Abstract
Amniotic epithelial cells (AECs) are ideal seed cells for tissue regeneration, but no research has yet been reported on their tendon regeneration potential. This study investigated the efficiency of AEC allotransplantation for tendon healing, as well as the mechanism involved. To this aim ovine AECs, characterized by specific surface and stemness markers (CD14-, CD31-, CD45-, CD49f, CD29, CD166, OCT4, SOX2, NANOG, TERT), were allotransplanted into experimentally induced tissue defects in sheep Achilles tendon. In situ tissue repair revealed that AEC-treated tendons had much better structural and mechanical recoveries than control ones during the early phase of healing. Immunohistochemical and biochemical analyses indicated that extracellular matrix remodeling was more rapid and that immature collagen fibers were completely replaced by mature ones in 28 days. Moreover, spatial–temporal analysis of cellularity, proliferation index, vascular area, and leukocyte infiltration revealed that AECs induced a specific centripetal healing process that first started in the tissue closer to the healthy portion of the tendons, where AECs rapidly migrated to then progress through the core of the lesion. This peculiar healing evolution could have been induced by the growth factor stimulatory influence (TGF-β1 and VEGF) and/or by the host progenitor cells recruitment, but also as the consequence of a direct tenogenic AEC differentiation resulting in the regeneration of new tendon matrix. These findings demonstrate that AECs can support tendon regeneration, and their effects may be used to develop future strategies to treat tendon disease characterized by a poor clinical outcome in veterinary medicine.
Introduction
The role of tendon is to transfer muscular forces to bone. For this reason, tendons are constantly exposed to large mechanical loads that may induce injuries. Therefore, tendons can be exposed to trauma during sports activities, but they can also be affected by overuse or aging. The most commonly injured are Achilles and patellar tendons with pathologies raging from degenerative tendinopathies, partial tears, up to complete ruptures (18,30). Although spontaneous healing can occur, more often tendinopathy results in scar formation, which compromises the biomechanical properties of the tissue (55,56,66). For this reason, tendon regeneration still remains a major goal to reach (2,48,50,54).
The poor spontaneous clinical outcome of tendinopathy is likely to be related to the limited regenerative capacity of tenocytes. Thus, a growing interest has been attracted by the use of stem cell-based therapies (33).
In this context, different tendon progenitor cells have been used so far, from autologous or allogeneic tenocytes (12,33) to adult mesenchymal stem cells (MSCs) (6,7,15,25,33,45 and 57, more recently, to embryonic (21,69) and fetal/placental stem cells (44,63,68). However, the cell type that is able to support biologically and mechanically the regeneration of a tissue has not been clearly identified yet.
The use of tenocytes results very difficult either for the invasiveness of the surgical tendon explants retrieval or for the low number of cells that can be isolated from them.
Adult MSCs have been shown to be capable of tenogenic differentiation in vitro (35,64) and to enhance tendon repair in animal models (7,14,25,33,57). However, while adult MSCs appeared to enhance the biomechanical properties of tendons, they did not induce any visible improvement of tissue microstructure (6) while, sometimes, they generated undesirable foci of bone and cartilage (24,26). An explanation for their incomplete healing resides in the inability of MSCs to directly participate to the process of tissue regeneration, a contribution that, on the contrary, can be achieved with genetically modified MSCs. An increased regenerative potential has been recorded, in fact, in MSCs that overexpress genes involved in the tenocyte lineage such as bone morphogenetic protein (BMP)-12 or transforming growth factor-β1 (TGF-β1) (28,29,67), or when the undesirable osteogenic differentiation is switched off by the active form of Mothers against decapentaplegic homolog-8 (Smad8) (26).
Embryonic stem cells (ESCs) have displayed tenogenic potential, but additional research is required to overcome the risks of tumor formation (21,69).
Amnion-derived stem cells are a promising alternative of stem cells to be used in tendon tissue engineering, since they conjugate a remarkable plasticity with safety properties (3,16,41,46). Although the ability of these cells to support tendon healing has not been proved yet, clear evidences indicate that amniotic epithelial cells (AECs) could be a source of pluripotent stem cells (41,60). In addition, amnion components that were derived from early epiblastic cells seem to retain also the immunomodulatory properties required for the maintenance of fetus–maternal tolerance. In fact, several evidences demonstrated that AECs display anti-inflammatory (23,31,43,58), antifibroblastic (62), and antimicrobial properties (59) together with a low immunogenicity and tumurogenicity (27,61). Experimental evidences have recently been provided showing the capacity of amnion and chorion cells to evade or actively suppress immune response in both allogeneic and xenogeneic models (5,34,53,70). Therefore, the present research was designed to assess the therapeutic potential of AECs allotransplanted in experimentally injured sheep Achilles tendons. The sheep was chosen as experimental model for its translational value, since it displays several similarities with horse and human in joint structure, cell turnover, and musculo-skeletal regenerative processes (10,40,65). The research demonstrates that transplanted ovine AECs are a practical strategy to stimulate the early phase of tendon regeneration. AECs are able to survive for up to 28 days in the tendon lesion providing a direct contribution in collagen I (COL1) deposition and a positive paracrine environment, thus accelerating the recovery of normal tendon microstructure and biomechanical properties.
Materials and Methods
AEC Expansion, Labeling, and Storage
AECs were collected from slaughtered sheep of Appenninica breed at 3 months of pregnancy (25–30 cm of length). The cells were obtained from the isolated amniotic epithelial layer after enzymatic digestion (trypsin-EDTA, Sigma Chemical Co., St. Louis, MO, USA). Cell suspensions were seeded in flasks in minimum essential medium Eagle-α modification (α-MEM) supplemented with 20% fetal calf serum (FCS), 1% ultraglutamine, 1% penicillin/streptomycin plus 10 ng/ ml epidermal growth factor (EGF) at a concentration of 3 × 103 cells/cm2. At 70% of confluence, the cells were dissociated by 0.05% trypsin-EDTA and plated again at 3 × 103/cm2 for three consecutive passages. At the end of the in vitro expansion, an aliquot of AECs was immunostained for cytokeratin-8 [diluted 1:200 PBS/1% bovine serum albumin (BSA); Abcam, Cambridge, UK] revealed with a secondary anti-mouse cyanine-3 (Cy3; diluted 1:500 PBS/1%BSA; Sigma-Aldrich, St. Louis, MO, USA) antibody, while cell nuclei were identified with DAPI. Then, 2.5 × 106 AECs were stored in liquid nitrogen in vials. Before transplantation, thawed AECs were stained with the red fluorescent cell linker dye PKH26, according to manufacturer's instructions (Sigma). The PKH26 fluorescent cell linker dye stably incorporates into lipid regions of the cell membrane. Due to this extremely stable fluorescence, PKH26 is the linker dye of choice for in vivo cell tracking and monitoring studies (http://www.sigmaaldrich.com/insite_pkh_fluorescent_cell; http://www.sigmaaldrich.com/etc/medialib/docs/Sigma/General_Information/vol5_issue3_cell_signal.Par.0001.File.tmp/vol5-issue3-cell-signal.pdf).
Cytofluorimetric Analysis
AECs were screened by flow cytometry for the surface molecules cluster of differentiation 14 (CD14), CD29, CD31, CD45, CD49, CD117, and CD166 and for intracellular stem cell markers [telomerase reverse transcriptase (TERT), octamer-binding transcription factor 4 (OCT4), sex-determining region Y box 2 (SOX2), NANOG]. The primary antibodies used are indicated in Table 1. The unconjugated primary antibodies were fluorescein isothiocyanate (FITC) marked by using the Zenon antibody labeling kit (Gibco, Invitrogen, Carlsbad, CA, USA). Staining was performed on 5 × 105 cells/sample with 100 μl of 20 mM EDTA at 37°C for 10 min. For surface antigen staining, cell samples suspended in 100 μl washing buffer containing the appropriate amount of surface antibody and then exposed to 1 ml 0.5% paraformaldehyde. For intracellular antigen staining, cells were suspended in 1 ml of FACS lysing solution (BD) and incubated in perm 2 (1 ml; BD). Cells were then suspended in appropriate amount of intracellular antibody and exposed to 1 ml 0.5% paraformaldehyde. Cells were analyzed on a FACSCalibur flow cytometer (BD), using CellQuest™ software (BD). As quality control, Rainbow Calibration Particles (6 peaks) and CaliBRITE beads (both from BD Biosciences) were used. Debris was excluded from the analysis by gating on morphological parameters (lymphocyte gate); 20,000 nondebris events in the morphological gate were recorded for each sample. Mean fluorescence intensity ratio (MFI ratio) was calculated, dividing the MFI of positive events by the MFI of negative events.
Summary of Primary Antibodies Used in Cytoflourimetric Analysis

CD14, cluster of differentiation 14; FITC, fluorescein isothiocyanate; SOX2, sex-determining region Y box 2; OCT4, octamer-binding transcription factor 4; TERT, telomerase reverse transcriptase; PE, phycoerythrin.
In Vitro Differentiation
The AECs' plasticity to differentiate into mesenchymal tissue lineages was preliminary tested in vitro. For osteogenic differentiation, AECs were cultured on 12-well plates until they reached the 70% confluence. In half of the plates, AECs were exposed to a differentiation medium of 50 μM ascorbic acid, 10 mM β-glycerophosphate, 0.2 μM dexamethasone, 10% FCS, 1% ultraglutamine, and 1% penicillin/streptomycin. Twenty-one days later, osteogenesis was assessed by evaluating Alizarin Red Staining, alkaline phosphatase (ALP) activity, and the expression of bone-related genes. In brief, fixed AECs (cold 4% paraformaldehyde) were incubated in 40 mM Alizarin Red S for 30 min at room temperature (RT) before stopping the reaction with 80% acetone. ALP activity was processed and analyzed according the manufacturer's instruction (BCIP/NBT Liquid Substrate System B1911, Sigma). RT-PCR analysis was performed in order to evaluate the expression of two bone-related genes [Runt-related transcription factor 2 (RUNX2) and osteocalcin (OCN)] as described below. For tenogenic differentiation, AECs were incubated in 12-well plates at 70% confluence and were exposed to α-MEM supplemented with 10% FCS and 50 ng/ml of BMP-12 (Tebu-Bio, Milan, Italy) for 21 days. Tenogenic differentiation was then documented by analyzing the morphological cell phenotype, the expression of COL1 evaluated by immunocytochemistry (see below), and finally by the analysis of COL1, scleraxis B (SCXB), and tenomodulin (TNMD) gene expression by reverse transcriptionpolymerase chain reaction (RT-PCR) as described below.
RNA Isolation and RT-PCR
Total RNA was extracted from samples by TriReagent (Sigma) following manufacturer's instruction, and its integrity was evaluated by 1% agarose gel electrophoresis and ethidium bromide staining. The RNA was treated with DNaseI digestion (Sigma) for 15 min at RT. One microgram of total RNA of each sample was used for reverse transcription reaction with the Oligo dT primer and BioScript™ (Bioline). 2X Ready mix™ Taq PCR reaction mix (Sigma) was used for PCR reaction using 3 μl of cDNA and 0.5 μM of each primer, in a final volume of 25 μl. The PCR gene primers, mRNA sequence GenBank number, product length, and cycles are shown in Table 2. Where ovine sequences of particular genes (OCT4A, TNMD, and SCXB) were unavailable, multiple alignments of various species were performed, and primers were designed to the consensus sequence within regions of high homology of Bos Taurus. The reaction mixtures were incubated for 5 min at 95°C, followed by 95°C for 30 s, 55°C for 30 s, 72°C for 45 s, and 72°C for 7 min. RT-PCR was normalized by the transcriptional levels of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The PCR products were separated on 2% agarose gel stained with ethidium bromide, visualized on a Gel Doc 2000 (Biorad), and analyzed with Quantity One 1-D Analysis software (Biorad). Each reaction was carried out in triplicate.
Primer Sequences and RT-PCR Conditions Applied to AECs

COL1, collagen 1; TNMD, tenomodulin; SCXB, scleraxis B; OCN, osteocalcin; RUNX2, Runt-related transcription factor 2; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
AEC Transplantation Technique
The experimental surgical procedure has been approved by the Ethical Committee CEISA of the University of Chieti and Teramo (approval ID 27/2010/ CEISA/COM of 11/11/2010). Twenty-three sheep of Appenninica breed with a mean weight of 40 kg were used. Under general anesthesia, the pelvic limbs of the animals were placed off with both tarsi under flexion. A 3-cm skin incision was made, starting at 4 cm proximal to the tuber calcis. The medial and more prominent component of Achilles tendon, that is, the tendon of m. flexor digitorum superficialis, was isolated. Using a sterile punch, a full thickness hole, 3 mm in diameter, was realized. Using a randomized choice, the hole of one limb was filled with a total volume of 30 μl composed of a suspension containing 4 × 106 AECs plus fibrin glue (1:1, v/v; Tissucol; Baxter Spa), while the contralateral was filled with fibrin glue alone (6,26,28,29). The paratenon and fascia were closed before skin suture. After surgery animals were kept in a small sheepfold until sacrifice.
Eighteen animals were randomly divided among the three time points (7, 14, and 28 days) in order to process the tendon explants (Ctr and AEC-allotransplanted tendons) for the morphological/molecular analyses. The remaining five animals were sacrificed 28 days after the surgical procedures to use the tendon explants for the biomechanical tests.
Morphological and Immunohistochemical Analysis
The explanted tendons were transversally cut far, at least 5 mm from the injured area. The specimens were placed in liquid nitrogen. Cryosections, 7 μm in thickness, were processed with hematoxylin–eosin (H&E), Herovici (HE; a polychrome stain for differentiating pre-collagen from collagen), and immunohistochemistry.
The H&E and HE staining were combined to collect information on the microarchitecture regeneration and collagen fibers reorganization, respectively, within the injured tendon districts during the different interval points (7, 14, and 28 days). The cellularity, blood vessels, and extracellular matrix fiber organization were used to generate a score in order to describe the different steps of the tendon healing process. In detail, three different scores were adopted to indicate:
a damaged tissue with hypercellularity, great vascular density characterized by blood vessels irregularly distributed within a faint extracellular matrix without organized fibers; an early stage of tendon healing displaying a decreased presence of cells showing round nuclei, abundant deposition of matrix with fibers that started to acquire a parallel orientation, and a widespread and scattered blood vessels network; an early stage of tendon regeneration showing a reduced concentration of cells with a prevalent flattened shape and fusiform nucleus, an extracellular matrix characterized by parallel fibers, and blood vessels both oriented along the longitudinal axis of the healthy portion of the tendon.
The immunohistochemical analyses were performed with the antibodies summarized in Table 3. In brief, the composition of the extracellular matrix was assessed for collagen I (COL1; 1:200/PBS/BSA1%) and III (COL3; 1:500/PBS/BSA1%). Moreover, cell proliferation (Ki-67; 1:50/PBS/BSA1%), vascular organization (von Willebrand factor, vWF; 1:400/PBS/BSA1%), leukocyte infiltration (CD45; 1:100/PBS/BSA1%), TGF-β1 (1:100/PBS/ BSA1%), and vascular endothelial growth factor (VEGF; 1:50/PBS/BSA1%) were analyzed. All antibodies used are sheep-specific. All antigens were revealed with secondary anti-mouse or anti-rabbit Alexa Fluor 488 (diluted 1:400 PBS/1%BSA; Molecular Probes) antibodies, while cell nuclei were identified with DAPI. Primary antibodies were replaced with nonimmune sera as negative controls.
Primary and Secondary Antibodies Used for Tendon Analysis

vWF, von Willebrand factor; TGF-β1, transforming growth factor-β1; VEGF, vascular endothelial growth factor.
Morphometric Analysis
Morphometric analyses were performed with an Axioscop 2plus epifluorescence microscope (Zeiss, Oberkochen, Germany) equipped with a cooled color charge-coupled device camera (CCD; Axiovision Cam, Zeiss) interfaced with an interactive and automatic image analyzer (Axiovision, Zeiss). The data were processed using a KS300 computed image analysis system (Zeiss) and quantified as previously described (38). Briefly, guided programs (macros for KS300) were created to count inside a standard field of 20,000 μm2:
cellularity (total number of DAPI stained nuclei in the field), proliferation index (PI; % Ki-67 green-stained cells/total number of nuclei in the field), total vascular area (VA; vWF-positive area in μm2 in the field), leukocyte infiltration (% CD45-positive cells/total number of nuclei in the field).
Analyses were carried out on at least five different sections of each tendon specimen analyzed (total tendon analyzed/group = 5). Morphometric analyses were performed at 200x of magnification by capturing five images from six contiguous areas starting from the healthy zone (area 0) and proceeding throughout the repairing zones for five consecutive areas. The extension of each zone analyzed was ~300 μm and, as a consequence, the total area analyzed within each section was ~1,800 μm.
Laser Capture Microdissection (LCM) Technique
The frozen sections were briefly air-dried on uncoated glass slides and washed with 70% ethanol. The sections were kept on dry ice at −80°C until they were subjected to LCM. Just before the procedure, the sections were fixed in 70% ethanol for 10 s and stained with H&E according to Hoffman et al. (26). LCM was performed by using a laser capture microdissection (MMI Cellcut device) apparatus. The settings of the laser were performed as follows: spot diameter 10 μm, pulse duration 50 ms, laser power 50 mW. The areas to microdissect were identified under a light microscope at 40x of magnification including a portion of the implantation site within the host tendon. Every section was dropped on a separate cap. Total RNA from microdissected sections (n = 25–35 sections) was extracted, and 1 μg was used for RT-PCR procedures as described above using specific tenogenic gene primer sequences (Table 2).
Biomechanical Testing
For biomechanical testing, five samples for each tendon group were considered after 28 days. Moreover, tendon biomechanical tests were performed also on five healthy sheep Achilles tendons collected at the local slaughterhouse as a control. The tendons were harvested at the calcaneus and musculo-tendinous junction and firmly ripped by serrated grips 2 cm away from the edge of the repair site. The maximum specific rupture force (N/mm2) and the maximum stiffness (expressed as percentage of preloaded sample) were evaluated using a digital dynamometer (Tinius Olsen H10KT dynamo meter, PA, USA). Before measurements, specimens were preloaded for 1 min with 10 N to provide initial tension (10). The displacement rate was 6 mm/min. Specimens were kept moist throughout the test period with isotonic NaCl solution prewarmed at 38°C.
AEC Detection in Engrafted Tendon
The PKH26 (excitation: 551nm, emission: 567nm) labeled AECs were analyzed with an Axioskop 2plus microscope (Zeiss). Sections with retrieved PKH26-positive cells were immunostained, as described above, for COL1.
Morphometric analyses were also adopted in order to:
quantify the PI of PKH26-positive cells by considering the percentage of proliferating PKH26 cells (Ki-67 and PKH26 double-stained cells) / total PKH26-positive cells, evaluate the percentage of PKH26-positive cells that coexpressed CD45 (CD45 and PKH26 double-stained cells) / total number of PKH26 cells.
The morphometric analyses were performed on at least five fields on all the consecutive sections that displayed engrafted PKH26-positive cells.
Statistical Analysis
The data were checked for normalcy by Shapiro–Wilk W test, and the data sets were compared by Kruskal–Wallis or Wilcoxon–Mann–Whitney test, followed by Dunn's post hoc test, when necessary. The differences were considered significant for p < 0.05, and data were expressed as median (5°–95° percentile).
Results
Characterization of Ovine AECs
The mechanical isolation of the amniotic epithelial layer allowed to isolate a purified population of AECs as documented by cytokeratin-8 distribution at first passage (Fig. 1A). The cells could be rapidly expanded in vitro for three successive passages with an average doubling time of ~25h.

Surface and stem cell markers in cultured AECs. (A) Images of AECs (I passage) visualized under a phase contrast microscope (left) or after immunocytochemistry (right) for cytokeratin-8. The insert shows the immunohistochemical detection for cytokeratin-8 in an amniotic membrane section. Scale bars: 50 μm (for larger images) and 100 μm (for the smaller corner image). (B) Histograms show the average of surface and stemness markers MFI ratio levels analyzed on 3 replicates ± SD of AECs by flow cytometry. (C) Expression of OCT4A, NANOG, SOX2, and TERT in in vitro expanded or cryopreserved AECs. (Left) An example of RT-PCR. (Right) Semiquantitative analysis normalized for GAPDH (values are mean of 3 replicates ± SD). (D) The top three panels indicate AEC osteogenic in vitro differentiation exposed for 21 days to the osteoinductive medium (DM): left, Alizarin Red Staining; middle, ALP activity. In the corner boxes, AECs maintained for 21 days in growth medium (GM) are shown. Right, bone-related genes RT-PCR. The bottom three panels indicate the tenogenic differentiation (DM) induced in AECs cultured for 21 days with BMP-12 factor: left, a phase contrast microscopy image; middle, COL1 immunocytochemistry (green), counterstained with DAPI. The corner box indicates AECs maintained for 21 days in the growth medium (GM). Right, image of RT-PCR analysis for tendon-related genes. Scale bars: 50 μm (for larger images) and 100 μm (for the smaller ones). AECs, amniotic epithelial cells; Fr/Tw, freezed and thawed AECs; I and III, relative in vitro passages; MFI, mean fluorescence intensity ratio calculated dividing the MFI of positive events/MFI of negative events; OCT4, octamer-binding transcription factor 4; SOX2, sex-determining region Y box 2; TERT, telomerase reverse transcriptase; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; ALP, alkaline phosphatase; RUNX2, Runt-related transcription factor 2; OCN, osteocalcin; BMP-12, bone morphogenetic protein 12; COL1, collagen 1; TNMD, tenomodulin; SCXB, scleraxis B.
Both in vitro expansion and cryopreservation did not modify the molecular profile of the cells (Fig 1B, C). In detail, flow cytometry showed that AECs did not display any hemopoietic markers (CD14, CD31, and CD45), expressed several surface adhesion molecules (CD29, CD49f, and CD166), and intracellular stemness markers (TERT, SOX2, OCT4, and NANOG); CD117 resulted always unexpressed. The RT-PCR confirmed the persistent expression of stemness markers TERT, SOX2, OCT4A, and NANOG after expansion and cryopreservation. The AECs exposed to osteogenic and tenogenic media were able to modify their phenotype assuming the specific morphological and tissues-related molecular profile (Fig. 1D).
AECs Accelerated Tissue Regeneration
All animals well tolerated the surgical procedure (Fig. 2A). Tendon explants showed a limited area of inflammation that progressively decreased, becoming particularly limited in the allotransplanted tendon after 28 days (Fig. 2B). The analysis of the Achilles tendon microarchitecture carried out with H&E and HE stainings showed that AECs markedly accelerated the early phase of tendon healing (Fig. 2C).
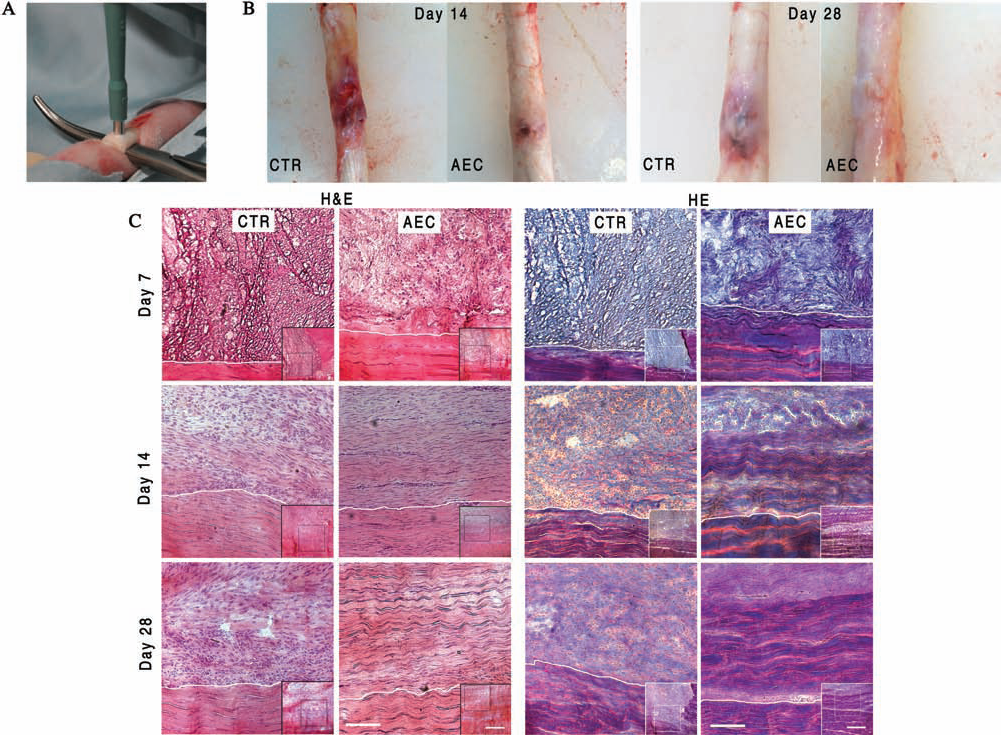
Morphological analysis of tendon healing. (A) An example of the experimental defect induced in the Achilles tendon. (B) Images of tendons explanted 14 and 28 days after treatment. (C) H&E and HE stains of injured Achilles tendon performed at 7, 14, and 28 days posttreatment. For each time point, six different animals were considered. The images in the lower right corners were photographed at 50x magnification. The healing tissue is in the top of the picture, while the healthy portion is in the bottom below the dotted line. The images show a more rapid and complete tissue organization in AEC-allotransplanted tendons. Scale bars: 100 μm (for the larger images) and 500 μm (for the smaller ones). AECs, tendon transplanted with amniotic epithelial cells; Ctr, contralateral tendon treated with only fibrin glue; H&E, hematoxylin and eosin staining; HE, Herovici staining.
The differences between AECs and control (Ctr) tendons became evident at day 14, when AEC tendon started to display an organized architecture. In detail, the lesion zone was filled with several fusiform cells surrounded by an abundant extracellular matrix organized in parallel fibers (score 2 in Table 4, Fig. 2C). The degree of tissue organization further increased later, when the density of the newly deposited extracellular matrix closely resembled that of a healthy tissue with fibers completely oriented along the longitudinal axis of tendon (score 3 in Table 4, Fig. 2C). By contrast, the repair sites of Ctr tendons maintained a lower level of tissue organization throughout the whole experimental period: a high incidence of cells and an abundant matrix with few new deposited fibers irregularly oriented until day 28 (score 1 in Table 4). No traces of fibrin glue persisted after day 14 in both Ctr and transplanted tendons. The score evolution of tissue regeneration of each Ctr and AEC tendon explants obtained by combining the results of H&E and HE staining were summarized in Table 4.
Tendon Microarchitecture Score Detected by Using Haematoxylin–Eosin and Herovici Staining During the Early Phase of Tendon Healing in Each Single Control (Ctr) and Amniotic Epithelial Cell (AEC)-Treated Tissue Explants

Score 1, a damaged tissue with hypercellularity, high density of blood vessels, and disorganized extracellular matrix. Score 2, an early stage of tendon healing with intermediate presence of cells, abundant extracellular matrix with parallel fibers, and the persistence of abundant scattered blood vessels. Score 3, the early stage of tendon regeneration with low concentration of fusiform cells displaying flattened nuclei, extracellular matrix showing parallel fibers, and blood vessels both oriented along the longitudinal axis of the healthy portion of the tendon. +, the relative score recorded in each single tendon explant obtained by six different animals.
AECs Modulated the Mechanisms Involved in Tendon Regeneration
The analysis of protein matrix composition (Fig. 3A), TGF-β1 and VEGF expression (Fig. 3B), and blood vessel remodeling (Fig. 4A) confirmed the AEC influence. COL3, normally localized in the paratenon of the healthy tendon, was found distributed within the repair site of AEC-allotransplanted tendons from day 7 until day 14, when it started to be replaced by COL1. By contrast, COL3 appeared in the repair sites of Ctr tendons only at day 14, where it remained irregularly distributed until day 28. COL1 was almost absent in both tendons at day 7. Then, it appeared in allotransplanted sites as fiber-like wires that, after 28 days, reached a greater density and a typical orientation along the longitudinal axis of the healthy tissue. By contrast, COL1 was barely detectable in Ctr tendons at day 28, where it displayed a scattered random distribution throughout the repair site.

Matrix protein composition and TGF-β1 and VEGF expression in normal (lower portion of the picture below the dotted line) and healing (upper portion) Achilles tendons. The images show the cell nuclei in blue (DAPI) and the different proteins analyzed in green (Alexa Fluor 488). (A) Representative immunofluorescent images of COL3 (left) and COL1 (right) showing the matrix protein remodeling 7, 14, and 28 days after treatment. (B) TGF-β1 (left) and VEGF (right) recorded 7, 14, and 28 days after the induction of tendon defects. Scale bars: 200 μm (for all images). AECs, tendon transplanted with amniotic epithelial cells; Ctr, contralateral tendon treated with fibrin glue only; TGF-β1, transforming growth factor-β1; VEGF, vascular endothelial growth factor.
Healing was accompanied in both allotransplanted and Ctr tendons by an increased expression of TGF-β1. This key growth factor was mainly recorded at the edges of the experimental lesion in Ctr tendons, while it was widespread in the whole repair sites of the AEC-treated tendons, where it reached higher concentrations. Interestingly, the production of this factor was switched off 28 days after the AECs introduction, while it persisted in Ctr tendons (Fig. 3B).
A marked VEGF synthesis was triggered by tendon injury. The angiogenic factor was distributed within the repair sites of Ctr and AEC-treated tendons until day 14. Then, the protein disappeared in allotransplanted tendons, while its production was maintained in the lesions of Ctr tendons (Fig. 3B).
The quantitative analysis of the vWF (total VA) revealed a wider angiogenic response in treated tendons at day 7 (Fig. 4B). Then, the VA progressively decreased, and the blood vessels were observed at day 28 distributed among the new deposited extracellular matrix and aligned along the longitudinal axis of the healthy tendon (Fig. 4A). By contrast, a significantly higher total VA was recorded in Ctr tissues at days 14 and 28 (Fig. 4B). Moreover, a constant blood vessel network was observed irregularly scattered among the repair site (Fig. 4A).

Morphometric analyses of total VA, PI, cellularity, and leukocyte infiltration in healing tendons. (A) Comparison of blood vessel network architectures in healthy and experimentally injured tendons at day 28. Blood vessels were identified by the anti-vWF antibody (green). The cell nuclei were counterstaining with DAPI (blue). Scale bars: 50 μm. (B) The table shows the effects of AECs on the total VA, PI, leukocyte infiltration, and cellularity in tendon healing at different time points posttreatment. The data are expressed as median (5°–95° percentile); n = 15 replicates/treatment/time point/animal. For each time point, six different animals were considered (n = 90; total replicates/time point/animal group). a,b,cSignificantly different values for p < 0.01. vWF, von Willebrand factor; AECs, tendon transplanted with amniotic epithelial cells; Ctr, contralateral tendon treated with fibrin glue only; Total VA, total vascular area calculated as vWF-positive area in μm2/standard field of 20,000 μm2 area; PI, proliferation index calculated as the percentage of Ki-67 green-stained cells/total number of nuclei in the standard field; Leukocyte infiltration, expressed as percentage of the CD45-positive cells/total number of nuclei in the field; Cellularity, expressed as the total number of nuclei (DAPI stained) in the field.
COL1 and COL3 mRNA levels selectively extracted from the defect sites during the different time points (Fig. 5A, B) using the LCM technique revealed that the expression of these tendon-related genes were rapidly upregulated in the tendon lesions containing AECs. COL3 mRNA content, in fact, increased more rapidly in allotransplanted tendons and returned on basal values at day 28. By contrast, the expression of this immature form of collagen started to increase in Ctr tendons only at day 14. Similarly, the increase in COL1 mRNA content was more evident in allotransplanted tendons where it reached levels that were similar to that recorded in the healthy tendons at day 28.
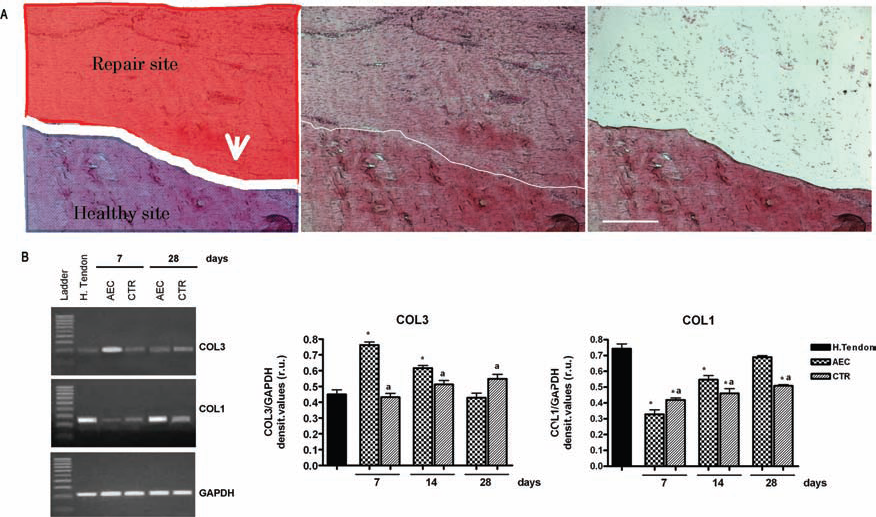
COL1 and COL3 expression in the defect area of healing tendons. (A) The left image shows a schematic representation of the different tissue portions (healthy and repair area) as visualized at 40x on a cryosection used for the LCM-mRNA extraction. The central image shows the same section after H&E staining. The right image depicts the histological section after the defect site was removed by LCM. Scale bars: 200 μm. (B) COL1 and COL3 mRNA expression. Left: An example of RT-PCR. Right: Semiquantitative analysis normalized for GAPDH (values are mean of 3 replicates ±SD). aSignificantly different values between tendon groups (p < 0.05); ∗significantly different values from the control healthy tendon (p < 0.05). LCM, laser capture micro dissection; COL3, collagen type III; COL1, collagen type I; H. tendon, healthy Achilles tendon; AECs, tendon transplanted with amniotic epithelial cells; Ctr, contralateral tendon treated with only fibrin glue.
Morphometric Analysis Revealed a Different Healing Evolution Between AEC-Transplanted and Ctr Tendons
The morphometric analysis of total VA, cellularity, PI, and leukocyte infiltration between healthy and healing tendons of Ctr and allotransplanted tissues was first carried out according to previous authors (6,28), with aggregated values recorded in the core of the repair sites (areas 4–5) (Fig. 4B). The cellularity in AEC-treated tendons was slightly higher than that of Ctr tendons until day 14; then, it progressively decreased while continued to increase in Ctr tissue.
The PI of AEC-treated tendons was significantly higher than that recorded in the healthy tissue at all the time points considered. However, the PI of AEC-treated and Ctr tendons was similar until day 14 to become significantly lower in Ctr tissues at day 28.
Seven days after tendon injury, leukocyte infiltration of AEC-treated tendons was higher than that of healthy tissues, but significantly lower than that of Ctr tendons. Then, leukocyte infiltration rapidly dropped in AEC-allotransplanted tissues to values that were similar to the healthy tissue, while it remained on significantly higher values in Ctr tendon lesions.
The spatial–temporal variations of these parameters were then recorded from the healthy portion of the tendon (area 0) to the center of the lesion zone (area 5) throughout five consecutive areas (areas 1–5) (Fig. 6). The cellularity of allotransplanted tendons showed a gradient distribution with higher values in the core of the defect (areas 4–5) and lower at the periphery (areas 1–3) where they reached levels that were similar to those recorded in the healthy portion of the tendon (area 0) at days 14 and 28. In Ctr tendons, the cellularity increase was less evident, slower, and widespread within the wound area.

Spatial–temporal variations of total VA, PI, leukocyte infiltration, and cellularity in healing tendons. The data are tabulated as median ± 25° and 75° percentile of 15 determinations/field/animal performed at 200x of magnification by capturing five images from six contiguous areas (indicated on x-axis as 0–5) starting from the healthy portion of the tissue (area 0) and proceeding throughout the repairing zone (1–3) to the core of the lesion (4–5). The side of each field analyzed was ~300 μm for an overall length of ~1800 μm. The final data considered for each time point are the median of 90 different determinations (15 determinations/field for 6 different animals/time point). AECs, tendon transplanted with amniotic epithelial cells; Ctr, contralateral tendon treated with only fibrin glue; Total VA, total vascular area calculated as vWF-positive area in μm2/standard field of 20,000 μm2 area; PI, proliferation index calculated as the percentage of Ki-67 green-stained cells/total number of nuclei in the field; Leukocyte infiltration, expressed as percentage of the CD45 positive cells/total number of nuclei in the field; Cellularity, expressed as the total number of nuclei (DAPI stained) in the field.
The PI showed a similar gradient distribution in the AEC-treated tendons: the PI that reached high values in the whole defect area at day 7 then decreased rapidly at the periphery of the lesion. By contrast, a high PI was retained in the core of the lesion until day 28. In Ctr tendons, the PI decreased uniformly in the defect area during the last 2 weeks.
Total VA showed a more intense angiogenic response at day 7 and a more rapid normalization in AEC-treated tendons. By contrast, the total VA recorded in the whole repair area (areas 1–5) of Ctr tendons remained on greater values from days 14 to 28.
AEC injection, in addition, strongly reduced the leukocyte infiltration within the wound area, which from day 14 became similar to that recorded in the healthy field.
AECs Improved the Mechanical Properties of Tendon Healing
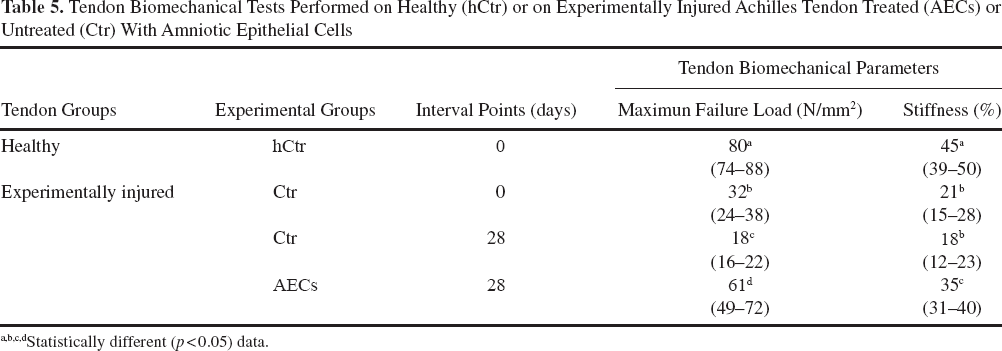
The biomechanical properties of experimentally injured tendons are summarized in Table 5. In detail, the mean maximum failure load [80 (74–88) N/mm2] and stiffness [45% (39–50%)] of healthy tendons were reduced immediately after the induced defect of ~2.5 and 2 times, respectively [32 (24–38) N/mm2 and 21% (15–28%)].
Tendon Biomechanical Tests Performed on Healthy (hCtr) or on Experimentally Injured Achilles Tendon Treated (AECs) or Untreated (Ctr) With Amniotic Epithelial Cells
Statistically different (p < 0.05) data.
The recovery of both parameters were significantly improved by the presence of AECs: 28 days after allotransplantation, the tendon maximum failure load [61 (49–72) N/mm2] and stiffness [35% (31–40%)] increased ~2 and 1.5 times, respectively, reaching values significantly higher than those recorded in Ctr tendons [18 (16–22) N/mm2 and 18% (12–23%): for both p < 0.05].
AECs Survived in Host Tissue and Contributed to Tendon Regeneration
Labeled AECs were always recovered within the engrafted tendon. As expected, the PKH26 fluorescent signal of the retrieved cells was observed around the nuclei, but it was not detected in the extracellular matrix (Fig. 7A, B). The majority of AECs rapidly migrated in proximity of the healthy portion of the tendon, as shown in Figure 7A. They maintained within the host tissue a constant PI (Fig. 7B) ranging between 15% and 25%. The AECs' persistence in the host tissue was guaranteed by the low incidence of phagocytosis that until day 14 involved only ~10% of them. Then, the percentage of phagocytosed PHK26-positive cells increased (Fig. 7B) but never exceeded 35%. Some of the identified AECs 14 and 28 days after transplantation were localized within the new deposited extracellular matrix. These cells displayed a fusiform shape and a high intracellular content of COL1. On the contrary, the COL1 protein was undetectable in all the PKH26-positive cells that did not assume a tenocyte-like morphology as well as in the AECs before allotransplantation (Fig. 7B). On the contrary, PHK26 cells never coexpressed with the endothelial marker vWF but were always positioned close to blood vessels (data not shown).

Detection of allotransplanted AECs within the host tendon. (A) Merged images showing the PKH26-labeled AECs (red fluorescence) within the host tissue. Cell nuclei were counterstained in blue (DAPI) and COL1 in green (Alexa Fluor 488 secondary antibody). The healing tissue is at the top of the picture at days 7 and 14, while on the right at day 28. PKH26 cells migrated at the periphery of the defect at day 7, while resulted close to the front of the new fiber deposition from day 14. Scale bars: 100 μm. (B) Merged images showing the proliferative activity (left image), phagocytosis (central image), and COL1 distribution (right image) in host cells and transplanted PKH26-labeled AECs (red). Left image: Immunofluorescent image obtained 28 days after transplantation showing in red the PKH26-labeled AECs, in blue the nuclei counterstained with DAPI, and in green the Ki67 proliferative marker. Central image: Immunofluorescent image acquired 28 days after transplantation showing in red the PKH26-labeled AECs, in blue the nuclei counterstained with DAPI, and in green the leukocytes identified by the surface marker CD45. The arrows indicate phagocytosed AECs that display an orange fluorescence for the merge of PKH26 and CD45 fluorescence. Right image: A representative fluorescent image achieved 14 days after transplantation showing in red the PKH26-labeled AECs, in green the COL1, and in blue the counterstaining nuclei. The AECs localized at the periphery of the COL1 fibers are poliedric and with a clear red fluorescence around the circle nuclei. By contrast, the AECs entrapped within the COL1 fibers displayed a fusiform shape, a flattened nuclei, and an orange color as a consequence of the merge of the green and red fluorescence. The inset in the corner shows the absence of COL1 in AECs before transplantation (in vitro culture, III passage), as indicated by immunocytochemistry. Scale bars: 50 μm (for all three images and for the inset).
Discussion
This investigation shows for the first time that AEC allotransplantation into a tendon lesion can significantly improve the healing process. AECs can theoretically differentiate into the cell types derived from all the three germ layers (41,60), and experimental evidence has demonstrated their great regenerative potential for several tissues (46). This research extends the potential therapeutic application of these cells to the tendon tissue that, despite the frequent mechanical injuries it is exposed to, it has a very limited repairing ability.
Cell therapy aimed at counteracting the scarce repairing ability of the tendon could theoretically indicate ESCs as the ideal stem cell source even if the high risk of teratoma formation strongly limited their use (41). To overcome this problem and promote a safe and practical strategy, a stepwise differentiation approach has been proposed in order to transform ESCs into tendon-like tissue before transplantation (69). This strategy was first proposed by Barberi et al. (9), who avoided teratoma by inducing ESCs to differentiate into neural progenitor cells before injection into nervous system. As indicated by several reports, the amniotic-derived cells, on the contrary, may represent an alternative and prompt stem cell source with a low teratoma risk (8,37,41,60). Consistent with these previous evidences, ovine allotransplanted AECs are able to safely engraft and survive into the injured ectopic sites for 4 weeks. In addition, the allotransplantation of AECs in the experimentally induced tendon lesion accelerates tissue regeneration and, most importantly, the recovery of mechanical properties during the early phase of healing. Such a remarkable effect is likely to be the results of a combination of factors generated by the presence of AECs in the lesion area. The engraftment of AECs, for instance, seems to stimulate the production of helpful growth factors, like VEGF and TGF-β1, that were widespread in the tendon defects where they reached high concentrations. On the contrary, TGF-β1 in Ctr injured tissues increased slowly, remaining localized on the edge of the lesion site without reaching the receptors evenly distributed in the wound (13). In order to overcome this limited endogenous production of this cytokine during the process of spontaneous tendon healing, TGF-β1-transfected bone marrow-derived stem cells have been produced to enhance the presence of the growth factor within the tendon defect, thus improving the early phase of healing (29,67). The insertion of AECs, without any transgenic transformation, produced the same effect. The production of TGF-β1 has been shown to be positively related to procollagen type I mRNA content and to collagen protein synthesis (1,32,39,67). According to these evidences, the expression of two tendon-related genes (COL1 and COL3) were rapidly upregulated in allotransplanted tendons, thus suggesting that these cells, directly or through a mediated action by local progenitor cells, stimulate the deposition of collagen fibers. These observations seems to contrast with the findings of a recent research that involved the use of fetal-derived embryonic-like stem cells in a large animal flexor tendonitis model (68), where the authors were unable to find differences in biochemical parameters (DNA and collagen content) between Ctr and treated tendons.
Apart from the different source of cells used, the upregulation of both collagen gene expressions were here recorded with the LCM technique that allowed us to determine the COL3 and COL1 mRNA contents exclusively within the injured tissue. In the absence of such a condition, as suggested by Watts et al. (68), strictly localized changes in gene expression can be missed since analyzed on homogenized tissue samples containing healthy and repairing portions of the tendon.
The prompt healing in AEC-treated tendons was documented by histological stainings and immunohistochemistry, showing the first signs of regeneration 14 days after transplantation, when a tendon-like microarchitecture became clearly detectable in the lesion zone. Histological examination showed a homogenous tissue within the repair sites where several tenocyte-like flattened cells resulted entrapped among the newly deposited fibers aligned along the longitudinal axis of the tendon. Immature fibers of COL3 accumulated during the first 2 weeks were replaced by COL1, the mature form of collagen that accounts for more than 90% of the extracellular matrix proteins in healthy tendons (22). After 4 weeks, the repair sites were characterized by an organized regular connective tissue consisting of aligned COL1 fibers with interspersed tenocytes showing a slightly lower density than that of the healthy portion of the tendon.
The mechanical recovery of healed tendons and other tissue healing systems is the result of a complex set of parameters (29,67,68) such as the amount of collagen (42,52), the diameter and alignment of fibrils (17,47), the degree of collagen cross-linking (19,20), and the ratio of proteoglycan/collagen (11). In the present research, the overall output of these factors was evaluated by measuring the biomechanical properties of tendons at the end of the experimental interval. AEC-treated tendons, consistent with the histological recordings, displayed a better mechanical recovery than Ctr tissues that showed lower maximum failure loads and stiffness.
The spatial–temporal analysis performed to compare cellularity, PI, leukocyte infiltration, and total VA from the healthy to the repairing portion of the tissue enabled us to reveal a different evolution of the healing process. AEC allotransplantation, in fact, produced a centripetal improvement of the cell parameters that first involved the areas closer to the healthy portion of the tissue where AECs rapidly migrated. The process of tendon regeneration rapidly proceeded along the healthy–repair tendon axis and brought leukocyte infiltration and total VA in the periphery of the lesion to normal values in 4 weeks. This peculiar healing evolution could be the consequence of the modulatory influence exerted by AECs addressed to modulate the production of growth factors, the recruitment of host progenitor cells, the remodeling of the blood vessel network, and the inflammatory host response, all processes recognized as crucial for tendon healing and regeneration (18,22,48,54–56).
In detail, angiogenesis is promptly activated in AEC-treated tendons as indicated by the high VEGF levels and by the widespread VA extension recorded in the wound area at 7 days. An active angiogenesis is, in fact, required for a rapid formation of the intravascular plexus during the granulation tissue development. However, a whole recovery of tendon biomechanical properties occurs only after the regression of the vascular plexus and, as a consequence, of a specific remodeling of the blood vessel network (54–56). An experimental research specifically performed on sheep Achilles tendon clearly demonstrated the existence of a positive correlation between the high vascularity and the incidence of degenerative tendon diseases (49). Analogously, adverse consequences on the recovery of biomechanical properties have been observed when the early phase of tendon healing occurred with a VEGF overexpression (28). In this context, the rapid regression and remodeling of blood vessels induced by AECs may be interpreted as a positive clinical sign that indicates as the accelerated process of healing was obtained following a normal regenerative program. Even if the mechanisms involved in AEC-induced vascular remodeling remain still unknown, they could be related either to the modulatory influence on VEGF synthesis or to the widespread anti-inflammatory effect recorded within the wound district. In parallel, the presence of AECs stimulated a rapid cell homing in the core of the defect area. The higher core cellularity did not appear related to the injection of AEC, since after 14 days, the cells migrated at the periphery of the wound area. Moreover, the PI stimulated by AEC never resulted strictly superimposable with the gradient distribution of cellularity. The higher cellularity recorded in the core of the lesion may be more easily interpreted as the consequence of the paracrine effects of AEC that could stimulate the recruitment of cells from paratenon, thus rapidly activating the process of collagen deposition and organization (28).
Apart from the paracrine effects of AECs, the results also indicate a direct involvement of the transplanted cells in the process of tendon regeneration.
AECs, mainly found at the edge of the repairing area at all-time points considered, progressively differentiated into cells able to support the formation of extracellular matrix characterized by typical tendon biomolecules starting from day 14. Several engrafted AECs assumed, in fact, a tenocyte-like shape remaining entrapped within the new deposited extracellular matrix and activated for the first time an intense COL1 production that will likely be used for tendon regeneration.
The finding that AECs formed tenocyte-like cells after transplantation confirmed the mounting evidence of in vivo transdifferentiation ability of human AECs (36). Unfortunately, we were unable to use the coexpression of PKH26 marker to investigate the contribution of the injected AECs in the synthesis of matrix/growth factors molecules since the ovine AECs showed immediately after isolation and maintained after transplantation a basal secretion for COL3, TGF-β1, or VEGF (data not shown). By contrast, PKH26-positive AECs recovered in the host tissues never coexpressed vWF, thus excluding a direct contribution of the amniotic cells towards angiogenesis. The acceleration of early phase of tendon healing in the sheep represents a preliminary positive step that will encourage us to carry out long-term investigations aimed to confirm that this cell therapy can lead to complete tendon repairing. The higher cellularity and PI recorded in AEC-treated tendons suggests that tissue remodeling could continue after 4 weeks, even if a well-organized blood vessel network and a mature tissue/matrix architecture have already been assembled.
The way AECs were well tolerated by the immune system of the host represents another important issue to be considered for the use of these cells in allotransplantation settings. Apparently, the transplantation of AECs elicited a low inflammatory reaction since the incidence of CD45-positive cell infiltration was significantly lower in cell-treated lesions than in Ctr. The immunomodulatory influence of AECs is in agreement with the results reported in other allo and xenograft models (8,46), and it is probably at the basis of the tissue environment that enables protracted cell survival and successful early tendon regeneration. In fact, the beneficial effects induced by human amniotic membranes on the myocardial infarction and liver fibrosis experimental models analogously have been related to the release of soluble factors supporting the survival, proliferation, and differentiation of host cells rather than to cell replacement of injured tissue. The mechanisms hypothesized on both these preclinical models reproduced (36), as observed in our experimentally injured tendon model, involved the release of potent immunomodulatory and anti-inflammatory cytokines, growth factors, associated with wound healing including angiogenetic factors (i.e., VEGF), inducing proliferation and differentiation molecules (i.e., TGF-β1). In particular, the anti-inflammatory response induced by AECs may have a direct role in the positive evolution of tendon healing by considering the higher tendon recovery obtained by treating the acute tendinopathy with anti-inflammatory drugs (51).
Altogether, these evidences confirmed that either ovine or human AECs are able to provide benefits in different experimentally induced tissue defects, thus suggesting their potential use in regenerative medicine. The efficiency of AECs in inducing tissue repairing could not be actually improved by a preliminary selection of the pluripotent stem cell subpopulation that is largely proposed in other stem cell sources (16). In fact, the epithelial layer of ovine and human amniotic membranes (36) did not express the CD117 marker that, on the contrary, is weakly expressed by amniotic mesenchymal cells (36) and that represents the selective surface marker successfully used for human amniotic fluid mesenchymal stem cells (4,16). In conclusion, the present study demonstrates that AECs can accelerate and positively modulate the process of tendon healing by stimulating a prompt recovery of microarchitecture and biomechanical properties of experimentally injured tendons. AECs, in fact, improved tendon repair and influenced the production of biomolecules crucial for tendon regeneration (VEGF, TGF-β1), modulating local inflammatory reaction, and blood vessel network remodeling. AECs stimulated the process of healing conjugating a strong paracrine role with a direct contribution in the synthesis of new tendon matrix. Altogether, these results encourage the use of AECs as a new cell source that did not require any preliminary in vitro differentiation or genomic transfection.
Footnotes
Acknowledgments
The support of Tercas Foundation grant is gratefully acknowledged. We thank Prof. Francesco Ciardelli and Dr. Stefania Savi of the Department of “Chimica e Chimica Industriale,” University of Pisa, for their technical support in the biomechanical tests and Dr. Delia Nardinocchi, Dr. Maura Turriani, and Dr. Domenica Cocciolone for their valuable technical assistance in cell culture and tissue morphological analyses. The authors declare no conflicts of interest.
