Abstract
Following spinal cord injury (SCI), both an inhibitory environment and lack of intrinsic growth capacity impede axonal regeneration. In a previous study, prevention of cyclic adenosine monophosphate (AMP) hydrolysis by the phosphodiesterase-4 inhibitor rolipram, in combination with Schwann cell (SC) grafts, promoted significant supraspinal and proprioceptive fiber growth and/or sparing and improved locomotion. In another study, transplanted SCs transduced to generate a bifunctional neurotrophin (D15A) led to significant increases in graft SCs and axons, including supraspinal and myelinated axons. Here we studied the growth and myelination of local and supraspinal axons and functional outcome following the combination of rolipram administration and neurotrophin-transduced SC implantation after SCI. Rolipram was administered subcutaneously for 4 weeks immediately after contusion at vertebral T8 (25.0-mm weight drop, MASCIS impactor). GFP or GFP-D15A-transduced SCs were injected into the injury epicenter 1 week after SCI. GFP-D15A SC grafts and GFP SC grafts with rolipram contained significantly more serotonergic fibers compared to GFP SCs. SC myelinated axons were increased significantly in GFP SC with rolipram-treated animals compared to animals receiving SCI alone. Rolipram administered with either GFP or GFP-D15A SCs significantly increased numbers of brain stem-derived axons below the lesion/implant area and improved hindlimb function. Compared to the single treatments, the combination led to the largest SC grafts, the highest numbers of serotonergic fibers in the grafts, and increased numbers of axons from the reticular formation below the lesion/implant area and provided the greatest improvement in hindlimb function. These findings demonstrate the therapeutic potential for a combination therapy involving the maintenance of cyclic AMP levels and neurotrophin-transduced SCs to repair the subacutely injured spinal cord.
Keywords
Introduction
The propensity of Schwann cells (SCs) to promote axonal regeneration in the peripheral nervous system (PNS) has led to experiments to test their ability to foster axonal regrowth in the central nervous system (CNS) (28,32,33). When SCs alone are implanted acutely into a complete transection gap in adult rat spinal cord, axons grow onto the SC bridge from both stumps, a mean of 2,000 myelinated axons is found on the bridge, and there are eight times more accompanying nonmyelinated (ensheathed) axons as well (46). In order to attain more regenerative growth, particularly from supraspinal axons, and to cause regenerated fibers to leave the bridge and grow into the neighboring spinal cord, investigation of combination strategies was initiated in this complete transection model. Combinations of SCs with methylprednisolone (10), neurotrophins presented in varying ways (5,25,47), olfactory ensheathing cells (OECs) (38), or OECs plus chondroitinase (14) all led to improved results. Overall, these results were higher numbers of myelinated axons on the bridge, increased numbers of regenerated axons from brain stem neurons on the bridge, exit of regenerated axons from the bridge into the spinal cord, and a significant improvement in locomotion (14).
Likewise, when combination strategies with SCs were tested in a rat spinal cord contusion model, the results were improved over the implantation of SCs only. With the introduction of SCs into contusion sites, the cavity formation that follows contusion is reduced, about 5,000 SC-myelinated axons are observed in the implant, significantly more axons (from spinal and supraspinal neurons) are present below the graft (35,42) and there is modest locomotor improvement (37,42). In a study of SCs transduced to secrete a bifunctional neurotrophin molecule (D15A) (43) with both neurotrophin-3 (NT-3) and brain-derived neurotrophic factor (BDNF) activity, there were up to fivefold increases in graft volume, SC number, and SC-myelinated axon counts (16). Also, there was a threefold increase in myelinated-to-unmyelinated axon ratios, revealing an enhancement of myelination. Lengths of axons from a number of brain stem neurons as well as sensory axons were increased fivefold within D15A grafts. Most notably, the total number of axons was estimated to be over 70,000 in the SC/D15A implants (16).
Another combination strategy that led to both significant histological and behavioral findings following contusive spinal cord injury (SCI) were SCs paired with elevation of cyclic adenosine monophosphate (cyclic AMP) (36). This intervention involved subcutaneous administration of rolipram, a phosphodiesterase-4 inhibitor, introduction of SCs into the contusion epicenter, and the injection of an analog of cyclic AMP above and below the SC implantation site. This strategy led to increased levels of cyclic AMP in the brain and spinal cord rostral to the implant, reversing the reduction that occurs after injury. The outcome was that lateral white matter sparing was increased 2.3-fold, myelinated axons in the graft were more numerous (fivefold), serotonergic fiber growth into and beyond the grafts was promoted, more axons from the reticular formation (125%) and red (73%) and raphe (42%) nuclei were present below the level of the implant, and functional recovery was very significantly improved as assessed by a number of behavioral tests (36). Whereas these results were obtained with SC transplants, rolipram administration, and the injection of the cyclic AMP analog, there were significant improvements in most of the parameters mentioned above, including aspects of locomotion, with the implantation of SCs plus rolipram without cyclic AMP injections. Total numbers of axons in the grafts were estimated to be 40,000 for the triple combination and 20,000 for the SC-implanted, rolipram-receiving animals.
It was the goal of the work reported herein to combine the transplantation of SCs transduced to generate D15A (to increase substantially the number and myelination of axons in the implant) with the administration of rolipram (which led to improvement in motor ability). Here we report that this full combination led to the largest SC grafts, the greatest amount of serotonergic fiber growth within the grafts, and the most improved locomotor function compared to the individual treatments (D15A SCs or rolipram with GFP SCs), as well as SC-only and SCI-only controls. Engineering of SCs to produce neurotrophic molecules in combination with agents that elevate cyclic AMP appears to provide a more effective approach for SCI repair.
Materials and Methods
Schwann Cell Culture
SCs were obtained from sciatic nerves of adult female Fischer rats (Harlan Co., Indianapolis, IN, USA) as described previously (26) but with additional modifications as later detailed in Meijs et al. (24). Briefly, nerves were cut into 1-mm explants, shifted to new dishes three times to remove fibroblasts, enzymatically and mechanically dissociated before culturing in Dulbecco's modified Eagle's medium (DMEM; Invitrogen, Carlsbad, CA, USA)/10% fetal bovine serum (FBS; Atlanta Biologicals, Inc., Flowery Branch, GA, USA) containing three mitogens [1 μM forskolin (Sigma-Aldrich, St. Louis, MO, USA), 20 μg/ml bovine pituitary extract (Biomedical Technologies, Inc., Stoughton, MA, USA), 2.5 nM heregulin (Genetex, Irvine, CA, USA)], and subjected to Thy 1.1 antibody (ATCC, Manassas, VA, USA) and complement (Sigma-Aldrich) to remove any remaining fibroblasts (16). SCs were passaged to polylysine-coated dishes (Sigma-Aldrich), fed mitogen-containing medium, and infected with virus (see below). Their purity for grafting, by a method described earlier (42), was 95–98%.
Lentiviral Vector Transduction of SCs
Vector preparation by the Miami Project Viral Vector Core was performed as previously described (4,13). Briefly, the genes coding for enhanced green fluorescent protein (EGFP) or the bifunctional neurotrophin molecule human D15A (43) (gift of Dr. Pantelis Tsoulfas, The Miami Project) were subcloned into a lentiviral vector plasmid, P156RRLsinPPThCMVMCSpre (generously provided by Dr. L. Naldini, University of Torino). For ease of use, EGFP will be subsequently referred to as simply GFP in the ensuing sections. This plasmid contained the cytomegalovirus (CMV) promoter to drive transgene expression and the woodchuck posttranscriptional regulatory element (WPRE) to enhance mRNA transport. Transfection by plasmids and viral harvesting were conducted in cultured 293T cells (ATCC) (2). Virus was concentrated by ultracentrifugation at 20,000 × g and resuspended in phosphate-buffered saline (PBS BDH/VWR, Radnor, PA, USA). After this, the viral vectors were titered for transducing units either with 293T cells or by using an enzyme-linked immunosorbent assay (ELISA; Perkin Elmer, Wellesley, MA, USA) quantifying p24 core protein concentrations, according to the manufacturer's instructions. SCs were transduced with LV GFP or LV GFP-D15A. Purified viral vector stocks were stored at −80°C until SC infection.
Virus concentrated in PBS was used to infect SCs. Cultures were infected by the addition of a predetermined volume of concentrated virus in DMEM/10% FBS/mitogens (2). A multiplicity of infection (MOI) of 30 was used in all cultures to ensure high transduction efficiency without toxicity. The infected SCs were passaged two more times; 95–98% of the SCs were infected. These cells were harvested by trypsinization (Invitrogen), centrifuged, resuspended, and counted. The SCs used in these experiments were from the same SC stock as those used in the Golden et al. study (16). After counting, SCs were resuspended in DMEM/F12 (Invitrogen) and kept on ice for no longer than 2 h before implantation.
Spinal Cord Contusion and SC Transplantation
Adult female Fischer rats (Harlan Co., 180–200 g; n = 105) were housed two per cage according to NIH guidelines for the care and use of animals. The University of Miami Animal Care and Use Committee approved all animal procedures. Prior to surgery, rats were anesthetized [45 mg/kg ketamine (Vedco, Inc., St. Joseph, MO, USA), 5 mg/kg xylazine (Lloyd, Inc., Shenandoah, IA, USA)] by intraperitoneal injection. A laminectomy performed at thoracic vertebra T8 exposed the dorsal surface of the spinal cord underneath (T9) without disrupting the dura mater. The exposed spinal cord was contused using the Multicenter Animal Spinal Cord Injury Study (MASCIS) weight drop device (New York University, New York, NY, USA) (17) by dropping a 10.0-g rod from a height of 25.0 mm. The contusion impact height, velocity, and compression were monitored. Animals (n = 5) were excluded immediately when height or velocity errors exceeded 7% or if the compression distance was not within the range of 1.75–2.25 mm. After injury, the muscles were sutured (Ethicon, Somerville, NJ, USA) in layers, and the skin was closed with metal wound clips (Fine Science Tools, Inc., Foster City, CA, USA).
Rolipram (Sigma-Aldrich) was delivered subcutaneously from two 2001 osmotic minipumps (Durect Corp., Cupertino, CA, USA) implanted in pockets within the rear flanks of the animal, starting within 30 min of injury (1 week delivery at 1.0 μl/h). Each pump contained 0.5 mg/kg rolipram in 1:1 ethanol/physiological saline. The animals tolerated this vehicle better than dimethyl sulfoxide (DMSO) (36). Pumps were exchanged weekly to permit delivery for 4 weeks. The rats were allowed to recover in a warmed cage with water and food, which were easily accessible. Gentamicin (5 mg/kg, intramuscular; Abbott Laboratories, North Chicago, IL, USA) was administered immediately postsurgery and then daily for 7 days. The analgesic Buprenex (0.3 mg/kg, SC; Reckitt Benckiser, Richmond, VA, USA) was delivered post-surgery and daily for 2 days. Lactated Ringer's solution (5 ml; 2×/day; Hospira, Inc., Lakeforest, IL, USA) was administered the first day and as needed thereafter. Bladders were expressed 2×/day until voiding returned. During the postoperative period, 10 animals exhibited either early morbidity or pump delivery failure, and thus data were not obtained from these animals.
At 1 week postinjury, rats were reanesthetized for exposure of the contusion site and injection of 2 million SCs into the injury center; SCs were in a 1:1 mix of SCs/DMEM/F12 and Matrigel (BD Biosciences, San Jose, CA, USA) for an 8-μl total volume as performed previously (29). Injections were performed using a 10-μl siliconized Hamilton syringe (Hamilton, Reno, NV, USA) with a pulled, beveled glass pipette tip (120 μm diameter; VWR), held in a micromanipulator with a microinjector at a rate of 2 μl/min (World Precision Instruments Ltd., Sarasota, FL, USA). The injection pipette was kept in place for an additional 3 min to minimize leakage upon withdrawal. Animals were assigned randomly into the following groups (Table 1): (i) injury + injection of 1:1 DMEM/F12/ Matrigel without cells (n = 18); (ii) injury + transplantation of LV/GFP SCs (n = 22); (iii) injury + transplantation of LV/D15A-transduced SCs (n = 16); (iv) injury + transplantation of LV/GFP SCs with rolipram (n = 18); and (v) injury + transplantation of LV/D15A SCs with rolipram (n = 16). The D15A SCs were double infected, first with GFP and then a second time with D15A, as performed previously (16). After the injections, the muscle layers and the skin were closed separately, and the animals received postoperative care as performed in Golden et al. (16).
Number of Animals Used for Each Outcome Measure

Behavioral testing was performed on all animals in each group, the number represented in brackets. GFP, green fluorescent protein; SC, Schwann cell; D15A, bifunctional neurotrophin molecule.
Retrograde Tracing
At 13 weeks postinjury, after endpoint behavioral testing, a subgroup of animals from each treatment cohort (n = 6–8/cohort) was retrogradely traced with 2% Luxol Fast blue (Sigma-Aldrich). Fast blue was injected at 7 mm distal to the caudal edge of the original laminectomy at four sites (0.5 μl per site) at depths of 0.5 and 1.0 mm, bilaterally. During the injection procedure, small pledgets of gelfoam (Pfizer, Inc., New York, NY, USA) were placed over the top of the injection sites so as to prevent any leakage of the dye. Injections of Fast blue were made far caudal to the implant/injury site so as to ensure that the dye would only label fibers beyond the implant (47). Perfusion and sectioning are described in the next section. Fast blue-labeled neuronal somata were counted in the reticular formation, red, raphe, and vestibular nuclei, and the motor cortex.
For analysis of Fast blue-labeled neuronal somata, sections through the brain (400-μm intervals) were first imaged using an Olympus BX51 microscope (Olympus America Inc., Center Valley, PA, USA) and Stereo Investigator's meander scan software (MicroBrightField, Williston, VA, USA) at 600× under oil. Labeled neuronal somata were counted according to bregma level using reference histological preparations from Paxinos et al. (31) and then digitally overlaid with corresponding bregma maps for quantitative analysis. Counts in specific neuroanatomical regions were normalized to volume using the formula: sample count × (neuroanatomical region thickness/section thickness) (34). The Abercrombie correction formula (1) was employed to correct for overcounting and to give final retrogradely labeled neuron counts as shown; section thickness/(section thickness + mean diameter of the counted object) where the section thickness was 20 μm and the object diameter, the average neuronal cell somata, was determined to be 15.26 μm (reticular formation), 16.78 μm (vestibular nuclei), 14.21 μm (raphe nuclei), or 15.06 μm (total brain stem) from the sampling of 20 labeled cells (19).
Histology
At 13 weeks after injury (12 weeks posttransplantation), all rats were deeply anesthetized (70 mg/kg ketamine, 10 mg/kg xylazine) and transcardially perfused with 4% paraformaldehyde (PFA, 0.1 M, pH 7.4; Sigma-Aldrich). The spinal cord and brain were removed and postfixed overnight in 4% PFA at 4°C. For cryostat sectioning and immunochemical staining, the PFA-fixed tissue was transferred to phosphate-buffered 30% sucrose (Sigma-Aldrich) for 48 h at 4°C. The T7–11 thoracic spinal cord (20-mm-long piece), which contained the complete lesion and graft site, was sagittally sectioned at 20 μm on a cryostat. Sections were collected into five series and stored at −20°C until further processing; the interval between sections was 100 μm. Brains were sectioned coronally at 20 μm into five series for analysis of retrogradely labeled neuronal somata; as every fourth section was examined, the interval between sections was 400 μm.
For counting central (spared)- and SC-myelinated (graft) axons in 1 μm toluidine blue (Sigma-Aldrich)-stained plastic sections, a 1-mm slice from the injury center from a subgroup of animals (n = 3–6/cohort) was prepared by electron microscopic techniques for plastic embedment following the initial PFA fixation (48). After sectioning and staining, counts were obtained from three 1-μm sections per animal.
Immunochemistry
Every other fifth sagittal section was immunochemically stained for fluorescence microscopy as previously described (16,41). The primary antibodies used were a mix of monoclonal antibody anti-glial fibrillary acidic protein (GFAP, SMI-22R, 1:1,000; Covance Inc., Emeryville, CA, USA; cat. #SMI-22R) and anti-serotonin [5-hydroxytryptamine (5-HT), 1:5,000; Immunostar Corp., Hudson, WI, USA; cat. #20080]. The GFAP antibody was raised against purified protein derived from the Bigner-Eng clones MAb1B4, MAb2E1, and MAb4A11 and visualizes immunostained astrocytes and astrocytic processes, as well as Bergman glia (12). The 5-HT antibody was generated in a rabbit against serotonin coupled to bovine serum albumin with paraformaldehyde. Quality control testing (Immunostar) has shown that this antibody reacts with serotonin but does not cross-react with 5-hydroxytryptophan, 5-hydroxyindole-3-acetic acid, and dopamine. The fluorescence secondary antibodies were a mix of Alexa 660-conjugated donkey anti-mouse (1:200; Invitrogen, Carlsbad, CA, USA) and Alexa 594-conjugated goat anti-rabbit (1:200; Invitrogen). Sections were mounted onto Snowcoat X-tra slides (Surgipath, Winnipeg, Manitoba, Canada) and coverslipped with Vectashield mounting medium (Vector Laboratories, Inc., Burlingame, CA, USA) containing the Hoechst nuclear dye and kept at 4°C.
Determination of Graft Volume and 5-HT Fiber Length
Graft volumes were estimated by 3D reconstruction of serial sections using Neurolucida software (MBF BioScience, Williston, VT, USA) as described elsewhere (16). The graft area in each 20-μm sagittal section (at intervals of 200 μm) was delineated by GFAP antibody and GFP SC fluorescence. Total fiber lengths of 5-HT-immunostained fibers in grafts were obtained using computer-assisted microscopy and stereological analysis via the isotropic virtual planes module (MicroBrightField Inc.). Random, blinded analysis at 600× was performed using an unbiased counting frame superimposed over the graft area (counting frame area, 3,600 μm2; sampling box height, 18 μm; top and bottom guard zones, 1 μm each; distance between isotropic planes, 15 μm; three mutually orthogonal orientations; sampling intervals, x = 175 μm, y = 350 μm). For each sampling site, the beaded axon morphology of each 5-HT-immunostained axon was first identified within the counting frame under fluorescent microscopy. Usually, zero to four distinct 5-HT axon profiles were visible within each counting frame. Next, a virtual plane was dynamically mapped inside this region by a sampling line that moved across the screen as the focal plane was moved through the section. Intersections between the sampling line, representing the isotropic virtual plane, and 5-HT axon profiles in focus were counted. The computer systematically selected a new isotropic random orientation of the virtual plane for each sampling site. The random orientation of the virtual planes allowed the use of the classical stereological formula to estimate length density, LV = 2QA, where QA is the number of transects of axon per unit test area. Total 5-HT axon length within the implant/lesion was then automatically computed by the Isotropic Virtual Planes software (MBF BioScience) based upon stereological formulae (44).
Image Processing
Images were taken using an Olympus BX51 microscope and meander scan software (MicroBrightField Inc.). The background was set to a single color (black or white), and the tonal range and sharpness (smart sharpen, 1.9 pixels) of the Tiff files were then normalized in range using Adobe CS2 (Adobe Systems Inc., San Jose, CA, USA).
Behavioral Testing
Gross hindlimb performance was evaluated in all animals weekly from 1 to 12 weeks postimplantation using the Basso, Beattie, Bresnahan locomotor rating scale (BBB) (3). A BBB subscore also was obtained as the recovery of some animals did not follow the progression of the BBB score, as described elsewhere (37). Footprint analysis was performed at endpoint using a procedure modified from de Medinaceli and colleagues (11,35). Deficits in descending motor control were examined by testing the animals on a custom-made horizontal grid-walk apparatus with irregularly spaced rungs (35). All behavioral assessments were performed by two individuals blinded to the animals' treatments.
Statistical Analysis
One-way ANOVA followed by the Bonferroni post hoc test was used for comparing results of axon lengths after immunohistochemical detection of 5-HT fibers, graft volumes, retrogradely traced neurons, and endpoint functional tests. A mixed factorial (repeated measures) ANOVA followed by the Tukey–Kramer test was used for comparison of weekly functional recovery patterns after injury. Differences were accepted to be statistically significant at p < 0.05, p < 0.01, and p < 0.001, compared to injury controls. All errors are given as standard errors of the mean.
Results
Graft Volumes Are Most Increased in the D15A-Rolipram SC Combination Group
The data revealed significant differences in graft volumes among treatment groups. For comparison of total graft volumes among the treatment groups by ANOVA, a significant treatment effect compared to GFP SCs alone was observed [F(3, 60) = 12.65, p < 0.001]. Compared to GFP SCs alone, the other three treatment groups (GFP-D15A SCs, GFP SCs + rolipram, and GFP-D15A SCs + rolipram) showed significant increases in SC implant size in immunostained preparations (Fig. 1A–E), in reconstructed images (Fig. 1F, G), and in the measured graft volumes {GFP-D15A SCs, 0.66±0.06 mm3 compared to 2.31±0.16 mm3, a 350% increase [t(63) = 34.72] GFP SCs + rolipram, 1.89±0.10 mm3, a 286% increase [t(63) = 28.73]; and GFP-D15A SCs + rolipram, 3.31±0.18 mm3, a 502% increase [t(63) = 60.71], respectively; one-way ANOVA, p < 0.001 for all comparisons} (Fig. 2). The combination of rolipram with D15A SCs resulted in markedly larger graft volumes than the GFP-D15A SC and GFP SC + rolipram groups by 1.4-fold [t(63) = 20.42, p < 0.001] and 1.8-fold [t(63) = 31.61, p < 0.001], respectively (Fig. 2). Analysis by 3D reconstructions of SC grafts further confirmed that the combination grafts of GFP-D15A + rolipram (Fig. 1G) exhibited significantly larger volumes than the GFP SC grafts alone (Fig. 1F).

GFP SC grafts were largest with the combination treatment. (A–E) Photomicrographs of the Schwann cell (SC) grafts (GFP, green fluorescent protein; green) that fill the injured host spinal cord as indicated by staining for astrocytes using glial fibrillary acidic protein (GFAP, red). The groups depicted are (A) injury control, (B) GFP SC graft, (C) GFP SC graft with rolipram administration, (D) GFP-D15A SC graft, and (E) GFP-D15A SC graft with rolipram administration. The graft was largest in group E. Scale bar: 500 μm. (F and G) Reconstructions emphasize the increased volume of the GFP-D15A + rolipram graft (G) compared to the GFP SC implant (F). D15A, bifunctional neurotrophin molecule.
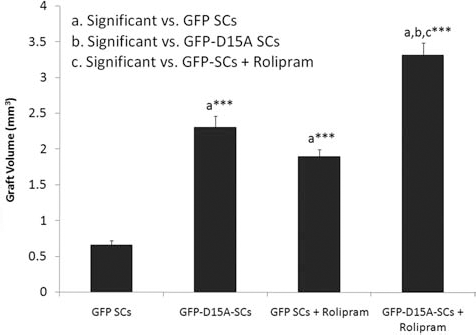
D15A- and rolipram-treated groups led to significant increases in graft volumes. GFP-D15A SC implantation plus rolipram administration resulted in graft volumes that were significantly larger than those containing GFP SCs, GFP-D15A SCs, and GFP SCs in combination with rolipram. Both GFP SCs + rolipram and GFP-D15A SC groups were larger than GFP SC controls. A statistically significant difference was accepted at values of p < 0.001 compared to either the GFP SC-only group or the individual treatment groups as indicated (by a, b, or c).
Central Oligodendrocyte and SC-Myelinated Axon Counts Are Not Enhanced by the D15A-Rolipram SC Combination Treatment
In low-magnification photographs of plastic sections (Fig. 3, top row), SC transplants are shown to reduce cavitation dramatically, particularly within the combination group. Myelinated axon counts were performed at higher magnification (Fig. 3, bottom row). For central oligodendrocyte-myelinated axons (CMAs), counted to assess white matter sparing, we found a significant treatment (all groups) effect compared to SCI [F(4, 18) = 3.070, p < 0.05]. CMA preservation in peripheral white matter surrounding the SC implants was not statistically increased with rolipram administration {GFP SCs + rolipram, 27,500±13,600 axons [t(22) = 2.032, p = 0.057]; GFP-D15A SCs + rolipram, 28,200±12,500 [t(22) = 2.106, p = 0.054]} compared to GFP SCs (5,190±1,430) and GFP-D15A SCs (4,300±2,440) without rolipram (Fig. 4A).

Cavitation, diminished by SC implantation, was further reduced or eliminated by rolipram. The implantation of SCs (either GFP or GFP-D15A) diminished cavitation. The boxed areas are shown at higher magnification in the next two rows. The middle row (dotted squares) illustrates peripheral regions of the spinal cord where central (spared) myelinated fibers were counted (see Fig. 4A.) The myelinated axons are seen as small dark rings. The bottom row (solid squares) illustrates graft areas where SC-myelinated axons were counted (see Fig. 4B). Toluidine blue-stained 1-μm plastic sections. C, cavity. Scale bars: 500 μm (top row), 20 μm (middle and lower rows). CMA, central myelinated axons; PMA, peripheral (SC)-myelinated axon.
The presence of D15A in SC implants did not lead to more myelinated axon counts than observed following implantation of GFP SCs or GFP-D15A SCs plus rolipram, a surprising finding. Rolipram was the most important component of the combination strategy in increasing white matter sparing or myelinated axon content in the SC graft.
A significant effect of cell implantation on SC-myelinated axon number was seen compared to SCI [F(4,18 = 5.929, p < 0.01]. These counts were highest in GFP SC implants with rolipram treatment (4,850±1,090 axons) compared to the injury control group (1,240±440 axons), a 3.9-fold increase [t(22) = 4.822, p < 0.01] (Fig. 4B).
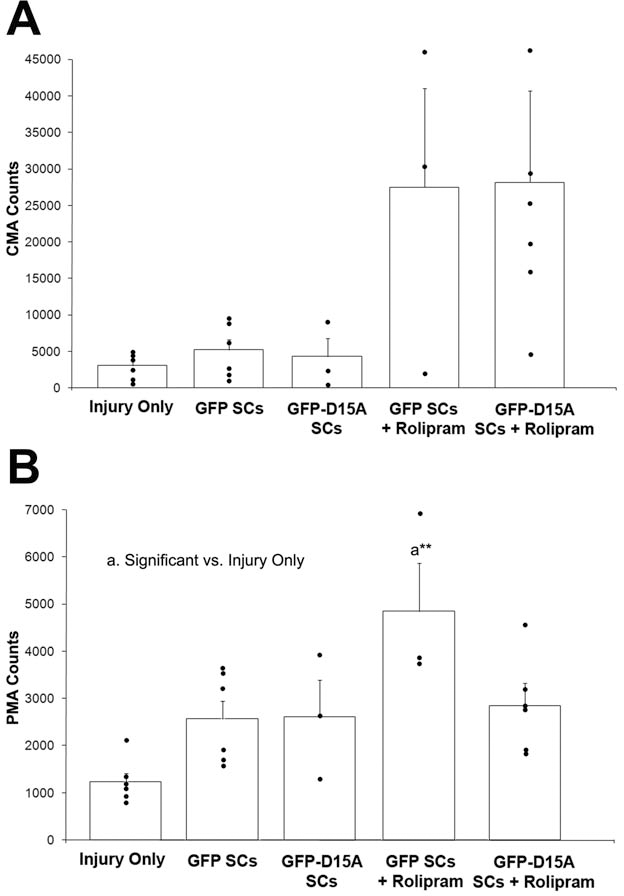
Central (spared) and SC-myelinated (graft) axon counts increased with D15A or rolipram treatment. (A) There was no significant difference among treatment groups for the central myelinated axon (CMA) counts. (B) There was a significant difference in SC-myelinated axon (PMA) counts, however, between the GFP SC with rolipram group compared to the injury control group (**p < 0.01).
An Additive Increase in Axon Supraspinal Sparing/Growth Occurs When Rolipram Is Combined with D15A SCs
Whereas we could not distinguish regenerated from spared axons in a contusion injury, we could assess the presence of specific types of axons caudal to the lesion/implant area by injecting a retrograde tracer caudally and counting labeled neuronal somata in the brain stem following transport in axons that are present in the caudal site. Following implantation of either GFP or GFP-D15A SCs alone, limited (<500) labeled neuronal somata were found in the reticular formation and raphe and vestibular nuclei. A significant treatment effect of combinatory cell implants was seen compared to SCI for numbers of retrogradely traced neurons in all brain stem regions: the reticular formation [F(4, 25) = 32.16, p < 0.001], the raphe [F(4, 25) = 16.88, p < 0.001], and the vestibular nuclei [F(4, 25) = 9.518, p < 0.001]. Figure 5A illustrates the presence of labeled somata in these areas when rolipram was administered with D15A SC implantation in contrast to significantly fewer labeled somata in GFP SC-transplanted animals. Figure 5B displays the numbers and positions of the labeled neuronal somata (dots) within a single, representative brain stem coronal section at bregma −11.96. The GFP SC + rolipram treatment led up to a 28-fold greater number of total labeled neurons in the above areas compared to the injury control, GFP SC-only, or GFP-D15A SC groups {comparison vs. GFP SCs: reticular formation [t(29) = 5.122, p < 0.001], raphe nuclei [t(29) = 4.289, p < 0.01], vestibular nuclei, no significance} (Fig. 6). The GFP-D15A SC + rolipram group exhibited up to a 40-fold greater number of labeled somata compared to these same control groups {comparison vs. GFP SCs: reticular formation [t(29) = 8.820, p < 0.001], raphe nuclei [t(29) = 6.148, p < 0.001], vestibular nuclei [t(29) = 5.042, p < 0.001]}. Again, rolipram was the most important part of the combination strategy. Only in the reticular formation was there a significant improvement of GFP-D15A SCs + rolipram treatment over GFP SCs + rolipram. Animals from all groups showed negligible (less than five) numbers of retrogradely labeled neurons in the M1/2 motor cortices (data not shown).

Supraspinal neuronal somata were labeled following introduction of tracer caudal to the lesion/implant region in the rolipram groups. (A) Fluorescent images show retrogradely labeled neuronal somata in the reticular formation and in the raphe and vestibular nuclei. The top row, depicting these areas in animals implanted with GFP SCs, shows few Fast blue-labeled neurons in these regions. The second row, however, demonstrates the presence of many more labeled neuronal somata in these areas of animals receiving GFP-D15A SCs with rolipram. Scale bar: 50 μm. (B) The number and position of the Fast blue-labeled neuronal somata are shown here by the green (reticular formation), yellow (raphe nuclei), and pink (vestibular nuclei) dots. Quantification of the tracing is displayed in Figure 6.

Administration of rolipram significantly increased the number of brain stem neuronal somata labeled by retrograde tracing from areas below the lesion/implant. The effect of rolipram to increase axonal sparing and/or growth is shown by retrograde tracing with Fast blue; the total numbers of neurons in the reticular formation and the raphe and vestibular nuclei are significantly greater with rolipram administration compared to the treatment groups lacking rolipram. In addition, labeling in the reticular formation was significantly increased in the GFP-D15A SC + rolipram group compared to the GFP SC + rolipram group. A statistically significant difference was accepted at values of ***p < 0.001 or **p < 0.01 compared to either the GFP SC-only group or the individual treatment groups as indicated (by a, vs. injury only; b, vs. GFP SCs; c, vs. GFP-D15A-GFP SCs; d, vs. GFP SCs + rolipram).
Serotonergic Axon Length Within SC Grafts Is Further Increased by Combining D15A and Rolipram
It is clear from 5-HT antibody staining that there were more serotonergic fibers in the full combination treatment than in the GFP SC implantation alone group (Fig. 7A, B). Statistical analysis revealed that the application of a combination treatment led to significant effects on the amount of serotonergic axon length within the implant compared to GFP SCs alone [F(3, 60) = 88.58, p < 0.001]. Axon length measurement using the isotropic virtual planes technique showed that the presence of serotonergic axons in SC grafts was increased in the GFP-D15A- and rolipram-treated SC groups (Fig. 7C). Compared to GFP SCs alone (21.54±3.83 mm), there was a 14.5-fold increase in 5-HT axonal length within the SC grafts with D15A {306.67±28.94 mm [t(63) = 7.717, one-way ANOVA, p < 0.001]} or with rolipram addition {306.80±28.14 mm [t(63) = 8.528, one-way ANOVA, p < 0.001]} (Fig. 7C). The use of GFP-D15A SCs and rolipram administration jointly (574.00±33.63 mm) was significantly better than either of the single treatments with a nearly twofold increase in 5-HT axon length within the implants compared to GFP SCs + rolipram [t(63) = 7.656, p < 0.001] or D15A SCs [t(63) = 6.987, p < 0.001].

5-HT axons were more numerous following the combination of D15A and rolipram treatment. (A, B) Immunostaining for 5-hydroxytryptamine (serotonin; 5-HT) demonstrated more numerous axons (red) after the combination treatment (B) compared to GFP SC (green) implants (A). Scale bar: 50 μm. (C) Measurements resulting from assessing isotropic virtual planes revealed that 5-HT axons were increased within SC grafts, particularly with the D15A and rolipram combination, which was significantly better than either D15A or rolipram with SCs. A statistically significant difference was accepted at values of ***p < 0.001 compared to either the GFP SC-only group or the individual treatment groups as indicated (by a, b, or c).
Hindlimb Movement Is Improved Following the Combination Treatment Compared to D15A SCs, But Not Rolipram with GFP SCs
There was an overall significant improvement in hindlimb function as evaluated by open-field locomotor testing using the BBB score with the addition of rolipram to GFP SCs. Locomotor assessment indicated that GFP-D15A SC grafts alone did not improve functional outcome significantly compared to GFP SC-only controls (Fig. 8A). But the addition of rolipram to either SC group significantly enhanced open-field locomotion [t(81) = 3.319, p < 0.05] (Fig. 8A) and reduced foot falls compared to GFP SC alone [t(81) = 3.352, p < 0.05] (Fig. 8C). The testing revealed a significant improvement in open-field locomotion (BBB score and subscore) and gridwalk ability with the combination therapy of D15A SCs and rolipram when compared to the GFP SC groups {vs. GFP SCs [t(81) = 3.735, p < 0.01], vs. D15A SCs [t(81) = 3.523, p < 0.01]}, but not the GFP SCs and rolipram group (p > 0.05). Animals that were unable to step (sweepers) (Table 2) were not included in the footprint analysis, which may have reduced the size of functional improvement provided by the combinatory approach. Indeed, many fewer animals were unable to step in the combination treatment group (6.3%) compared to the injury-only (61.1%) and GFP SC (18.2%) controls. This improved combination treatment group showed significantly (i) less foot exorotation, (ii) improved base of support, and (iii) fewer foot fall errors on an irregular horizontal ladder test compared to the SCI and GFP SC control groups (Fig. 8C).

Hindlimb function and foot positioning are improved by rolipram administration. Basso, Beattie, Bresnahan (BBB) locomotor testing revealed that the addition of rolipram resulted in a significant improvement in hindlimb function. The GFP-D15A SCs with rolipram showed the greatest amount of improvement throughout the testing. Improved functional recovery in the BBB score (A) and subscore (B) was observed early in assessment and was maintained thereafter. The results for stride length, base of support, foot rotation, and footfall errors are presented. (C) GFP-D15A SC grafts did not improve foot positioning compared to the GFP SC controls. On the other hand, the addition of rolipram to either SC group significantly improved foot placement compared to GFP SCs alone. Letters and colors correspond to the treatment groups being compared: letter a and dark blue, injury control; letter b and green, GFP SCs; letter c and blue, GFP-D15A SCs; letter d and red, GFP SCs + rolipram; letter e and purple, GFP-D15A SCs + rolipram. A total of 21 of 90 animals were removed from the footprint and gridwalk tests due to an inability to perform three consecutive steps (see Table 2 for a breakdown of these animals according to group). In the injury-only group, most of the animals exhibited an inability to perform consecutive stepping (sweeping) and therefore are not shown in (C). A statistically significant difference was accepted at values of ***p < 0.001, **p < 0.01, or *p < 0.05 compared to either the GFP SC-only group or the individual treatment groups as indicated (by a, vs. injury only; b, vs. GFP SCs; c, vs. GFP-D15A-GFP SCs; d, vs. GFP SCs + rolipram).
Number of Animals That Were Removed Due to Sweeping (Inability to Perform Three Consecutive Steps)Group Sweepers/Total Number of Animals in Group

Discussion
Based on previous reports of successful interventions (16,36), we designed the project reported here to test the combinatorial strategy of implanting SCs transduced to generate a bifunctional neurotrophin, D15A, and the systemic administration of rolipram to prevent the drop in cyclic AMP levels that occurs after contusion injury. The combination led to the largest SC grafts, the highest number of serotonergic fibers in the graft, the greatest numbers of axons from the reticular formation and raphe and vestibular nuclei below the lesion/implant area, and the most significant improvement in hindlimb function. There was a trend toward increased white matter sparing, but it did not reach significance. Rolipram with GFP SCs led to a significant increase in graft volume (but not as much as when combined with D15A), a significant increase in SC-myelinated axon number in the graft (in contrast to the combination), a significant increase in the number of reticulo-, raphe- and vestibulospinal fibers below the lesion/implant, an increase in serotonergic fibers in the graft (but not as much as in the combination), and improved hindlimb function. D15A with grafts led to a significant increase in graft size (but not as much as with the combination), a significant increase in graft serotonergic fibers (but not as high as in the combination), although white matter sparing, SC-myelinated axon counts, supraspinal fibers below the lesion/implant, and hindlimb function were not significantly improved. The transplantation of SCs, as in former studies (35,37,42), greatly abated cavitation, which was further improved in the combination.
How do these results compare with the earlier studies? First, it must be stated that there were considerable differences in the experimental design compared with the earlier investigations. The vehicle for rolipram was changed from DMSO to 1 ethanol:1 saline, with the idea of reducing vehicle toxicity. Rolipram was administered for 4 weeks rather than 2 weeks to potentially increase its salutary effect. There were no accompanying cyclic AMP injections into the spinal cord to increase clinical relevance of the approach by simplifying the treatment. The contusion injury severity was increased from a 12.5-mm to a 25-mm weight drop SCI. Finally, Matrigel, rather than culture medium, was chosen as the vehicle for the SCs because Matrigel has been found to improve cell survival, graft vascularization, and axonal growth into the graft (29). Also, the SCs were not GFP labeled in the earlier Pearse et al. study (36).
Here the combination of D15A SCs + rolipram significantly increased SC graft volume, nearly fivefold compared to GFP SCs or 1.8× larger than GFP SCs + rolipram or 1.4× larger than D15A SCs. In the Golden et al. (16) work, the addition of D15A also increased graft size. While NT-3 and BDNF have been shown to reduce SC proliferation and enhance their differentiation (15), elevated levels of neurotrophins could increase graft size through an enhancement of SC proliferation indirectly via the mitogenic signals (45) provided by a larger number of axons that are able to penetrate the D15A SC implants (22). D15A, by acting through BDNF-TrkB signaling, can also increase axon caliber and myelination (8) as well as revascularization (30), which may contribute to increased graft size. Additionally, and more likely, neurotrophins could have enhanced SC survival, as blocking neurotrophins, such as NT-3, either in vitro with antibodies (23) or through a knockout approach in vivo (39), triggers significant SC death.
In this study, there was a trend toward increased spared CMAs with rolipram and SCs; previously, we have shown that rolipram only (without SCs) significantly improved CMA sparing (36,40). White matter sparing was not improved in the presence of D15A compared to GFP- or SCI-only animals in the Golden et al. study (16) and confirmed in the present work. In the Golden et al. (16) work, D15A increased SC-myelinated axon number in grafts 3.5- to 5-fold. The Pearse et al. (36) study showed that rolipram alone or rolipram + SCs increased SC-myelinated axon numbers. Here SC-myelinated axon numbers were not significantly different among SC treatment and control groups. We do not know the reason(s) for this difference compared to earlier work, but it might be due to the presence of Matrigel (29), the lack of cyclic AMP injection into the cord, the injury severity, or negative interactions of the neurotrophins, specifically NT-3, with SC myelination (9,18), which may have been more pronounced in the context of Matrigel or cyclic AMP elevation.
Rolipram administration led to a significant increase in serotonergic fibers in GFP SC + rolipram and D15A SC + rolipram grafts compared to GFP SC-only implants. The increase was more significant in the combination than in D15A SC or GFP SC + rolipram grafts. Increased numbers of 5-HT-positive axons were also detected when rolipram was combined with SC grafts in the Pearse et al. study (36). D15A also supported serotonergic fiber growth in the Golden et al. investigation (16). Thus, both rolipram and D15A enhance serotonergic fiber growth into SC transplants, and the effects appear additive.
Earlier work has shown that when rolipram was introduced with or without SCs, the number of retrogradely labeled neurons with axons projecting caudal to the lesion increased significantly in the reticular formation and raphe nuclei compared to the injury-only control or SC transplant (36). In the current study, provision of rolipram to GFP SC- or D15A SC-transplanted animals yielded similar results. It has been previously reported that the combination of cells, neurotrophic factors, and cyclic AMP elevation can promote greater axonal regeneration after SCI (21), but this is the first study that has combined the use of the bifunctional neurotrophin D15A and the phosphodiesterase inhibitor rolipram to increase levels of cyclic AMP. Although the use of rolipram seems to be the critical factor for increasing axon growth/sparing across the injury, there was a significant additive effect provided by D15A. Prior studies have demonstrated neurotrophin-induced axon growth after SCI when neurotrophins are either directly infused into the spinal cord (47) or expressed by transduced cells (20,25) after SCI.
In the combination study reported here, hindlimb movement in the open field, stride length, base of support, foot rotation, and foot fall errors on a horizontal grid walk were evaluated. The BBB test scores showed significant improvements in 1) all groups compared to injury only, 2) GFP SCs + rolipram and the combination compared to GFP SCs, and 3) the combination versus D15A SCs. Thus, the combination was significantly more effective than D15A SCs but not rolipram with SCs. In comparison, stride length, base of support, foot rotation, and hindpaw placement on an irregularly spaced gridwalk test were improved with rolipram and GFP SCs but were further enhanced with the combination of rolipram + D15A SCs. Examination of the temporal recovery pattern of locomotor recovery revealed that the observed functional improvements occur very soon after treatment. When viewed in light of the most pronounced anatomical effect of the best combination treatments, rolipram + SCs (GFP or D15A), the enhanced numbers of retrogradely labeled neuronal somata in the brain stem with axon projections caudal to the lesion/implant, the mechanism of such functional improvement is likely due, in major part, to enhanced axonal sparing. Preservation of descending projections beyond the lesion/implant site would be expected to limit the degree of neurological deficit following SCI.
In sum, the current study demonstrates that the combination of D15A SCs and rolipram led to the largest SC grafts, the highest numbers of serotonergic fibers in the grafts, increased numbers of axons from the reticular formation below the lesion/implant area, and the greatest improvement in hindlimb function. Individually, rolipram and D15A have been shown previously to improve the functionality of SC implants (16,36) or other cellular approaches (6,7,27) for SCI repair. The present findings demonstrate the therapeutic potential for a combination therapy involving the maintenance of cyclic AMP levels and neurotrophin-transduced SCs to repair the subacutely injured spinal cord.
Footnotes
Acknowledgments
We gratefully acknowledge The Miami Project Viral Vector Core and Dr. B. Blits for lentiviral vector preparation; Dr. P. Tsoulfas (University of Miami) for providing the D15A plasmid and Dr. L. Naldini (University of Torino) for providing the lentiviral vector plasmid; staff in The Miami Project Animal Surgery and Behavior Core including Dr. A. Marcillo and P. Diaz for spinal cord contusions as well as D. Koivisto, M. Stagg, M. Garcia, I. Oropesa, and A. Maldonado for BBB scoring and postoperative animal care; Y. Pressman for SC culture; G. Alex, J. Ferro, N. Varshist, D. Bustin, and D. Maggio for tissue preparation; R. Bhatia, S. Nevavetla, S. Zhuchenko, and A. Patel for frozen sectioning and digital overlays; B. Frydel for helpful discussions on image analysis; R. Puzis and L. Tuesta for immunohistochemistry; M. Bates and R. Puzis for plastic sectioning; and W. Chan for proofreading the manuscript. This research was funded by NIH NINDS grants 05628 (D.D.P. and M.B.B.), 09923 (M.B.B.), and 38665 (M.B.B.), The Christopher and Dana Reeve Foundation International Research Consortium, The Bryon Riesch Paralysis, Craig H. Neilson, Fa Bene, and Daniel Heumann Foundations, The Miami Project to Cure Paralysis, and The Buoniconti Fund. The authors declare no conflicts of interest.
