Abstract
Skeletal muscles are the largest organs in the human body, and several therapeutic trials have been conducted that included stem cell transplantation to regenerate damaged or wasted muscles. It is well known that it is essential to make a favorable microenvironment (stem cell niche) to induce the proper differentiation of the transplanted stem cells. Some drugs, such as losartan (angiotensin II type I blocker), enhance the therapeutic effects of transplanted stem cells by inhibiting fibrosis. In this study, we hypothesized that another substance, vitamin C (ascorbic acid), might improve the niche for stem cell transplantation based on its potent antioxidant effects. In both gross and microscopic observations, vitamin C-depleted mice exhibited more incomplete regeneration of damaged muscles than those treated with vitamin C. Carbonylated protein groups, which are the end products of oxidative stress, were detected in all experimental groups; however, the vitamin C-depleted groups exhibited a more potent positive reaction than that of the vitamin C-supplied groups. The difference is clearer in the presence of transplanted stem cells. Moreover, the serum total vitamin C level and the ascorbic acid (AA) to dehydroascorbic acid (DHA) ratio also were decreased in the presence of transplanted adipose-derived stem cells (ASCs). Taken together, these data can be considered as proof of vitamin C utilization by cells in vivo. The vitamin C-supplied groups displayed more severe fibrosis than that of the vitamin C-depleted groups. Since vitamin C is a major cofactor for the collagen synthesis, its deficiency resulted in reduced fibrosis. In conclusion, we demonstrated that vitamin C not only has a positive effect on adjusting the stem cell niche to boost muscle regeneration but also has an adverse aspect due to its profibrotic effect.
Introduction
Skeletal muscles are the largest organs in the human body, occupying about 40% of the entire body mass (13). Given the current trend in global aging of the human population, sarcopenia, the loss of skeletal muscles, is becoming a vital concern. Moreover, other progressive and intractable muscular diseases, such as Duchenne muscle dystrophy (DMD), remain unsolved problems because of the absence of effective cures.
Therefore, there have been a lot of trials to treat muscle wasting, and stem cell transplantation is one of them. Cells of various types (muscle satellite cells, myoblasts, mesoangioblasts, and so on) have been used in transplantation (6, 8, 10, 17, 23, 26, 31) so far; however, we have used adiposederived stem cells (ASCs) (2, 3, 7, 20) in this experiment for the following reasons. First, ASCs can differentiate into multiple types of cells including satellite cells and myocytes, which are directly needed for muscle regeneration, and endothelial cells, which can support muscle regeneration by supplying adequate blood flow to the damaged area (1–3, 6). Second, ASCs also can be obtained easily in large amounts by lipectomy. Finally, the most critical benefit of ASCs is that they are considered to have immunomodulatory properties (27), and consequently, allogenic ASCs can be transplanted with less concern of major histocompatibility complex (MHC) incompatibility between donor and recipient.
Stem cell transplantation is, however, not a master determinant. Since stem cells have the potential to differentiate into multiple lineages, this characteristic is both an advantage and a disadvantage. In other words, it is essential to make a favorable microenvironment (stem cell niche) to induce proper differentiation of the transplanted stem cells toward the specific desired cells.
Previous studies demonstrated that specific substances, such as losartan (angiotensin II type I blocker), enhanced the therapeutic effects of transplanted stem cells by inhibiting fibrosis (4, 8, 35). In this study, we hypothesized that another substance, vitamin C (ascorbic acid), might also induce a better niche for stem cell transplantation. Vitamin C is known as a potent antioxidant (24), and for this reason, it can increase the viability of the transplanted cells by reducing oxidative and inflammatory stress. Based on these facts, we hypothesized that vitamin C can play a pivotal role in muscle regeneration through stem cell transplantation.
Vitamin C is also crucial for collagen synthesis (30). Thus, in the case of vitamin C deficiency, we expected that fibrosis would be diminished due to the reduced synthesis of collagen fiber. Here, we additionally investigated in the present report whether the decreased fibrosis had a positive effect on muscle regeneration.
To verify the role of vitamin C, we used senescence marker protein 30 (SMP30) knockout (KO) mice in the experiment. Since SMP30 functions as gluconolactonase in vitamin C biosynthesis, its knockout mice cannot synthesize enough amount of l-ascorbic acid, which is required for the maintenance of normal health condition (15).
Materials and Methods
Animals
Eight- to 9-week-old, male, specific pathogen-free, SMP30 knockout mice (n = 48) weighing 19–23 g were used in this study. Mice were initially obtained from the laboratory of Dr. Akihito Ishigami in Tokyo Metropolitan Institute of Gerontology (TMIG), Tokyo, Japan, and bred in a specific pathogen-free facility. The SMP30-KO mice were maintained in a room at 22 ± 3°C with a relative humidity 50 ± 10% and a 12-h light–dark cycle. The mice were given food and water ad libitum. The genomic DNA of the SMP30-KO mice was purified from the tissue of the mouse tails using several combined procedures, which have been used traditionally. The genotypes were identified by PCR using the sense primer TS3 (5′-CTAGCCATGGTGGATGAAGAT-3′), the antisense primer TA4 (5′-CAAGTAACTCTAGGTATGGAC-3′), and the inserted positive selection marker NEO (5′-TCGT GCTTTACGGTATCGCCGCTCCCGATT-3′), which was described elsewhere (12), showing how the knockout in the SMP30 gene was made in the mice by inserting a targeting vector into the third exon of the SMP30 gene (data not shown). Animal experiments were performed in accordance with the NIH guidelines for the care and use of laboratory animals and with approval from the institutional animal care and use committee of Kyungpook National University (KNU 2011-19).
Isolation and Culture of Mouse Adipose-Derived Stem Cells (mASCs)
Mouse abdominal fat tissue specimens were obtained from inguinal fat pads of donors under 1 year of age [male and female C57BL/6 mice from Japan SLC, Inc., Hamamatsu, Japan, and green fluorescent protein-positive (GFP+) transgenic mice from Dr. Masaru Okabe in Osaka University, Osaka, Japan]. Harvested fat tissues were briefly disinfected with 70% ethanol and extensively washed with equal volumes of Dulbecco's phosphate-buffered saline (DPBS; Linz, PAA). After that, fat tissues were digested with 0.2% collagenase type I (Worthington Biochemical, Lakewood, NJ, USA) at 37°C for 10 min. Digested fat tissues were filtered through a 70-μm nylon cell strainer (SPL, Naechon-myeon, Korea) to separate dissociated cells from undigested tissues. After centrifugation of the filtered tissue, the liquid was separated into three layers: an upper layer of yellow fat, an intermediate layer of adipose tissue, and a bottom layer of pelleted stromal vascular fraction (SVF). The pellet in the bottom layer was carefully collected and washed with PBS twice, and then seeded onto a culture dish (SPL) with complete medium consisting of low-glucose Dulbecco's modified Eagle medium (DMEM-LG) (Gibco, Gaithersburg, MD, USA) containing 10% fetal bovine serum (FBS) (Gibco) and 1% penicillin-streptomycin solution (WelGENE, Daegu, Korea). Cells were placed in a 37°C, 5% CO2 conditioned incubator. After a 2-h incubation, the culture dish was washed with PBS to remove nonadherent cells and red blood cells, and then the media were changed every 2 days. Cells were subcultured to three to four passages (P3-P4) before transplantation.
Muscle Laceration, ASC Transplantation, and Muscle Specimen Collection
During anesthesia by intraperitoneal injection of a Zoletil (Virbac, Carros cedex, France) and Rompun (Bayer, Leverkusen, Germany) combination, focal disc-shaped muscle laceration followed by ASCs (5 × 105 cells/mouse) or PBS injection was performed in the right legs of all mice. Details are shown in Figure 1A. Two or 4 weeks after the laceration, both lacerated right (Rt.) and intact left (Lt.) legs of all mice (except for the left leg of the ASC-treated mice) were collected as shown in Figure 1B.

Illustration of surgery and specimen collection. (A) Skeletal muscle removal procedures. (1) Thoroughly shave the hair on back of the calf of a mouse leg. (2) With a surgical blade, make a horizontal incision on the center of the back calf. (3) Open up the incision to make enough space for the operation and use a Bovie (Aaron Medical Industries, St. Petersburg, FL, USA) to give hemostasis along the muscle. (4) Perform a biopsy by vertically placing a 4-mm biopsy punch on the exposed muscle and remove a part of gastrocnemius muscle and underlying soleus muscle. (5) After hemostasis, inject adipose-derived stem cells (ASCs) or PBS directly to the damaged area using a micropipette. (6) Make a simple continuous suture with a 3/0 black silk suture (Ethicon, Livingston, Scotland, UK). (7) Finish the operation by tying both ends. (B) Skeletal muscle specimen collection procedures. (1) Perform a blunt dissection on the gastrocnemius muscle and underlying soleus muscle by slipping a pair of forceps under the Achilles' tendon. (2) Place a scissor under the muscle that has been bluntly dissected and incise the origin of the muscles and Achilles tendon, respectively. (3) Completely separate the muscle and weigh before it dries.
Experimental Design
SMP30-KO mice were divided into four groups: (i) laceration without treatment (N group) (n = 12), (ii) laceration with ASC transplantation (C group) (n = 12), (iii) laceration with vitamin C supplement (V group) (n = 12), and (iv) laceration with combinatorial therapy of ASCs and vitamin C (VC group) (n = 12). Prior to muscle laceration, vitamin C (l-ascorbic acid, Sigma-Aldrich, St. Louis, MO, USA; 1.5 g/L) was supplied in the drinking water for a week and was maintained for the rest of the experimental period (2 or 4 weeks) after surgery in the V and VC groups. During the same period, vitamin C depletion was carried out by supplying tap water and an autoclaved diet to the N and C groups. Among the 12 mice per group, 10 mice received wild-type (WT)-ASC transplants and the other 2 received GFP-positive ASC transplants to visualize regenerated myofibers differentiated from the transplanted cells. Because of the different stages of muscle healing after injury (11), half of the mice were sacrificed at 2 weeks after laceration when muscle regeneration was maximized, and the rest were sacrificed at 4 weeks after laceration when muscle fibrosis was matured. From each mouse, a blood sample was collected from the caudal vena cava, and collection of a muscle sample was also done as described above.
Measurement of Serum Vitamin C by HPLC
The serum vitamin C level was measured with the high-performance liquid chromatography (HPLC)-electrochemical detection method as previously described (16). Blood samples from the sacrificed mice were centrifuged at 3,000 × g (Eppendorf 5810R, Hamburg, Germany) for 15 min at 4°C. The supernatant serum was carefully retrieved and 100 μl of serum was mixed with 450 μl of 3% metaphosphoric acid (Sigma-Aldrich). After centrifugation at 10,000 × g for 10 min, the supernatant was retrieved, and the serum vitamin C level was measured as both the reduced form (ascorbic acid, AA) and oxidized form (dehydroascorbic acid, DHA) with the Shodex-5SIL-4E column (4.6×250 mm; Showa Denko, Tokyo, Japan).
Histopathology and Immunohistochemistry
Mice were anesthetized with Zoletil and Rompun and then rapidly exsanguinated through the caudal vena cava. Muscle specimens were quickly removed, cut transversely at the healing portion, and fixed in 10% neutral buffered formalin (DC Chemical Co. Ltd., Seoul, Korea), then processed routinely and embedded in paraffin wax. The sections were cut to 4-μm thickness and then deparaffinized in toluene (Duksan Pure Chemicals, Gyeonggi-do, Korea) and rehydrated in a graded alcohol series. The sections were stained with hematoxylin (Sigma-Aldrich) and eosin (Junsei chemicals, Tokyo, Japan) (H&E) to evaluate the degree of regeneration and with Masson's trichrome stain (Sigma-Aldrich) to evaluate the degree of muscle fibrosis. The number of regenerative (centronucleated) myofibers in the healing area was counted in 400× fields of each muscle tissue. The size of the regenerated myofibers was quantified in 400× fields, and the fibrotic area was also examined in 200× fields for each muscle with the Leica Application Suite software (LAS version 2.8.1, Leica Microsystem, Seoul, Korea).
For immunohistochemistry (IHC), other muscle samples were fixed in methacarn fixative [methanol (Duksan Pure Chemicals)/chloroform (Sigma-Aldrich)/acetic acid (Duksan Pure Chemicals), 6:3:1]. After fixation, sectioning, deparaffinization, and rehydration were done as for H&E above. IHC was done using the Protein Carbonyls Immunohistochemical Staining Kit (Cosmo Bio Co., Ltd., Tokyo, Japan) according to the manufacturer's instruction.
Immunofluorescence Observation
The isolated muscle tissues were embedded in freezing medium (Leica, Wetzlar, Germany) and sectioned at 8-μm thickness. The sectioned muscle tissues were fixed in 4% paraformaldehyde (Yukari Pure Chemical, Tokyo, Japan) at 4°C for 15 min and immunostained with GFP (Santa Cruz Biotechnology, Santa Cruz, CA, USA) as the primary antibody and Tyramide Signal Amplification Kit (T20922, Invitrogen, Carlsbad, CA, USA). 4′, 6-Diamidino-2-phenylindole (DAPI, dihydrochloride; Molecular Probes, Leiden, the Netherlands) was used as a nuclear counterstain.
Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted from frozen muscle tissues using Trizol reagent (Invitrogen). Total RNA was separated from DNA and proteins by adding chloroform and precipitated by mixing with isopropanol. The concentration of the total RNA was measured with a Quanti-iT RNA assay kit (Invitrogen) and a Qubit fluorometer (Invitrogen). The extracted RNA was then used as a template for reverse transcription into cDNA using RT & GO mastermix (MP Biomedicals, Santa Ana, CA, USA) following the manufacturer's instructions. Synthesized cDNA was amplified with quantitative real-time PCR using Rotor-Gene Q (Qiagen, Hilden, Germany) with the Rotor-Gene SYBR Green PCR kit (Qiagen) and the following primers: collagen type I [5′-TGCAACATGGAGACAGGTCAGA-3′ (sense), 5′-GA ACGGGAATCCATCGGTCAT-3′ (antisense) (8)], forkhead box O3 (FoxO3) SYBR Green QuantiTect Primer Assay (QT01777867), mammalian target of rapamycin (mTOR) SYBR Green QuantiTect Primer Assay (QT00118734), myogenic differentiation (MyoD) SYBR Green QuantiTect Primer Assay (QT00101983), paired box protein 7 (Pax7) SYBR Green QuantiTect Primer Assay (QT00147728), Regulatory Associated Protein Of mTOR, (Raptor) SYBR Green QuantiTect Primer Assay (QT00145502), transforming growth factor-β1 (TGF-β1) SYBR Green QuantiTect Primer Assay (QT00145250), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) SYBR Green QuantiTech Primer Assay (QT01658692, all from Qiagen). The results were evaluated by Rotor-Gene Q. The threshold cycle (Ct) was determined, and the relative gene expression ratio was calculated as follows: fold change = 2ΔΔCt.
Statistical Analysis
All values are presented as mean ± SEM. Statistical analyses were determined using Student's t test or one-way analysis of the variance (ANOVA) followed by Bonferroni post hoc test for multiple comparisons. The value of statistical significance was set at p < 0.05 or p < 0.01.
Result
Gross Assessment of Muscle Wound Healing
All the treatment groups, either ASCs or vitamin C, showed better regeneration in the gross morphology of the mice, sacrificed 2 weeks after laceration (2 weeks), and the V and VC groups exhibited improvement in the gross morphology of the mice, sacrificed 4 weeks after laceration (4 weeks) (Fig. 2A). The size of the wound was largest in the right leg of the N group (N-Rt.) both at 2 and 4 weeks; however, especially in the V-Rt. and VC groups, the severity of the wound was diminished upon gross examination. This result agrees with the data on the moist muscle weight shown in Figure 2B and C.

Gross assessment of muscle healing. Experimental groups are as follows: laceration without treatment (N group), laceration with ASC transplantation (C group), laceration with vitamin C supplement (V group), and laceration with combinatorial therapy of ASCs and vitamin C (VC group). Lt., left legs that served as intact controls; Rt., right legs that are lacerated. (A) Gross morphology of muscle specimens. Dashed circles indicate lacerated lesions. (B) Moist muscle weight of muscle specimens of the mice sacrificed 2 weeks after laceration. (C) Moist muscle weight of muscle specimens of the mice sacrificed 4 weeks after laceration. Data are shown as mean ± SEM (*p < 0.05, **p < 0.01).
Validation of the Antioxidative Function of Dietary Vitamin C Intake
We performed immunohistochemical staining to detect carbonylated protein groups, which are the end products of protein oxidation by free radicals, with a commercial IHC kit. Since protein carbonyls are considered as useful markers of oxidative stress, vitamin C-depleted groups were expected to exhibit strong positive reactions. All groups exhibited positivity for protein carbonyls (Fig. 3). However, the N-Rt. and C groups displayed a stronger positivity than that of the V-Rt. and VC groups as expected. In the case of the C group, the transplanted cells worsened the microenvironment by causing severe inflammatory stress in addition to the existing oxidative stress. Thus, the positive reaction was strongest in the C group (Fig. 3K, M) rather than the N-Rt. group (Fig. 3C, E).

Representative images for detecting the end product of oxidative stress by protein carbonyls immunohistochemical staining. (A) Positive control [normal kidney, 2,4-dinitrophenylhydrazine (DNPH+)]; (B) negative control (normal kidney, DNPH–); (C) N-Rt., 2 weeks; (D) N-Lt., 2 weeks; (E) N-Rt., 4 weeks; (F) N-Lt., 4 weeks; (G) V-Rt., 2 weeks; (H) V-Lt., 2 weeks; (I) V-Rt., 4 weeks; (J) V-Lt., 4 weeks; (K) C, 2 weeks; (L) VC, 2 weeks; (M) C, 4 weeks; (N) VC, 4 weeks. All groups exhibited distinct positivity for carbonylated protein groups, a useful index of oxidative stress. Among them, vitamin C-depleted groups showed more potent positivity, which was maximized in the presence of transplanted cells. Original magnification: 200×. Scale bars: 100 μm.
Decreased Level of Serum Total Vitamin C and the Ratio of AA (Ascorbic Acid) to DHA (Dehydroascorbic Acid) in the ASC-Transplanted Groups
Since the SMP30-KO mice exhibit vitamin C deficiency naturally, in this experiment, the supply of vitamin C was highly dependent on the food and drinking water; moreover, since the food and drinking water of the N and C groups were depleted for vitamin C during the entire experimental period, their serum vitamin C levels were nondetectable (N.D.). However, despite both the V and VC groups being given the same amount of vitamin C with all the other conditions the same except for the transplantation of ASCs, they exhibited quite different values at 2 weeks after laceration not only in their serum total vitamin C levels but also in the AA (ascorbic acid)/DHA (dehydroascorbic acid) ratios (Fig. 4A). The total vitamin C level was diminished in the ASC-transplanted group, the VC group, and this meant that the ASCs utilized the vitamin C themselves through cellular uptake and/or absorption. In addition, ascorbic acid, the reduced form of vitamin C, increased in the presence of ASCs, whereas dehydroascorbic acid, the oxidized form of vitamin C, decreased with cell transplantation. Consequently, the AA/DHA ratio was also lower in the VC group than in the V group, indicating that vitamin C functioned as a reducing agent, which reduced oxidative and inflammatory stress potentiated by cell transplantation. There was no significant difference between the two groups at 4 weeks after laceration (Fig. 4B).

Serum vitamin C level after muscle laceration ± cell transplantation. (A) Two weeks after laceration. (B) Four weeks after laceration. Vitamin C level of the N and C groups was nondetectable. The ratio of ascorbic acid (AA, reduced form) to dehydroascorbic acid (DHA, oxidized form) and total vitamin C level was decreased by cell transplantation in early phase (2 weeks). Later, however, there was no significant difference between two groups. Data are shown as mean ± SEM. (*p < 0.05, **p < 0.01).
Confirmation of the Differentiation of Transplanted ASCs Into Muscle Fibers
To ascertain whether the transplanted ASCs differentiated to muscle fibers as intended, immunofluorescence was done for anti-GFP on the frozen tissue sections of the GFP-positive ASC-transplanted mice, with the slides observed under a fluorescence microscope. Several myofibers that express green fluorescence were identified at a wavelength of 488 nm, showing that these fibers were derived from GFP-positive ASCs. In addition, ASCs that are fusing onto an existing myofiber were also observed (Fig. 5).

Immunofluorescence observation to confirm myogenic differentiation of transplanted ASCs. (A) GFP; (B) 4′,6-diamidino-2-phenylindole (DAPI) for nuclear counter stain; (C) merged image. Myogenic differentiation of green fluorescent protein-positive (GFP+) ASCs was confirmed by immunofluorescence based on the fact that several myofibers (asterisk) and ASCs that are fusing onto an existing myofiber (arrow) displayed bright green fluorescence. Original magnification: 200×. Scale bars: 100 μm.
Histopathological Evaluation of Muscle Regeneration
To evaluate the degree of muscle regeneration, H&E staining was performed in the 2-week muscle samples. All groups exhibited regenerative (centronucleated) myofibers around the lacerated area. However, there was a significant difference between the vitamin C-depleted and vitamin C-supplied groups. The left legs of the N and V groups had no centronucleated myofibers, since they were intact; however, we measured the sizes of their myofibers to assess the effect of vitamin C on normal muscles. Even though vitamin C did not affect the normal size of the myofibers in the intact controls, myogenic progenitors seemed to differentiate so well that they could form larger and more mature myofibers under the influence of vitamin C during regeneration from damage (Fig. 6).

Histopathological evaluation by H&E staining of muscle regeneration 2 weeks after muscle laceration. (A) N-Rt., (B) V-Rt., (C) C, (D) VC. (E) The average size of centronucleated myofibers of vitamin C-supplied groups was larger than that of vitamin C-depleted groups. Vitamin C did not affect the normal size of myofibers when comparing peripherally nucleated mature myofibers of intact controls (N-Lt. and V-Lt.). Original magnification: 400×. Scale bars: 50 μm. Data are shown as mean ± SEM. (*p < 0.05, **p < 0.01).
Increased Expression of mTOR and Raptor in the Vitamin C and ASC Cotreated Group
We quantified the expression levels of mTOR and Raptor mRNAs (which are closely related to the increase in muscle mass) in the 2-week muscle samples using qRT- PCR to infer the protein expression level of mTOR complex 1 (mTORC1), which is composed of mTOR, Raptor, and mammalian lethal with SEC13 protein 8 (mLST8). The VC group exhibited the highest expression levels of mTOR and Raptor mRNAs. Although the V-Rt. and C groups also had a tendency for increased expression of both genes, there was no significant difference compared to the N-Rt. group. Only the VC group showed significant increases in both mTOR (Fig. 7A) and Raptor (Fig. 7B) mRNAs. Based on these results, mTORC1 was assumed to be upregulated under the combinatorial treatment of vitamin C and ASCs.

Real-time PCR analysis in the 2-week samples for relative expression levels of several genes that are related to the change in the muscle mass. (A) Mammalian target of rapamycin (mTOR); (B) regulatory associated protein of mTOR (Raptor); (C) Forkhead box protein O3 (FoxO3a), normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH), respectively; and (D) the relative expression ratio of mTOR complex 1 (mTORC1) components to FoxO3a. Vitamin C did not affect the expression levels of mTOR and Raptor by itself as seen in the comparison of intact controls (N-Lt. and V-Lt.), but it can upregulate both genes by the combined treatment with ASCs. In addition, not only FoxO3a expression level but also the relative expression ratio of mTORC1 components to FoxO3a was downregulated by vitamin C, even in comparison to the intact controls. Data are shown as mean ± SEM. (*p < 0.05, **p < 0.01).
Decreased Expression of FoxO3a in the Vitamin C-Supplied Groups
Since FoxO3a has the opposite function of mTORC1 on muscle mass, we also quantified the expression level of FoxO3a mRNAs in the 2-week muscle samples. All vitamin C-supplied groups exhibited a lower expression of FoxO3a than that of their vitamin C-depleted counterparts (Fig. 7C). Thus, this demonstrated that vitamin C is intimately involved in the regulation of FoxO3a.
Vitamin C Induced a Higher Expression Ratio of mTORC1 to FoxO3a
We calculated the ratio of the mTORC1 components (mTOR and Raptor) to FoxO3a in each group based on the results described above. The formula was [(“comparative expression level of mTOR” + “comparative expression level of Raptor”)/“comparative expression level of FoxO3a”]. The ratios presented a similar pattern with the FoxO3 expression levels. In other words, the ratio was higher in the vitamin C-supplied groups than in the vitamin C-depleted groups (Fig. 7D).
Increased Expression of Pax7 and MyoD in the ASC-Transplanted Groups
Pax7 and MyoD mRNA expression levels were examined in the 2-week muscle samples to compare the degree of proliferation of satellite cells and myogenic determination in each group. Both genes were upregulated when the ASCs were transplanted, and the C group exhibited higher expression levels of Pax7 and MyoD than that of the VC group (Fig. 8).

Real-time PCR analysis in 2-week samples for relative expression levels of Pax7 and MyoD, Values are normalized to GAPDH. (A) Paired box protein 7 (Pax7), (B) myogenic differentiation (MyoD). ASC transplantation induced upregulation of both Pax7 and MyoD. However, vitamin C rather decreased MyoD expression, when comparing the C and VC groups. Data are shown as mean ± SEM. (*p < 0.05, **p < 0.01).
Histopathological Evaluation of Muscle Fibrosis
Masson's trichrome staining was done to analyze the degree of fibrosis in the 4-week muscle samples. Since vitamin C is essential for collagen synthesis, fibrosis was also severe in the vitamin C-supplied groups (Fig. 9) as expected.

Representative images for histopathological evaluation by Masson's trichrome stain of fibrosis 4 weeks after muscle laceration. (A) N-Rt., (B) V-Rt., (C) C, (D) VC. Collagen fibers are more abundant in vitamin C-supplied groups than vitamin C-depleted groups. Original magnification: 200×. Scale bars: 100 μm.
Increased Expression of TGF-β1 and Collagen Type I in Vitamin C-Supplied Groups
To validate the relationship between vitamin C and fibrosis, qRT-PCR was done with TGF-β1 and collagen type I primer sets in the 4-week muscle samples. TGF-β1 was upregulated by vitamin C. The V-Rt. group had a higher expression of TGF-β1 than that of the N-Rt. group, and the VC group expressed much more TGF-β1 than the C group. Furthermore, the higher TGF-β1 expression levels of the vitamin C-supplied groups were not restricted to the damaged portion (the V-Lt. group exhibited higher expression of TGF-β1 than that of the N-Lt. group), indicating that vitamin C may enhance fibrogenic potentials (Fig. 10A). The collagen type I expression pattern also had the same tendency as TGF-β1, except for the intact left legs (Fig. 10B).
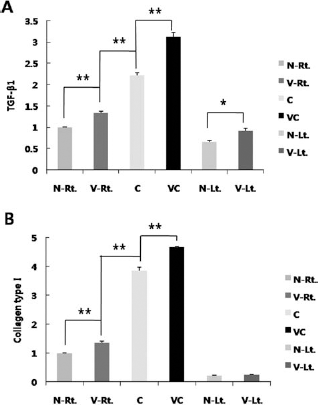
Real-time PCR analysis. (A) Relative expression level of transforming growth factor-β1 (TGF-β1), normalized to GAPDH, (B) Relative expression level of collagen type I, normalized to GAPDH. TGF-β1 and collagen type I presented the same pattern except for intact control. Vitamin C-supplied groups and ASC-transplanted groups exhibited higher expression levels of both genes. Consequently, the synergistic effect of vitamin C and ASCs made the VC group express both genes more. Data are shown as mean ± SEM. (*p < 0.05, **p < 0.01).
Discussion
Vitamin C is a well-known antioxidant, and it also plays a crucial role in proline hydroxylation, which is required for the synthesis of collagen (25). Since collagen is a prominent structural protein found in connective tissues such as bones, tendons, ligaments, teeth, skin, cartilage, and blood vessels (25), it is essential for reconstructing damaged tissues. Thus, vitamin C may help tissue regeneration by decreasing the level of oxidative stress and inflammatory stress, caused by tissue damage and nonautologous (allogenic) cell transplantation, and supplying the basic materials to rebuild connective tissues around the parenchyma. However, collagen is also a main component of fibrotic tissue; it may promote fibrosis in the damaged area via increased collagen synthesis.
This double-sidedness of vitamin C means that it is quite confusing as to whether vitamin C makes a better niche for muscle regeneration. Our experimental results showed that it really ameliorates the engraftment of transplanted ASCs into host muscle. However, the results also implied the potential long-term adverse outcome of vitamin C treatment because of the profibrotic effect apart from its beneficial effect.
Normally, SMP30 is not expressed in skeletal muscles even in normal WT mice (22); these transgenic mice were used only for the evaluation of the effects of vitamin C deficiency in muscle regeneration with or without stem cell transplantation.
Grossly, vitamin C-supplied groups exhibited a better morphological recovery and an increased moist muscle weight, but we could not exclude the possibility that the increased moist muscle weight was due to the replacement of the parenchyma by the fibrotic tissues, in other words, pseudohypertrophy. To elucidate the precise mechanism of the increased moist muscle weight in the vitamin C-supplied groups, the following parameters were examined.
First, we verified that orally treated vitamin C worked effectively as an antioxidant agent. The decreased amount of total vitamin C in the VC group could be proof of vitamin C uptake by the cells. The lower ratio of AA to DHA in the VC group than in the V group also indicated that vitamin C was utilized to reduce inflammatory and oxidative stress during active regeneration. In addition, differentiation of the transplanted ASCs into the skeletal muscles, as we intended, was confirmed by visualization.
After verifying the intended function of dietary vitamin C and transplanted ASCs, microscopic observation and qRT-PCR analyses were conducted to reveal the mechanism for the increase in muscle mass. The average size of the centronucleated myofibers of the vitamin C-supplied groups was bigger than that of vitamin C-depleted groups. Counting the number of regenerative myofibers per field was meaningless, since it correlated with the size of the myofibers. As described above, vitamin C-supplied groups had larger and more mature myofibers than the vitamin C-depleted groups, and the reason of this phenomenon could be explained as follows. First, there are two types of transporters that carry vitamin C into cells. The first one is the glucose transporter (GLUT), which transports dehydroascorbic acid (oxidized form of vitamin C), and the other is sodium-dependent vitamin C transporter 1 (SVCT), which transports ascorbic acid (reduced form of vitamin C) (34). Savini et al. reported that the expression levels of both types of vitamin C transporters decreased during differentiation of the muscles, but increased with severe oxidative stress (32). It is assumed that myogenic progenitors of the vitamin C-depleted groups were blocked from completing their differentiation and maturation following determination of their lineage to muscle cells by oxidative stress. Since they expressed less vitamin C transporters when differentiated, the blockage of complete myogenic differentiation was ascribed to their decreased resistance to oxidative stress.
Second, mTORC1 and FoxO3a expression levels, which are closely related to the control of muscle mass during the developmental period, were also estimated indirectly by analyzing their mRNA levels using real-time qPCR. mTOR and Raptor, the constituents of mTORC1, did not show any obvious correlation with vitamin C; however, FoxO3a exhibited a distinctive decline in expression by the supply of vitamin C. Based on a previous study, which revealed that there is a positive dose–response association of dietary vitamin C intake with the plasma insulin-like growth factor (IGF1) concentration (36), the declined expression of FoxO3a was construed as the result of activated IGF1/ phosphoinositide-3-kinase (PI3K)/V-Akt murine thymoma viral oncogene homolog (Akt) signaling due to the intake of dietary vitamin C. The reason that the mTOR and Raptor expression levels did not show any significant changes by vitamin C was considered as follows. Activated Akt may directly control FoxO3a, and not mTORC1. To promote mTORC1 expression, several additional steps, such as the inhibition of tuberous sclerosis 1 and 2 (TSC1 and TSC2) and Ras homolog enriched in brain (Rheb) activation, are needed following Akt activation (33). Thus, those intermediate steps could be influenced by vitamin C; mTOR and Raptor expression levels did not increase, although vitamin C was assumed to increase the plasma IGF1 level. mTOR and Raptor seemed to be increased by the stem cell transplantation rather than by the supply of vitamin C, and that would be a result of the various growth factors secreted by the ASCs or the role of ASCs as a source of amino acids, which are known to be a direct stimulator of mTORC1 (33). To link these two factors, we calculated the ratio of the relative expression level of mTORC1 constituents (mTOR and Raptor) to FoxO3a. The ratio was higher in the vitamin C-supplied groups than in the vitamin C-depleted groups, and this meant that vitamin C supported protein synthesis essential for muscle regeneration. Therefore, this expression pattern of the signaling pathway may contribute to the different sizes of the regenerated myofibers. Although there were significant differences not only between the N-Lt. and V-Lt. groups (intact controls) but also between the V-Rt. and VC groups in the mRNA expression level of the mTORC1 constituents (mTOR and Raptor) to FoxO3a, there were no differences in the moist muscle weights or microscopic observations. Hence, we believe that the IGF1/PI3K/Akt pathway, which is a major regulator of developing muscles (33), is not closely connected with controlling muscle mass during regeneration in the skeletal muscle of adult mice. Nonetheless, vitamin C itself is involved in the inhibition of FoxO3a, a sensor of the redox state (5); therefore, not only can it prevent muscle atrophy by inhibiting the expression of atrogenes (29) but it can also block apoptosis (5). Thus, it is plausible that vitamin C positively affects muscle regeneration following stem cell transplantation by boosting the settlement of the transplanted stem cells via its viability enhancing effect.
We also verified the mRNA expression levels of Pax7, a satellite cell marker, and MyoD, a myoblast marker, to verify whether vitamin C had a direct influence on increasing the satellite cell fraction or on stimulating myogenic determination. Significant differences were detected in the ASC-transplanted groups rather than in vitamin C-supplied groups. In other words, ASC-transplanted groups showed higher expression levels of both genes, and this may result from ASC differentiation to satellite cells. ASCs have the potential to differentiate into several cell types, and their differentiation to satellite cells has already been reported (20). Since activated satellite cells differentiate into myoblasts, the expression pattern of MyoD was supposed to be similar to that of Pax7. However, vitamin C did not induce a clear correlation between the mRNA expression levels of both Pax7 and MyoD. Because of this expression pattern, the positive effects of vitamin C on muscle regeneration appear ambiguous; however, considering transplanted ASCs and cell sensitivity to oxidative stress, the effect of vitamin C is not harmful. In the case of the satellite cells, their quiescent form is more resistant to oxidative stress than their activated form (28). Furthermore, even in myogenic cells, the lower their differentiation, the more resistant to oxidative stress they are (28). Therefore, the effect of vitamin C on regulation of Pax7 and MyoD might not be positive, although it could be helpful in establishing a more favorable microenvironment like preparing a reservoir for regeneration, even though the onset point of regeneration would be delayed. Accordingly, the C group that displayed the highest mRNA expression of Pax7 and MyoD did not exhibit a significant difference with the N-Rt. group in moist muscle weight 4 weeks after laceration, the approximate time point for the completion of regeneration. This result indicates that myogenic specification is crucial for muscle regeneration without a doubt; however, in the case of stem cell transplantation, other factors involved in securing the viability of transplanted cells are also important.
Factors related to fibrosis should be checked, since vitamin C is an essential cofactor for the synthesis of collagen fibers, and the fibrotic scar tissue accumulated within the injury site interrupts muscle regeneration and functional recovery (18). TGF-β is thought to play a pivotal role in muscle fibrosis by triggering muscle cells to differentiate into fibrotic cells during muscle healing (18); therefore, we analyzed the degree of fibrosis with samples collected 4 weeks after laceration by checking the mRNA expression level of collagen type I along with that of TGF-β and by microscopic observation after special staining for differentiating collagen fibers. As expected, more collagen fibers were observed in the vitamin C-supplied groups based on the results of the Masson's trichrome stain, and the same tendency was exhibited in the real-time PCR results. However, cell transplantation affected more closely the increased expression levels of collagen type I and TGF-β than the supply of vitamin C, and this result could mean that the ASCs secrete TGF-β as a cytokine and collagen as an extracellular matrix component (14). Putting various reports together, TGF-β inhibits muscle differentiation through functional repression of myogenic transcription factors by mothers against decapentaplegic homolog 3 (Smad3) (19), and disrupts an IGF2-stimulated autocrine amplification cascade that is required for muscle differentiation (9). In addition, it also inhibits satellite cell proliferation (28) and induces transdifferentiation of myogenic progenitors to myofibroblasts (18, 21). Therefore, the supply of vitamin C could be considered as harmful for muscle regeneration in this respect because of its influence on TGF-β upregulation, which leads to more fibrogenic potentials. In the present study, we adopted a surgical laceration method for assessing muscle regeneration after damage; therefore, the increase in TGF-β in this study was neither severe nor persistent as is in the case of degenerative muscular disease models such as mdx mice. Hence, the negative effect of vitamin C on muscle regeneration through TGF-β upregulation was not strong in the present study, but it would be potentiated and overwhelm the positive effect of vitamin C under chronic inflammatory conditions; thus, it would be better to use vitamin C with TGF-β blockers such as losartan to utilize vitamin C in muscle regeneration more effectively.
In addition, both myofibroblast activation and collagen are certainly needed at a level suitable for wound healing. If the rate of collagen synthesis by myofibroblasts exceeds that of collagen degradation, then it results in fibrosis (37). Otherwise, in normal wound healing, activated myofibroblasts promote wound contraction, and this process is followed by division and migration of epithelial cells and/or endothelial cells over the basal layers to regenerate the damaged tissue, and then wound healing process is completed (37). Furthermore, since collagen presumably serves as a scaffold to maintain the shape of the newly formed blood vessels (25), it may assist in providing a good blood supply to the damaged area for regeneration. Taken together, vitamin C has advantages and disadvantages, and for this reason, its influence on muscle regeneration can vary depending on the diverse circumstances where it is used. However, at least in the present study that adopted a therapeutic approach by stem cell transplantation in the muscle laceration model, the beneficial effects of vitamin C outweighed its detrimental effects, based on several reasons discussed above.
In conclusion, we demonstrated that vitamin C not only positively affects the stem cell niche via its potent antioxidant effect through which it boosts muscle regeneration, but it also might hamper muscle regeneration via its profibrotic effect.
Through the present study, we reconfirmed that it is essential to create proper stem cell niche to improve the efficiency of stem cell transplantation for muscle, and these results might be presented as a basis for clinical applications.
Footnotes
Acknowledgments
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (2011-0025973) and the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A111551). The authors declare no conflict of interest.
