Abstract
We investigated the effects of environmental enrichment (EE) on the function of transplanted adipose stem cells (ASCs) and the combined effect of EE and ASC transplantation on neurobehavioral function in an animal model of chronic hypoxic-ischemic (HI) brain injury. HI brain damage was induced in 7-day-old mice by unilateral carotid artery ligation and exposure to hypoxia (8% O2 for 90 min). At 6 weeks of age, the mice were randomly injected with either ASCs or PBS into the striatum and were randomly assigned to either EE or standard cages (SC), comprising ASC-EE (n = 18), ASC-SC (n = 19), PBS-EE (n = 12), PBS-SC (n = 17), and untreated controls (n = 23). Rotarod, forelimb-use asymmetry, and grip strength tests were performed to evaluate neurobehavioral function. The fate of transplanted cells and the levels of endogenous neurogenesis, astrocyte activation, and paracrine factors were also measured. As a result, EE and ASC transplantation synergistically improved rotarod latency, forelimb-use asymmetry, and grip strength compared to those of the other groups. The number of engrafted ASCs and βIII-tubulin+ neurons derived from the transplanted ASCs was significantly higher in mice in EE than those in SC. EE and ASC transplantation also synergistically increased BrdU+βIII-tubulin+ neurons, GFAP+ astrocytic density, and fibroblast growth factor 2 (FGF2) level but not the level of CS-56+ glial scarring in the striatum. In conclusion, EE and ASC transplantation synergistically improved neurobehavioral functions. The underlying mechanisms of this synergism included enhanced repair processes such as higher engraftment of the transplanted ASCs, increased endogenous neurogenesis and astrocytic activation coupled with upregulation of FGF2.
Keywords
Introduction
Multipotent adipose stem cells (ASCs) are abundantly harvested from lipoaspirate and may be useful for angiogenesis and tissue engineering (39). Recent evidences suggest that these cells can ameliorate ischemic tissue injury and produce an appropriate environment for promoting angiogenesis (7,29,34). ASCs also secrete several potentially beneficial growth factors such as hepatocyte growth factor (HGF), insulin-like growth factor 1 (IGF1), vascular endothelial growth factor A (VEGFA), and fibroblast growth factor 2 (FGF2) (6,31,35,37). These factors promote endogenous repair processes and functional recovery. However, stem cell replacement therapy alone is not likely sufficient to optimize outcome (19). Mesenchymal stem cells (MSCs) are emerging as a good source for cell-based therapy (12), but transplantation of MSCs derived from bone marrow, umbilical cord blood, or adipose tissue alone is not sufficient for functional recovery in neurological diseases. The beneficial effects of MSCs seem to be caused by indirect paracrine mechanisms rather than cell replacement and direct tissue regeneration (8,10,11,17,18,28). Therefore, transplantation of adipose-derived MSCs may result in limited and transient effects instead of long-lasting permanent improvement.
In experimental models, environmental enrichment (EE) including a running wheel, novel objects, and social interactions strongly affect neural plasticity for restoration after injury (23). EE also increases the expressions of growth factors that play an important role in angiogenic and neurotrophic activities (33). EE can enhance proliferation of resident neural stem/progenitor cells in the subventricular zone (SVZ) and their migration into the ischemic lesion, thereby contributing to behavioral recovery (23,24,33). However, the effects of EE on transplanted stem cells vary depending on experimental conditions (19,20). In a rat model of stroke, EE was found to augment migration of transplanted mouse neural stem cells and to enhance functional recovery (19). In contrast, the survival, migration, and differentiation of transplanted human neural stem cells were not affected by EE (20).
The effects of EE on the function of transplanted ASCs in an animal model of cerebral palsy (CP) have yet to be studied. We postulated that the combination of MSC transplantation and EE would synergistically improve the motor functions of subjects with chronic hypoxic-ischemic (HI) brain injury. Therefore, we investigated (1) whether EE after transplantation of ASCs could synergistically enhance functional recovery, (2) the therapeutic mechanism of combined ASCs and EE in an animal model of chronic HI brain injury, and (3) whether EE affects the engraftment and fate of transplanted ASCs. We found that EE after transplantation of ASCs elicited synergistic neurorestorative effects through enhanced engraftment and neuronal differentiation of transplanted cells, and the promotion of endogenous repair processes such as neurogenesis, astrocytic activation, and upregulation of FGF2 in the brain tissue.
Materials and Methods
Isolation, Culture, and Characterization of ASCs
The use of human cells in this study was approved by the Institutional Review Board (IRB No. 4-2008-0277), and human lipoaspirate was obtained from a volunteer (a 68-year-old woman) who provided informed consent. Fat tissue was treated with 0.075% collagenase A (Roche, Indianapolis, IN, USA) for 40–50 min in an incubator at 37°C CO2. Separated adipose-derived cells were seeded at 1–5 × 104 cells/cm2 in T25 or T75 culture flasks (BD Falcon, Franklin Lakes, NJ, USA) in low- glucose Dulbecco's modified Eagle's medium (LG-DMEM; Invitrogen-Gibco, Rockville, MD, USA) containing 10% fetal bovine serum (FBS; Invitrogen-Gibco) and 100 U/ml penicillin/streptomycin (Invitrogen-Gibco) in a humidified 5% CO2 atmosphere at 37°C. Adherent MSCs were subcultured when they reached 70–80% confluence and used for transplantation after three to four passages. The cells were harvested with 0.25% trypsin/EDTA (Invitrogen-Gibco), centrifuged at 1,300 rpm with a rotor size of 196.5 mm (371.27×g) for 3 min and resuspended in phosphate- buffered saline (PBS; Invitrogen-Gibco) before transplantation as well as flow cytometry. To investigate multipotent characteristics of MSCs, the osteogenic, adipogenic, and chondrogenic differentiation were confirmed by staining with von Kossa, Oil red O, and Safranin O (all from Sigma-Aldrich, St. Louis, MO, USA) after 2–3 weeks incubation in the respective differentiation culture media of human MSC Osteogenic SingleQuots (Cat. No. PT-4120), Adipogenic Induction SingleQuots (Cat. No. PT-4135), and Chondrogenic SingleQuots (Cat. No. PT-4121) (all from Lonza, Walkersville, MD, USA). For flow cytometry, a total of 5 × 105 cells were resuspended in 0.2 ml PBS and were stained with anti-mouse monoclonal antibodies for cluster of differentiation 14 (CD14), CD31, CD34, CD45, CD29, CD44, CD90, and CD73 (all from BD Biosciences, San Jose, CA, USA) on ice for 30 min. After staining with primary antibody, cells incubated with secondary antibodies conjugated with fluorescein isothiocyanate (FITC; 1:200, Beckman Coulter, Brea, CA, USA) or phycoerythrin (PE; 1:200, Beckman Coulter) for 20 min at room temperature. The fluorescence intensity of the cells was evaluated by flow cytometry (FACScan; Becton Dickinson, Franklin Lakes, NJ, USA). To compare the characteristics of MSCs, the same method described above was used for human bone marrow-derived MSCs (Lot No. B100420-06; from Dr. Kim, Yonsei Cell Therapy Center, Severance Hospital, Seoul, Korea). To prelabel the cells before transplantation, a portion of the cells were either incubated with 3 μg/ml 5-bromo-2-deoxyuridine (BrdU; Sigma-Aldrich) for 3 days, 1 μM chloromethyl-1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine perchlorate (CM-DiI; Invitrogen, Carlsbad, CA, USA) for 5 min at 37°C and then for 15 min at 4°C or were infected with retrovirus expressing red fluorescent protein (dsRFP) for 3 days. The retrovirus expressing dsRFP was generated by the transfection of pMX-dsRFP plasmid (from Dr. Srivastava, Gladstone Institute of Cardiovascular Disease, University of California, San Francisco, CA, USA) into cells stably transfected with the group antigens (GAG) and polymerase (POL) gene as well as the G protein of vesicular stomatitis virus (VSV-G) (293 GPG) cells (from Dr. Park, Hanyang University, Seoul, Korea) using Fugene HD (Roche).
Neonatal Hypoxic-Ischemic Brain Injury
In 7-day-old CD-1® (ICR) mice (Orient, Seongnam, Korea), permanent ischemic brain damage was induced by unilateral right carotid artery ligation. Hypoxic brain injury (8% O2 for 90 min) was also induced as previously described (Fig. 1A) (22,41,44). Body temperature was maintained at 37°C within the hypoxic chamber. One week after the HI brain injury, a scalp incision was made in order to identify the brain lesion in the posterolateral area of the right hemisphere. The presence and extent of brain injury in all subjects were assessed with a light through the semitransparent skull. As determined by discoloration, animals with severe brain lesions covering more than 50% of the unilateral hemisphere were excluded to eliminate the sampling errors inherent in the condition due to volumetric changes in the neostriata (22).

Animal model and experimental design. (A) The hypoxic chamber. After ischemic brain damage was induced by unilateral right carotid artery ligation in 7-day-old CD-1® (ICR) mice, hypoxia was induced at 8% O2 for 90 min using nitrogen gas (N2) and monitored using oxymetry. (B) At postnatal week 6 (P42), adipose stem cells (ASCs) were intrastriatally injected using stereotaxic coordinates. (C) Standard cage (SC; 27 × 22.5 × 14 cm). (D) Enriched environment (EE; 86 × 76 × 31 cm) including tunnels, shelters, toys, running wheels for voluntary exercise, and social interaction. (E) Schematic timeline of the experimental procedures. PBS, phosphate-buffered saline (vehicle).
Cell Transplantation
At postnatal week 6 (P42), the mice were anesthetized with ketamine (100 mg/kg; Huons, Gyeonggi-do, Korea) and xylazine (10 mg/kg; Bayer Korea, Seoul, Korea) by intraperitoneal (IP) injection and were randomly assigned to one of two groups for treatment with either ASCs or PBS. The mice received an intrastriatal injection of ASCs (1 × 105 cells, 2 μl volume, 0.01 μl/s infusion rate) or PBS using stereotaxic coordinates (AP +0.5 mm from bregma; ML −1.5 mm from bregma; DV −3.5 mm from dura) (Fig. 1B). Body temperatures were maintained at 37°C in a heating chamber during the recovery period.
Animals and Experimental Housing
After stereotaxic surgery, a total of 89 mice (44 male and 45 female) were randomly transferred to EE or standard conditions (SC) and assigned to one of five groups: ASC-EE (n = 18), ASC-SC (n = 19), PBS-EE (n = 12), PBS-SC (n = 17), and untreated controls (n = 23). Whereas SC controls were housed for the same duration in a standard cage (27 × 22.5 × 14 cm) without social interaction (3–4 mice/cage) (Fig. 1C), EE mice were housed in a huge cage (86 × 76 × 31 cm) containing novel objects such as tunnels, shelters, and toys, and running wheels for voluntary exercise, allowing for social interaction (12–15 mice/cage) for up to 2 months (Fig. 1D). All animals were housed in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) and were given food and water ad libitum with alternate 12-h light/dark cycles, according to animal protection regulations. The experimental procedure was approved by the Institutional Animal Care and Use Committee (IACUS No. 2010-0252). A schematic timeline of this experiment from birth to 14 weeks of age is provided in Figure 1E.
Behavioral Assessment
Rotarod Performance
A rotarod test was performed to assess motor coordination and balance using a rotarod treadmill (No. 47600; UGO Basile, Comerio, VA, Italy). All animals received a preoperative performance evaluation at 5–6 weeks of age. Rotarod tests were then performed at 2-week intervals until 8 weeks posttransplantation using constant speed (48 rpm) and accelerating speed (4–80 rpm) paradigms. The latency of mice falling from the rod was measured twice during each test, and individual tests were terminated at a maximum latency of 300 s.
Forelimb-Use Asymmetry Test
To evaluate functional asymmetry resulting from a unilateral brain lesion and consequent hemiplegia, the cylinder test and ladder walking test were performed 8 weeks after treatment. In the cylinder test, the number of times each forelimb contacted the cylinder wall (Jeung Do B&P, Seoul, Korea) while the mouse was rearing straight was evaluated over a period of 5 min. The percentage of contacts with hemiplegic forelimb use was evaluated by the following formula (22): [number of contacts with contralateral limb + 1/2 (number contacts with both limbs)] / (number contacts with ipsilateral limb + number contacts with contralateral limb + number contacts with both limbs) × 100 (%).
The difference (Δ) in the percentage of cylinder wall contacts by the contralateral limb relative to the preoperative evaluation was calculated.
In the ladder walking test, the mice were required to walk a distance of 1 m three times on a horizontal ladder with metal rungs (Jeung Do B&P) located differing distances apart. The number of slips from the transverse rungs with each forelimb was measured by videotape analysis. The difference (Δ) in the percentage of slips on the transverse rungs of the ladder relative to the total number of steps taken by the hemiplegic forelimbs compared to the preoperative evaluation was calculated.
Grip Strength Test
A grip strength test was performed using the SDI Grip Strength System (San Diego Instruments Inc., San Diego, CA, USA), which includes a push–pull strain gauge. A 2-mm-diameter triangular piece of metal wire was used as the grip bar. Each animal was held near the base of its tail and approached the bar until it was able to be gripped with the forepaw. Peak force was automatically registered in gram-force by the apparatus. The mean peak force of three trials was used for analysis.
Immunohistochemistry
Immunohistochemistry was performed as previously described (10). After transplantation of ASCs prelabeled with BrdU (3 μg/ml) for 3 days, engrafted cells were identified by immunostaining against BrdU at posttreatment 2 weeks (n = 3 each) and 8 weeks (n = 3 each). An additional group of subjects received an IP injection of BrdU (50 mg/kg) once a day for 12 days, beginning 1 day after stereotaxic surgery, to evaluate endogenous cell genesis and neurogenesis in the striata. Two weeks after treatment, new neurons were evaluated in the neostriata of three subjects from each group (n = 3 each). Eight weeks after treatment, another matched group of three mice was also included to evaluate the long-term survival of newly generated striatal neurons (n = 3 each). Sections were stained with the cell proliferation marker rat anti-BrdU (1:400, Abcam, Cambridge, UK) and the neuronal marker βIII-tubulin using mAb clone TuJ1 (mouse IgG, 1:400, Covance, Princeton, NJ, USA). The sections were incubated with Alexa Fluor® 488 goat anti-rat (1:400, Invitrogen) and Alexa Fluor® 594 goat anti-mouse (1:400, Invitrogen) secondary antibodies, then covered with Vectashield® mounting medium with 4′,6-diamidino-2-phenylindole (DAPI; Vector, Burlingame, CA, USA). Double-labeled BrdU+/βIII-tubulin+ cells were then assessed by confocal imaging (LSM700, Zeiss, Gottingen, Germany). The area of the neostriatum was obtained using the MetaMorph® Imaging System (Molecular Device, Sunnyvale, CA, USA) and converted to volume. The number of newly generated neurons was quantified as density (/mm3). Total numbers of newly generated cells and new neurons were then estimated by multiplying the densities of BrdU+ cells and BrdU+/βIII-tubulin+ cells by the total volume of the striatum. Total volume was calculated by multiplying the summed areas of every cross-section of the striatum by the section thickness (16 μm) in each sample. Individual sections were also costained for neuronal nuclei (NeuN; 1:400, Chemicon, Pemecula, CA, USA) or glial fibrillary acidic protein (GFAP; 1:400, Chemicon) to confirm the identity of the specific neural lineage cells. In addition, immunohistochemistry was performed with GFAP or CS-56 (anti-chondroitin sulfate antibody, 1:200, Abcam, Cambridge, MA, USA) (n = 3 each). GFAP+ and CS-56+ cell densities (%) in the striatum (/mm2) were quantified using the MetaMorph® Imaging System (Universal Imaging Corporation, Downingtown, PA, USA).
Assessment of Growth Factors in the Brain
To identify growth factors that are regulated by ASC transplantation and/or EE (n = 3 each), neostriata separated from brain were lysed in 200 μl of cold radio-immunoprecipitation assay (RIPA) buffer [50 mM Tris–HCl, pH 7.5, 1% Triton X-100, 150 mM NaCl, 0.1% sodium dodecyl sulfate (SDS), 1% sodium deoxycholate] with a protease inhibitor cocktail (both from Sigma-Aldrich). Tissue lysates were then centrifuged at 13,000×g for 15 min at 4°C. The supernatant was harvested, and the protein concentration was determined using a protein assay kit (Bio-Rad, Hercules, CA, USA). An array-based multiplex ELISA assay (Quantibody® array, RayBiotech, Norcross, GA, USA) was used to determine which of the following 10 cytokines or growth factors were detectable in the neostriata: FGF2, epidermal growth factor (EGF), granulocyte colony-stimulating factor (CSF3 or G-CSF), HGF, IGF1, leptin (LEP), matrix metalloproteinase-2 (MMP2), stromal cell-derived factor-1 [SCF1; chemokine (C-X-C motif) ligand 12; CXCL12], vascular cell adhesion protein-1 (VCAM1), and VEGFA. Expression of angiogenic factors was detected using an array scanner (Gene PIX” 4000B, Axon Instruments, Foster City, CA, USA).
Western Blot Analysis
To confirm the expression of FGF2 level by ASC transplantation and/or EE (n = 3 each), 40 μg of extracted proteins was dissolved in sample buffer (60 mM Tris–HCl, pH 6.8, 14.4 mM β-mercaptoethanol, 25% glycerol, 2% SDS, and 0.1% bromophenol blue; Invitrogen), incubated for 10 min at 70°C, and separated on a 10% SDS reducing polyacrylamide gel (Invitrogen). Separated proteins were then equally loaded and transferred onto polyvinylidene difluoride membranes (Invitrogen) using a trans-blot system (Novex® Mini-Cell; Invitrogen). Blots were blocked for 1 h in Tris-buffered saline (TBS) (10 mM Tris–HCl, pH 7.5, 150 mM NaCl) containing 5% nonfat dry milk (BioRad) at room temperature, washed three times with TBS, and incubated at 4°C overnight with a FGF2 (1:1,000, Abcam) antibody in TBST (10 mM Tris pH 7.5, 150 mM NaCl, and 0.02% Tween 20) containing 3% nonfat dry milk. The next day, blots were washed three times with TBST and incubated for 1 h with horseradish peroxidase-conjugated secondary antibodies (1:3,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) at room temperature. After washing three times with TBST, the protein was visualized with an ECL detection system (Amersham Pharmacia Biotech, Little Chalfont, UK).
Statistical Analysis
The effects of EE on the endogenous repair process and functional recovery after transplantation of ASCs were evaluated for each group. The numbers of transplanted ASCs, BrdU+ cells and BrdU+/βIII tubulin+ cells, GFAP+ or CS-56+ cell density (/mm3), as well as expressions of paracrine factors in the neostriata and the results of behavioral tests were analyzed using one-way ANOVA followed by a post hoc Tukey's comparison or independent t test using SPSS (version 18.0; Armonk, NY, USA). A statistically significant level was defined when p < 0.05.
Results
Cultured ASCs Show Characteristics of MSCs
ASCs isolated from adipose tissue showed typical spindle-shaped, fibroblast-like morphology (Fig. 2A) (10,11). ASCs were also induced to undergo differentiation into mesenchymal lineage such as osteogenic, adipogenic, and chondrogenic differentiation (Fig. 2B–D). In addition, flow cytometry showed that these cells were strongly positive for MSC-specific markers such as CD90, CD73, CD44, and CD29 (Fig. 2E), but were negative for CD14 (monocyte-macrophage marker), CD31 (endothelial cell marker), CD34 (hematopoietic stem cell marker), and CD45 (panhematopoietic cell marker) (Fig. 2E), as observed in bone marrow-derived MSCs (Fig. 2F). We therefore concluded that cultured ASCs had the characteristics of MSCs.

The characteristics of adipose stem cells. (A) The cultured adipose stem cells show typical spindle-shaped, fibroblast-like morphology. Scale bar: 200 μm. (B–D) The adipose stem cells were induced for osteogenic (B), adipogenic (C), and chondrogenic (D) differentiation, and the differentiation was confirmed by von Kossa (B), Oil red O (C), and Safranin O (D) staining. Scale bar: 100 m. (E, F) Flow cytometry of cultured adipose stem cells (E) shows the characteristics of mesenchymal stem cells, similar to those of bone marrow-derived mesenchymal stem cells (F). Positive cells for each antigen are marked with horizontal bars. FITC, fluorescein isothiocyanate; PE, phycoerythrin; CD, cluster of differentiation.
Rotarod Performance Shows That EE Synergistically Improved Neurobehavioral Function After Transplantation of ASCs
We first determined whether EE and ASCs could restore neurobehavioral function using rotarod tests at both constant (48 rpm) and accelerating (4–80 rpm) speeds every 2 weeks after HI brain injury. When rotarod performance was assessed prior to the induction of HI brain injury, no differences were observed among the groups (data not shown). A significant improvement in rotarod performance in the ASC-EE mice emerged compared with that in the PBS-SC and untreated controls at 4 weeks after treatment at the constant speed (F = 4.534, p < 0.05) (Fig. 3A) and at 2 weeks after treatment at an accelerating speed (F = 3.725, p < 0.05) (Fig. 3B). The significant neurobehavioral improvement in ASC-EE mice was maintained throughout the study period until 8 weeks after treatment. Importantly, 8 weeks after treatment, ASC-EE mice (156.8 ± 18.1 s) exhibited significantly longer latency than the other four groups: ASC-SC (69.4 ± 18.0 s, post hoc comparison of p < 0.05), PBS-EE (80.5 ± 20.2 s, p < 0.05), PBS-SC (54.1 ± 15.7 s, p < 0.001), and untreated controls (51.2 ± 13.3 s, p < 0.001) at a constant speed (F = 6.695, p < 0.001) (Fig. 3A).
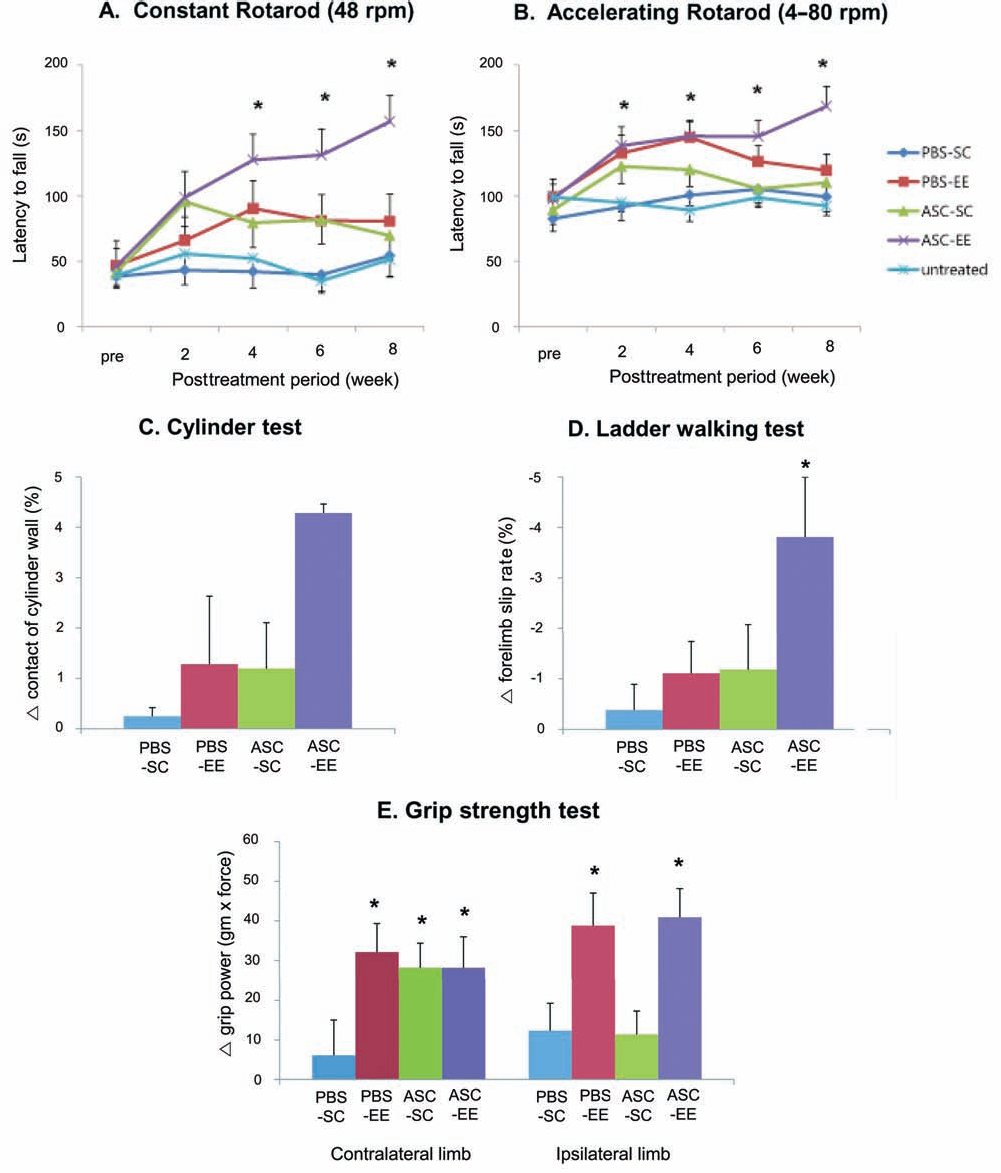
Enriched environment synergistically improved neurobehavioral function. (A, B) Rotarod tests were performed at 2-week intervals. Rotarod tests at constant (A) and accelerating (B) speeds showed that neurobehavioral activity of mice treated with EE after ASC transplantation (ASC-EE, n = 18) was significantly improved compared to those of ASC-SC (n = 19), PBS-EE (n = 12), PBS-SC (n = 17), and untreated controls (n = 23) (*p < 0.05). (C–E) Other behavioral tests such as cylinder (C), ladder walking (D), and grip strength (E) tests were performed before and at 8 weeks after ASC transplantation. Only ASC-EE mice consistently performed better in cylinder (C) and ladder walking (D) tests than did the PBS-SC controls. (E) EE increased grip strength in both limbs, while ASC transplantation increased the grip strength only in the contralateral hemiplegic limb. PBS, phosphate-buffered saline; ASC, adipose stem cell; SC, standard cages; EE, environmental enrichment.
Similarly, rotarod function of ASC-EE mice was also the highest (168.5 ± 15.1 s) among the groups at an accelerating speed at 8 weeks after treatment: ASC-SC (110.2 ± 10.2 s, p < 0.05), PBS-EE (119.5 ± 12.2 s, p < 0.05), PBS-SC mice (99.3 ± 11.1 s, p < 0.001), and untreated controls (93.3 ± 7.1 s, p < 0.001) (F = 7.496, p < 0.001) (Fig. 3B). In contrast to the robust and persistent neurobehavioral improvement in ASC-EE mice, ASC-SC and PBS-EE mice exhibited a modest tendency of transient rotarod improvement during the first 2–4 weeks after treatment even if PBS-EE mice showed a significantly longer latency at accelerating rotarod (144.7 ± 11.5 s) over untreated controls (89.2 ± 8.0 s) at 4 weeks of treatment (p < 0.05) (Fig. 3B). However, this tendency was not sustained through 6 weeks posttreatment. Neither ASC-SC nor PBS-EE enhanced rotarod latency at both constant and accelerating speeds compared with those in PBS-SC mice and untreated controls 8 weeks after treatment (Fig. 3A, B), suggesting that only ASC-EE can induce sustained neurobehavioral improvement.
The Combination of EE and ASCs Attenuated Forelimb-Use Asymmetry
To evaluate whether the EE or ASC transplantation ameliorates the asymmetry caused by unilateral brain damage, both cylinder and ladder walking tests were performed at 8 weeks after treatment. The cylinder test revealed a strong tendency toward improvement in the percentage of cylinder wall contacts with the hemiplegic forelimb in ASC-EE mice (4.3 ± 0.2%) compared to those in the other groups including ASC-SC (1.2 ± 0.9%), PBS-EE (1.3 ± 1.4%), and PBS-SC (0.3 ± 0.2%) (Fig. 3C). Wall contacts with the hemiplegic forelimb did not differ among the PBS-SC, PBS-EE, and ASC-SC groups.
In the ladder walking test, the percentage of slips on the transverse rungs of the ladder relative to the total number of steps by the hemiplegic forelimbs were significantly decreased only in the ASC-EE mice (F = 3.071, p < 0.05). In particular, forelimb-use asymmetry of ASC-EE (–3.8 ± 1.2%) was improved compared to that in the PBS-SC controls (–0.4 ± 0.5%, p < 0.05), while these slip percentages were similar among ASC-SC (–1.2 ± 0.9%), PBS-EE (–1.1 ± 0.6%), and PBS-SC (–0.4 ± 0.5%) mice (Fig. 3D). Taken together, cylinder and ladder walking test results suggested that only the combination of EE and ASC injection, but not each alone, can improve forelimb-use asymmetry.
EE Improves Bilateral Grip Strength, Whereas ASC Transplantation Causes Hemiplegic Grip Strength Recovery
We next evaluated whether EE or ASCs can strengthen the motor power weakened by unilateral ischemic brain damage. For this, we measured the grip strength at 8 weeks after treatment. The grip power relative to preoperative evaluation in the contralateral hemiplegic limb showed significant improvements in ASC-EE (28.2 ± 7.8 gram × force), ASC-SC (28.2 ± 6.2), and PBS-EE (32.2 ± 7.2) mice compared to that in the PBS-SC controls (6.1 ± 9.0, p < 0.05) (Fig. 3E), suggesting that EE, ASC transplantation, and the combination of these two treatments are all effective in improving the motor function hindered by unilateral ischemic brain damage.
Interestingly, the grip power of the ipsilateral control limb also significantly increased in ASC-EE (41.0 ± 7.2 gram × force) and PBS-EE (38.8 ± 8.3) mice compared to those in ASC-SC (9.1 ± 5.9) and PBS-SC (12.4 ± 6.9) mice, suggesting that EE, but not ASC transplantation, can also improve motor function of nonhemiplegic limbs in controls. Taken together, EE resulted in systemic improvement in the bilateral motor strength of hemiplegic and nonhemiplegic forelimbs, whereas ASC transplantation improved only the hemiplegic forelimbs. In addition, given that the ASCs were transplanted only into the controlling area of the hemiplegic forelimbs, the therapeutic effect of ASC transplantation may be limited to the transplanted area (Fig. 3E).
EE Enhances the Engraftment of Transplanted ASCs
To determine the engraftment and fate of transplanted ASCs, we prelabeled the ASCs with BrdU for 3 days. The density of the transplanted BrdU+ cells in the striatum, a nonneurogenic area, was 2.34-fold significantly higher in ASC-EE mice (921 ± 172 cells/mm3) than in ASC-SC mice (394 ± 70.5 cells/mm3) at 2 weeks after treatment (t = 2.834, p < 0.05) (Fig. 4A), suggesting that EE increased the engraftment of the transplanted ASCs. This enhanced engraftment of the transplanted ASCs by EE might be more significant given that the mean striatal volume of ASC-EE mice (46.5 ± 1.3 × 10−3 mm3) was significantly larger by 1.2-fold than that of ASC-SC mice (38.6 ± 1.2 × 10−3 mm3) (t = 4.464, p < 0.001).

The fate of transplanted adipose stem cells. ASCs were transplanted into the striata of mice after prelabeling with bromodeoxyuridine (BrdU) (A–E), chloromethyl-1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine perchlorate (CM-DiI) (F–H), or red fluorescent protein (RFP)-expressing retrovirus (I–N). Scale bar: 10 μm. (A) The number of engrafted ASCs prelabeled with BrdU 2 weeks after transplant was higher in mice treated with ASC-EE than it was in ASC-SC mice (*p < 0.05, n = 3). (B–E) When the fate of transplanted cells were evaluated, BrdU-labeled ASCs expressed βIII-tubulin (B, C), neuronal nuclei (NeuN) (D), or glial fibrillary acidic protein (GFAP) (E). (F–H) When cells were prelabeled with CM-DiI, most of the transplanted cells were negative for βIII-tubulin. (I–N) However, three-dimensional confocal microscopy showed that ASCs infected with RFP-expressing retrovirus were triple-labeled for βIII-tubulin and 4′,6-diamidino-2-phenylindole (DAPI). (O) The number of neurons colabeled with BrdU and βIII-tubulin that are derived from transplanted ASCs was higher in ASC-EE mice than ASC-SC mice (*p < 0.05, n = 3). (P, Q) However, most of the grafted cells observed at 2 weeks that differentiated into mature neurons stained with NeuN, no longer remained by 8 weeks posttransplantation. ASC, adipose stem cell; SC, standard cages; EE, environmental enrichment.
The Number of Neurons Derived From the Transplanted ASCs Is Higher in the EE Mice
We next determined the fate of transplanted ASCs. Three-dimensional confocal microscopy showed that a fraction of the BrdU-labeled ASCs were costained for neuronal markers such as βIII-tubulin (Fig. 4B, C) and NeuN (Fig. 4D), suggesting that these transplanted ASCs differentiated into neurons. While a fraction of ASCs expressed neuronal phenotypes such as βIII-tubulin and NeuN, some ASCs were positive for GFAP, suggesting that some ASCs could also differentiate into astroglial lineage cells (Fig. 4E). When cells were prelabeled with CM-DiI, most of the transplanted cells were negative for βIII-tubulin (Fig. 4F–H). Since the signal strength of cells prelabeled with BrdU and CM-DiI is attenuated as the cells divide, we also labeled the cells by infection with RFP-expressing retrovirus. In agreement with BrdU-labeled cells, retrovirus-infected ASCs were also positive for βIII-tubulin and were triple-labeled with DAPI, corroborating neuronal differentiation of the transplanted ASCs (Fig. 4I–N).
To determine the effect of EE on neuronal generation from the transplanted ASCs, we quantified the number of BrdU prelabeled cells that stained positive for βIII-tubulin in the neostriata. The density of BrdU-labeled cells costained for βIII-tubulin was significantly higher by 2.51-fold in the ASC-EE group (168 ± 57.0 cells/mm3) than that in the ASC-SC group (66.7 ± 23.5 cells/mm3) at 2 weeks after treatment (t = 1.634, p < 0.05), demonstrating that EE increases neuronal differentiation from the transplanted ASCs (Fig. 4O). However, most of the grafted cells that differentiated into mature neurons stained with NeuN no longer remained by 8 weeks posttransplantation (Fig. 4P, Q). In addition, the grafted cells did not differentiate into astrocytes or myelin-forming oligodendrocytes at 8 weeks posttransplantation (data not shown).
The Combination of EE and ASCs Enhances Endogenous Neurogenesis
Transplantation of ASCs combined with EE to induce endogenous neurogenesis in the neostriatum was assessed immunohistologically by counting the number of newly generated neurons colabeled with BrdU and βIII-tubulin in mice injected with BrdU for 12 days (Fig. 5A–C). Two weeks after treatment, EE typically potentiated mitogenic neurogenesis that had been modestly induced by transplanted ASCs. Mice treated with EE after transplantation of ASCs showed significant induction of mitosis in the neostriatum (426 ± 161 BrdU+ cells/mm3) of the ipsilateral hemisphere compared with that in the PBS-SC controls (76.8 ± 31.4, p < 0.05) (Fig. 5D). Furthermore, striatal neurogenesis in ASC-EE mice (114 ± 62.2 BrdU+ βIII-tubulin+ cells/mm3) was significantly increased compared to that in the PBS-SC controls (32.2 ± 12.8) in the damaged striata (p < 0.05) (Fig. 5E), although the contralateral hemisphere did not show a statistical difference among the groups. However, when a separate cohort was assessed 8 weeks after treatment, most of the new striatal neurons stained with BrdU+ βIII-tubulin+ did not survive (data not shown).

The combination of EE and ASCs enhanced endogenous neurogenesis. After transplantation of ASCs, mice were injected daily with BrdU for 12 days. (A–C) Mitotic marker BrdU+ or neuronal marker βIII-tubulin+ cells were determined using three-dimensional confocal microscopy. Scale bar: 10 μm. (D, E) The number of newly generated cells labeled with BrdU (D) and the number of new neurons colabeled with BrdU and βIII-tubulin (E) were significantly higher in ASC-EE than in PBS-SC mice (*p < 0.05, n = 3 each), suggesting that EE characteristically potentiated mitogenic neurogenesis that was induced modestly by transplanted ASCs. PBS, phosphate-buffered saline; ASC, adipose stem cell; SC, standard cages; EE, environmental enrichment.
The Combination of EE and ASCs Induces Astrocytic Activation
Since a growing body of evidence suggests that astrocytes play crucial roles in neural functions, we evaluated the levels of reactive astrocytes after treatment with ASCs and/or EE. Striatal GFAP+ cell density (%) was calculated as the GFAP+ area (/mm2) divided by the striatal area (/mm2). Although the number of newly generated astrocytes that can be identified as BrdU+GFAP+ cells was not distinguishable in the neostriata of mice given BrdU for 12 days (data not shown), ASC-EE mice started to show a higher GFAP+ cell density than the other groups at 2 weeks posttreatment (F = 18.186, p < 0.001) (Fig. 6A–J): ASC-EE (12.5 ± 1.2%), ASC-SC (8.2 ± 0.7%) (p = 0.001), PBS-EE (5.7 ± 0.5%) (p < 0.001), and PBS-SC (4.9 ± 0.6%) (p < 0.001) (Fig. 6K). At 8 weeks after treatment, ASC-EE (5.5 ± 0.7%) still showed a significant increase in GFAP+ cell density compared with that of PBS-SC (3.2 ± 0.5%) (p < 0.05) (F = 3.047, p = 0.031) (Fig. 6K). Interestingly, transplantation of ASCs alone did not elicit a long-lasting effect of astrocyte activation (8.2 ± 0.7% at 2 weeks after treatment; 3.6 ± 0.6% at 8 weeks after treatment), even though mice treated with ASCs had significantly higher GFAP+ densities compared to those of PBS-SC mice at 2 weeks posttreatment (p < 0.05). In addition, the levels of the glial scar marker CS-56 did not differ among the groups (Fig. 6L), demonstrating that the combination of EE and ASCs do not increase the detrimental glial scar formation which inhibits neuroregeneration after damage. This result suggests that EE after transplantation of ASCs was able to sustain endogenous astrocytic activation in a synergistic manner.

The combination of EE and ASCs induced astrocyte activation. (A–J) Two and 8 weeks after ASC transplantation and exposure to EE, the amounts of GFAP+ cells (A–H) and CS-56+ cells (I, J) were determined using fluorescence microscopy. Scale bar: 100 μm. (K) The density of striatal GFAP+ cells (%) was significantly higher in ASC-EE mice than in the other groups 2 weeks after treatment (*p < 0.001, n = 3) and than in PBS-SC mice 8 weeks after treatment (*p < 0.05, n = 3). The amount of GFAP+ cells in ASC-SC mice was marginally but significantly higher at 2 weeks (*p < 0.05, n = 3) but not at 8 weeks after treatment. (L) The CS-56+ cell density did not differ between groups. PBS, phosphate-buffered saline; ASC, adipose stem cell; SC, standard cages; EE, environmental enrichment.
Synergistic Effect of ASCs and EE Strongly Correlates with FGF2 Upregulation
To identify the growth factors associated with the repair process of functional recovery induced by ASC transplantation and EE, we measured the protein levels of 10 specific candidate factors using an array-based multiplex ELISA assay. Among these factors, the level of FGF2 was significantly elevated in mice with EE after transplantation of ASCs (908 ± 162 pg/ml) compared with those in the ipsilateral striata of ASC-SC mice (350 ± 38.2 pg/ml) (p < 0.001), PBS-EE (174 ± 37.1 pg/ml) (p < 0.001), and PBS-SC mice (148 ± 36.5 pg/ml) (p < 0.001), as well as those in the contralateral striata of ASC-EE (359 ± 28.9 pg/ml) (p < 0.001) and ASC-SC mice (315 ± 67.6 pg/ml) (p < 0.001) at 2 weeks after treatment (F = 11.249, p < 0.001) (Fig. 7A, B). Furthermore, this FGF2 upregulation was sustained until 8 weeks after treatment in mice with EE after transplantation of ASCs (970 ± 240 pg/ml) (F = 4.633, p < 0.05) compared to those in the ipsilateral striata of ASC-SC mice (406 ± 105 pg/ml) (p < 0.05), PBS-EE (373 ± 116 pg/ml) (p < 0.05), and PBS-SC mice (307 ± 69.2 pg/ml) (p < 0.05), as well as those in the contralateral striata of ASC-EE (429 ± 18.9 pg/ml) (p < 0.05) and ASC-SC mice (214 ± 22.1 pg/ml) (p < 0.05). Western blotting confirmed this FGF2 upregulation (Fig. 7C). However, other factors such as EGF, CSF3, HGF, IGF1, LEP, MMP2, CXCL12, VCAM1, and VEGFA were not synergistically elevated in mice treated with a combination of ASCs and EE compared to the levels in the other groups. This result suggests that FGF2 may have a key role in neurorestoration at 8 weeks when functional outcomes were maximized in mice with EE after transplantation of ASCs.

The FGF2 protein level dramatically increased only in the combination treatment with EE and ASCs. Two and 8 weeks after ASC transplantation and exposure to EE, the neostriata were lysed, and the levels of fibroblast growth factor 2 (FGF2) protein were determined by multiplex ELISA assay (A, B) and Western blotting (C). (A) Representative images of multiplex ELISA assays. (B) The level of FGF2 was significantly higher in the right neostriatum of ASC-EE mice than it was in the other tissues at 2 weeks after treatment (*p < 0.05, n = 3). The significant increase in FGF2 was sustained at 8 weeks after treatment in mice with EE after transplantation of ASCs compared to that in the ipsilateral striata of ASC-SC mice, PBS-EE, and PBS-SC mice, as well as that in the contralateral striata of ASC-EE and ASC-SC mice (*p < 0.05, n = 3). (C) Representative images of Western blotting. PBS, phosphate-buffered saline; ASC, adipose stem cell; SC, standard cages; EE, environmental enrichment; GAPDH, glyceraldehyde 3-phosphate.
Discussion
HI brain injury is a major cause of damage to the fetal and neonatal brain, resulting in considerable incidence of morbidity from CP (41,43,44). The majority of affected children exhibit neurodevelopmental impairment (3,9,40). The beneficial effects of rehabilitation approaches such as cardiorespiratory exercise (5), constraint-induced movement therapy (38), and body weight-supported treadmill training (13) have been reported in children with CP. However, the treatments for adults with CP have usually restricted to supportive cares including the prevention of complications, or a modest reduction of spasticity, abnormal movement patterns and musculoskeletal pain, because of a prevailing view that regeneration of the adult brain in chronic stage is extremely limited. Recently, the focus of rehabilitation strategy is shifting in response to increasing evidence for neurorestoration (1).
Since a paucity of effective treatment is available for adults who sustained neonatal HI brain injury, therapies must be developed that address the needs of neurotrophic factors or stem cells (18). Cell-based therapy has emerged as a potential treatment option to be investigated (32). Among a variety of cell sources, transplantation of ASCs is an option that could potentially reverse the damaged functions. The use of ASCs for prompting angiogenesis and tissue repair has generated interest in its potential therapeutic use (15,27,30), and clinical trials involving ASC-mediated enhancement of bone and adipose regeneration and angiogenesis are under way (15,25,45). In particular, paracrine effects account for improved vascularity in ischemic lesions treated with ASCs (31,35). Treatment with adipose-derived MSCs has therefore been attempted for acute therapy in ischemic stroke. They markedly attenuated brain infarct size and improved neurological function, even more than did bone marrow-derived stem cells (21,26). However, the behavior of the transplanted cells was found to change in the damaged environment, and they showed limited survival and migration in previous studies (10,11).
Several studies have revealed that EE can induce proliferation of endogenous neural stem/progenitor cells in the SVZ or migration of grafted stem cells (19,20,23,24,33). Although the number of patients in chronic phase of brain injury is higher than that in acute phase, the majority of previous studies involving cell transplantation have been performed in acute models (20,23,24,33). Thus, in this study, we intentionally applied EE in the chronic stage. We studied the synergistic effects of EE after transplantation of stem cells at 6 weeks of adult age, 5 weeks after brain injury. Therefore, the main purpose of our study was to verify the therapeutic mechanism of functional recovery in response to EE after transplantation of ASCs in an adult animal model of neonatal HI brain injury in the chronic phase.
We demonstrated that EE after transplantation of ASCs elicited synergistic neurorestorative effects and maximal functional outcomes in various behavioral assessments of rotarod performance and forelimb-use asymmetry tested 8 weeks after treatment. The grip strength test revealed that EE had systemic effects on bilateral grip power, while transplanted ASCs improved only contralateral hemiplegic grip strength 8 weeks after treatment. However, ASCs or EE alone did not facilitate rotarod behavioral recovery during the later period of 6–8 weeks after treatment, despite their transient effect on rotarod performance 2–4 weeks after treatment. In addition, most of these newly generated cells did not survive up to 8 weeks after treatment, even if the grafted cells were more differentiated into neurons, and endogenous neurogenesis was also synergistically enhanced in the neostriata of mice treated with ASCs and EE at 2 weeks after treatment. This finding suggests that the peak functional outcome of rotarod performance was not mediated by neuronal replacement.
One of the limitations of our study is that we adopted intrastriatal injection of the cells instead of systemic administration. Given the invasiveness of intrastriatal approach, systemic administration such as intravenous injection would be a preferred way to deliver the cells in human patients. Systemic administration, however, may lead to the distribution of transplanted cells into other tissues as well as the damaged brain tissue, raising the possibility that fewer number of cells engraft into the damaged brain tissue if the same number of cells are injected. Furthermore, as the blood–brain barrier is relatively intact in chronic model of HI brain injury as compared to the acute phase, effective homing of the transplanted cells into the damaged brain tissue after the systemic administration might require other additional interventions. To facilitate the clinical application of our study, we will investigate to develop intravenous or intra-arterial injection approach which can lead to the behavioral improvement in the model of chronic HI injury. Another limitation of our study is the relatively short-term evaluation period (8 weeks after cell transplantation). Determination of long-term therapeutic effects and safety would facilitate clinical application of our approach to human patients. In addition, the relatively small number of animals we used for mechanism study is also one of the limitations of our study. Although we used a large number of animals for behavioral evaluation, we used only three animals for histological assessment and protein analysis as these evaluations cost significant amount of efforts.
Our results are compatible with previous findings that EE increases endogenous SVZ cell migration toward an ischemic injury and facilitates rotarod function during the relatively earlier period after injury (24). Although the majority of transplanted ASCs did not survive to 8 weeks posttreatment in our study, transient engraftment may have contributed to brain plasticity and better functional outcomes by enhancing endogenous repair processes in the chronic HI brain injury. On the other hand, FGF2 was synergistically enhanced in the striata of mice treated with EE after transplantation of ASCs. These findings coincided with significant functional recovery 8 weeks after treatment. The role of FGF2 as a mediator of the effects of exercise on the brain is supported by demonstrations that FGF2 is not only a strong proangiogenic factor (14) but also a neurotrophic factor which is widely synthesized by astrocytes in the adult brain (2,36,42). The FGF2 can be induced by physical exercise and regulated in an activity-dependent fashion, raising the possibility that FGF2 is involved in behavioral function (16). Since FGF2 was initially required for migration of neural progenitors (46), neuronal recruitment into the striatum may have been potentiated by elevated FGF2 in our study. In addition, neurogenic repair processes largely depend on FGF2, which is predominantly expressed by reactive astrocytes in the injured brain. EE has been shown to increase overall expression of GFAP similar to the pattern of FGF2 (4), raising the possibility that astrocyte activation can mediate functional improvement through an FGF2-dependent mechanism. We propose that the increase in FGF2, but not glial scar marker CS-56, may provide a favorable microenvironment for repair processes and thus contribute to functional recovery.
Conclusion
In a chronic HI brain injury model, EE and transplantation of ASCs synergistically promoted functional recovery through enhanced engraftment and neuronal differentiation of transplanted ASCs, induction of endogenous neurogenesis, and astrocyte activation coupled with upregulation of FGF2. The sustained astrocyte activation and augmentation of FGF2 throughout the study period only in mice with a combination of ASCs and EE suggest that the synergistic function of these two treatment mechanisms may be critical to functional recovery after transplantation. Collectively, our results suggest that a rehabilitative strategy combined with cell-based therapy could be effective for the treatment of CP and that it may also be applied to the treatment of other neurological diseases including adult stroke.
Footnotes
Acknowledgments
This study was supported by grants from the National Research Foundation (NRF-2010-0020408; NRF-2010-0024334), the Stem Cell Research Center of the 21st Century Frontier Research Program (SC-4160), the Yonsei University College of Medicine (6-2012-0025), the Chyung Ki Lee Research Fund, and the Converging Research Center Program (2012K001482) funded by the Ministry of Education, Science and Technology, Republic of Korea. Author contributions: Seo, J.H.: Conception and design, collection and/or assembly of data; Kim, H.B.: Data analysis and interpretation, manuscript writing, final approval of manuscript; Park, E.S., Lee, J.E., Kim, D.W.: Administrative support, data analysis and interpretation; Kim, H.O.: Provision of study material or patients; Im, S.H.: Financial support, data analysis and interpretation; Yu, J.H., Kim, J.Y., Lee, M.Y.: Collection and/or assembly of data; Kim, C.H: Data analysis and interpretation; Cho, S.R.: Conception and design, data analysis and interpretation, manuscript writing, final approval of manuscript. The authors declare no conflict of interest.
