Abstract
Niemann Pick disease type C1 (NPC) is an autosomal recessive disease characterized by progressive neurological deterioration leading to premature death. In this study, we hypothesized that human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs) have the multifunctional abilities to ameliorate NPC symptoms in the brain. To test this hypothesis, hUCB-MSCs were transplanted into the hippocampus of NPC mice in the early asymptomatic stage. This transplantation resulted in the recovery of motor function in the Rota Rod test and impaired cholesterol homeostasis leading to increased levels of cholesterol efflux-related genes such as LXRα, ABCA1, and ABCG5 while decreased levels of 3-hydroxy-3-methylglutaryl coenzyme A reductase were observed in NPC mice. In the cerebrum, hUCB-MSCs enhanced neuronal cell survival and proliferation, where they directly differentiated into electrically active MAP2-positive neurons as demonstrated by whole-cell patch clamping. In addition, we observed that hUCB-MSCs reduced Purkinje neuronal loss by suppression of inflammatory and apoptotic signaling in the cerebellum as shown by immunohistochemistry. We further investigated how hUCB-MSCs enhance cellular survival and inhibit apoptosis in NPC mice. Neuronal cell survival was associated with increased PI3K/AKT and JAK2/STAT3 signaling; moreover, hUCB-MSCs modulated the levels of GABA/glutamate transporters such as GAT1, EAAT2, EAAT3, and GAD6 in NPC mice as assessed by Western blot analysis. Taken together, our findings suggest that hUCB-MSCs might play multifunctional roles in neuronal cell survival and ameliorating motor deficits of NPC mice.
Keywords
Introduction
Niemann-Pick disease type C1 (NPC) is a fatal genetic disorder with an incidence estimated at between 1:120,000 and 1:150,000 live births (33,44). NPC is caused by the mutation of NPC1 gene (95% of cases) or NPC2 gene (5), which encodes a transmembrane protein of the late endosome and lysosome. The NPC1 gene, a major protein of NPC, facilitates the movement of unesterified cholesterol derived from the cellular uptake of lipoproteins (30) and its mutation results in excessive accumulation of unesterified cholesterol in the late endosome and lysosome (31). In the aspect of neurodegenerative disease, the phenotype of NPC is characterized by distended neurons, demyelinated axons, reactive gliosis, and neuronal loss, eventually leading to dystonia, ataxia, seizures, dementia, and premature death (1,38,42). To date, some chemicals, such as allopregnanolone (13) or miglustat (24,37), have been useful in handling this fatal disease; however, it is still necessary to search for alternative treatment strategies.
The therapeutic strategy of stem cell transplantation is known to replace damaged cells and to provide a better microenvironment for endogenous cells by paracrine effects. Indeed, stem cell transplantation has emerged as a promising therapeutic approach for neurodegenerative disorders. A number of studies have shown that not only neural stem cells but also embryonic or mesenchymal stem cells can contribute to the correction of neurological abnormalities (7,12,28,29,43). Recently, we have demonstrated that engrafted Oct4-expressing human adipose tissue-derived stem cells efficiently migrate and differentiate into action potential-exhibiting, functional neurons in the hippocampus and eventually promote the amelioration of lesion cavities in a brain trauma model (18).
In this study, we are using human umbilical cord blood (hUCB) as a unique isolation source of stem cells. Leung et al. have reported that hUCB contains highly immature cells with extensive proliferation capacity and a remarkable proliferative response (27). Consistently, our previous studies have shown that hUCB mesenchymal stem cells (MSCs) maintain stemness and multipotency and Oct4A contributes to this property (41). Many researchers have recently suggested that administration of whole hUCB cells (hUCBCs), hUCB-MSCs, or hUCBC-derived mononuclear cells provide beneficial effects to animal models with neurodegenerative diseases: in Alzheimer's disease (AD) transgenic mice, hUCBCs reduce AD-like pathology (34); in Parkinson's disease and amyotrophic lateral sclerosis mice, they significantly delay the onset of symptoms and the death end point (3,6,10,11); and in rat ischemic models, hUCBCs improve neurobehavioral function and reduced infarct volume (22).
However, very few studies relating to NPC have dealt with the therapeutic application of stem cells, including hUCB-MSCs. Therefore, it would be of interest to determine whether hUCB-MSCs can improve NPC symptoms in both in vitro and in vivo models. For this, hUCB-MSCs were transplanted into the hippocampus of NPC mice at the asymptomatic stage. After transplantation, the mice performed better in the Rota Rod test than control NPC mice and their cholesterol deposition reduced compared to that of controls. Interestingly, we found that neuronal cell survival increased not only in hippocampal regions but also in the cerebellum. Some of the transplanted cells directly differentiated into neuronal cells with functional synapses. Furthermore, hUCB-MSCs activated PI3K/AKT and JAK2/STAT3 pathway signaling and altered neurotransmitter transporter expression toward reducing the excitotoxic cytotoxicity. The overall results support the availability of hUCB-MSC- based therapy in a NPC mouse model and in other neurodegenerative diseases.
Materials and Methods
Isolation and Maintenance of hUCB-MSCs
Isolation, culture, and characterization of hUCB-MSCs were performed as described previously (36,41). hUCB samples were obtained from the Seoul City Borame Hospital Cord Blood Bank. Samples from term and preterm deliveries were harvested at the time of birth with the mother's informed consent. This work was approved by the Borame Hospital Institutional Review Board and Seoul National University (IRB No. 0603/001-002-07C1). The blood samples were processed within 24 h of collection. The mononuclear cells were separated from the UCB using Ficoll-Paque TM PLUS (Amersham Bioscience, Uppsala, Sweden) and were suspended in a culture medium DMEM (Gibco, Grand Island, NY, USA), containing 20% fetal bovine serum (FBS), 100 I/ml penicillin, 100 mg/ml streptomycin, 2 mM L-glutamine, and 1 mM sodium pyruvate.
In Vitro Differentiation and Flow Cytometric Analysis of hUCB-MSCs
For osteogenic differentiation, hUCB-MSCs were maintained in DMEM, containing 100 nM dexamethasone, 50 μM ascorbic acid, 10 mM β-glycerophosphate supplemented with 10% FBS, and then they were immunostained with Alizarin red S. For adipogenic differentiation, hUCB-MSCs were cultured in DMEM supplemented with 5% FBS, 1 μM dexamethasone, 10 μM insulin, 200 μM indomethacin, and 0.5 mm isobutylmethylxanthine for 3 weeks followed by Oil Red O staining. For chondrogenic differentiation, hUCB-MSCs were cultured with DMEM containing 3.5 g/ml glucose, 1% (v/v) ITS (Sigma-Aldrich), 2 mM L-glutamine (Invitrogen), 100 μg/ml sodium pyruvate, 0.2 mM ascorbic acid 2-phosphate (Sigma-Aldrich), 10–7 M dexamethasone (Sigma-Aldrich), and 10 ng/ml transforming growth factor-β3 (TGF-β3) (R&D Systems) medium. After 3–4 weeks, the differentiated cells were immunostained with toluidine blue. For flow cytometric analysis, the cells were detached and fixed with 70% ethanol for 10 min at 4°C. They were stained with each FITC- or phycoerythrin-conjugated antibodies (BD Bioscience) at 4°C for 30 min. Thereafter, the cells were washed twice and resuspended in 1 ml of PBS and were immediately analyzed using flow cytometry (FACSAria, Becton Dickinson).
Cytokine Array Analysis
At 80% confluence, the culture medium of the hUCB-MSCs was switched to serum-free culture medium DMEM (Gibco) for 48 h and the conditioned medium was collected for analysis of the cytokine expression. Cytokines, secreted by the hUCB-MSCs, were determined using RayBio® Human cytokine Antibody Array 3 and 5 (RayBiotech, Norcross, GA, USA) according to the manufacturer's instructions. The positive controls on the membrane were used to normalize the signal intensity of individual cytokines.
Animal Models
NPC mice were obtained from breeding pairs of Balb/c NPC1 NIH mice purchased from Jackson Laboratories (Bar Harbor, MA, USA). The genotyping was performed as previously described (45). All experiments were approved and followed by the regulations of the Institute of Laboratory Animals Resources (SNU-090819-3, Seoul National University, Korea).
Transplantation of the hUCB-MSCs Into NPC Mice
The mice (4 weeks old) were randomly divided into four groups: hUCB-MSC-transplanted NPC mice (n = 16), vehicle-injected control (VC) NPC mice (n = 10), untreated control NPC mice (n = 11), and wild-type control (WT) mice (n = 12). Anesthesia was induced with 5% isoflurane and maintained with 2% isoflurane administered through a facial mask. The hUCB-MSCs were transplanted into the hippocampus using the stereotaxic apparatus and Ultra-micropump (World Precision Instruments, Sarasota, FL, USA). To track injected hUCB-MSCs, three NPC mice were given cells labeled with a membrane-bound fluorescent marker PKH26. The injection coordinates were 2.00 mm posterior, 1.40 mm bilateral to bregma, and injection depth was 2.00 mm. Each recipient received approximately 1 × 106 cells in 2 μl of cell suspension with PBS at a rate of 500 nl per minute. For sham control, animals underwent the same transplantation procedure but they received a vehicle infusion of an equal volume of PBS. After transplantation, the scalp was closed by suture and the animals were allowed to recover from the anesthesia.
Rota Rod Test
Motor coordination test was assayed using a Rota Rod treadmill (7650 Accelerating model, Ugo Basile Biological Research Apparatus, Comerio, Italy). The mice, 3 weeks old, were first trained during 1 week by habituating. After the training, the mice, 5–7 weeks old, were subjected to the Rota Rod test at 10 rpm. The test was performed once per week and the mean record was adopted as the performance time of three attempts.
RNA Extraction and RT-PCR
Total RNA was extracted from mice brain using Easy-spin total RNA extraction kit (Intron Biotechnology, Seoul, Korea) according to the manufacturer's protocol. The cDNA was then amplified in a PCR reaction containing 5–10 pM of specific primers. Primer sequences were designed using Primer Express software (PE-Applied Biosystems, Warrington, UK) with gene sequences obtained from the GeneBank database. The primer sets used were as follows: ATP-binding membrane cassette transporter A1 (ABCA1) (F: 5′-CGTTTCCGGGAAGTGTCCTA-3′; R: 5′-GCTAGAGATGACAAGGAGGATGGA-3′), ATP-binding membrane cassette transporter G5 (ABCG5) (F: 5′-GTCCTGCTGAGGCGAG-TAAC-3′; R: 5′-GCAGCATCTGCCACTTATGA-3′), Liver X receptor α (LXRα) (F: 5′-AGGAGTGTCGACTT-CGCAAA-3′; R: 5′-CTCTTCTTGCCGCTTCAGTTT-3′), and GAPDH (F: 5′-TGAAGCAG-GCATCTGAGGG-3′; R: 5′-CGAAGGTGGAAGAGTGGGAG-3′).
Protein Extraction and Western Blot Analysis
The brains of mice 7–8 weeks old were taken and divided into cerebrum/cerebellum or cortex, striatum, hippocampus, and cerebellum following experimental purposes. Each tissue was homogenized independently in lysis buffer containing protease inhibitor cocktail (Roche Applied Science, Mannheim, Germany), followed by vigorous shaking for a few minutes in the presence of 20 μl of 10% Nonidet-P40. The total protein content of each lysate was quantified using a Bio-Rad protein assay kit (Bio-Rad Laboratories, Foster City, CA, USA). For Western blotting, an equal amount (30 μg) of protein extract was subjected to 8–15% SDS-PAGE analysis and transferred to a nitrocellulose membrane for incubation with primary antibodies. After immunoblotting with secondary antibodies, proteins were detected with enhanced chemiluminescence reagent (Intron Biotechnology). The relative band intensities were determined using NIH ImageJ version 1.34e software. One representative band from three independent experiments was chosen for presentation. The following primary antibodies were used: anti-phosphoinositide3-kinase (PI3K), anti-phosphoPI3K (pPI3K), anti-AKT, anti-phosphoAKT (pAKT), anti-phospho Janus kinase2 (pJAK2), anti-phospho signal transducers and activators of transcription protein3 (pSTAT3) Tyr705, anti-pSTAT3 ser727, anti-glycogen synthase kinase3β (GSK3β), anti-pGSK3β Ser9 (Cell Signaling, Danvers, MA, USA); anti-pSTAT3 Ser 727, anti-excitatory amino acid transporter2 (EAAT2), anti-microtubule associated protein2 (MAP2), anti-glial fibrillary acidic protein (GFAP), anti-doublecortin (DCX), anti-calbindin (CBD), anti-3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGR) (Millipore, Billerica, MA, USA); anti-glutamic acid decarboxylase65 (GAD6), anti-GABA transporter1 (GAT1) (Abcam, Cambridge, MA, USA); anti-JAK2, anti-excitatory amino acid transporter3 (EAAT3) (Santa Cruz Biotechnology Inc., Delaware, CA, USA); and anti-β-actin (Sigma-Aldrich).
Immunohistochemistry
Mice were perfused with 0.1 M PBS (pH 7.4), followed by 4% paraformaldehyde in 0.1 M PBS, and brains were isolated carefully and soaked in 4% paraformaldehyde in 0.1 M PBS overnight for postfixation. Sections (4 μm thick) were cut and used for H&E staining. For immunohistochemistry, the brain tissues were transferred to a mold filled with infiltration mixture (OCT compound; Sakura Finetek, Tokyo, Japan) and kept at −70°C overnight until cryosection on a cryostat (CM 3050, Leica, Wetzlar, Germany). Primary antibodies incubated with the sections were: anti-DCX, anti-MAP2, anti-GFAP, anti-human mitochondria (HuMi), anti-CBD (Millipore). Subsequently, the sections were extensively washed with PBS and 1-h incubation with anti-mouse or rabbit secondary antibodies conjugated with Alexa Fluor 488/594 (Molecular Probes, Eugene, OR, USA), followed by Hoechst 33238 (1 μg/ml, Sigma-Aldrich) staining in order to visualize the cell nuclei. Images were captured and merged on a confocal microscope system (Eclipse TE200, Nikon, Nagano City, Japan). To compare the degree of cholesterol accumulation, we performed filipin staining as previously described (19,21), and then depicted the hippocampal, cortical, and striatal regions from each group (n = 5). NIS element software (Nikon) was used to analyze the intensity of filipin staining.
Whole-Cell Patch Clamp Recordings
Four weeks posttransplantation, the brains were placed in ice-cold, oxygenated (95% O2/5% CO2) low-Ca2+/high-Mg2+ slicing solution composed of (in mM): 5.0 KCl, 7.0 MgCl2, 28.0 NaHCO3, 1.25 NaH2PO4, 0.5 CaCl2, 7.0 glucose, 3.0 pyruvic acid, 1.0 ascorbic acid, and 234.0 sucrose. After sectioning, 300-μm-thick slices were preincubated in the artificial cerebrospinal fluid. For recordings, a slice was transferred to a submersion-type recording chamber continuously perfused with the 95% O2/5% CO2-saturated standard artificial cerebrospinal fluid at 34–35°C. Individual hippocampal cells were viewed with an Olympus microscope fitted with a 40× water-immersion objective and differential interference contrast optics (Olympus, Tokyo, Japan). Whole-cell patch clamp recordings were made using an Axopatch 200B patch clamp amplifier (Axon Instruments, Foster City, CA, USA). The recording pipettes were pulled from borosilicate capillaries (1.5 mm OD, 1.0 mm ID; WPI) using a micropipette puller (Narishige, Tokyo, Japan). The resistance of the pipette was 4–6 MΩ when it was filled with a solution containing (in mM): 130.0 potassium gluconate, 1.0 MgCl2, 10.0 HEPES, 10.0 EGTA, 1.0 CaCl2, and 4.0 adenosine triphosphate-Mg adjusted to pH 7.25 with 1 M KOH (290–320 mOsm). The solutions were perfused at a rate of 3–4 ml/min at 33–35°C. A tight GΩ seal was obtained on the neuron identified. Recordings began ~5 min after whole-cell access was obtained and the current reached a steady state. The series resistance ranged from 15 to 30 mΩ as estimated directly from the amplifier. The data were digitized by Digidata 1200B (Axon Instruments). The recording was abandoned if the input resistance changed more than 15% during the recording.
Neural Progenitor Cell Culture and Generation of hUCB-MSC-Derived Conditioned Medium
To prepare neural progenitor cells, the subventricular zones were collected in 4-week-old mice, and then the tissues were minced, washed in PBS, and digested in Hank's balanced salt solution containing 0.125% trypsin and 0.01% DNase for 30 min at 37°C. The cells (5 × 105) were plated in 100-mm dishes and cultured in Neurobasal medium supplemented with 2% B27, 10 ng/ml basic fibroblast growth factor (bFGF), and 10 ng/ml epidermal growth factor (EGF). Neural stem cells (NSCs) were grown as spheres and they were used up to passages 5 and 6. To determine a potential preventive effect of hUCB-MSCs, NSCs were pretreated with hUCB-MSC-derived conditioned medium (CM, 1 μg/ml) for 6 h, and the cells were treated with hydrogen peroxide (H2O2, 0.3 mM) for an additional 6 h. The optimal concentrations of hUCB-MSC CM and H2O2 were determined by cytotoxicity test.
Statistical Analysis
Results are shown as means ± SE. Statistical analysis of significance was calculated by one-way ANOVA followed by Bonferroni post hoc test for multigroup comparisons (StatView 5.0; SAS Institute, Cary, NC). Statistical significance is indicated in the figure legends.
Results
Characterization of hUCB-MSCs
To determine the multilineage differentiation potential of hUCB-MSCs, hUCB-MSCs were controlled under lineage-specific culture conditions. The hUCB-MSCs were able to differentiate into multilineages which are osteogenic, adipogenic, and chondrogenic lineages (Fig. 1A, C, E, control; Fig. 1B, D, F, induction of multilineage differentiation). We then determined the proliferation and immunological phenotypes of hUCB-MSCs. In a cumulative population doubling assay, hUCB-MSCs were able to achieve more than 35 cumulative population doublings (Fig. 1G). In a karyotype test, no chromosomal abnormalities were found (Fig. 1H) and in cell surface antigen profiles, hUCB-MSCs were positive for CD24, CD29, CD44, CD73, CD90, and CD105, and negative for CD10, CD14, CD31, CD34, CD45, CD62P, CD133, and HLA-DR (Table 1). To determine whether particular cytokines are released from hUCB-MSCs, we performed a quantitative cytokine analysis. The result of cytokine array revealed that hUCB-MSC conditioned medium contained considerable amounts of immune modulatory and neurotrophic growth factors, such as interleukin-10 (IL-10), tissue inhibitor of metalloproteinases 1 and 2 (TIMP1 and 2), FGF, glial-derived neurotrophic factor (GDNF), brain-derived neurotrophic factor (BDNF), and nerve growth factor (NGF) (Fig. 2). These data suggest that hUCB-MSCs could be readily expanded and have multilineage differentiation potential with cytokine release.

The characterization of human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs). hUCB-MSCs differentiated into osteogenic (B), adipogenic (D), and chondrogenic (F) lineages. The undifferentiated controls are shown in (A), (C), and (E), respectively. Scale bars: 200 μm. (G) The cumulative population doubling level (CPDL) were achieved more than 35 cumulative population doublings. (H) In a karyotype test, no chromosomal abnormalities were found.

hUCB-MSCs released various immumodulatory substance, neurotrophic cytokines, and growth factors. Released soluble factors of hUCB-MSCs were analyzed by cytokine array. The positive controls on membrane were used to normalize the signal intensity of individual cytokines. GRO, growth regulated oncogene; IL, interleukin; EGF, epidermal growth factor; IGF, insulin-like growth factor; VEGF, vascular endothelial growth factor; PDGF, platelet-derived growth factor; BDNF, brain-derived neurotrophic factor; FGF, fibroblast growth factor; Flt-3, FMS-like tyrosine kinase-3; GDNF, glial-derived growth factor; HGF, hepatocyte growth factor; LIF, leukemia inhibitory factor; NT, neurotrophin; PIGF, placental growth factor; TIMP, tissue inhibitor of metalloproteinases.
The Phenotype Determination of hUCB-MSCs Using Flow Cytometry

hUCB-MSCs Ameliorated Altered Cholesterol Metabolism in Cerebrum of NPC Mice
To determine whether the lysosomal cholesterol accumulation is ameliorated by engrafted hUCB-MSCs, filipin staining was performed in NPC mice. The quantitative analysis of the filipin-positive area in hippocampus, motor cortex, and striatum at postnatal day 56 revealed that hUCB-MSC-treated NPC mice showed significantly decreased cholesterol deposition (Fig. 3A, B). Each area occupied by filipin was 2.99 ± 0.5%, 2.05 ± 0.35%, 4.49 ± 0.85% in NPC mice, while hUCB-MSC-treated NPC mice had, respectively, 2.33 ± 0.59%, 1.12 ± 0.38%, and 2.56 ± 0.93% filipin-positive area (n = 3 per group). To explain what led to this reduction, we evaluated intracellular cholesterol transport molecules, LXRα and its target molecules, such as ABCA1 and ABCG5, which govern sterol transport metabolism. As shown in Figure 3C, mRNA levels of ABCA1, ABCG5, and LXRα were hardly detectable in NPC, whereas they were upregulated after hUCB-MSC treatment. Moreover, hUCB-MSCs could lower cholesterol synthesis by reducing protein expression of the rate-limiting enzyme of cholesterol synthesis, HMGR (Fig. 3D).
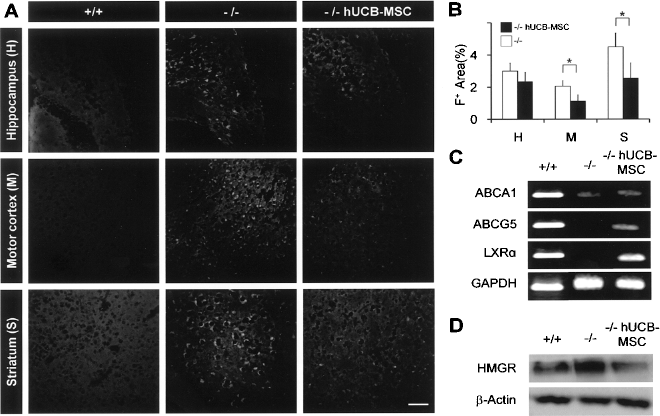
hUCB-MSCs restored the altered cholesterol metabolism and trafficking in Niemann Pick type C1 (NPC) mice. (A) The cholesterol accumulation was determined by anti-filipin staining images. Scale bar: 50 μm. (B) Quantitative analysis of the filipin-positive areas between untreated and hUCB-MSC-treated NPC mice (mean ± SD, *p > 0.05) (n = 3 for each category). (C) mRNA level of ATP-binding membrane cassette transporter A1 (ABCA1), ATP-binding membrane cassette transporter G5 (ABCG5), Liver X receptor α (LXRα), and GAPDH using RT-PCR. (D) Protein level of 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGR) using Western blot analysis. +/+, wild type mice; –/–, NPC mice; –/–hUCB-MSCs, hUCB-MSC-transplanted NPC mice.
The Engrafted hUCB-MSCs Improved Motor Deficits in NPC Mice
To evaluate motor function of the mice, the Rota Rod test was performed. The performance was assessed once per week for 3 weeks after transplantation. In every challenge, WT mice completed the task for 180 s (data not shown). As shown in Figure 4, hUCB-MSC-transplanted NPC mice could endure on the machine for 155.25 ± 25.89, 128.41 ± 22.26, and 89.83 ± 11.88 s at 7, 14, and 21 days posttransplantation, respectively, whereas lower records were obtained from the VC-NPC mice (91.86 ± 16.17, 74.38 ± 9.32, 41 ± 7.23 s) and untreated NPC mice (130.63 ± 24.5, 98.88 ± 17.46, 56.75 ± 15.38 s). These data suggest that hUCB-MSCs improved the impaired motor function of NPC mice.
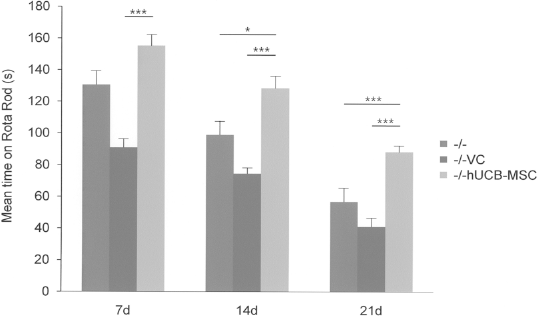
hUCB-MSCs improved motor function in NPC mice. Locomotion ability was assessed by the Rota Rod test. hUCB-MSC-transplanted NPC mice showed significant improvement (*p < 0.05; ***p < 0.001). The test was performed once per week for 3 weeks posttransplantation. d, days; –/–VC, NPC mice treated with vehicle (PBS).
Cerebellar Purkinje Cell Survival Was Alleviated by hUCB-MSC Transplantation
The cerebellum is one of the most severely affected areas in a murine model of NPC and Purkinje cells are exceedingly susceptible to neurodegeneration (35,40). Therefore, we determined whether transplanted hUCB-MSCs prevented Purkinje cell loss in the cerebellum of NPC mice. The H&E staining in Figure 5A showed a significant reduction in the number of Purkinje cells observed in both untreated and VC-NPC mice, while hUCB-MSCs prevented this Purkinje cell loss. This phenomenon was reconfirmed by immunohistochemistry and Western blotting using CBD as a Purkinje cell marker (Fig. 5B–D). We next investigated whether recruitment of Purkinje cells is associated with apoptotic and inflammatory signaling modulation. As depicted in Figure 5E, protein levels of proapoptotic Bax and the active form of caspase-3 were moderately attenuated and inflammatory molecules including induced nitric oxide synthase (iNOS), F4–80, and cycloxygenase 2 (COX2) were also decreased after hUCB-MSC transplantation. Our data suggest that hUCB-MSCs protect Purkinje cells by providing antiapoptotic and anti-inflammatory effects in NPC mice.
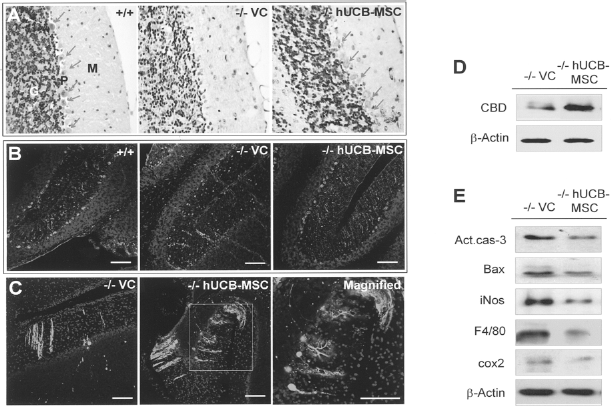
hUCB-MSCs prevented Purkinje cell loss and cerebellar inflammation. (A) Purkinje neurons are indicated by arrows in hematoxylin and eosin (H&E) staining. (B, C) Purkinje cells were labeled with anti-calbindin (CBD) in immunohistochemistry. Nuclei were counterstained with Hoechst 33258. Scale bars: 50 μm. (D) Protein level of CBD was determined by Western blot analysis. (E) Western blot analysis of activated caspase-3, Bcl-2 associated X-protein (BAX), inducible nitric oxide synthase (iNOS), F4/80, and cyclooxygenase2 (COX2) were decreased when the hUCB-MSCs were transplanted in NPC mice.
The hUCB-MSCs Drove Neurogenesis and Differentiated Into Functional Neurons
Because the dentate gyrus is known as a site of neurogenesis in the brain (23), we examined the influence of hUCB-MSCs on in vivo neurogenesis at 3 weeks after transplantation using immunohistochemical analysis. More DCX-positive neural progenitor cells were found in the dentate gyrus of the hUCB-MSC-treated NPC mice compared to the other groups (Fig. 6A). Further, protein levels of DCX and mature neuronal marker MAP2 were increased in hUCB-MSC-transplanted samples whereas the levels of GFAP were unchanged (Fig. 6B). To evaluate whether the phenotype was acquired by the transplanted cells, HuMi-positive cells were determined by immunohistochemistry. As shown in Figure 7A, HuMi revealed the presence of transplanted cells in the cortex and they coexpressed MAP2 immunoreactivity, whereas cells simultaneously expressing GFAP and HuMi were hardly detectable (Fig. 7B). We next determined whether transplanted hUCB-MSCs derived neurons had electrical and physiological activities using whole-cell patch clamp recordings of hippocampal slices. The recording site was the hippocampal CA1 region and PKH26-labeled hUCB-MSCs were visually identified (Fig. 7C, D). A train of action potentials was fired by injecting a depolarizing current (0.2 nA, 600 ms, upper panel in Fig. 7E) into the PKH26-labeled cells and it was blocked by 1 μM of tetrodotoxin (TTX), a voltage-dependent Na channel blocker (lower panel in Fig. 7E). In addition, miniature excitatory postsynaptic currents, recorded in labeled neurons tested at a holding potential of −70 mV, were blocked by 6-cyano-7-nitroquinoxaline-2, 3-dione (CNQX) and 2-amino-5-phosphopentanoic acid (AP5), specific antagonists for AMPA and NMDA receptors, respectively (Fig. 7F). These data suggest that transplanted hUCB-MSC-derived cells were able to differentiate into synapse associated with receptor-dependent neuronal cells.

hUCB-MSCs increased neural progenitor cells. (A) Doublecortin (DCX) + neural progenitor cells were determined by confocal microscope in dentate gyrus. Nuclei were counterstained with Hoechst 33258. Scale bar: 50 μm. (B) Proteins level of DCX at 3 weeks and microtubule associated protein 2 (MAP2) and glial fibrillary acidic protein (GFAP) at 4 weeks posttransplantation were determined for neural progenitor cells, mature neurons, and astrocytes, respectively, using Western blot analysis. The immunoblots were quantified by densitometry and normalized β-actin levels in the same lane. *p < 0.05; **p < 0.01. H, hippocampus; M, motor cortex; S, striatum.

hUCB-MSCs differentiated into mature neurons with functional synapse in NPC mice. (A) Asterisks indicate hUCB-MSCs labeled with anti-human mitochondria (HuMi). The neurons were labeled with anti-MAP2. Arrows indicate differentiated neurons derived from hUCB-MSCs. (B) Asterisks indicate hUCB-MSCs labeled with anti-HuMi and astrocytes were labeled with anti-GFAP in the cortex. Magnified image of square is also shown. Scale bars: 50 μm (A, B), 10 μm (magnified images). (C) PKH26-labeled testing cells (red) are shown around the transplantation site. Scale bars: 250 μm (left, upper box), 20 μm (lower box). (D) A picture of the cell injection by pipette into the pyramidal CA1 region of the hippocampus. Scale bar: 10 μm. (E) Action potentials in donor-derived neurons induced by depolarization (upper panel) and inhibited by tetrodotoxin (TTX, lower panel). (F) The miniature excitatory postsynaptic currents (upper panel) were abolished by 6-cyano-7-nitroquinoxalin-2, 3-dione (CNQX) and 2-amino-5-phosphopentanoic acid (AP5, lower panel).
hUCB-MSCs Promoted Neuronal Survival via Upregulation of PI3K/AKT and JAK2/STAT3 Pathways in NPC Mice
As we have shown that hUCB-MSCs contributed to neuronal protection in NPC mice, we examined several signaling pathways known to be involved in neuronal protection such as the PI3K/AKT and JAK2/STAT3 cascade to identify possible hUCB-MSC-induced neuroprotective mechanisms (9,16). By Western blot analysis, PI3K/AKT signaling pathway was shown to be activated, followed by GSK3β inactivation via phosphorylation at Ser9 residue after hUCB-MSC transplantation in both the cerebrum and cerebellum of NPC mice 4 weeks after transplantation (Fig. 8). Moreover, JAK2/STAT3 phosphorylation was also increased in hUCB-MSC-transplanted NPC mice. We next determined whether hUCB-MSCs could contribute to the protection of neurons in response to oxidative stress as described previously (17); neural stem cells were maintained in hUCB-MSC-derived conditioned medium (CM) and treated with hydrogen peroxide (H2O2). As we described previously (Fig. 8), CM rapidly induced PI3K/AKT activation in neural stem cells in a time-dependent manner and the pathway was also activated by CM following H2O2-induced oxidative stress (Fig. 9). To confirm the CM induced effect, LY294002, a potent inhibitor of PI3K, was applied to NSCs exposed to CM and H2O2 for the last 2 h, leading to decreased protein levels of PI3K and pAKT (Fig. 9). These data suggest that the PI3K/AKT and JAK2/STAT3 signaling pathways activated by hUCB-MSCs mediates their neuroprotective effect in NPC mice.

hUCB-MSCs promoted neuronal cell survival via upregulation of phosphoinositide3-kinase/AKT (PI3K/AKT) and Janus kinase2/signal transducers and activators of transcription protein3 (JAK2/STAT3) signaling pathways in NPC mice. Protein levels of each signaling-related molecules and β-actin were determined in cerebrum and cerebellum of NPC mice by Western blot analysis.

hUCB-MSC-derived conditioned medium activated PI3K/AKT pathway under the H2O2-induced oxidative stress state. Protein levels of PI3K and pAKT are determined by Western blot analysis. PI3K and pAKT levels of neural stem cells increased after conditioned medium (CM, 1 μg/ml) treatment in a time-dependent manner. Then H2O2-treated neural stem cells were cocultured with CM, with or without LY294002, a PI3K inhibitor, followed by Western blot. One representative from three independent experiments is shown.
hUCB-MSCs Restored Disrupted Homeostasis of Neurotransmitters
Previously, we have shown that the expression of glutamate and GABA transporter was altered in NPC mice compared with wild-type littermates (4). It has been shown that inadequate or unbalanced neurotransmitter levels can cause neurotoxicity. We therefore performed Western blot analysis to determine the effect of hUCB-MSCs in cellular homeostasis due to impaired transport in NPC mice. One of the GABA transporters, GAT1 protein level, was found to be decreased; however, intracellular glutamate transporter-related GAD6, EAAT2, and EAAT3 protein levels were increased in hUCB-MSC-transplanted NPC mice compared with VC-NPC mice (Fig. 10). These data suggest that hUCB-MSCs might modulate transporter expression to reduce excitotoxicity in the brain of NPC mice.

hUCB-MSCs modulated the GABA transporters in NPC mice. Protein levels of GABA transporter 1 (GAT1), excitatory amino acid transporter 2 (EAAT2), excitatory amino acid transporter 2 (EAAT3), and glutamic acid decarboxylase 65 (GAD6) were determined by Western blot analysis. Immunoblot was quantified by densitometry. *p < 0.05; **p < 0.01, ***p < 0.001.
Discussion
In this study, we investigated whether hUCB-MSCs could differentiate into neuronal cells and how hUCB-MSCs could protect against neuronal cell death in NPC mice. Lee and colleagues reported that bone marrow-derived mesenchymal stem cells (BM-MSCs) modulated sphingolipid metabolism of endogenous NPC Purkinje cells, resulting in their survival possibly due to the release of bioactive trophic factors from the BM-MSCs (26). Consistent with the effect of BM-MSCs in NPC mice, we demonstrated the dynamic role of hUCB-MSCs, via activation of the PI3K/AKT and JAK2/STAT3 signaling pathways, and their differentiation into functional neurons in cerebrum, which promoted Purkinje cell survival in the cerebellum. Recently, we found that the transcription factor REX1 regulates the proliferation/differentiation of human MSCs through the suppression of p38 mitogen activated protein kinase (MAPK) signaling via the direct suppression of mitogen activated protein kinase kinase 3 (MKK3) (2). Moreover, decreased histone deacetylase (HDAC) activity is important for MSC self-renewal by balancing polycomb group genes and jumonji domain containing 3 expression (14). These findings suggest that transcriptional regulation and chromatin modification might define the cellular microenvironment in which a deficiency of NPC1 leads to disease endpoints.
Glutamate toxicity has been posited to play a role in AD, and reduced glutamate transporter and glutamate uptake were decreased in brains from patients with AD (32). Na+-dependent transporters for glutamate exist on astrocytes (EAAT2 and GAD6) and neurons (EAAT3). EAAT2 could be covalently modified by a lipid peroxidation product, 4-hydroxynonenal, and EAAT2 immunoprecipitated from AD brain had significantly higher levels of 4-hydroxynonenal modified protein (25). In this study, we have described that a deficiency of NPC1 decreased the levels of EAAT2, GAD6, and EAAT3, and hUCB-MSCs increased the levels of glutamate transporters, suggesting that hUCB-MSCs might maintain the glutamate concentration to reduce neuronal excitotoxicity. Further research is needed to evaluate the modulation of glutamatergic signaling in hUCB-MSCs transplanted NPC mice.
Cholesterol transport in the brain is essential for various functions including neuronal survival and the synthesis of neurosteroids. Aberrant cholesterol transport is linked to the early onset of NPC and the lipid accumulation has been detected in neurons of the cerebral cortex, cerebellum, and hippocampus (38). In this study, we have shown that hUCB-MSCs ameliorated altered cholesterol metabolism in the hippocampus, motor cortex, and striatum of NPC mice by increasing cholesterol efflux of transporters such as ABCA1, ABCG5, and their upstream receptor, LXR, and by decreasing HMGR to reduce cholesterol. Recently, Karasinska et al. reported that ABCA1-B/-B mice have reduced plasma high-density lipoprotein (HDL) cholesterol levels associated with decreased brain cholesterol content and enhanced brain uptake of esterified cholesterol from plasma HDL, leading to motor and sensorimotor behavioral and synaptic changes (15). Such an exaggerated cholesterol transport mechanism has been described in NPC mice. Oral administration of LXR agonist slows the progress of NPC symptoms through the activation of ABCA1 and low-density lipoprotein receptor cholesterol transporters (39). Therefore, our findings suggest that cholesterol efflux transporters might play a critical role in regulating cholesterol flux uptake and it might be associated with Purkinje cell loss due to mistaken cholesterol demand in the endosomal/lysosomal compartment.
With the results of the present study, we have shown that the PI3K/AKT signaling cascade and phosphorylation of GSK3β were increased in hUCB-MSC-transplanted NPC mice. In addition, we found that the changes in the PI3K/AKT pathway occurred concurrently with disturbances in lipid storage and cholesterol deposits as detected by filipin staining and Purkinje cell loss. Moreover, a sustained increase in the levels of GSK3β through tau hyperphosphorylation has previously been shown in neurofibrillary tangles in AD, suggesting that phosphorylation of GSK3β might occur in anticipation of tangle formation in NPC. Previously, we have shown that self-renewal of NSCs was increased by inhibition of p38 MAPK and ERK1/2 (45) and the inhibition of nitric oxide restored the self-renewal of NSCs via inhibition of both GSK3β and caspase-3 in NPC mice (20). Hence, our studies suggest that several kinases including p38 MAPK and GSK3β might precede tangle formation in NPC mice.
Transplantation of MSCs has attracted interest because of the harnessing of their immunosuppressive properties. It has been shown that MSCs inhibit the activation and proliferation of T cells, mediated by various soluble factors, including transforming growth factor-β, prostaglandin E2, and indoleamine 2,3-dioxygenase in vitro (8). However, further study is required to investigate the molecular mechanisms to regulate the immune response during transplantation.
Taken together, hUCB-MSCs ameliorated altered cholesterol metabolism by activation of cholesterol transporters, increased neuronal survival via activation of PI3K/AKT and JAK2/STAT3 signaling, and prevented excitotoxicity in NPC mice. Moreover, hUCB-MSC transplantation significantly increased neuronal progenitors, which differentiated into functional neurons in NPC mice. The transplanted hUCB-MSCs implied beneficial effects in preventing neuronal cell loss in the cerebrum and promoting Purkinje cells in the cerebellum, and eventually these effects improved motor function in NPC mice. Therefore, hUCB-MSCs could be a beneficial source for stem cell therapy, and these studies could lead to a better understanding in a variety of neurodegenerative diseases.
Footnotes
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MEST, 2010-0020265) and the 21st Century Frontier/Stem Cell Research Committee (SC5110).
