Abstract
Bladder outlet obstruction (BOO) caused by collagen deposit is one of the most common problems in elderly males. The present study is to investigate if human mesenchymal stem cells (MSCs) are capable of inhibiting collagen deposition and improve cystometric parameters in bladder outlet obstruction in rats. Human MSCs were labeled with nanoparticles containing superparamagnetic iron oxide (SPION), and transplanted in rat BOO lesion site. Forty 6-week-old female Sprague-Dawley rats were divided into four groups (group 1: control, group 2: sham operation, group 3: BOO, and group 4: BOO rats receiving SPION-hMSCs). Two weeks after the onset of BOO, 1 × 106 SPION-hMSCs were injected into the bladder wall. Serial T2-weighted MR images were taken immediately after transplantation of SPION-labeled human MSCs and at 4 weeks posttransplantation. T2-weighted MR images showed a clear hypointense signal induced by the SPION-labeled MSCs. While the expression of collagen and TGF-β protein increased after BOO, the expression of both returned to the original levels after MSC transplantation. Expression of HGF and c-met protein also increased in the group with MSC transplantation. Maximal voiding pressure and residual urine volume increased after BOO but they recovered after MSC transplantation. Human MSCs transplanted in rat BOO models inhibited the bladder fibrosis and mediated recovery of bladder dysfunction. Transplantation of MSC-based cell therapy could be a novel therapeutic strategy against bladder fibrosis in patients with bladder outlet obstruction.
Keywords
Introduction
Bladder outlet obstruction (BOO) caused by collagen deposit is one of the most common problems in elderly males. The collagen deposition in the bladder occurs frequently during development of various pathological processes and eventually cumulates in bladder fibrosis. This bladder fibrosis adversely affects the smooth muscle function and the capacity of the bladder. Although numerous treatments have been developed, the recovery of voiding dysfunction is insufficient. Reducing the collagen deposition could improve voiding dysfunction in bladder fibrosis.
Mesenchymal stem cells (MSCs) are self-renewing cells with pluripotent capacity to differentiate into various cell types. Therapeutic potential of bone marrow-derived MSCs on experimental cardiac and liver fibrosis has previously been reported (1,19,24). Thus, MSC transplantation into the bladder may have the potential to reduce collagen deposition of the bladder. Inhibition of collagen deposition using MSCs could also lead to functional restoration. This approach may provide a new and effective avenue for the treatment of bladder fibrosis and bladder outlet obstruction.
Reliable in vivo monitoring of transplanted cells should enable a systematic investigation of cell therapy. In vivo monitoring of transplanted cells has been performed by immunohistochemistry in tissue sections and this procedure necessitates sacrifice of experimental animals. Using molecular imaging with nanoparticles, migration and location of metastatic cancer cells and transplanted MSCs can be monitored noninvasively and evaluation can be done in the same host (3,4,8,16,29). Among several molecular imaging methods, including optical imaging and magnetic resonance imaging (MRI), superparamagnetic iron oxide nanoparticles (SPION) have been used for intracellular labeling of stem cells and cancer cells and facilitate the detection of labeled cells using MRI (8,16,28,29). We have reported previously that the SPION-labeled human MSCs were detected by MRI in the rat bladder following transplantation (29).
The aim of the present study is to investigate the repair of bladder fibrosis and restoration of bladder function in obstructed rat bladder outlet as monitored by molecular MRI after transplantation of human MSCs labeled with SPION.
Materials and Methods
Preparation of Human Mesenchymal Stem Cells (MSCs)
Human MSCs were isolated and culture expanded according to the method described previously (22). Briefly, 10—20 ml of bone marrow aspirate was obtained under sterile conditions by puncture of the posterior iliac crest of bone marrow transplantation donors after receiving informed consent. Mononuclear cells were isolated from the bone marrow using Ficoll-Hypaque (Sigma, St. Louis, MO) density centrifugation (400 x g for 25 min). The interface mononuclear cells were collected and washed twice with phosphate-buffered saline (PBS). The cells were resuspended, counted, and plated at 2 × 105/cm2. The cells were cultured in hMSC medium composed of Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum, 100 U/ml penicillin, and 100 μg/ml streptomycin. Cells were plated into T75 flasks and the cultures were incubated at 37°C in 5% CO2 in air and 95% relative humidity. The medium was changed every 3—4 days. When the cultures reached approximately 90% of confluence, MSCs were detached with 0.05% trypsin-EDTA solution (Gibco-BRL) and replated into passage culture at a density of 1 × 106 cells in T75 flask. The MSCs were confirmed to be negative for hematopoietic markers by flow cytometry and capable of differentiating into osteocytes, chondrocytes, and adipocytes in vitro (data not shown).
Labeling of hMSCs with Iron Oxide Nanoparticles
In vitro labeling of MSCs with superparamagnetic iron oxide nanoparticles (SPION) was performed using GenePORTER (GTS, San Diego, CA) as the transfection agent. Cultures were incubated in medium containing 1:250 GenePORTER and 25 μg/ml SPION (Feridex; AMI, Cambridge, MA) for 60 min at room temperature (RT) on a rotating shaker, and then were kept for 4 h in a 5% CO2 incubator at 37°C. Labeled MSCs were harvested for transplantation by gentle trypsinization. To remove excess iron oxide nanoparticles, trypsinized cells were washed in PBS and concentrated by centrifugation, resuspended in PBS, and kept on ice. The labeling efficiency of the SPION-labeled hMSCs was examined with Prussian blue staining. The labeling viability was done with trypan blue staining. For Prussian blue staining, which indicates the presence of iron, the cells were fixed with methyl alcohol, washed, incubated for 30 min with 2% potassium ferrocyanide in 6% hydrochloric acid, washed again, and counterstained with nuclear fast red.
In Vivo MRI
MR examinations were performed after transplantation of SPION-labeled MSCs. MR image more than 1 × 106 of SPION-labeled MSCs was taken on the back muscle of rats. MR image up to 48 days of SPION-labeled MSCs on the back muscle of rats was taken.
Transplantation of Human MSCs Into the Bladder of Rats
All procedures were conducted in accordance with the National Institute of Health guidelines for the care and use of laboratory animals and were approved by the Institutional Animal Care and Use Committee of the hospital. Forty 6-week-old female Sprague-Dawley rats (200 g, n = 40) were used in this study (group 1: control, n = 10; group 2: sham operation, n = 10; group 3: BOO, n = 10; and group 4: BOO rats receiving SPION-MSCs, n = 10). Rats were anesthetized intraperitoneally with 1% ketamine (30 mg/kg) and xylazine hydrochloride (4 mg/kg) and BOO operation was conducted.
The lower abdominal skin incision was made and the urethra was dissected and 4-0 silk sutures were placed around the urethra, including a length of metal rod with an outer diameter of 1 mm, which was positioned extraluminally. After the suture was tied the rod was removed. The abdominal wall was then closed after the removal of the rod. At 2 weeks after the onset of BOO, saline was injected into the rat bladder using a 500-μl syringe with 26-gauge needle and the incised abdominal skin was sutured in group 3. SPION-labeled MSCs (1 × 106 cells) were transplanted into the rat bladder using a 500-μl syringe with a 26-gauge needle and the incised abdominal skin was sutured in group 4. Injection of Flomoxef (10 mg/kg, cephalosporin; Ildong, Seoul, Korea) was administered IP daily to prevent infection. The bladder was harvested 4 weeks after the cell transplantation.
MRI of Transplantation of SPION-Labeled hMSCs Into the Rat Bladder
MR examinations were performed after transplantation of SPION-labeled hMSCs. Follow-up serial MRI was performed up to 4 weeks.
Histology
At the end of the in vivo MRI experiments, each animal was anesthetized and perfused through the heart with 100 ml cold saline and 100 ml of 4% paraformaldehyde in PBS. After 24 h of fixation in 4% paraformaldehyde, the bladder was cryprotected in 30% sucrose for 24 h, cut into 20-μm sections in a cryostat (Leica CM 1900), and stained with H&E, Prussian blue, and Masson's trichrome. Each slide was inspected microscopically and 10 randomly chosen representative areas from light microscope images were captured (200x magnification). Captured video images were then displayed on a color monitor and simultaneously digitized and analyzed using an IBM computer. To evaluate the results of Masson's trichrome staining representative portions of each slide were calculated in blinded fashion with a square micrometer and the mean area was expressed as the relative percent. The mean percent collagen area was defined according to the formula: (collagen)/(collagen + muscle). This technique was predicated on the area calculation of the smooth muscle, which stained red, and connective tissue, which stained blue. Quantitative image analysis was done using OPTIMAS, version 6.1 (Media-Cybernetics).
For immunofluorescence microscopy, adjacent serial sections were processed for fluorescent staining with human nuclear matrix antigen (hNuMA, 1:100, mouse monoclonal, Oncogene) to identify the transplanted human MSCs.
Western Blot Analysis
Bladder tissues were homogenized in radioimmunoprecipitation assay lysis buffer on ice. After 20 min of centrifugation at 10,000 x g at 4°C the supernatants were collected. The tissue homogenates were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis with 15% polyacrylamide for transforming growth factor-β (TGF-β) and 8% for all other proteins, followed by transfer to polyvinylidene difluoride membranes. The membranes were blocked for 1 h with 5% nonfat dry milk and PBS containing 0.05% Tween-20 and then incubated overnight with primary antibody at 4°C. The primary antibodies used were anti-rat collagen I (1:1000, rabbit polyclonal antibody NB600-408, Novus Biologicals, Littleton, CO), anti-rat TGF-β (1: 500, rabbit polyclonal antibody SC-146, Santa Cruz Biotech, Santa Cruz, CA), anti-rat hepatocyte growth factor (HGF; 1:500, goat polyclonal antibody SC-1357, Santa Cruz), and anti-rat c-met (1:250, rabbit polyclonal antibody SC-162, Santa Cruz). The blots were washed in PBS containing 0.05% Tween-20 and incubated for 1 h in horseradish peroxidase-conjugated secondary anti-rabbit or anti-goat antibodies at room temperature, after which detection was done by enhanced chemiluminescence (GE Healthcare, Piscataway, NJ).
Quantitative analysis of several animals was provided by scanning of the blots and calculating the relative intensities of collagen I, HGF, c-met, and TGF-β in relation to the corresponding actin signal.
Recovery of Voiding Function After Transplantation
Voiding response was assessed at 4 weeks after transplantation. Female Spraue-Dawley rats were anesthetized with isoflurane (2% oxygen) for surgical insertion of bladder catheter. The bladder was exposed via a midline abdominal incision. A catheter (PE-50), the bladder end of which was heated to create a collar, was inserted through a small incision in the bladder dome, and a suture was tightened around the collar. The other end of the catheter was passed through subcutaneous tissue and exited through the skin. After closing the abdominal incision by suturing the muscle and skin, rats that were to be studied without anesthesia were placed in a restraining cage for 5—6 h (including 2 h of recovery from isoflurane anesthesia) that was large enough to permit them to adopt a normal crouching posture but narrow enough to prevent them from turning around. The rats were subsequently allowed to recover from isoflurane anesthesia. The bladder catheter was connected via a T-stopcock to a pump for continuous infusion of physiological saline and to a pressure transducer. Physiological saline was infused at room temperature into the bladder at a constant rate of 0.04 ml/min to elicit repeated voiding responses. The volume evoked micturition reflexes were studied in urethane-anesthetized restrained rats. The intravesical pressure to induce micturition, pressure threshold (PT), maximal voiding pressure (MVP), and intercontraction interval (ICI; the interval between voids or reflex bladder contractions) were measured.
Statistical Analysis
Two-way ANOVA and the post hoc Tukey test were used for analysis of stem cell transplantation. Data are presented as means ± SE. A value of p < 0.05 was considered statistically significant.
Results
Cell Labeling and Viability
Prussian blue staining of SPION-labeled MSCs revealed abundant uptake of the SPION-GenePORTER complex in the MSC cell cytoplasm. However, no stainable iron was detected in the nonlabeled control MSCs (Fig. 1). SPION-labeled MSCs were cultured for 6 days and the viability of the labeled cells measured by using trypan blue staining. The viabilities of MSCs with and without SPION labeling were 98% and 95%, respectively.

Prussian blue staining of human mesenchymal stem cells (MSCs) with or without nanoparticles containing superparamagnetic iron oxide (SPION). (A) Control human MSCs without SPION labeling. (B) Human MSCs labeled with SPION. Note the abundant iron particles in the cytoplasm of the cells (arrows). Scale bar: 20 μm.
In Vivo MRI
In vivo MRI according to the concentration of SPION-labeled MSCs showed a clear hypointense signal at all concentrations greater than 1 × 106 MSCs/ml on the back muscle of rats. In vivo MRI of SPION-labeled MSCs on the back muscle of rats showed clear hypointense signal intensities after transplantation. The distribution of the hypointense signal intensity was located in the bladder. On follow-up serial MRI, hypointense signal intensity faded but persisted until 48 days posttransplantation (Figs. 2 and 3).
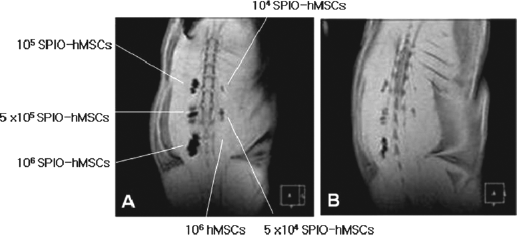
In vivo MRI of SPION-labeled human MSCs on the back muscle and bladder of rats. MRI showed a clear hypointense signal intensity until 48 days after transplantation. (A) Immediately after transplantation of SPION-labeled human MSCs, MRI showed a clear hypointense signal at all concentrations greater than 1 × 106/ml SPION-labeled human MSCs on the back muscle of rats. (B) The hypointense signal intensity faded but persisted until 48 days after transplantation.

In vivo MRI of SPION-labeled human MSCs into the bladder of BOO model of rats. MRI showed a clear hypointense signal intensity and the areas of decreased MR signal intensity in the bladder were confined locally (arrows). (A) Before transplantation of SPION labeled human MSCs and (B) immediately after transplantation of SPION labeled human MSCs. (C) At 4 weeks after transplantation of SPION-labeled human MSCs. (D) Schematic drawing of bladder, urethra, and transplanted MSCs. B, bladder wall; M, injected MSCs; U, urethra.
Body and Bladder Weight
The mean of body weight (g) in groups 1—4 was 238 ± 1 (control), 228 ± 1 (sham), 223 ± 2 (BOO), and 241 ± 2 (BOO + MSCs), respectively. There was no significant difference of body weight between groups. The mean of bladder weight (mg) was 89.2 ± 1.9, 73.3 ± 5.8, 630.5 ± 156.5, and 445.0 ± 5.0 in groups 1—4, respectively. The group with BOO showed higher bladder weight than the groups of sham operation and controls. The MSC transplantation group showed lower bladder weight compared with the BOO group without MSC transplantation (Fig. 4).
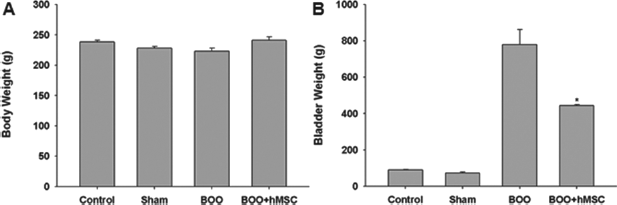
Changes in weight of body and bladder after human MSC transplantation. There was no significant difference of body weight between groups. The group with BOO showed increased bladder weight compared with the sham operation group. The group with human MSC transplantation showed decreased bladder weight compared with the BOO group (*p < 0.05).
Histology
The presence of SPION was found at 4 weeks after transplantation of SPION-labeled MSCs into the bladder with Prussian blue. MSCs positive for human nuclear antigen were found at the transplantation site indicating that SPION-MSCs survived in the bladder at 4 weeks posttransplantation (Fig. 5).

Histologic findings of SPION-labeled human MSCs in rat bladder. Prussian blue staining at 4 weeks after MSC transplantation. (A—C) Intracytoplasmic SPION particles (blue dots) are clearly visible. Scale bars: 20 μm (A), 40 μm (B), and 80 μm (C). (D) The presence of human MSCs in rat bladder at 4 weeks after transplantation was confirmed by immunostaining with anti-human nuclear matrix antibody that is specific for human cells. Scale bar: 50 μm (D). U, urothelium of bladder; I, intraluminal side of bladder; E, extraluminal side of bladder.
Change of Collagen Deposition After Transplantation
The ratio of collagen/collagen + muscle (%) was 11.4 ± 0.4 (control), 13.0 ± 0.6 (sham), 27.1 ± 0.8 (BOO), and 21.2 ± 0.7 (BOO + MSCs) in groups 1—4, respectively. The group with BOO showed higher ratio than the group with sham operation (p < 0.05). The group with transplantation of MSCs after BOO showed lower ratio than the group with BOO (p < 0.05: Sham vs. BOO, BOO vs. BOO + MSCs, p < 0.001: Sham vs. BOO) (Fig. 6). The collagen deposition was higher in the group with BOO and was lower after transplantation of MSCs as shown by Masson's trichrome staining (Fig. 7).

Change of collagen deposition after human MSC transplantation. The group with BOO showed increased percent collagen area and it decreased after transplantation of MSCs. Sham, sham operation; BOO, bladder outlet obstruction; BOO + MSCs, human MSC injection at 2 weeks after bladder outlet obstruction (*p < 0.05: Sham vs. BOO, BOO vs. BOO + MSCs, **p < 0.001: Sham vs. BOO).
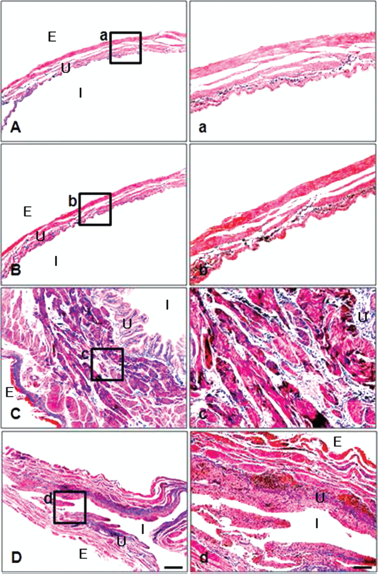
Histologic change of collagen deposition after human MSC transplantation. The collagen deposition increased in the group with BOO and decreased after transplantation of human mesenchymal stem cells. (A—D) Masson's trichrome staining. (A) Control, (B) Sham, (C) BOO, (D) BOO + SPION-labeled human MSCs. Scale bar: 20 μm. (a—d) Masson's trichrome staining. (a) Control, (b) Sham, (c) BOO, (d) BOO + SPION-labeled human MSCs. Scale bar: 20 μm. Sham, sham operation; BOO, bladder outlet obstruction; BOO + SPION-labeled MSCs, human MSCs injected at 2 weeks after bladder outlet obstruction.
Western Blot Analysis of Collagen, HGF, TGF-β, and c-met
The group with BOO showed higher expression of collagen and TGF-β than the control group and sham-operated group. The group with SPION-MSC transplantation after BOO showed lower amount of collagen and TGF-β than the BOO group without cell transplantation. The group with BOO-SPION-MSC transplantation showed higher levels of HGF and c-met than the groups with control, sham, or BOO alone (Fig. 8). Semiquantitative analysis of collagen, HGF, c-met, and TGF-β in membrane fraction from the bladders of sham, BOO, and BOO-MSCs indicated that the levels of collagen, HGF, and c-met were significantly increased in BOO-MSC group (p < 0.05: BOO vs. BOO + MSCs) (Fig. 8).

Western blot analyses of collagen I, transforming growth factor-β (TGF-β), hepatocyte growth factor (HGF), and c-met in bladder. (A) The group with BOO showed increased expression of collagen and TGF-β compared with the group with sham operation. The group with BOO-human MSC transplantation showed decreased levels of collagen and TGF-β compared with the group with BOO alone. The group with BOO-human MSC transplantation showed increased levels of HGF and c-met compared with the group with control, sham, or BOO. (B) Semiquantitative analysis of level of collagen, HGF, c-met, and TGF-β in membrane fraction from the bladder of sham, BOO, and BOO-MSCs. Levels of collagen, HGF, and c-met were significantly increased in BOO-MSCs. *p < 0.05: BOO versus BOO + MSCs.
Improvement of Voiding Function After Transplantation
Intercontraction interval (ICI, s) was 337 ± 26 (control), 346 ± 54 (sham), 300 ± 35 (BOO), and 485 ± 107 (BOO + SPION-MSCs) in groups 1—4. ICI was lower after BOO and improved after transplantation of MSCs (p < 0.05). Pressure threshold (PT, cm H2O) was 8.1 ± 0.3, 9.6 ± 1.2, 8.1 ± 0.7, and 7.6 ± 0.4 in groups 1—4. PT had no change among groups. Maximal voiding pressure (MVP, cm H2O) was 78.8 ± 14.9, 51.0 ± 15.6, 40.6 ± 3.4, and 43.6 ± 2.0 in groups 1—4. Residual urine volume (RU, ml) was 0.51 ± 0.23, 0.37 ± 0.06, 0.05 ± 0.03, and 0.03 ± 0.03, in groups 1—4. ICI was lower after BOO and improved after transplantation of SPION-MSCs (p < 0.05). MVP and RU were higher after BOO and improved after transplantation of SPION-MSCs (Fig. 9).

Recovery of cystometric parameters after transplantation of human MSCs. Sham, sham operation; BOO, bladder outlet obstruction; BOO + human MSCs, human MSCs injected at 2 weeks after bladder outlet obstruction; ICI, intercontraction interval; MVP, maximal voiding pressure; PT, pressure threshold; RU, residual urine volume. *p < 0.05.
Discussion
MSCs are self-renewing adult stem cells with various differentiation potential (22). The relative ease of isolating MSCs from bone marrow and the great plasticity of the cells make them ideal tools for the stem cell-based therapy. Recent studies have shown that in vivo human MSCs transplanted into fetal sheep are able to differentiate into cells of various tissues (14). Transplanted human MSCs or neural crest stem cells showed differentiation into endothelial or smooth muscle cells in vivo following engraftment in corpus cavernosum (30,31). MSCs can prevent deleterious remodeling and improve recovery after myocardial infarction (20,23). Thus, MSCs fulfill all criteria of in vivo cellular source for tissue reconstitution.
The potential mechanism proposed for the therapeutic effects by MSCs is their being a source for cell replacement (25). Although stem cells were present at lower frequency in damaged liver or lung tissues, MSCs are known for reducing fibrosis in liver or lung (6,21). MSCs could secrete growth factors, and contribute to reduce fibrosis through paracrine mechanisms rather than by cell incorporation (11,27). The elevation in the recipient's blood HGF after transplantation of MSCs and promoting remnant liver regeneration at the initial stage was reported (10,15). MSCs may protect against injury by altering the microenvironment of injury site at sites of engraftment. In a previous study, we have shown that the human MSCs secrete many growth factors including nerve growth factor (NGF), brain-derived growth factor (BDNF), neurotrophin-3 (NT3), insulin-like growth factor (IGF), HGF, glial-derived growth factor (GDNF), ciliary neurotrophic factor (CNTF), and vascular endothelial factor (VEGF) (18).
In the present study of bladder sections transplanted with MSCs, the validation of MSCs was made by use of Prussian blue staining and anti-human nuclear antibody. The ratio of collagen/collagen + muscle was lower in rats transplanted with MSCs in bladder outlet obstruction model compared with animals without transplantation. The lower ratio resulted from inhibition of collagen deposition following MSC transplantation.
In a previous study, liver cirrhosis in rats was treated by transplantation of hepatocyte growth factor (HGF)-expressing MSCs (32). Expression of HGF protein increased in liver after MSC transplantation and the expression of c-met, HGF receptor, also increased. In the present study, expression of collagen protein increased markedly after BOO operation and recovered to normal level after transplantation of MSCs. Expression of TGF-β protein also increased after BOO and recovered after transplantation of MSCs. The complex of HGF and c-met activates tyrosine kinase (9) and HGF suppresses expression of TGF-β1 and its type I receptor TGFR-1 (17). It is suggested that HGF released from MSCs may be mediated by the specific targeting and blocking of TGF-β1 expression and signaling (9).
In the present study, ICI was lower after BOO and improved after MSC transplantation. MVP and RU were higher after BOO and improved after MSC treatment. It is suggested that the inhibition of bladder fibrosis improved cystometric parameters in bladder outlet obstruction in rats.
For the investigation of the implantation, survival, and functioning of cells, the animals should be sacrificed, which does not allow an opportunity to investigate transplanted cells in vivo in the same host in continuation. In contrast, a reliable in vivo imaging method to localize transplanted cells and monitor their restorative effects should enable a systematic investigation of cell therapy. Molecular imaging aims to visualize targeted cells in living organisms. Such cell trafficking studies would be a valuable tool for development and evaluation of cell-based treatment strategies. In order to identify migration of labeled cells by MR imaging, the contrast produced by the label must be sufficient enough to detect small clusters of cells and simultaneously must not be toxic to the cells or to the host. Cells labeled with superparamagnetic nanoparticles (SPION) that contains iron oxide particles exhibit much higher stability in vivo and stronger contrast (3,13).
Iron oxide-labeled cells appear as hypointense areas in tissues with an associated decreased signal intensity on iron-sensitive T2-weighted and T2-weighted gradient echo images. Iron oxide particles are an FDA-approved MR contrast agent, and therefore, quality control, sterility, and stability have all been well documented. SPION particles that are used for cellular labeling have not demonstrated any adverse effects in cell viability and differentiation of MSCs (2,3,12).
In this study, we confirmed the ability of in vivo MRI for detection of labeled stem cells in the bladder of rats with BOO. The validation of SPION-labeled MSCs was made using Prussian blue staining and anti-human nuclear antibody in rat bladder sections. Transplanted SPION-labeled MSCs were observed in vivo until 4 weeks posttransplantation. Serial imaging of the hypointense MRI due to the presence of SPION-labeled MSCs decreased over time. This could possibly be due to dilution of iron after cell division or metabolism of iron oxide particles. In summary, SPION-labeled MSCs transplanted into rat bladder survived 4 weeks posttransplantation and their movement monitored using MRI. Transplanted MSCs inhibited the bladder fibrosis and mediated improvement of bladder dysfunction in the rat BOO model.
Bladder outlet obstruction secondary to benign prostatic hyperplasia is one of the most frequently noted urological problems in elderly male patients. Current approaches to BOO treatment have classically focused primarily on reducing prostate size and decreasing smooth muscle cell tone. However, abnormal accumulations of extracellular matrix are frequently observed during the development of pathological processes. This fibrosis can adversely affect smooth muscle functioning as well as the capacity of the bladder to empty properly. With time, the obstructed bladder deteriorates due to alterations in its physiology and structure. Prolonged BOO progresses from a compensated state, where emptying remains normal, to a decompensated state by reduced flow, interrupted urination, and finally a postvoid residual volume.
Mechanisms responsible for the bladder dysfunction induced by BOO remain poorly understood. Bladder dysfunction after BOO are related to a primary neurologic event but mediated by alterations in ultrastructural properties of the smooth muscle and collagen deposition in obstructed bladders have been well documented. Compensated bladder dysfunction with overactive bladder is expected after 6 weeks BOO and the restoration of bladder dysfunction is expected by the inhibition of collagen deposition using a 4-week period of transplantation of MSCs in rat model of obstructed bladder outlet (5).
MSC recruitment to the bladder after BOO appears to be associated with increased blood flow and decreased tissue hypoxia, which may contribute to improvement in histopathological and functional parameters (33). In contrast to local injection of MSCs, intravenously administered MSCs are distributed throughout the whole animal. Furthermore, there is concern of causing capillary clogging when larger cell types such as MSCs are infused, a complication that could result in hemodynamic compromise, interference with pulmonary gas exchange, and respiratory distress (7). Intravenously injected MSCs are localized mainly to the pulmonary capillary bed (25). Local injection may have a better effect than intravenous injection.
In conclusion, the present study showed that transplantation of SPION-MSCs in a rat BOO model resulted in improvement of bladder fibrosis and improvement of bladder dysfunction in rat model of obstructed bladder outlet as monitored by the molecular MRI. Results of the present study indicate a future direction that patients suffering from bladder fibrosis could be treated by transplantation of autologous MSCs and attain recovery of bladder dysfunction.
Footnotes
Acknowledgments
This research was supported by grant from the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (KRF-2008-313-E00407). The following author contributionsare acknowledged: Conceived and designed the experiments (H.J.L., Y.S.S., S.U.K.), performed the experiments (H.J.L., J.H.W., S.H.D., J.H.K., K.I.S., S.J.L.), analyzed the data (H.J.L., J.H.W., I.J.L., K.T.C), wrote the paper (H.J.L., J.H.W., Y.S.S., S.U.K.). The authors declare no conflicts of interest.
