secretion of inflammatory mediators by various human cells (monocytes, eosinophils, B cells, and DCs), and B- and T-cell differentiation (24). Whether CD23 is implicated in the functions of the amniotic cell stimulated monocytes remains to be clarified.
Within DC functions, the ability to stimulate T cells and to produce inflammatory cytokines has a central role in promoting the immune response of effector lymphocytes (16). In this study, we have shown that hAMTC-DCcont and hAMTC-DCtw had an impaired ability to stimulate allogeneic T-lymphocyte proliferation. We suggest that the altered immunophenotype of the resulting cells corresponded to an alteration in their functions, as previously demonstrated (47). In contrast, only the hAEC-DCcont (and not hAEC-DCtw) had a significantly reduced ability to stimulate T-cell proliferation compared to mDCs. These results provide additional evidence of the different effects of hAMTCs versus hAECs in the control of monocyte-DC differentiation. Moreover, we observed a differential ability of hAMTC-DC and hAEC-DCs to affect CD4+ and CD8+ T-cell proliferation. hAMTC-DCcont almost completely impaired CD4+ and CD8+ T-cell proliferation, whereas hAMTC-DCtw and hAEC-DCcont reduced only CD8+ T-cell proliferation. Since CD8+ T cells play a central role in transplantation rejection these data suggest that amniotic cells could play an important role during cotransplantation for the improvement of cell engraftment and prevention of graft versus host disease. Monocytes derived from coculture with hAMTCs and hAECs showed some differences in phenotype and T-cell stimulatory properties, in accordance with the cytokine/chemokine profile measured.
It is known that upon differentiation/maturation, DCs produce several types of cytokines, which will influence the outcome of the immune response. In particular, fully mature DCs produce proinflammatory cytokines such as IL-12p40/p70 and TNF-α that might be crucial to induce T-cell immunity (44). We have previously reported that the presence of hAMTCs cultured in Transwell conditions with monocytes differentiated toward fully mature DCs significantly blocked the production of proinflammatory cytokines, such as TNF-α, MIG/CXCL9, CXCL10/IP-10, and CCL5/RANTES (47). Here we also show that exposure to hAMTCs (both in contact and Transwell conditions) significantly reduced IL-12p70, IL-8, and MIP-1α. Moreover, supernatants from cocultures of hAMTCs and monocytes contained higher concentrations of IL-10 compared to hAMTCs alone or mDCs, suggesting that IL-10 was produced by monocytes after exposure to hAMTCs. Consistent with other reports (35,55), this indicates that monocytes differentiated into DCs in the presence of amniotic membrane-derived cells become IL-10-producing cells. Other inflammatory cytokines such as IL-8, CXCL9, and MIP-1α were again significantly reduced. Thus, the inhibition of Th1 inflammatory cytokines (in particular IL-12, TNF-α, and CXCL9) and the increased levels of IL-10 and CCL2 (a chemokine associated with Th2 polarization) (26) support the data obtained from the phenotype of monocytes differentiated in the presence of hAMTCs, indicating a switch of monocytes toward M2 macrophages, cells that participate in polarized Th2 reactions, promote tissue repair and remodeling, and have immunoregulatory functions (49). When monocytes were differentiated toward DCs in the presence of hAMTCs (in both contact and Transwell conditions), in addition to the increased levels of IL-10 and CCL2, we also observed a significant increase in IL-1β, in accordance with a previous report regarding the effect of passaged amniotic mesenchymal cells on monocyte-DC differentiation (35). Interestingly, the production of IL-1β by monocytes was proposed to be one of the activating signals that BM-MSCs require to secrete T-cell inhibitory molecules (25). In the case of monocytes differentiated with hAECs (in both contact and Transwell conditions), we observed a significant inhibition of IL-12p70 and TNF-α and the increase in IL-10, indicating the ability of hAECs to also promote the induction of monocytes with anti-inflammatory properties. To the contrary, the inhibition of the proinflammatory cytokines IL-8, CXCL9, and MIP-1α was significantly decreased only in hAEC-DCcont conditions compared to mDCs, confirming the need of cell-to-cell contact between monocytes and hAECs to play a more important role during the differentiation and maturation of DCs. The different ability of hAECs to modulate the cytokine secretion by monocyte-derived DCs is in agreement and could account for the different T-cell stimulatory ability discussed above.
Conclusion
In conclusion, our data show the contribution of both the mesenchymal and epithelial populations derived from the amniotic membrane in the dendritic maturation process, and we demonstrate that hAMTCs and hAECs do not have the same potential to inhibit the generation of mDCs. By comparing their effects in different culture settings and even by using CM derived from the culture of these cells in nonstimulating conditions, we clearly show the higher modulatory capacity exerted by hAMTCs compared to hAECs. Therefore, this study provides data on the capacity of these cells and of their mechanisms of immunomodulatory activity that could be relevant for defining their applications in clinical use.
Footnotes
Acknowledgments
The authors thank the physicians and midwives of the Department of Obstetrics and Gynecology of Fondazione Poliambulanza-Istituto Ospedaliero, Brescia, Italy, and all of the mothers who donated placenta and the volunteers who donated blood. The authors wish to thank the personnel of Department of Radiation Oncology of Fondazione Poliambulanza-Istituto Ospedaliero, Brescia, Italy, for the assistance with cell irradiation. The authors thank Dr. Antonietta Silini for help in editing the manuscript. This study was supported by Fondazione Cariplo, grant No. 2011-0495 and Competitiveness ROP ERDF 2007-2013 of Lombardy Region (Regional Operational Programme of the European Regional Development Fund–Progetto NUTEC NUove TECnologie ID No. 30263049). U.M. funded by a Monash Researcher Accelerator Award from Monash University and supported by the Victorian Government's Operational Infrastructure Program. The authors declare no conflicts of interest.
