Abstract
The development of small diameter vascular grafts with a controlled pluricellular organization is still needed for effective vascular tissue engineering. Here, we describe a technological approach combining a tubular scaffold and magnetically labeled cells to create a pluricellular and organized vascular graft, the endothelialization of which could be monitored by MRI prior to transplantation. A novel type of scaffold was developed with a tubular geometry and a porous bulk structure enabling the seeding of cells in the scaffold pores. A homogeneous distribution of human mesenchymal stem cells in the macroporous structure was obtained by seeding the freeze-dried scaffold with the cell suspension. The efficient covering of the luminal surface of the tube was then made possible thanks to the implementation of a magnetic-based patterning technique. Human endothelial cells or endothelial progenitors were magnetically labeled with iron oxide nanoparticles and successfully attracted to the 2-mm lumen where they attached and formed a continuous endothelium. The combination of imaging modalities [fluorescence imaging, histology, and 3D magnetic resonance imaging (MRI)] evidenced the integrity of the vascular construct. In particular, the observation of different cell organizations in a vascular scaffold within the range of resolution of single cells by 4.7 T MRI is reported.
Keywords
Introduction
While synthetic materials like Dacron® or expanded polytetrafluoroethylene (ePTFE) have been successfully used for over 40 years in bypass surgeries of large diameter vessels (>6 mm), few synthetic alternatives to the autologous vein and artery tissues currently exist for small diameter applications (5,41). The failure of small diameter graft is mainly due to thrombosis, which results from the host response to foreign material. In order to avoid this response and thus improve the graft integration, research approaches aimed at combining synthetic grafts with biological entities. For example, they focused on endothelial cell recruitment and adhesion on the graft lumen prior or after implantation [increase of graft permeability (13,17,30), gelatin, fibronectin, or laminin coatings (13), addition of growth factors like fibroblast growth factor-1 (FGF-1), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF) (67)]. However, these approaches faced the issue of coating stability and long-term attachment of endothelial cells on synthetic surface. More integrative approaches were then followed, with the goal to imitate the structure and composition of native arteries in order to reproduce their functional properties (35,62). Recently, organized patterns mimicking the layer by layer structure of native arteries were obtained (23,28,36). For such purpose, cells were cultivated to form sheets further assembled into a tubular structure, which was finally endothelialized (36). This type of tubular grafts was reported to handle a burst pressure superior to the one of the saphenous vein, the gold standard in terms of autologous grafts (31). In another study (19), allogeneic smooth muscle cells were cultured on degradable polyglycolic acid (PGA) tubular scaffolds to support synthesis of a collagenous extracellular matrix. A subsequent decellularization process removed antigenic cells from the graft, thereby allowing the use of banked allogeneic cells to produce vascular grafts that are nonimmunogenic. This vascular graft can be seeded with autologous endothelial cells to reduce the risk of thrombosis associated with small diameter vascular grafting in peripheral or coronary arteries. Alternatively, two groups have pioneered the use of a radial magnetic force to form a tubular endothelium. First, the combination of magnetic cell sheet engineering and cylindrical magnet led to tubular constructs made of heterotypic layers of endothelial cells, smooth muscle cells, and fibroblasts (25). Also a magnetic radial force was used to coat a collagen membrane with several layers of smooth muscle cells followed by one layer of endothelial cells (44,45).
The goal of the present study was to create a small diameter vascular graft mimicking the first two layers of a native artery (the endothelium and the muscle layer) using a scaffold based approach, so that a rapid assembly of the graft could be achieved. The scaffold material developed is a tubular polysaccharide hydrogel based on pullulan and dextran. This type of polysaccharide has been previously successfully tested for cell viability and adhesion (15,59) and the implantation of nonporous tubes for 8 weeks in a rat aorta showed neotissue formation and no inflammatory response (15). These 3D structures with biodegradable polymers could thus be proposed as mimics of the extracellular matrix. The originality of the polysaccharide tubes presented in this study stands in their macroporous structure, which makes cell colonization possible. Furthermore, the desiccated structure enables a rapid and efficient cell seeding by a spongelike effect upon hydration with the cellular suspension of interest. Such a method is proposed here to introduce human mesenchymal stem cells (MSCs) in the bulk of the tube. These cells could then further differentiate into smooth muscle cells (SMCs), which compose the media layer of native arteries. The mimicry of the arterial intima also requires coating the luminal surface with endothelial cells. This tricky step remained unsuccessful using conventional static or rotary seedings. Here, we aim at taking advantage of magnetic endothelial cells to force the luminal endothelialization.
Magnetic nanoparticles ranging from a few to tenths of nanometers are increasingly being used in the biomedical field. They are routinely applied in the field of diagnostic imaging as contrast agents in magnetic resonance imaging (MRI). Besides their use as probes for cell biology (20,38,52), their therapeutic applications include the antitumoral magnetic hyperthermia (8,60) or the magnetic guidance of drugs to target tissues in vivo (49,50). During the last decade, in vitro cell labeling with magnetic nanoparticles has become a common method for in vivo cell imaging, the labeled cell being detectable by MRI (9,11,29). Noncytotoxic labeling methods of cells with iron oxide (maghemite, magnetite) nanoparticles have consequently been extensively developed and studied with a large range of cell types (10,65), including endothelial cells (3,40,64). More recently, magnetic forces were used in vivo to retain or to capture labeled cells to injured sites for localized cell-based therapies (16,37), in particular with targeting endothelial cells to the surface of stents (46,48). Recently, magnetic techniques have been proposed in the framework of tissue engineering, towards the in vitro generation of predictable and controllable tissues. Such methods were demonstrated to enable cell manipulation with magnetic devices and the creation of highly controlled cell patterns (22,27,51,53). The use of magnetic forces to promote the endothelialization of a tubular scaffold suitable for internal stem cell seeding has not yet been reported.
The present study therefore took advantage of the intrinsic characteristics of the scaffold material and of the remote control over cell organization offered by the magnetic force to create a pluricellular graft with a defined structure. In particular, the vascular patterns successfully generated through magnetic attachment of endothelial cells were evaluated in terms of homogeneity and cellular organization using complementary imaging modalities: histological staining, confocal fluorescence microscopy, and 3D noninvasive MRI cell imaging.
Materials and Methods
Scaffolds
Polysaccharide-based scaffolds were prepared using a mixture of pharmaceutical polysaccharides (Mw 200,000, Hayashibara, Okayama, Japan) and dextran (Mw 500,000, Pharmacosmos, Holbaek, Denmark) at a ratio 75:25 with a cross-linking agent sodium trimetaphosphate (STMP, Sigma-Aldrich, Saint-Quentin Fallavier, France) used in the food industry. The STMP is used at 11% (w/v) under alkaline conditions as previously described (6,34,47). For enhancement of endothelial cell adhesion, 1% gelatin (Sigma-Aldrich) was directly added to the polysaccharide solution. Pores were created by a gas-foaming technique. Scaffolds were given a tubular shape during synthesis, coated with a 0.2% gelatin solution for enhancement of endothelial cell adhesion, and then were freeze-dried for 48 h afterwards for complete removal of water. The obtained desiccated tubes had inner and outer diameters of around 2 and 8 mm while desiccated. After full hydration and cell seeding of the tubes, inner and outer diameters of 2.5±0.5 mm and 8.7±0.6 mm were measured. Storage occurred at room temperature until use.
Cell Culture
Human umbilical vein endothelial cells (HUVEC-Cs, ATCC, GC Standards S.a.r.l. Molsheim, France) and human endothelial progenitor cells isolated from cord blood (EPCs provided by G. Uzan and M. Lavergne, Inserm, U972, Hôpital Paul Brousse, Villejuif—all patients gave informed consent for all procedures—sex unknown) were cultured in endothelial basal medium [EBM-2 (Lonza, Verviers, Belgium)] supplemented with endothelial growth mediamicrovascular (EGM)-2MV SingleQuots (Lonza; CC-4147). Human mesenchymal stem cells (MSCs, Lonza; Batch 7F3675, female, 22 years) were cultured in mesenchymal stem cell basal media (MSCBM; Lonza). Cells were cultured in a cell incubator at 37°C with 5% CO2. All cell types were adherent during cell culture. After detachment, they were rounded with a mean diameter of 17.5±2.2 μm, 13.5±2.1 μm, and 18±4.2 μm for HUVEC-Cs, EPCs, and MSCs, respectively.
Magnetic Cell Labeling
The magnetic nanoparticles were synthesized following Massart's method (39). They have a magnetic core made of maghemite (γ-Fe2O3) of 8 nm diameter and bear negative charges due to carboxylate groups on their surface, ensuring their stability in aqueous solution.
Endothelial cells and endothelial progenitor cells were incubated for 30 min at 37°C with a labeling solution consisting of a sterile suspension of nanoparticles in a serum-free culture medium (RPMI; PAA, Les Mureaux, France) supplemented with sodium citrate (Sigma-Aldrich) at 5 mM. The extracellular iron concentrations were 2 mM for HUVEC-Cs and 5 mM for EPCs. The labeling medium was subsequently removed and replaced by a nanoparticlefree culture medium (EBM-2) for 2 h, ensuring that all nanoparticles adsorbed on cell membranes were internalized. The seeding into scaffolds was systematically performed right after this 2-h process.
The magnetic particle uptake was quantified by single-cell magnetophoresis (66). The velocity of labeled cells in suspension upon application of a well-calibrated magnetic field gradient was measured and the magnetic moment of each tracked cell was deduced by balancing the magnetic force with Stokes viscous drag. This method allowed for both the determination of the iron mass per cell and the distribution of this mass among the cell population (n = 120).
Fluorescent Cell Labeling
The cells were detached with trypsin (PAA) and labeled with a fluorescent membrane marker. PkH26 (red) and pkH67 (green) dyes (Sigma-Aldrich) were respectively used for endothelial cells (HUVEC-Cs or EPCs) and MSCs. Cell seeding of the scaffolds occurred just afterwards.
Cell Proliferation Assay
A CyQuant assay (Invitrogen, Life Technologies SAS, Saint Aubin, France), which is based on the production of a fluorescent product upon reagent binding with nucleic acids, was used (1) to assess endothelial cell viability after magnetic labeling and during culture with or without the application of a magnetic force; (2) to measure the number of HUVEC-Cs in the tubular gels after cell seeding. A standard curve was first established for cell numbers ranging from 500 to 2 × 105 cells.
For the proliferation assays, 1,000 cells were seeded in 48-well plates (BD Falcon, Becton Dickinson France SAS, Le Pont-De-Claix, France), and the cell number was monitored for 6 days. Three conditions were tested: nonlabeled cells and magnetically labeled cells cultured under magnetic force or not. The force (~10 pN) was created by a permanent neodymium magnet (SupermagnetE, Uster, Switzerland) put under the bottom of the culture wells. The conditions were run in quadruplicate and the experiment was repeated three times.
Transmission Electron Microscopy (TEM)
Magnetically labeled HUVEC-Cs ([Fe] = 5 mM, 30-min incubation) were washed with 0.1 M cacodylate buffer (Sigma-Aldrich) and incubated in 2% glutaraldehyde (Sigma-Aldrich) in cacodylate buffer for 1 h at 4°C. Samples were postfixed with 1% osmium tetroxide containing 1.5% potassium cyanoferrate, followed by uranyl acetate staining (all Sigma-Aldrich). Finally, samples were gradually dehydrated in ethanol (30–100%) and embedded in Epon (Delta Microscopie, Ayguesvives, France). Thin sections of 70 nm were examined with a Zeiss EM902 transmission electron microscope at 80 kV (INRA, Plateform MIMA2, Laboratoire de Genomique et Physiologie de la Lactation, Jouy en Josas, France).
Capillary Network Formation
For analysis of capillary tube formation, 1 ml Matrigel (Becton Dickinson) was laid into a 35-mm diameter Petri dish and incubated at 37°C for 30 min. Control and magnetically labeled HUVEC-Cs were trypsinized (at days 0, 1, and 3 after labeling), and 5 × 105 cells were plated onto Matrigel. Patterns were observed by videomicroscopy.
Cell Seeding of the Tube Bulk
Cells can be incorporated into the desiccated scaffolds upon their hydration with the appropriate cell suspension, which are in this case MSCs. In order to visualize the cell distribution in the porous structure of the scaffold after cell seeding, MSCs were magnetically labeled with iron oxide nanoparticles ([Fe] = 0.5 mM, 30-min incubation time) and 1-cm-long tubes were seeded with 5 × 105 cells. The seeded tubes were then cultivated for 3 days before being fixed in 4% paraformaldehyde and visualized using confocal fluorescent imaging, MRI, or a Prussian blue staining performed on cryosections (see MRI and Histological Analysis sections). In dual seedings, the MSCs were not magnetically labeled.
Magnetic Cell Patterning
Two types of neodymium magnets (SupemagnetE) were used for the cell patterning experiments: (1) a plain cylindrical magnet (22 mm diameter and 10 mm height), for the asymmetric patterning of cells, and (2) a holed cylindrical one (10/20 mm inside/outside diameters and 10 mm height) for the symmetric patterning of cells. The magnetic force exerted on labeled cells by the plain magnet was evaluated by measuring the magnetic field at various distances with a teslameter (RS Components SAS, Beauvais, France) and retrieving the magnetic field gradient. The holed cylindrical magnet was more finely calibrated using a viscous drag approach similar to magnetophoresis. The lumen was divided in concentric circles with an increment of 150 μm, and cells were tracked in each region (20 < n < 114, mean value = 73) to deduce the magnetic force as a function of the distance from the magnet symmetric axe.
For endothelial cell patterning, an inox (stainless steel) well (home-made) was put in the lumen of each cylindrical magnet (symmetric cell patterning) or attached to a plain magnet surface (asymmetric cell patterning), and both were sterilized by autoclaving. The tubes were then cut in 1.2-cm-long segments and submitted to UV light before being introduced in sterile wells. They were then hydrated with EBM-2, and placed in the incubator. In the meantime, magnetically labeled HUVEC-Cs were trypsinized, labeled with pkH26 dye, and resuspended in EBM-2 at a concentration of 2 × 105 cells/50 μl. The culture medium in the lumen of the tubes was then replaced by 50 μl of this cell suspension. Culture medium was then added in each well to reach the top of the well. Wells were placed for 2 h in the incubator. Subsequently, the magnets were removed, and the tubular scaffolds were put in 24-well plates (BD Falcon) with 1.5 ml medium and cultured for 1–7 days before being fixed in 4% paraformaldehyde and analyzed in confocal microscopy, MRI, and/or histology. For pluricellular scaffolds, the desiccated tubes were not hydrated with EBM-2 but with a suspension of 5 × 105 MSCs in MSCBM labeled with pkH67 dye before magnetic patterning, and EPCs labeled with pkH26.
The seeded grafts were cultured in EBM-2 for 1–7 days.
Confocal Microscopy Imaging
Samples were fixed in 4% paraformaldehyde and images were acquired with an inverted Olympus microscope at 4× magnification, associated with a spinning disc confocal unit (Andor Technologies, Belfast, UK), and a cooled CCD camera. Image analysis and reconstruction were performed with ImageJ (NIH, Bethesda, MD, USA).
Magnetic Resonance Imaging (MRI)
Samples were fixed in 4% paraformaldehyde and stored at 4°C in PBS (PAA) before use. A 3-mM gadolinium solution (Doctarem®, Guerbet, Villepinte, France) was added in the lumen of the tube before imaging to enhance the contrast and assess scaffold architecture. MRI was performed on a 4.7 T small animal research imaging system (Bruker, Fällanden, Switzerland) interfaced to ParaVision software for preclinical MR imaging research (PARCC, HEGP, Paris, France). To detect with a high spatial resolution, a cryogenic probe was used. A flash-3D sequence was chosen with a voxel size of 50 × 50 × 50 μm3, a repetition time (TR) of 20 ms, and an echo time (TE) of 5 ms. Images were analyzed with ImageJ software.
Histological Analysis
After cell seeding, the tubes were cultured for 3 days in a cell culture incubator. They were then fixed in a 4% paraformaldehyde solution before being embedded in OCT (Optimal Cutting Temperature compound; VWR International S.A.S., Fontenay-sous-Bois, France) and frozen in isopentane (Sigma-Aldrich) cooled by a liquid nitrogen bath. Prussian blue staining was used to reveal the presence of intracellular iron. Cryosections (8 μm) were incubated for 20 min with 2% potassium ferrocyanide in 2% hypochloric acid, washed again, and counter-stained with nuclear fast red (all from Sigma-Aldrich), dehydrated, and mounted.
Statistics
Data are presented as mean±standard error. Results are analyzed using a one-way analysis of variance (ANOVA) with Bonferroni's post hoc test. A value of p < 0.05 is considered as statistically significant.
Results
Endothelial Magnetic Cell Labeling
Endothelial cells and endothelial progenitor cells were incubated with a suspension of anionic iron oxide (maghemite) nanoparticles (8 nm). As a result, the labeled cells bear a magnetic moment, which depends on the internalized iron mass and can be determined by a viscous drag approach (Stokes law) (66). The iron mass per cell is tailored by varying the incubation time or the extracellular iron concentration during incubation (Fig. 1A, B). In the present study, endothelial cells were incubated for 30 min at [Fe] = 2 mM, which led to a mean iron mass per cell of 9.2±0.6 pg. Endothelial progenitors were incubated for 30 min at [Fe] = 5 mM, corresponding to an iron content of 10±0.4 pg per cell. Corresponding electron microscopy images are shown in Figure 1C, demonstrating the packing of nanoparticles in endosomes.

Magnetic labeling of endothelial cells. (A, B) Human umbilical vein endothelial cells (HUVEC-Cs) (A) and endothelial progenitors (EPCs) (B) were incubated at 37°C for 30 min with iron oxide nanoparticles at different concentration ([Fe] = 0, 0.5, 1, 2, 5, or 10 mM). Magnetophoresis enabled us to determine the mean iron mass per cell as a function of the extracellular iron concentration during the incubation period. (C) Transmission electron microscopy (TEM) image of an EPC incubated with iron oxide nanoparticles at [Fe] = 5 mM for 30 min at 37°C. The nanoparticles, black points on the image, are packed within intracellular endosomes and the extracellular space is free of any. (D, E) The proliferation [N(t)/N(t = 0)] of the HUVEC-Cs (D) and EPCs (E) was monitored over 6 days in a nanoparticle-free medium. For HUVEC-Cs, three conditions were compared: Control HUVEC-Cs (no label, no force); labeled HUVEC-Cs (9.2 pg iron, no force), labeled HUVEC-Cs under magnetic force (9.2 pg iron, 10 pN force). For EPCs, control cells and labeled ones ([Fe] = 5 mM) were compared. (F) Control and labeled EPCs ([Fe] = 5 mM) were deposed on Matrigel, in a magnetic field gradient of 10–20 mT/mm for the labeled ones, creating a cellular force of 10 pN. Pictures taken at different time points show that control cells and labeled cells experiencing a force form the same types of capillary networks and at the same speed. (G) Magnetically labeled ([Fe] = 2 mM, 30-min incubation) HUVEC-Cs were seeded in Matrigel at days 0, 1, and 3 after labeling, and images of the capillary structures obtained 16 h after seeding are shown on the images. No differences can be observed concerning the organization of these networks.
The proliferation curves of HUVEC-Cs and EPCs after labeling show no significant difference between the labeled and the nonlabeled groups (Fig. 1D, E). Cell proliferation under magnetic force was also studied for HUVEC-Cs. This force was orthogonal to the adhesion plane and of the same order of magnitude than the one applied on cells during magnetic patterning. Wells containing labeled HUVEC-Cs submitted to the magnetic force and the control ones (no label, no force) showed a similar increase in cell population (Fig. 1D).

Polysaccharide-based tubular scaffold for optimal cellularization. (A) Porous tubular scaffold for a seeding of mesenchymal stem cells (MSCs) in the bulk and endothelial cells in the lumen. (B) The tube is put in a chamber fitting the dimension of a hollow cylinder magnet. (C) The video-tracking of magnetically labeled endothelial cells enabled us to derive the map of the radial magnetic cellular forces in the tube lumen, which ranges from 1 to 2 pN. (D) Sequential seeding of cells. (D1) Cell seeding of the tube bulk was achieved through its hydration with a MSC suspension. (D2) Magnetically labeled endothelial cells were then added in the lumen of the tube so that they experienced an increasing radial magnetic force promoting their attachment to the luminal surface. (D3) Finally, the cell proliferation during the graft culture led to the complete tube endothelialization.
The mass of iron per cell was simultaneously quantified for HUVEC-Cs at day 1 (6±0.4 pg) and day 3 (1.1±0.1 pg) during proliferation and was found inversely proportional to the rate of cell proliferation, demonstrating that no nanoparticles are exocytosed by the cells and that each cell's iron mass was equally divided between its daughter cells.
Magnetically labeled HUVEC-Cs and EPCs were also plated on Matrigel, and the subsequent formation of capillary networks monitored. For EPCs, capillary tube formation was observed by videomicroscopy during 18 h after seeding with or without the application of a magnetic force (10 pN) (Fig. 1F). The capillaries patterns were not affected by the magnetic labeling and force. For HUVEC-Cs, we verified that the magnetic labeling did not alter as well the ability of the cells to form network, but also that the cells are still capable in forming such patterns at day 1 or day 3 after labeling (Fig. 1G).
Polysaccharide-Based Tubular Scaffold for Optimal Cellularization
For the engineering of a small diameter vascular graft, a polysaccharide scaffold was developed. It was given a tubular shape with an inner diameter of 2 mm after lyophilization (Fig. 2A). This scaffold has an average pore size of 200 μm to make cell colonization possible. To apply a radial magnetic force at the tube lumen site, a home-made container was adapted to a hollow cylinder magnet (Fig. 2B). The subsequent magnetic forces experienced by cells in the lumen were quantified by tracking their movements towards the lumen walls. Force mapping is presented in Figure 2C, in the range from 1 pN in the center of the lumen to 2 pN on the luminal surface of the tube. Figure 2D finally describes the cell seeding protocol. The first step consists of the hydration of the desiccated tube with a MSC suspension to perform cell seeding in the bulk of the tube. In the second step, magnetically labeled endothelial cells are added in the tube lumen and a radial magnetic force is applied in order to promote cell attachment to the intimal surface. The seeded tubes are then cultivated for 1–7 days to make possible cellular proliferation and maturation. Here, cell seeding of the tube bulk (step 1) and cell coating of lumen (step 2) were first studied independently before they were combined to obtain a pluricellular vascular graft.
Imaging of the Cellular Organization in the Tube Bulk
The seeding with MSCs of the tube bulk was first performed without magnetic patterning of endothelial cells on the tube lumen. Similarly to endothelial cells, the MSCs were labeled with two different imaging probes, namely a fluorescent marker and iron oxide nanoparticles. The scaffolds were then hydrated with a MSC suspension to allow for cell seeding. Fluorescent imaging shows the incorporation of the cells (green dots) into the tube pores (Fig. 3A). MSCs labeled with iron oxide nanoparticles were detected with a Prussian blue staining (iron imaging) on histological slices (blue spots, arrows) revealing the same distribution of the cells through the scaffold pores (Fig. 3B). Magnetically labeled MSCs were also seen on highresolution MRI images (black spots) showing a large covering of the pores in the whole tube (Fig. 3C).

Imaging of the cellular organization in the tube bulk. Desiccated tubes were hydrated with a cell suspension of MSCs, resulting in a cell distribution in the tube bulk. Cells were both fluorescently and magnetically labeled to allow fluorescent imaging (A) and iron oxide imaging (B, C). (A) On confocal images, MSC appear as green spot in the tube bulk. (B) A Prussian blue staining reveals the iron content of the cells which in turn appear in a blue color in the gel pores (as indicated by the arrows). (C) High-resolution magnetic resonance imaging (MRI) enabled us to get a global view of the MSC distribution over the whole scaffold.
Endothelial Cell Attachment Through Magnetic Patterning
The magnetic cell patterning of endothelial cells was first performed without seeding of MSCs. After magnetic labeling, endothelial cells (HUVEC-Cs) were labeled with a fluorescent membrane marker and seeded in the lumen of the tubular scaffolds. When no external force was used, some very low fluorescence was detected by confocal microscopy, which demonstrates that cells did not attach to the tube surface. Other controls (magnetically labeled cells with no magnet and nonlabeled cells with magnet) showed similar behaviors, with very low fluorescence at the lumen. On the other hand, the transient presence of a magnet led to the formation of a cell pattern reflecting the symmetries of the magnetic force field: a cylindrical magnet around the tube resulted in a homogenous coating of endothelial cells all over the luminal surface of the tube, while a magnet put on the side of the tube drove cells in a limited region of the surface (Fig. 4).

Endothelial cell attachment through magnetic patterning. The tubes were hydrated with culture medium. HUVEC-Cs were labeled both magnetically and with a red fluorescent membrane tag before being seeded in the tube lumens. (A) When no magnetic force was applied, the cells did not attach to the tube surface and a very low fluorescent signal was detected (A3). (B) On the contrary, the use of a cylindrical magnet around the tube resulted in a homogenous coating of the surface with cells (B3, B4). (C) A magnet placed at one side of the tube resulted in an asymmetric coating (C3, C4). For each condition (control, symmetric, and asymmetric coating): (1) is an image of the experimental set up, (2) is a bright field view, (3) is a fluorescent view of a single Z plane of the tube lumen, (4) represents a 3D reconstruction performed with ImageJ, corresponding to 50 planes with an interval of 20 μm, and (5) is a schematic view of the 3D reconstruction.
In the case of symmetric magnetic patterning, histological studies were conducted to analyze the cell pattern and to reveal cellular structures. Cell nuclei were found to be rarely superimposed, which therefore suggested that the pattern mostly consisted of a cell monolayer. Prussian blue staining demonstrated the presence of intracellular iron, which appears as blue spots on Figure 5A.
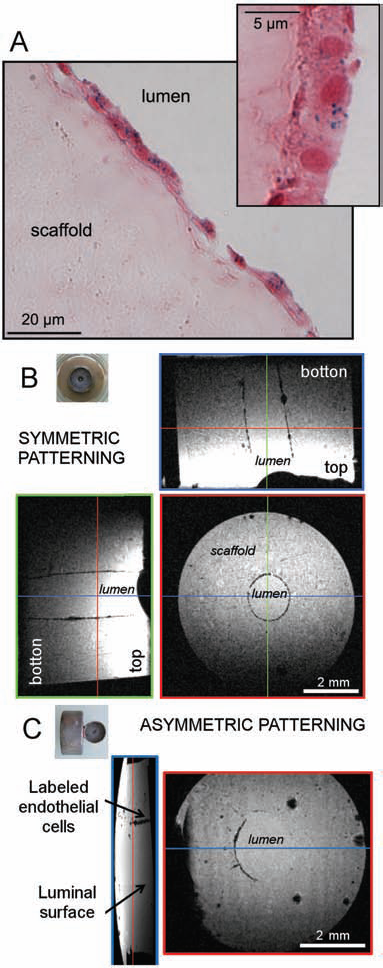
Iron oxide imaging. (A) The observation (40× magnification) of a histological section of a tube patterned with magnetic HUVEC-Cs reveals the presence of cells attached to the lumen. The Prussian blue staining of this section also demonstrate the intracellular localization of magnetic nanoparticles identified as blue dots within the cells. (B, C) On MRI, magnetically labeled cells appear as dark spots, because of their iron content. Views from different planes are displayed. For each plane of interest, the position of a slice according to a perpendicular plane is indicated by a color line and the corresponding view of this slice is framed by a bold line of the same color. (B) Tubes were hydrated and placed in the center of a cylindrical magnet before magnetically labeled HUVEC-Cs were added in the tube lumen. The homogeneity of the created cell pattern can be assessed both in the XY plane and in the Z direction. (C) The magnet was put at one side of the tube to create an asymmetric pattern: cells concentrate in a localized region.
MRI of the Magnetic Endothelial Layer
MRI was used to evaluate 3D magnetic patterning of cells (Fig. 5B, C). Magnetically labeled cells clearly appear as black dots on MR images. Orthogonal slices further assess the homogeneity of the pattern in the Z direction over the whole height of the imaged sample (5 mm). In the case of an asymmetric magnetic field (Fig. 5C), only one side of the surface is covered with magnetic cells, the other side providing a control image of the scaffold, as it appears without seeded cells. Conversely, the use of a cylindrical magnet leads to coverage of the whole surface with labeled cells (Fig. 5B).
Graft Endothelialization and Implementation to Biomimetic Vascular Grafts
The cylindrical magnet enabled to create a pattern of endothelial cells on the lumen for blood vessel engineering. After the transient application of the magnetic force (2 h), the magnet was removed and the tubes were cultured for 3–7 days. The cell population was monitored and showed a global increase over the 7 days of culture, suggesting an evolution toward a continuous endothelium (Fig. 6). On the contrary, a very low number of cells was detected for the control tubes with nonlabeled cells.

Graft endothelialization. Evolution of the cell number per tube at days 1, 3, and 7 after an initial symmetric magnetic patterning of 2 × 105 HUVEC-Cs with a cylindrical magnet at day 0: (white bars) magnetically labeled HUVEC-Cs; (black bars) nonlabeled HUVEC-Cs (control). A value of p < 0.05 was considered as statistically significant. *p < 0.05, ***p < 0.005.
Afterwards, a dual cell seeding was performed as described in Figure 2C. The tubes were first hydrated with a MSC suspension labeled with pkH67 and green spots could be observed in the bulk of the tube on confocal images (Fig. 7). The luminal surface was then magnetically covered with EPCs. For this final step of dual cell seeding, human EPCs were chosen as a challenging experimental model with great potential for biomedical applications. In particular, EPCs have the capacity to differentiate into cells of the endothelial lineage but have not undergone any specialization (arterial or venous), they possess a capacity to proliferate higher than that of mature endothelial cells (HUVEC-Cs), while exhibiting senescence after much more population doubling. These magnetically labeled EPCs were tagged with pkh26 dye, and they appeared as red spots on confocal images and covered the tube luminal surface. The combination of a novel type of macroporous tubular polysaccharide scaffolds and a magnetic cellular engraftment of the tube luminal surface allows the rapid construction of a tissue engineering blood vessel.
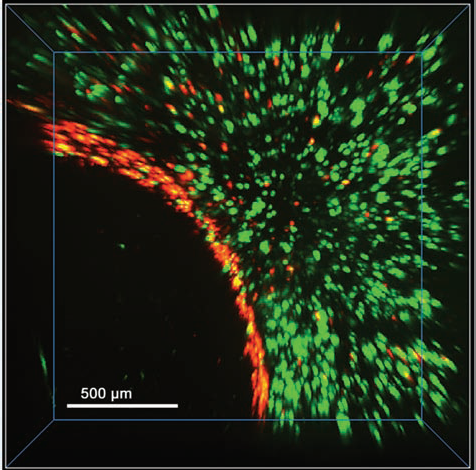
Toward vascular grafts with a pluricellular organization. The tubes were hydrated with a suspension of MSCs before the magnetic patterning of the lumen was performed with magnetically labeled EPCs. The cells were labeled with a green (MSCs) and red (EPCs) fluorescent membrane marker. These pluricellular grafts were imaged at day 3. This figure represents a 3D reconstruction of a stack composed of 50 slices of 10 μm. The specific localization of MSCs in the tube bulk and EPCs on the lumen is clearly visible.
Discussion
A native artery is composed of three main concentric layers. The inner continuous endothelium intima is responsible for the exchanges between the inside and the outside of the vessel and is involved in the regulation of several mechanisms included homeostasis, vasomotricity, inflammation, and smooth muscle cell phenotype (1,14,57). The second layer, the media, consists in a muscle tissue which enables the vasoconstriction and dilatation. The cellular organization and the orientation of the fibers composing the extracellular matrix are implied in the mechanical resistance of the vessel. Finally, the adventitia, a connective tissue, supports the whole structure. The goal of the present study was to engineer a vascular graft mimicking the first two layers of a native artery. The tube bulk was seeded with mesenchymal stem cells, which possess the potential to differentiate into smooth muscle cells (61,63). Any other cells could be similarly seeded in the tube bulk. An equivalent of the media layer could thus be obtained after a differentiation step. The intima was obtained by the magnetic patterning of endothelial cells on the tube lumen leading to the endothelialization of the graft.
The choice for an external force to drive the endothelialization was motivated by the obstacles usually encountered in endothelial cell attachment as well as the poor control over the endothelial layer thickness offered by existing techniques. This choice implied the magnetic labeling with iron oxide nanoparticles of the endothelial cells. As a consequence, the effect of this labeling technique was studied as a prerequisite to further use in cell patterning.
Cytocompatibility of Magnetic Labeling
Cytotoxicity of iron oxide nanoparticles is a fundamental issue, especially because of their clinical applications as diagnostic contrast agents for MRI. Beyond the physical and chemical characteristics of the nanoparticles (material and surface properties, size, and aggregation state), other parameters may affect the results on nanoparticle toxicity: the cell model chosen, the method used for cell transfection, the dose, and exposure time (21,55). All of these four parameters influence the final cellular iron concentration. For cellular iron contents similar to the ones obtained here, studies conducted with another type of magnetic nanoparticles, magnetite cationic liposomes (MCL), reported no effect on the proliferation of various cell types (human endothelial and smooth muscle cells, MSCs among others) and on three differentiation pathways of MSCs: adipo-, osteo-, and chondrogenesis (53). With the same type of nanoparticles but at higher cellular iron content, cellular stresses characterized by a transiently reduced proliferation and a remodeling of the actin cytoskeleton were observed (56). The effect of magnetic labeling on MSC differentiation was also studied using the commercially available ferrumoxide suspension, Ferridex, that is the association of dextran-coated iron oxide nanoparticles with a transfection agent. The labeled cells retained their differentiation capacities for the osteogenesis and adipogenesis pathways but in some cases chondrogenesis was inhibited (4,12,32).
In the present study, the labeling medium consisted of nonaggregated nanoparticles, cell exposure was limited to a short incubation period (30 min) without a transfection agent, and the nanoparticles remained in intracellular vesicles after internalization. The chase period in a nanoparticle-free medium ensured that all the nanoparticles initially adsorbed on the cell membrane could be internalized. By the end of this process, the cell membrane is completely cleared of nanoparticles (65). Besides, it is demonstrated here that the endothelial cells confine the magnetic nanoparticles in endosomes, that no nanoparticles are exocytosed, that each cell share its magnetic content equally between two daughter cells during cell division, and that the magnetic daughter cells retain the ability to form capillary networks in matrigel. In a previous study (64) conducted with the same labeling conditions, the expression of endothelial markers [cluster of differentiation 62 E (CD62E), kinase inert domain receptor (KDR), tunica interna endothelial cell kinase (TIE2), CD31, CD144] was shown to remain unchanged after labeling, which supports the safety of the magnetic labeling.
One should also consider the potential effects on a whole organism of the implantation of such a graft containing cells with intracellular iron oxide. Even if such studies go far beyond the scope of this work, the iron mass introduced through this mean is reasonable. Given a 10-cm vascular graft that would be magnetically patterned, 1 million cells would initially attach and the total iron mass contained in the implanted graft would be of the order of 0.01 mg. This represents 0.25 × 10−3% of the total iron mass of the body (3–4 g) and only 0.01% of the iron mass that is renewed every day (1–2 mg/day). In comparison, injection of iron oxide nanoparticles as contrast agents for MRI results in addition of 0.1 g of iron in the body (clinical dose: 20–50 μmol/kg).
Magnetic Patterning of Cells
The present work reports the creation of 3D cell patterns using magnetic force. These cell patterns are rapidly created through a transient application of the magnetic force, and they can be easily tailored upon variation of magnet geometry, which makes magnetic patterning a versatile method to easily obtain controlled cell patterns in the three dimensions (24,26).
Magnetic cell patterning was applied here to the endothelialization of vascular grafts. Avoiding thrombosis of small diameter vascular grafts indeed remains a challenge, and a continuous endothelium is known to have an antithrombotic effect in vivo (7). Peptide binding and growth factor incorporation have been used for in vivo endothelial cell recruitment (67). In vitro endothelialization is mostly performed in bioreactors and relies on cell sedimentation (43). In addition to the long culturing times required, this method offers a poor control over the cell distribution. In comparison, the radial symmetry of the magnetic force used for patterning ensures that cells distribute homogeneously over the luminal surface (XY plane and Z direction). The magnetic patterning method also offers a better control over the thickness of the endothelial layer: the grafts are seeded initially with a single to a few layers of endothelial cells (at most two or three). In vitro maturation in bioreactors or directly in vivo should be studied to validate obtaining a continuous endothelial cell monolayer, either for use as vascular grafts or in pharmacological assays.
3D Noninvasive MRI Cellular Imaging
Magnetic labeling of cells not only allows for cell manipulation but also for scaffold imaging with high-resolution MRI. Because of its noninvasive characteristics, MRI could be implemented to assess complete endothelialization of the graft prior to implantation as a quality control, or later on, to monitor the endothelium in vivo. Here we show that the use of high magnetic field (4.7 T) combined with a cryogenic probe successfully identified magnetically labeled cells in the bulk of the scaffold (labeled MSCs) or in the lumen (labeled HUVEC-Cs) with a resolution close to individual cells (Fig. 4). In the lumen, we demonstrated the continuity of the endothelium created, and inside the scaffold, we observed an excellent seeding of the MSCs in the internal pores of the construct. High-resolution single-cell MRI has recently being successfully used to detect cells in vivo with a resolution of tens of micrometers (54), including stem cells (2,18,33,58). This observation of different cell organizations in a vascular scaffold within the range of resolution of a cell monolayer opens new ways in the field of vascular tissue engineering towards generation of vascular grafts with in vivo monitoring of the integrity of the layer after implantation.
Pluricellular Organized Vascular Graft
Cell seeding of a polymer scaffold was combined with magnetic cell patterning to create a pluricellular and organized vascular graft of 2 mm in diameter. The control over the cellular organization of the graft was made possible through both the scaffold properties and the implementation of a magnetic-based patterning technique. The usually poor cell seeding efficiency of polymer-based scaffolds was here circumvented by building on a sponge effect: the polysaccharide tube was first desiccated before being hydrated with the required cell suspension, this hydration providing the necessary forces to suck up the cells in the scaffold pores. The full hydration of the tube and the tight structure at the luminal surface ensured that endothelial cells did not enter the tube bulk during magnetic patterning. In comparison to cell-based techniques [cell sheet engineering (23,25,28,36), bioprinting (42), magnetic cell seeding (45)], a long maturation period is not necessary to obtain a cohesive structure. Indeed, the strategy implemented here is based on the assembly of the different elements composing the graft: the endothelial progenitors and mesenchymal stem cells on the one hand, and the scaffold on the other. Each of these elements can be processed and prepared separately, which enables a rapid final assembly. It is also possible to finely tune the number of endothelial cells that will attach to the lumen and, thereby, the thickness of the endothelium. Moreover, each element and the final construct could be imaged using MRI to evidence the integrity of the vascular graft. This would be particularly advantageous over other methods that require a lengthy and destructive histological analysis.
Conclusion
The ability to promote a confluent endothelium is of crucial importance to achieve high vascular graft patency. For such purpose, we tested a seeding method in which a magnetic force is used to generate, within a porous polysaccharide tubular scaffold, an endothelialization approaching that of native vessels. Small diameter vascular grafts were then successfully obtained in which endothelial cells (precursors or mature) are located on the tube lumen, and smooth muscle cell precursors (mesenchymal stem cells) are located within the tube. The combination of imaging modalities (histology, fluorescence microscopy, and 3D MRI) demonstrated the integrity of the vascular construct for up to 7 days in culture.
Magnetic cellular patterning therefore opens new windows to create tissue substitutes with optimized geometries and cellular organizations, possibly by combining several cell types.
Footnotes
Acknowledgments
The authors thank A. Al Faraj, G. Autret, and O. Clément (PARCC, Hôpital Européen Georges Pompidou, Paris) for assistance in MRI experiments; Christine Ménager (Li2C, CNRS UMR 7612, Université Paris 6 Pierre et Marie Curie) for providing magnetic nanoparticles; and L. Réa (MSC, UMR 7057, CNRS and Université Paris 7 Denis Diderot) for mechanical support. We extend our appreciation to G. Uzan and M. Lavergne (Inserm, U972, Hôpital Paul Brousse, Villejuif) for providing us with EPCs. This work was supported by the French Research Funding Agency (Agence Nationale de la Recherche, ANR) in the frame of the projects 3D Cell (ANR-09-EBIO-001), NMVASC (ANR-10 INTB-1502-01), and MagStem (ANR-11 SVSE5). D. Fayol was financed by Délégation Générale pour l'Armement (DGA). J. Ino is a recipient of the French Ministry of Higher Education and Research Scholarship (ED Galilée, University Paris 13). The authors declare no conflicts of interest.
