Abstract
Stem cell therapy is a potential treatment for spinal cord injury (SCI), and a variety of different stem cell types have been grafted into humans suffering from spinal cord trauma or into animal models of spinal injury. Although several studies have reported functional motor improvement after transplantation of stem cells into injured spinal cord, the benefit of these cells for treating SCI-induced neuropathic pain is not clear. In this study, we investigated the therapeutic effect of transplanting human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs) or amniotic epithelial stem cells (hAESCs) on SCI-induced mechanical allodynia (MA) and thermal hyperalgesia (TH) in T13 spinal cord hemisected rats. Two weeks after SCI, hUCB-MSCs or hAESCs were transplanted around the spinal cord lesion site, and behavioral tests were performed to evaluate changes in SCI-induced MA and TH. Immunohistochemical and Western blot analyses were also performed to evaluate possible therapeutic effects on SCI-induced inflammation and the nociceptive-related phosphorylation of the NMDA NR1 receptor subunit. While transplantation of hUCB-MSCs showed a tendency to reduce MA, transplantation of hAESCs significantly reduced MA. Neither hUCB-MSC nor hAESC transplantation had any effect on SCI-induced TH. Transplantation of hAESCs also significantly reduced the SCI-induced increase in NMDA receptor NR1 subunit phosphorylation (pNR1) expression in the spinal cord. Both hUCB-MSCs and hAESCs reduced the SCI-induced increase in spinal cord expression of the microglial marker, F4/80, but not the increased expression of GFAP or iNOS. Taken together, these findings demonstrate that the transplantation of hAESCs into the injured spinal cord can suppress mechanical allodynia, and this effect seems to be closely associated with the modulation of spinal cord microglia activity and NR1 phosphorylation.
Keywords
Introduction
Spinal cord injury (SCI), resulting from direct traumatic damage to the spinal cord, has been associated with a number of clinical complications that include functional disability, autonomic dysreflexia, urinary tract problems, altered sensations, and both acute and chronic pain (28,42,55). Central chronic pain syndrome, also known as below-level central neuropathic pain, develops in a majority of patients following SCI, and the pain and dysesthesias associated with this syndrome severely compromise the quality of life in nearly 70% of these patients. Often these patients experience (1) persistent or spontaneous pain, which occurs independent of peripheral stimuli, and (2) stimulus-evoked pain, which occurs in response to either a normally nonnoxious (allodynia) or noxious (hyperalgesia) stimuli and is one of the most common reasons for a reduced quality of life (9,57,59).
Recent efforts to improve treatment for SCI-induced disabilities have included stem cell transplantation, drug therapy, and cytokine therapy using a variety of model systems (1,44,53). However, SCI remains one of the most devastating types of trauma for patients due to the resulting long-lasting disability and the limited response to acute drug administration and efforts at rehabilitation. Although many studies have shown statistically significant functional motor recovery following stem cell implantation (5,45), it is not clear whether stem cell transplantation can influence the development or severity of SCI-induced neuropathic pain.
Two studies have demonstrated that the transplantation of embryonic neural stem cells can actually produce or potentiate thermal hyperalgesia (TH) and mechanical allodynia (MA) in SCI rats with a contusion at the T8 spinal segment (26,34). The authors of these studies suggested that the modification of embryonic stem cells by transfection with glial-derived neurotrophic factor or transduction with neurogenin-2 could modulate these neuropathic pain behaviors. On the other hand, other researchers have reported that serotonergic neural precursor cell grafts attenuate the hyperexcitability of dorsal horn neurons observed following spinal hemisection (22). In addition, predifferentiated embryonic stem cells have been shown to relieve chronic pain behaviors and restore sensory function following chemical-induced spinal cord injury in mice (24). Recently transplantation of bone marrow stromal cells (BMSCs) has also shown analgesic efficacy in diabetic or peripheral neuropathic pain rats (18,41). These findings indicate that the specific effect of stem cell transplantation on SCI-induced pain is dependent on the type and source of stem cells used and on the type of chronic pain being treated.
Among the multiple types of stem cells used for trans plantation, human umbilical cord blood-derived cells (hUCB-MSCs) and amniotic epithelial stem cells (hAESCs) are multipotent adult progenitor cells that can differentiate into various cell types, including osteoblasts, adipocytes, chondrocytes, fibroblasts, and endothelial cells (18,30). These stem cells have been recognized as potential candidates for cell-based therapy because they are easy to isolate and expand in vitro. Moreover, these cells have a major advantage over embryonic or fetal stem cells, because while they are still immature cells with wide differentiation potential, they do not carry the same ethical barriers as embryonic or fetal cells and thus can be used for both basic studies and clinical applications (30). While spinal cord transplantation of hUCB-MSCs has been used to examine their effect on functional motor recovery following spinal cord injury (47), the effect of transplantation of hUCB-MSCs or hAESCs on central neuropathic pain has not been examined.
The present study was designed to determine if the spinal cord transplantation of hUCB-MSCs or hAESCs will reduce SCI-induced MA and TH in a rodent model of spinal cord injury. In addition, the potential effects of these stem cells on the SCI-induced increases in N-methyl-d-aspartate (NMDA) receptor NR1 phosphorylation (pNR1, a chronic pain-related factor), induced nitric oxide synthase (iNOS) and cyclooxygenase 2 (COX2) (inflammatory mediators), glial fibrillary acidic protein (GFAP; a marker of astrocytes), and epidermal growth factor-like module-containing mucin-like, hormone receptor-like 1 (EMR1; F4/80) (a marker of microglia) expression were evaluated at the T13 site of injury as well as at the L4-5 spinal segments located below the injury site.
Materials and Methods
Animals
Experiments were performed on 30 male Sprague– Dawley rats weighing 180–200 g. All experimental animals were obtained from the Laboratory Animal Center of Seoul National University. They were housed in colony cages with free access to food and water and maintained in temperature- and light-controlled rooms (24 ± 2°C, 12/12 h light/dark cycle with lights on at 07:00) for at least 1 week prior to the study. All of the methods used in the present study were reviewed and approved by the Animal Care and Use Committee at Seoul National University and conformed to US National Institutes of Health guidelines. Every effort was made to minimize animal distress and discomfort and to reduce the number of animals used in the present study.
Spinal Cord Hemisection Surgery
A number of animal models of SCI exist and have been primarily used to determine mechanisms of motor dysfunctions (54,61). Recently, these various SCI animal models have been utilized for pain studies (40), and in particular the partial spinal transection injury model (hemisection) has become popular in neuropathic pain studies (20,32). Thus, this model was used in the present study. A spinal cord hemisection was performed according to the method described by Christensen et al. (9). Briefly, rats were anesthetized with 3% isoflurane (Hana Pharm., Seoul, Korea) in a mixture of N2/O2 gas, and the T11–12 vertebrae were identified by counting spinous processes from the sacrum. A laminectomy was performed between the T11–12 vertebral segments, and the lumbar enlargement was identified with the accompanying dorsal blood vessel. The spinal cord was then hemisected just cranial to the L1 dorsal root entry zone with a No. 15 scalpel blade (Feather Safety Razor Co., Osaka, Japan) without damaging the major dorsal vessel or its vascular branches. The musculature and the fascia were then sutured (Won Ind., Siheung, Korea), and the skin was apposed. Sterile procedures were used throughout the surgery. Sham surgery consisted of exposing the spinal cord in the same manner, but without hemisection (n = 6). During recovery, animals were separately housed in a clear plastic cage with a thick layer of sawdust bedding and were monitored for locomotor recovery.
Isolation and Culture of hUCB-MSCs and hAESCs
Isolation and culture of hUCB-MSCs were performed as described previously (46,58). Human umbilical cord blood samples were obtained from the Seoul City Borame Hospital Cord Blood Bank. Samples from term and preterm deliveries were harvested at the time of birth with the mother's informed consent. This work was approved by the Borame Hospital Institutional Review Board and Seoul National University (IRB No. 0603/001-002-07C1). The blood samples were processed within 24 h of collection. The mononuclear cells were separated from the UCB using Ficoll-Paque TM PLUS (Amersham Bioscience, Uppsala, Sweden) and were suspended in a Dulbecco's modified Eagle's medium (DMEM; Gibco, Grand Island, NY, USA), containing 20% fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 100 I/ml penicillin (Gibco), 100 mg/ml streptomycin (Gibco), 2 mM l-glutamine (Invitrogen, Carlsbad, CA, USA), and 1 mM sodium pyruvate (Invitrogen).
Isolation and culture of hAESCs were performed as described previously (37,38). Human amniotic tissue was obtained from Kangpuk Samsung Hospital (Seoul, Korea) with informed consent, and isolation and culture were performed with the approval of the Seoul National University Institutional Review Board (IRB No. 0611/001-002). The amniotic tissue was washed several times with phosphate-buffered saline (PBS; EMD Millipore Corp., Billerica, MA, USA) to remove blood and incubated with 0.05% trypsin-EDTA (Invitrogen) for 1 h. The amniotic epithelial cells were collected and suspended in standard culture media, consisting of K-SFM (keratinocyte-serum-free media) supplemented by 0.031 μg/ml human recombinant epidermal growth factor (EGF), 12.4 mg/ml bovine pituitary extract (all from Invitrogen), and 10% FBS.
hUCB-MSC and AESC Transplantation
Two weeks after SCI surgery (Fig. 1A), rats were randomly assigned to groups receiving PBS (n = 8), hUCB-MSCs (n = 8), or hAESCs (n = 8). Following a laminectomy at the same site as the injury, four injections of either PBS, hUCB-MSCs, or hAESCs were made in the following four locations: (1) 1 mm cranial to the lesion site, (2) 1 mm caudal to the lesion site, and (3) two injections were made 1 mm lateral to the lesion site in the contralateral spinal cord (these two contralateral injections were separated by a small distance from each other; see Fig. 1B).

The experimental design and timeline, the spinal cord location of stem cell injections, and the morphological appearance of the cultured stem cells. (A) The experimental design and time point of stem cell injection [day 14 after spinal cord injury (SCI) surgery] are depicted in this schematic drawing. (B) This schematic drawing depicts the approximate spinal cord location of the stem cell injection sites with respect to the T13 hemisection area. (C, D) Representative pictures of human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs) and human amniotic epithelial stem cells (hAESCs), respectively, in culture. Scale bar: 50 μm.
At each site, 2.5 μl of hUCB-MSCs, hAESCs, or PBS was infused for 60 s. Thus, a total of 1×106 hUCB-MSCs or AESCs (10 μl) were delivered to the spinal cord. Following the injection, the syringe was held in place for an additional 10 s before removal to prevent outflow of the injected cells. To allow tracking of the injected hUCB-MSCs and hAESCs, the cells were labeled with a membrane-bound fluorescent marker PKH26 (Sigma-Aldrich, St. Louis, MO, USA) prior to injection.
Behavioral Assessments
Following spinal cord hemisection, rats were examined for motor function in an open-field test space using the Basso, Beattie, and Bresnahan (BBB) locomotor rating scale (6). Briefly, the BBB scale ranges from 0 (no hindlimb movement) to 21 (normal movement, including coordinated gait with parallel paw placement). Scores from 0 to 7 indicate the return of isolated movements in the three joints (hip, knee, and ankle). Scores from 8 to 13 indicate the return of paw placement and coordinated movements with the forelimbs. Scores from 14 to 21 show the return of toe clearance during stepping, predominant paw position, trunk stability, and tail position. Only the scores of the ipsilateral hind limb on the hemisected side were recorded, because there were no observable differences in locomotor function of contralateral hind limb throughout the experimentation period.
Pain behavioral tests were performed 1 day before hemisection surgery on all animals to obtain normal baseline values of withdrawal response to mechanical and heat stimuli. Thirteen days after surgery (D+13), rats were again tested and the development of MA and TH were confirmed for each rat prior to randomly assigning each rat to one of the cell treatment groups (PBS, hUCB-MSCs, or hAESCs). All behavioral tests were performed blindly.
MA can be measured in various ways using von Frey filaments. In the present study, the number of paw withdrawal responses to 10 applications of a normally innocuous mechanical stimulus produced by a von Frey filament with a force of 2.0 g (North Coast Medical, Morgan Hill, CA, USA) was measured. Rats were placed on a metal mesh grid under a plastic chamber (Yeong Seong Bio Tech., Seoul, Korea), and the von Frey filament was applied from underneath the metal mesh flooring to each hind paw. The von Frey filament was applied 10 times to each hind paw, and the number of paw withdrawal responses out of 10 was then counted. The results of this mechanical behavioral testing for both the ipsilateral and contralateral hind limbs were expressed as a “percent withdrawal response frequency” (PWF, %), which represented the percentage of paw withdrawals (calculated from the number times the rat withdrew the paw in response to application of the von Frey filament out of the maximum of 10 applications) as previously described (51,52).
To assess nociceptive responses to heat stimuli (i.e., TH), we measured paw withdrawal response latency (PWL) to a heat stimulus using the plantar paw-flick latency test as previously described by Hargreaves et al. (23). Briefly, rats were placed in a plastic chamber with a glass floor (IITC Life Science Inc., Woodland Hills, CA, USA) and were allowed to acclimate for 10 min before testing. A radiant heat source was positioned under the glass floor beneath the hind paw to be tested, and withdrawal latency was measured using a plantar analgesia meter (IITC Life Science Inc.). The test was performed on both the ipsilateral and contralateral hind paws of each animal. Cutoff time in the absence of a response was set at 20 s to prevent any potential skin damage from the radiant heat source.
Immunohistochemistry
Protein phosphorylation occurs on the NMDA receptor subunits, which represents an imperative mechanism for the posttranslational modulation of the properties of the receptor. Overall, NMDA receptor phosphorylation studies suggest that phosphorylation of spinal cord NR1 subunit leads to enhanced NMDA receptor function and central sensitization. Since neuropathic pain is associated with increased phosphorylation of the NR1 subunit in the spinal cord (48,49), we wanted to examine the effect of transplantation of hUCB-MSCs and hAESCs on SCI-induced NR-1 phosphorylation. At the end of the behavioral experiment, rats (n = 3 in each group) were euthanized, and the L4-5 segments of the spinal cord were removed for pNR1 immunohistochemistry. Animals were first deeply anesthetized with 5% isoflurane, perfused transcardially with calcium-free Tyrode's solution (116.4 mM NaCl, 5.4 mM KCl, 1.6 mM MgCl2•6H2O, 0.7 mM MgSO4•H2O, 1.2 mM NaH2PO4•H2O, 5.6 mM glucose, 26.2 mM NaHCO3; all reagents were purchased from Sigma-Aldrich), followed by a fixative containing 4% paraformaldehyde and 0.2% picric acid (Sigma-Aldrich) in 0.1 M phosphate buffer (pH 6.9; Santa Cruz Biotech. Inc., Dallas, TX, USA). The spinal cord was then removed immediately, postfixed at 4°C overnight in the same fixative, and then cryoprotected in 30% sucrose (Junsei Chemical Co., Tokyo, Japan) in PBS (pH 7.4) for 48 h. Frozen serial frontal sections (40 μm) were cut through the lumbar L4-5 spinal cord using a cryostat (Microm, Walldorf, Germany). After elimination of endogenous peroxidase activity with 0.3% hydrogen peroxide (Junsei Chemical Co.) and preblocking with 1% normal donkey serum (Vector Laboratories, Burlingame, CA, USA), 0.3% Triton X-100 (Sigma-Aldrich), and 1% bovine serum albumin (Zymed, San Francisco, CA, USA) in PBS, the free-floating sections were incubated with a polyclonal rabbit anti-pNR1 antibody (1:1,000; Upstate, Lake Placid, NY, USA) at 4°C for 48 h. After washing with PBS, the sections were reacted for 2 h at room temperature in a dark chamber with a fluorescent secondary antibody, cyanine 3 (Cy3)-conjugated anti-rabbit IgG (1:200; Jackson ImmunoResearch Laboratories, West Grove, PA, USA). Some sections were processed in an identical fashion, but without the primary antibody, and these sections served as an immunohistochemical control for nonspecific binding. No positive immunofluorescent staining was detected in tissues processed in this manner. The spinal cord dorsal horn was divided into the following three areas for subsequent immunohistochemical analysis: (1) the superficial dorsal horn (SDH, laminae I and II), (2) the nucleus proprius (NP, laminae III and IV), and (3) the neck of dorsal horn (NECK, laminae V and VI). Five spinal cord sections from the L4-5 lumbar spinal cord segments were randomly selected from each animal. Visualization and localization of pNR1 cells in these spinal cord sections was performed using a confocal laser microscope system (Eclipse TE200, Nikon, Nagano City, Japan). The average number of pNR1-ir cell profiles per section from each animal was obtained, and these values were averaged again across each group and presented as group data (n = 3 in each group). For PKH26 detection, nuclei were counterstained with Hoechst 33238 (1 μg/ml, Sigma-Aldrich). All images per section were also captured and merged by a confocal laser microscope system (Eclipse TE200). To examine the number of surviving/remaining hUCB-MSCs and hAESCs after spinal cord injection, we counted the number of PKH26-labeled cells among the Hoechst-positive cells.
Protein Extraction and Western Blot Assay
The T13 segment and the L4-5 segments of the spinal cord were obtained at the end of the experiment for protein extraction and western blot analysis (n = 3 in each group). The experiments were performed on the following four groups of rats: (1) a sham group, (2) a control PBS injection-SCI group, (3) a hUCB-MSC-treated SCI group, and (4) a hAESC-treated SCI group. Spinal cord tissue from each animal was homogenized independently in lysis buffer containing protease inhibitor cocktail (Roche Applied Science, Mannheim, Germany), followed by vigorous shaking for 2–3 min in the presence of 20 μl of 10% Nonidet-P40 (Sigma-Aldrich). The total protein content of each lysate was quantified using a Bio-Rad protein assay kit (Bio-Rad Laboratories, Foster City, CA, USA). For Western blotting, an equal amount (30 μg) of protein extract was subjected to 8–15% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE; Amersham, Piscataway, NJ, USA) analysis and transferred to a nitrocellulose membrane (Amersham) for incubation with primary antibodies. After immunoblotting with secondary antibodies, proteins were detected with an enhanced chemiluminescence reagent (Intron Biotechnology, Kyungki-Do, Korea). The relative band intensities were measured using NIH ImageJ version 1.34e software (Bethesda, MD, USA). One representative band from three independent experiments was chosen for presentation. The following primary antibodies were used: anti-F4/80 (Abcam, Cambridge, UK), anti-glial fibrillary acidic protein (GFAP, Millipore, Billercia, MA, USA), anti-myelin basic protein (MBP), anti-neuron-specific βIII tubulin (antibody clone TUJ1), anti-inducible nitric oxide synthase (iNOS), anti-cyclooxygenase (COX2, all Abcam), and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Sigma-Aldrich).
Statistical Analysis
All values are expressed as the mean ± SEM. Statistical analysis was performed using Prism 5.0 (Graph Pad Software, San Diego, CA, USA). A repeated-measures two-way analysis of variance (ANOVA) was performed to determine overall differences in PWL (s) and PWF (%). A one-way ANOVA was also used to determine differences in the number of spinal pNR1-ir cells in spinal cord sections across all experiment groups. Post hoc analysis after repeated-measures two-way or one-way ANOVA was performed using the Bonferroni's multiple comparison test to determine the p value. A value of p < 0.05 was considered statistically significant.
Results
Morphologic Characteristics of hUCB-MSCs and hAESCs
The morphological appearance of the hUCB-MSCs and hAESCs was characteristic of that reported in the literature (27,30). The hUCB-MSCs were spindle shaped and showed a fibroblast-like appearance (Fig. 1C), while the hAESCs were polygonal in shape and exhibited a cobblestone-like morphology (Fig. 1D).
Effect of hUCB-MSCs and hAESCs on SCI-Induced Below-Level Neuropathic Pain
The effect of transplantation of hUCB-MSCs or hAESCs on below-level MA and TH in SCI rats is illustrated in Figure 2. The development of MA (Fig. 2A, B) and TH (Fig. 3A, B) was robust by day 13 postinjury, stem cells were implanted on day 14, and their potential effects on ipsilateral and contralateral MA and TH were evaluated for 28 days posttransplantation. While the transplantation of hUCB-MSCs into the spinal cord showed a trend toward gradually reducing PWF in both hind paws from day 10 to day 28 posttransplantation, this effect did not reach statistical significance. Conversely, the transplantation of hAESCs significantly reduced the SCI-induced increase in PWF from days 13 to 24 in the ipsilateral hind paw and on day 24 in the contralateral hind paw, respectively (*p < 0.05 as compared to PBS-treated SCI rats) (Fig. 2A, B). The initial suppressive effect of hAESCs occurred at approximately 2 weeks after cell transplantation. While transplantation of hAESCs significantly reduced MA, neither hUCB-MSC nor hAESC transplantation had any effect on SCI-induced TH as measured by changes in paw withdrawal latency in the Hargreaves test (Fig. 3A, B).
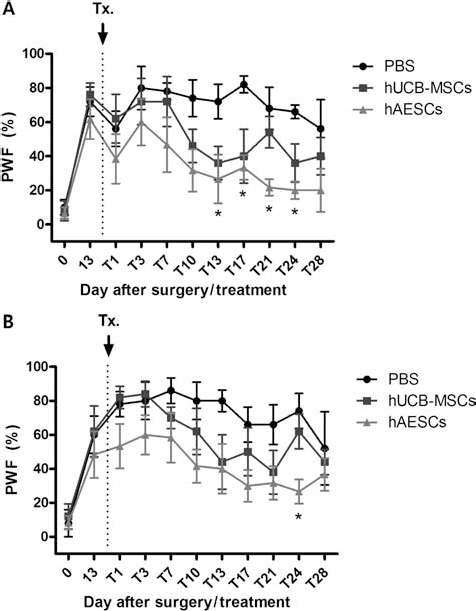
The effect of transplantation of hUCB-MSCs and hAESCs into the spinal cord on SCI-induced mechanical allodynia in the ipsilateral and the contralateral hind paws of rats with a T13 injury. While transplantation of hUCB-MSCs gradually reduced “percent withdrawal response frequency” PWF (%) in both the ipsilateral (A) and contralateral hind paws (B) from day 10 to day 28 following stem cell transplantation, the effect did not attain statistical significance. Conversely transplantation of hAESCs significantly reduced mechanical allodynia as measured by PWF (%) from day 13 to day 24 in the ipsilateral hind paw and at day 24 in contralateral hind paw, respectively (*p < 0.05 as compared to PBS-treated rats) (n = 8 in each group).
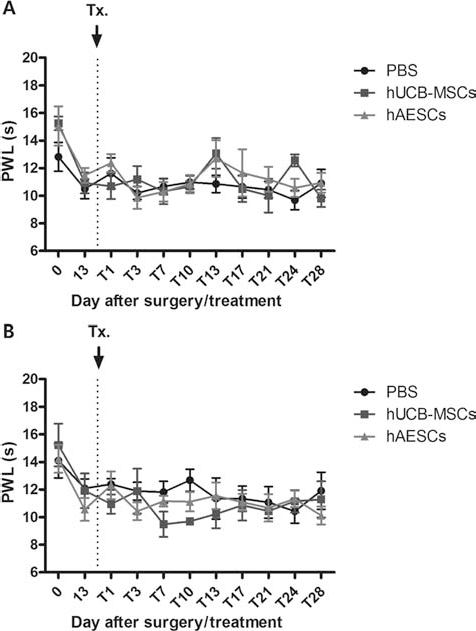
Graphs summarizing the effect of transplantation of hUCB-MSCs and hAESCs on thermal hyperalgesia in the ipsilateral and contralateral hind paws of SCI rats. Neither hUCB-MSC nor hAESC transplantation altered the SCI-induced decrease in ipsilateral (A) or contralateral (B) paw withdrawal response latency (PWL; s) (n = 8 in each group).
Effect of hUCB-MSCs and hAESCs on pNR1 Expression in Lumbar Dorsal Horn After SCI
SCI caused a significant increase in the number of phosphorylated NMDA receptor NR1 subunit immunoreactive (pNR1-ir) cell profiles in the SDH (laminae I and II), NP (laminae III and IV), and in the NECK region (laminae V and VI) of both the ipsilateral and contralateral spinal cord dorsal horns (*p < 0.05 as compared to that of sham surgery animals) (Fig. 4A–D). An increase in the number of pNR1-ir cells in the spinal cord dorsal horn is thought to be a major mechanism contributing to the induction of the central sensitization phenomenon in chronic pain states, and thus, the SCI-induced increase is consistent with the development of central sensitization. While the transplantation of hUCB-MSCs showed a trend toward suppressing the SCI-induced increase in the number of pNR1-ir cells in the NECK regions of ipsilateral and contralateral spinal dorsal horn (Fig. 4A–E), this did not reach statistical significance. On the other hand, transplantation of hAESCs significantly reduced the SCI-induced increase in the number of pNR1-ir cells in the NP and the NECK regions of ipsilateral and contralateral spinal dorsal horn (#p < 0.05 and ##p < 0.01 as compared with those of PBS-treated animals) (Fig. 4A–D, F).

The number of phosphorylated NMDA receptor NR1 subunit immunoreactive (pNR1-ir) cell profiles in different laminae of the spinal cord dorsal horn following transplantation. Graphs illustrating the number of phosphorylated N-methyl-d-aspartate (NMDA) receptor NR1 subunit immunoreactive (pNR1-ir) cell profiles in different laminae of the ipsilateral (A) and contralateral (B) spinal cord dorsal horn and photomicrographs (C–J) of representative pNR1-ir cells in the spinal cord following treatment with hUCB-MSCs and hAESCs. SCI caused a significant increase in the number of pNR1-ir cell profiles in the superficial dorsal horn (SDH, laminae I and II), the nucleus proprius (NP, laminae III and IV), and the neck of the dorsal horn (NECK, laminae V and VI) regions of both the ipsilateral (A) and contralateral spinal cord dorsal horns (B) (*p < 0.05 as compared to that of sham surgery animals). The transplantation of hAESCs significantly reduced the SCI-induced increase in the number of pNR1-ir cells in the NP and the NECK regions of ipsilateral and contralateral spinal dorsal horn (A, B; #p < 0.05 and ##p < 0.01 as compared with those of PBS-treated animals). (C–F) Representative pictures showing the effect of hUCB-MSCs and hAESCs on pNR1 expression. (G–J) The magnified images of rectangular in (C–F), respectively. Scale bar: 100 μm.
hUCB-MSCs and hAESCs Detected in Transplanted Spinal Cord
To determine the position of the injected stem cells in SCI rats, we performed cell tracking using confocal imaging in conjunction with the fluorescent cell dye PKH26, which incorporates a yellow–orange fluorescence into the lipid regions of the stem cell membrane prior to transplantation. PKH26-labeled hUCB-MSCs and hAESCs were present around the site of spinal cord injury (Fig. 5). Hoechst staining of cell nuclei was also conducted for detecting cell nuclei, and the image of the nuclear staining was merged with that of the PKH26-labeled cells (Fig. 5). This allowed confirmation that the injected labeled hUCB-MSCs and hAESCs were present within the injection sites near the site of spinal cord injury. To determine the number of surviving hUCB-MSCs and hAESCs following injection, we performed an analysis of the number of PKH26-labeled cells among the entire cell population (Hoechst-positive cells) at the injection site. PKH26-labeled hUCB-MSCs and hAESCs were counted in each section and measured by percentage. We found that the number of hUCB-MSCs at the injection site represented approximately 12% of the total cell population, and similarly the number of hAESCs at the injection site represented approximately 10% of the total cell population. Thus, the distribution of surviving hUCB-MSCs was comparable to that of surviving hAESCs at the injection site (Fig. 5D).

Representative confocal images of PKH26-labeled hUCB-MSCs (A) and hAESCs (B) near the injury site. No labeled cells were present in PBS injected SCI rats. (C) Hoechst staining was conducted for detecting nuclei and the confocal images of cell nuclear staining were then merged with those of the PKH26-labeled cells. These merged images illustrate the nuclei of the transplanted stem cells. Scale bar: 50 μm in low magnification (200×) and 100 μm in high magnification (400× and zoom-merge). (D) Ratio of cell distribution. Detected PKH26-labeled cells are presented as percentage of the total cell population. The percentage of PKH26-labeled surviving cells in hUCB-MSC-treated animals is similar to that in the hAESC-treated group.
Effect of hUCB-MSC and hAESC on iNOS and COX Expression in the Dorsal Horn of At-Level and Below-Level Spinal Cord Segments Following SCI
Since we have shown that the transplantation of hUCB-MSCs and hAESCs reduce the SCI-induced MA component of neuropathic pain, we next examined the spinal cord expression levels of several proteins including F4/80, GFAP, MBP, neuron-specific βIII tubulin (antibody clone TUJ1), iNOS, and COX2 that are known to be involved in spinal cord injury, inflammation, and/or chronic pain. We conducted a Western blot analysis of both the T13 (“at-level”) and L4-5 (“below-level”) spinal cord segments in each experiment group. At the site of spinal cord injury (T13), the expression of F4/80, GFAP, MBP, COX2, and iNOS were increased in PBS-treated SCI animals. While the SCI-induced increase in the expression levels of GFAP, COX2, and iNOS were not changed in the stem cell treatment groups, the SCI-induced increase in F4/80 expression was decreased in the hAESC treatment group compared with that of the PBS-treated SCI group (Fig. 6). Stem cell transplantation produced no changes in the expression of MBP and neuron-specific βIII tubulin (Fig. 6A). In addition, we conducted a Western blot analysis of the expression of these same proteins at the L4-5 (below-level) segments of the spinal cord. SCI increased the expression of both F4/80 and COX2 within the L4-5 spinal cord segments as compared with rats in the sham control group. Importantly, treatment with either hUCB-MSCs or hAESCs dramatically decreased the expression of F4/80 as compared with PBS-SCI rats (Fig. 6B). Conversely, transplantation of either hUCB-MSCs or hAESCs had no effect on the expression of GFAP, MBP, neuron-specific βIII tubulin, iNOS, or COX2 within below-level spinal cord segments (Fig. 6B).

Western blots depicting the effect of hUCB-MSCs and hAESCs on neuronal, inflammation, and immune-associated markers. At the site of spinal injury (T13), epidermal growth factor-like module-containing mucin-like, hormone receptor-like 1 (EMR1; F4/80), glial fibrillary acidic protein (GFAP), Myelin basic protein (MBP), cyclooxygenase 2 (COX2), and induced nitric oxide synthase (iNOS) were increased in PBS-treated SCI animals (A). Although the expression levels of GFAP, COX2, and iNOS did not change in the stem cell treatment groups, transplantation of hAESCs caused a decrease in the SCI-induced increase in the expression of F4/80 as compared with that of the PBS-treated group. In addition, within the L4-5 spinal cord segments (below-level site), F4/80 and COX2 also increased in SCI animals as compared to sham animals. This SCI-induced increase in F4/80, but not COX2, was reduced by transplantation of either hUCB-MSCs or hAESCs as compared to PBS-injected SCI rats (B). TUJ1, antibody clone for neuron-specific βIII tubulin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Effect of hUCB-MSC and hAESC on Motor Function After SCI
Prior to hemisection, the BBB scores were 21 in all SCI groups (Fig. 7). Immediately upon emerging from anesthesia, hemisected animals displayed loss of ipsilateral hindlimb function as indicated by the BBB scores of 0 for each group. At day 14 after hemisection, the transplantation of either hUCB-MSCs or AESCs produced no effects on motor function (Fig. 7).

A graph illustrating the effect of transplantation of hUCB-MSCs and hAESCs on motor function following SCI. Prior to hemisection, the Basso, Beattie, and Bresnahan (BBB) scores were 21 in all SCI groups. Immediately upon emerging from anesthesia, hemisected animals displayed loss of ipsilateral hindlimb function as indicated by BBB scores of 0 for each group. By day 14 posthemisection, the BBB scores were increased indicating partial recovery and the transplantation of hUCB-MSCs or AESCs had no significant effect on the further recovery of BBB scores indicating of a lack of effect on motor function.
Discussion
The present study demonstrated that the transplantation of hAESCs into the spinal cord reduced SCI-induced MA bilaterally in the rat hind paws. However, the transplantation of hUCB-MSCs or AESCs had no effect on SCI-induced TH. In addition, the SCI-induced increase in the expression of pNR1 in the L4-5 spinal cord segments was significantly inhibited in hAESC-treated animals, whereas it was only partially decreased in hUCB-MSC-treated animals. These findings indicate that the transplantation of hAESCs produce an antinociceptive effect on mechanical stimuli, but not heat stimuli, in SCI-induced central neuropathic animals. Moreover, this stem cell-induced modulation of MA was closely associated with the suppression of spinal cord pNR1, and since the phosphorylation of this receptor plays a role in the development of central sensitization associated with neuropathic pain (16), it is likely that the stem cell-induced reduction in spinal cord pNR1 is one mechanism by which hAESCs reduce MA as discussed below. Our study clearly shows that the effects of transplantation of hAESCs on both SCI-induced MA and pNR1 expression is more potent than that of hUCB-MSCs, and thus, our data would favor the use of hAESCs for treating SCI associated pain.
Traumatic injuries to the spinal cord lead to severe and permanent neurological deficits. Although no effective therapeutic approach is currently available, recent animal studies have shown that cellular transplantation strategies can enhance functional recovery following SCI (25,30). Researchers have reported that serotonergic neural precursor cell grafts attenuated the hyperexcitability of dorsal horn neurons bilaterally after spinal hemisection (22). Moreover, predifferentiated embryonic stem cells have been shown to reduce chronic pain behaviors and restored sensory function following chemically induced spinal cord injury in mice (24). Recently transplanted bone marrow stromal cells (BMSCs) have also shown analgesic efficacy in both diabetic and peripheral neuropathic pain rats (18,41). However, the present study is the first to evaluate whether the transplantation of either hUCB-MSCs or hAESCs produce antinociceptive effects on SCI-induced MA and TH. Our data show that hAESCs significantly reduce MA, but not TH, in the spinal cord injury model used and that while hUCB-MSCs also showed a tendency to reduce MA, this did not reach statistical significance. These findings suggest that transplantation of AESCs can be a potential therapy for SCI-induced central neuropathic pain, particularly the MA component of this pain.
Although there are some basic differences between hUCB-MSCs and hAESCs, including their cellular morphology and intrinsic characteristics, they both have the capacity for self-renewal and differentiation into a variety of diverse cell types (37,58). AESCs express surface markers normally present on embryonic stem and germ cells, and several studies have examined the use of these cells for treatment of a variety of conditions including burns (10,11), defects in the abdominal wall and ruptured spleens (15), and oral wounds (31). Additionally, there are several reports that suggest the use of hAESCs for a number of therapeutic applications (36,62). Similarly, a number of studies have recently reported success with the transplantation of hUCB-MSCs for treating neurodegenerative diseases in mouse models of Alzheimer's disease (43), Parkinson's disease (14), amyotrophic lateral sclerosis (8), and Niemann Pick disease (58).
In the present study, we demonstrated that transplantation of hAESCs significantly reduces the SCI-induced increase in pNR1 expression in the spinal cord dorsal horn and that this effect is much more robust than that observed following transplantation of hUCB-MSCs. This implies that hAESCs have a much more potent modulatory effect on spinal cord pNR1 expression, and this is consistent with the anti-allodynic effect that hAESCs have on SCI-induced central sensitization. It has been well recognized that the enhanced expression of pNR1, which occurs via intracellular signaling pathways, is a major mechanism contributing to the regulation of NMDA receptor function and to the induction of the central sensitization phenomenon in the spinal cord (16,50–52). It has previously been demonstrated that there is a significant increase in the number of pNR1-ir cells in response to quisqualic acid-induced excitotoxic lesions of or T13 hemisection injury to the spinal cord (7,58) and that the increase in pNR1 expression is associated with the induction of chronic pain. Accordingly, we postulate that the antiallodynic effects of transplanted hAESCs are mediated in part by modulation of spinal cord pNR1 expression.
Previously, we reported that the spinal lamina-dependent increase of pNR1 expression in laminae I and II versus III and IV in peripheral neuropathic rats is closely associated with the development of TH or MA, respectively (51,52). The increased expression of pNR1 in spinal laminae I–II and V–VI is dependent on activation of capsaicin sensitive primary afferents, which ultimately contribute to the development of thermal hyperalgesia, whereas the increase of pNR1 in spinal laminae III–IV and V–VI is related to the development of mechanical allodynia. In this regard, we believe that the effect of hAESCs on mechanical allodynia, but not thermal hyperalgesia, is associated with the regulation of pNR1 in laminae III–VI of SCI rats. Currently, it is not clear why the cell grafts had a greater influence on pNR1 expression in laminae III–VI compared to laminae I–II, but this will be the focus of a future study. In addition, in the present study, we did not determine the precise mechanisms by which hAESC transplantation results in the suppression of pNR1 expression, but one possibility is by modifying SCI-induced glial activation.
In this regard, we examined the effect of stem cell transplantation on SCI-induced glia activation including astrocytes and microglia, which are possible candidates that could potentially modulate pNR1 expression (13,17). It is notable that treatment with hAESCs significantly reduced the SCI-induced increase in F4/80 expression, a marker of microglia activation at both the site of spinal damage and the L4-5 segments (below-level) of the spinal cord. Transplantation of hUCB-MSCs also decreased F4/80 expression in the L4-5 segments of the spinal cord below the site of injury. On the other hand, neither AESC nor hUCB-MSC treatment had any effect on the SCI-induced increase in GFAP expression, a marker of astrocyte activation. Several studies have documented that activated microglia and astrocytes release a variety of proinflammatory cytokines including tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6. The increased release of these proinflammatory mediators enhances dorsal horn neuronal activity, which substantially contributes to the development of chronic pain states (29,39,63). In this regard, it has been demonstrated that the increased expression of TNF-α following SCI is associated with the development of below-level neuropathic pain and that reverse signaling through the full-length transmembrane form of TNF-α or administration of the TNF inhibitor, etanercept, reduces both cellular markers of the inflammatory response and pain-related behaviors (35,49). Moreover, it has been shown that IL-1 receptor antagonist treatment suppresses both mechanical hyperalgesia and the increases in pNR1 expression in rat models of bone cancer pain and arthritic pain (64,65). Recently Daulhac et al. also reported that increased expression of pNR1 is regulated by extracellular signal-regulated kinase (ERK) and the NMDA receptor via a feed-forward mechanism in spinal neurons and microglia in diabetic neuropathic pain (12). These findings demonstrated that glia activation, particularly microglia in the context of the present study, could increase pNR1 expression via cytokine signaling in spinal dorsal horn at sites below the site of spinal injury and that ultimately this could contribute to SCI-induced MA. Therefore, it is conceivable that hAESC transplantation blocks abnormal microglia activation in both the at-level and below-level regions of the spinal cord and that this indirectly reduces the SCI-induced increase in NMDA receptor function by modulating the phosphorylation state of the NR1 in spinal dorsal horn neurons.
Both hUCB-MSCs and AESCs significantly reduced F4/80 expression in below-level spinal cord, while transplanted hAESCs produced a more potent antiallodynic effect as compared to that of hUCB-MSCs. We believe that this phenomenon may be caused by a reduced effectiveness of hUCB-MSCs on F4/80 expression in the “at-level” site, which may relate to the response to the direct effects of spinal cord injury at the site. It is known that inhibition of early neuroimmune events including microglial inhibition can have a powerful impact on the development of long-term pain phenomena following SCI (60). Moreover, there are differences in mediator release at the site of injury versus below-level injury. Thus, it has been reported that there is a significant increase in the expression of monocyte chemoattractant protein-1 (MCP-1), TNF-α, tissue inhibitor of metalloproteinase 1 (TIMP-1), and IL-1 in the injury epicenter, whereas only TIMP-1 is upregulated in the area below the injury epicenter in a rat SCI model (56). Since microglia located at the below-level segments of the spinal cord appear to produce less mediators than those at the site of the injury, it is possible that the transplanted hUCB-MSCs and AESCs have a greater effect at the below-level segments, where microglia are perhaps not as engaged in the inflammation and apoptotic processes associated with the site of injury (19,21). However, the possible direct or indirect relationship between below-level microglia regulation and neuropathic pain, and the underlying mechanisms of the effect of stem cells needs to be investigated in future studies to get a better mechanistic picture of these events.
Microglia activation is closely associated with the inflammatory response in the spinal cord, and so we also hypothesized that hUCB-MSC and hAESC transplantation would modify SCI-induced changes in inflammatory mediators, including iNOS and COX2 expression at both the at-level and below-level regions of the spinal cord. However, based on the results in the present study, we found that the SCI-induced increase in iNOS and COX2 expression was not reduced by hUCB-MSC and hAESC transplantation. A recent study showed that transplantation of canine hUCB-MSCs 1 week after SCI was effective in improving clinical signs and neuronal regeneration and reducing fibrosis formation in canine spinal cord injury (48). Although these investigators reported that COX-2 expression was modified by canine UCB-MSC transplantation, we believe that the difference between their results and ours is due to the use of different stem cells (canine-derived vs. human-derived) or to differences in the animal models (rat vs. dog). In addition, the time point at which stem cell transplantation occurs following SCI could be a critical factor in the stem cell's ability to modulate inflammatory signaling and this requires additional research. Even though microglia activation can participate in the spinal cord inflammatory response, the present study demonstrates that the inhibition of microglia activity evoked by hUCB-MSC and hAESC transplantation is not directly involved in modulating the SCI-induced increase in iNOS and COX2 expression.
Finally, the effect of hUCB-MSC and hAESC transplantation on the potential recovery of motor function following SCI was also examined in the present study. Although recovery of motor function is a major concern in stem cell therapy studies related to neuronal regeneration, several studies have previously reported the effect of stem cell transplantation on functional motor recovery following spinal cord injury (4,33). We focused on the therapeutic effect of two different stem cell populations on central sensitization after the time point at which chronic pain had already developed in SCI animals, and so we did not investigate the potential use of these stem cells at earlier time points. Thus, we transplanted the stem cells 2 weeks after SCI surgery, at a time when the SCI animals had already begun to recover some motor function based on the BBB test (the BBB score was 14–16). Based on the time point selected for transplantation, it is not surprising that the implantation of these stem cells did not modify motor function to any significant degree. Importantly, however, these results also demonstrate that hUCB-MSC and hAESC transplantation did not delay the recovery of motor function, even though hAESCs simultaneously reduced below-level SCI-induced MA.
On the other hand, it is possible that multiple conflicting host responses (an inflammatory response to the SCI, a host response to the implanted stem cells, below-level responses to the injury, etc.) are occurring simultaneously, in which case the stem cells may have differential effects on each response. However, several studies including our previous findings have reported that the transplantation of UCB-MSCs or AESCs does not induce a host immune reaction in a variety of experimental conditions (2,3,27,58). Thus immunosuppression is not necessary in the current SCI model. Moreover, we believe that if immunosuppressive agents were given, this would not affect the antiallodynic effect of stem cells, since the SCI-induced increase in F4/80, a marker of microglia or macrophage activation, was suppressed, not enhanced, by the application of UCB-MSCs or AESCs in the present study. Based on this evidence, we suggest that the lack of acute immune rejection makes the consideration of transplanting UCB-MSCs or AESCs an attractive alternative for the potential treatment of neuropathic pain conditions particularly as they relate to spinal cord injury.
In conclusion, the present study demonstrates the therapeutic effect of hAESCs on SCI-induced MA, but not TH, in a rodent model of spinal cord injury. While hUCB-MSC transplantation produced a weak antiallodynic effect, transplantation of hAESCs attenuated MA in both hind paws. Consistent with this finding, the SCI-induced increase in spinal pNR1 and microglia activation was also significantly suppressed in hAESC-treated animals. Collectively, these findings demonstrate that hAESC transplantation can significantly reduce SCI-induced MA, and mechanistically this effect is associated with a decrease in spinal cord microglia activity and NMDA receptor NR1 phosphorylation. Thus, transplantation of hAESCs may have therapeutic value in treating below-level neuropathic pain that often accompanies spinal cord injury. However, while spinal cord injury is common in humans, spinal cord hemisection is not, and thus, rodent models have limitations in this regard. In addition, inflammatory and immune responses differ between primates and rodents, which also represents a limitation of the current study. On the other hand, in the context of potential stem cell therapy, there are several questions for which studies in rodents can be very informative, including which cell types and how many cells should be implanted, what percentage of cells survive after implantation, and with regard to the present study can stem cell transplantation alter SCI-induced pain. The results support a potential beneficial effect of stem cell transplantation on SCI-induced neuropathic pain. Moreover, our data together with that of other studies (20) in SCI models demonstrate that dysfunctional glia, a condition that Gwak and coworkers term “gliopathy,” is a key contributor in the underlying cellular mechanisms contributing to neuropathic pain in SCI.
Footnotes
Acknowledgments
This research was supported by a grant (2009K001256) from Brain Research Center of the 21st Century Frontier Research Program funded by the Ministry of Education, Science and Technology, the Republic of Korea. This work was also supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST, 2010-0020265). The authors declare no conflicts of interest.
