Abstract
Transplantation of olfactory ensheathing cells (OECs) has emerged as a very promising therapy for spinal cord injury (SCI). Also, local delivery of NT-3 can counteract pathological events and induce a regenerative response after SCI. Supplement of exogenetic NT-3 might be a new approach to SCI repair. In this study, we examined the therapeutic effect of rat NT-3 gene-modified OECs transplantation on SCI. Rat NT-3 gene was transfected into OECs using a retroviral system. The engineered NT-3-OECs were tested for their ability to express and secrete biologically active NT-3 in vitro. Then NT-3-OECs were implanted into contused T9 spinal cord of the adult rats. Their ability of survival and NT-3 production was examined. The effect of axon regeneration was evaluated at the morphological level and promotion of locomotor functional recovery were assessed. The result showed that genetically modified OECs were capable of surviving and producing NT-3 in vivo to significantly improve the recovery after SCI.
Keywords
Introduction
Spinal cord injury (SCI) is one of the most devastating forms of trauma experienced by humans. After such damage, due to the limited neuronal survival, the lack of spontaneous axonal regeneration, and the inhibitory glial environment, self-repair after damage of the mammalian central nervous system (CNS) is rather poor, resulting in a permanent loss of function and paralysis. Therefore, the repair of brain and spinal cord injury thus far is still a major therapeutic challenge, although recently dramatic progress in cellular transplantation, gene therapy, and molecular treatment has heightened optimism about future cures for such injuries (4, 5, 10, 14, 22, 29).
Due to their ability both to support CNS axon regeneration and to remyelinate the regenerated axons, olfactory ensheathing cells (OECs) have attracted more attention as potential therapeutic agents for the treatment of SCI. Research performed over the past decades demonstrated that the presence of OECs would create a more favorable and supportive environment for axonal growth, guide axon regeneration through the glia scar to the CNS, and promote the recovery of sensorimotor functions in paraplegic rats. Furthermore, the available evidence supports the view that OECs are more likely to integrate into the CNS microenvironment and migrate long distances within the neuropil of the CNS after grafting to repair the injured spinal cord. These cells uniquely present both Schwann cell-like and astrocyte-like characteristics (21) and thus OECs transplantation seems to have great potential to repair the damaged spinal cord (1,13,14,17,20,21).
Neurotrophin-3 (NT-3), a member of the neurotrophin superfamily, is well known for its beneficial effects on survival of several neuronal cells, neuroprotection, and neurite outgrowth. Local delivery of NT-3 can counteract pathological events and induce a regenerative response after SCI (8,11,27). Supplement of exogenetic NT-3 might be a new approach to SCI repair.
At present, little of these strategies alone have been sufficient in the repair of the adult CNS; two or more strategies used in combination might bring about adequate structural and functional recovery. During the past two decades, the dramatic progress in therapeutic alliance and in vivo and ex vivo gene therapy has heightened optimism about future cures for injury of the CNS. In the present experiment, we examined the effect of rat NT-3 gene-modified OECs transplantation on SCI. The NT-3 gene was transfected into OECs using a retroviral-based system. The engineered OECs were first characterized by their ability to express and secrete biologically active NT-3 in vitro. After implantation into the spinal cord of adult rats with contused T9 spinal cord, their ability to survive and produce NT-3 was examined. The locomotor functions of animals were assessed, and axon regeneration was evaluated at the morphological level. Our results demonstrated that the genetically modified OECs are capable of surviving and producing NT-3 in vivo to significantly improve the recovery after SCI.
Materials and Methods
Primary Culture and Purification of OECs
The method was modified from the protocol of Ramon-Cueto et al. (21). Primary olfactory bulb cultures were set up from adult Sprague-Dawley rats (2.5 months old). All animal studies were approved by the Ethical Committee of Second Military Medical University, China. Briefly, olfactory nerve layer was peeled away from the rest of the olfactory bulb, and then dissociated with 0.25% trypsin and 0.03% collagenase at 37°C for 30 min. After washing with DMEM/F-12/10% FBS, cells were scattered and plated on uncoated dishes two times, each for 12 h at 37°C in 5% CO2. The suspension was collected and 5 ml suspension of about 300,000 cells was planted on 100-mm petri dishes for 45 min at 37°C in 5% CO2. These dishes were incubated sequentially with anti-rabbit IgG antibody, 1.5 μg polyclonal rabbit p-75 NGFR antibody (Santa Cruz Biotechnology) for 12 h at 4°C, and PBS/5% BSA for 4 h at room temperature. PBS was used to wash the dishes after each step. Unbound cells were washed off and the attached cells were collected using a cell scraper. Then the cells were seeded onto a poly-l-lysine-treated 25-mm2 flask, and incubated with DMEM/F-12/10% FBS containing 2 μM forskolin (Sigma) and 20 μg/ml pituitary extract (Sigma) as mitogen. The purity of the cultured OECs was determined by comparing the number of Hoechst-labeled nuclei with the number of p-75 NGFR antibody (Santa Cruz Biotechnology) immunoreactive cells under fluorescent microscopy.
Retroviral Preparation and Infection of OECs
The cDNA encoding rat prepro NT-3 was isolated by using RT-PCR method with total RNA extracted from rat brain. Specific primers (forward, 5′-ACGGATC CATGTCCATCTTGTTTTATGTGATA-3′; reverse, 5′-ATCTCGAGTCATGTTCTTCCGATTTTTCTTGA-3′) for PCR were designed to amplify prepro NT-3 cDNA that yield 777 bp amplified products. Then NT-3 cDNA was inserted into U3 area of 3′ long terminal repeat (LTR) of the replication defective recombinant retroviral pN2A (7) by BamHI and XhoI enzyme sites (named pN2A-NT-3). As shown in Figure 1, the unique feature of this double-copy vector is that the transfected gene inserted in the 3′ LTR was able to duplicate and transfer to the 5′ LTR in the infected cell, which can improve the expression of transfected genes (Fig. 1). The construct was confirmed by restriction analysis and sequencing.
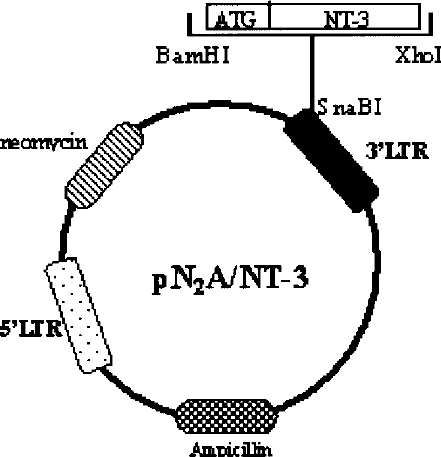
Schematic map of double-copy retroviral plasmid pN2A-NT-3. The original Moloney murine leukemia virus-based retroviral vector pN2A was inserted with a NT-3 gene in the U3 region of the 3′ long terminal repeat.
Lipofectamine (Invitrogen) packaged pN2A-NT-3 was used to transfect the packaging cell line, PA317 (19). The neomycin analogue, G418, was used to isolate resistant colonies. Viral supernatants from these colonies were titrated on NIH 3T3 cells as described previously (32). The cell line (PA317-NT-3) with the highest titer (7 × 104 cfu/ml) was used to infect the purified adult rat primary OECs.
The most effective method for infecting dividing OECs was a 2-h pretreatment with 8 pg/ml polybrene followed by an overnight infection with the recombinant retrovirus in medium containing mitogens. The same procedure was repeated the next day, but polybrene was added for only 1 h. Four to 5 days after the last infection, the OECs were selected with 200 μg/ml G418. Once the selected population grew in a stable manner, the cells were expanded using the same selection medium, with a maximum of six to seven total passages, to produce enough OECs for experiments.
Biological Activity of NT-3-OECs
Primary cultures of adult rat normal OECs and NT-3-OECs were fixed for 10 min with 4% paraformaldehyde. After washing in PBS and blocking with 1% BSA for 30 min, the cultures were incubated overnight at 4°C with the rabbit polyclonal anti-human NT-3 antibody (1/200, Promega), anti-p-75 NGFR rabbit antibody (5 pg/ml, Santa Cruz Biotechnology), monoclonal mouse anti-GFAP (1:1000, Sigma), and anti-S-100 mouse antibody (1:2000, Sigma) diluted in PBS containing 1% BSA. The next day the cultures were first washed in PBS and then incubated for 40 min at 37°C with FITC-labeled secondary antibodies (Promega). They were then washed and examined by Olympus BX-50 fluorescent microscopy.
Assay of NT-3 Production
The amount of NT-3 secreted by NT-3-OECs was measured by ELISA using NT-3 Emax ImmunoAssay Systems (Promega). According to the manufacturer's instructions, ELISA plates (96 wells) were coated with an anti-NT-3 monoclonal antibody (pH 8.2) overnight at 4–8°C. Plates were then blocked for 1 h at room temperature with blocking buffer. NT-3 standards ranging from 0 to 1000 pg/ml were prepared using recombinant NT-3 and applied to the wells. Conditioned medium (CM) was obtained by 2 × 106 cells in 2 ml medium, but with only 1% FBS for 24 h, then added to the wells (100 μl) undiluted or diluted 1:10 or 1:100. Samples and standards were incubated at room temperature for 6 h on a shaker. The plate was then incubated sequentially with chicken anti-human NT-3 polyclonal antibody overnight at 4°C, HRP-conjugated anti-chicken antibody (1:5000) at room temperature for 2 h, enzyme substrate TMB for 15 min at room temperature. PBS was used to wash the plates after each step. The enzyme reaction was stopped by adding 100 ml of 1 M phosphoric acid per well and the absorbance was measured at 450 nm. Sample values were calculated from the standard curve in the linear range.
The biological activity of the secreted NT-3 was tested using a PC12 cell line that stably expresses NT-3 receptor TrkC (15). Briefly, 2 × 104 PC12-TrkC cells were added to each well of a 24-well plate (Costar) that had been coated with poly-l-lysine. After attachment, the cells were exposed to CM from OECs or NT-3-OECs. PBS was used as negative control and 100 ng/ml NT-3 (CalBiochem) was positive control. Five days later, effects of NT-3 on cell differentiation were determined. Cells possessing one or more neurites of a length more than twice the diameter of the cell body were scored as positive. Each value is the mean ± SEM sampled from three independent experiments.
Hoechst Labeling of OECs
When cells reached confluence, monolayer of purified OECs or NT-3-OECs were incubated at 37°C in 1.5 μg/ml nuclear fluorochrome bisbenzimide (Hoechst 33342, Sigma) for 15 min. After several washes in DMEM, cells were trypsinized and collected for transplantation.
Surgical Procedures
Animal care and use followed recommended NIH guidelines. Female adult Sprague-Dawley rats received T9 spinal cord contusion injury by modified Allen method (12). In brief, all rats were anesthetized with 2% pentobarbital sodium (0.2 ml/kg) intraperitoneally. As for the spinal cord, laminectomy was performed to expose the dorsal surface of the T8–10 segment. The spinous processes of vertebrae above and below the lesion are secured in clamps to reduce motion of the spinal column during the procedure. After identification of the level and midline of the spinal cord, a 10 g rod was dropped from heights of 50 mm onto the exposed dorsal surface of the T9 spinal cord (10 × 5 g cmf). Rats then received 10 μl of stereotaxic injection into three sites of the subarachnoid space (8 μl at injury site, 1 μl at 1 mm from proximal and distal of injury site) (21), using sterile glass needles. A total of 24 animals were operated: (i) 8 animals received no grafted OECs, each received 10 μl normal saline; (ii) 8 received a graft of normal OECs, each received 10 μl OECs suspension containing about 105 cells; (iii) 8 received a graft of NT-3-OECs, each site received 10 μl cell suspension containing about 105 cells. After removal of the injector, muscles and skin were closed in separate layers and rats were kept at 22–25°C on highly absorbent bedding, injected with cefazolin sodium (40 mg/day) for up to 1 week, and received bladder expression twice daily until normal function returned. Regular histological HE staining procedure was performed according to an earlier report (28).
RT-PCR Analysis
Eight weeks after cell transplantation, animals from OECs or NT-3-OECs group were euthanized with a lethal dose of pentobarbital sodium and the T8–10 spinal cord tissue was collected. Total RNA was isolated from the tissue using Trizol (Invitrogen) and the RNA concentration was measured photometrically. After RNA extraction, the samples were digested with RNase-free DNase I, and cDNA was synthesized using an Omniscript TM PT kit (Qiagen). For PCR, specific primers (forward, 5′-AGCCGATGATTGCAACAGACAC-3′; reverse, 5′-CCAGCGCCAGCCTACGAG-3′) were designed to amplify NT-3 cDNA that yield 473-bp amplified products. Forty picomoles of each primer and 1 μg DNA were used for PCR. PCR was carried out in a programmable heating block using cycle consisting of denaturation at 95°C for 1 min, following by annealing at 55°C for 1 min and DNA extension at 72°C for 1 min. After 30 cycles of PCR, samples were electrophoresed on 1.5% agarose gel. Gels were stained with ethidium bromide and photographed under ultraviolet light.
Retrograde Tracing with HRP
Four animals of each group were used for HRP retrograde tracing. Eight weeks after surgery, an aqueous suspension of 30% HRP (Sigma, RZ > 3.0) and 2% DMSO (Sigma) was injected bilaterally three to four segments caudal to the injury segment to avoid diffusion of HRP into the transplant. After injection, the surgical exposure was closed and the animals were maintained for 36 h before being perfused by buffed 1% paraformaldehyde and 1.25% glutaraldehyde. The brain and the spinal cord were removed and stored in 20% sucrose in 0.1 M PBS at 4°C overnight. Then the sensory motor cortex in the forebrain and the magnocellular portion of the RN in the midbrain were cut transversely and serially at 30 μm. Every third section in RN or cortex was collected and stained with tetramethylbenzidine (TMB, Sigma) and hydrogen peroxide according to the method of Mesulam (18). After counterstaining with neutral red, the sections were observed under a light microscope. The distal spinal cord including the transected area was cryosectioned sagittally to ensure the injection was confined and no spread of dye to the transplant. Animals that did not meet these criteria were eliminated from the study.
For identification of neurons in the RN and cortex, the caudal most section through the nucleus magocellularis and sensory motor cortex where HRP-labeled neurons could be observed was designated as the first section for analysis. The numbers of HRP and the neutral red-labeled RSN and CSN on both sites were counted separately using a digitizing tablet and PC-based software (Metamorph). The criterion for a CSN was a HRP-filled pyramidal-shape >4 μm in diameter. For the RSN, only neurons with clearly seen nucleus were counted.
Immunohistochemistry
Rats were perfused with 4% paraformaldehyde in 0.1 M ice-cold phosphate buffer. The spinal cord was removed, postfixed for 5 h, and placed in 30% sucrose/PBS before preparing 30-μm sagittal cryosections. Hoechst-labeled OECs were visualized with fluorescent microscope equipped with a 365-nm excitation filter and a 420-nm emission filter. For immunofluorescence, sections were permeabilized and blocked with 0.3% Triton X-100/10% normal goat serum in 0.1 M PBS for 15 min. Primary antibodies were then applied to the sections overnight at 4°C. Slides were washed, mounted, and examined by Olympus fluorescent microscopy.
Functional Recovery
Functional tests were performed before operation and at 2 h, 3 days, and 1, 2, 3, 4, 5, 6, 7, and 8 weeks postoperation. Locomotor activity was evaluated using the open field walking scoring system. One animal at a time was allowed to move freely inside a circular plastic tray (90 cm diameter, 24 cm wall height) for 5 min. Behavioral recovery was scored according to BBB scale (2), which is composed of 21 different criteria of the movement of the hind limb from complete paralysis to complete mobility. As a second test for hind limb function, animals were subjected to an inclined plane test (23). The maximum angle at which the animal could maintain a stable position for 5 s on the inclined plane was recorded. Before each evaluation, we carefully examined the rats for perineal infection, wounds in the hind limbs, or tail and foot autophagia.
Statistical Analysis
All the data were statistically analyzed by factorial ANOVA.
Results
Characterization of NT-3-OECs
OECs cultures consisted of mainly long processing cells, which displayed p-75 NGFR and S-100 binding. The immunofluorescent staining demonstrated that the purity of OECs was more than 95%. To further ascertain that insertion of a new gene using viral vector-mediated gene transfer did not interfere with normal cell functioning, NT-3-OECs cultures were phenotypically examined for expression of the general cell markers low-affinity neurotrophin receptor p-75 NGFR, Schwann cell marker S-100, and glial-fibrillary acidic protein (GFAP) (Fig. 2). More than 85% of NT-3-OECs in primary cultures showed spindle-like morphology with two or three processes, described as Schwann-cell-like phenotype (3,6, 13,31), while less than 15% of cells had flat appearance.
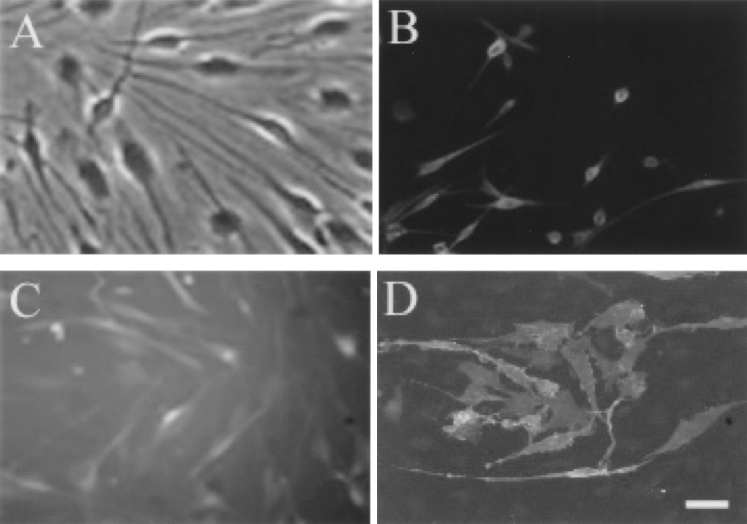
Characterization of an NT-3-overexpressing OECs cell line. Most NT-3-OECs in primary cultures showed spindle-like morphology, while a minority of them had flat appearance (A). NT-3-OECs were phenotypically assayed for expression of S-100 (B), GFAP (C), and p-75 NGFR (D). Scale bar: 100 μm.
Ex Vivo NT-3 Secretion and Biological Effect
The amount of NT-3 secreted by NT-3-OECs was determined by ELISA. It revealed that NT-3 production by control uninfected OECs was not detectable, whereas OECs transfected with NT-3 gene produced an average of 30 ng NT-3/106 cells/24 h.
The biological activity of the secreted NT-3 was examined using PC12-TrkC cells. There were 9.4 ± 1.34% cells that differentiated after 72 h of culture in CM from normal OECs; in CM from NT-3-OECs, cell differentiation was threefold greater (Fig. 3). This bioassay confirmed that NT-3 secreted from NT-3-OECs was capable of promoting differentiation of PC12-TrkC cells and was biologically active.
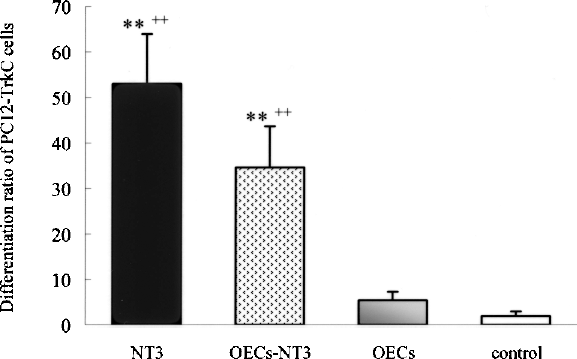
Differentiation ratio of PC12-TrkC cells, which were incubated for 5 days with DMEM (control), NT-3 50 ng/ml, conditioned media obtained from NT-3-OECs, or wild-type OECs. **p < 0.05 versus control group; ++p < 0.0 versus wild-type OECs (factorial ANOVA).
In Vivo NT-3 Expression
Eight weeks after surgery, RT-PCR was used to measure the mRNA level of NT-3 in the injured spinal cord (Fig. 4). Significantly higher levels of NT-3 mRNA expression in the injured spinal cord injected with NT-3-OECs were detected compared with that in the spinal cord of rats injected with normal OECs. This result demonstrated that ex vivo transduction of OECs using retroviral vectors resulted in a persistent increased NT-3 expression in vivo, at least up to 2 months after implantation.

Detecting rat NT-3 mRNA level by RT-PCR 8 weeks after transplanting NT-3-OECs into the injured rat spinal cord. Lane 1: DL2000 marker; lane 2: RT-PCR products of rat NT-3.
When prelabeled with bisbenzimide, both transplanted NT-3-OECs and normal OECs were recognized by their strongly blue fluorescent nuclei. Two months following implantation, Hoechst-labeled OECs were visualized as a dense mass of elongated, brightly fluorescent cells extending from the lesion site (Fig. 5A, B). These results demonstrated that the implanted OECs and NT-3-OECs were capable of surviving in the injured spinal cord at least 2 months after implantation.

Morphology and staining of spinal cord at the injured site of SD rats 8 weeks after spinal cord contusion injury. Hoechst-labeled OECs survive (B is enlargement of graph A) at injured site. Scale bars: 400 μm. Spinal cord morphology (C), H&E staining (D).
Axonal Regeneration
Spinal cord contusion lesion was characterized by an obvious scar at the T9 lesion epicenter and neuronal necrosis, reactive gliosis, and cavitation in adjacent rostral and caudal regions (Fig. 5C, D). As shown in Figure 6, the percentage of spared tissue of the lesioned spinal cord was obviously different in each group (p < 0.05). It revealed that OECs and NT-3-OECs transplantation, especially NT-3-OECs transplantation, may have provided protective and recovery promotion effect to the injured spinal cord.
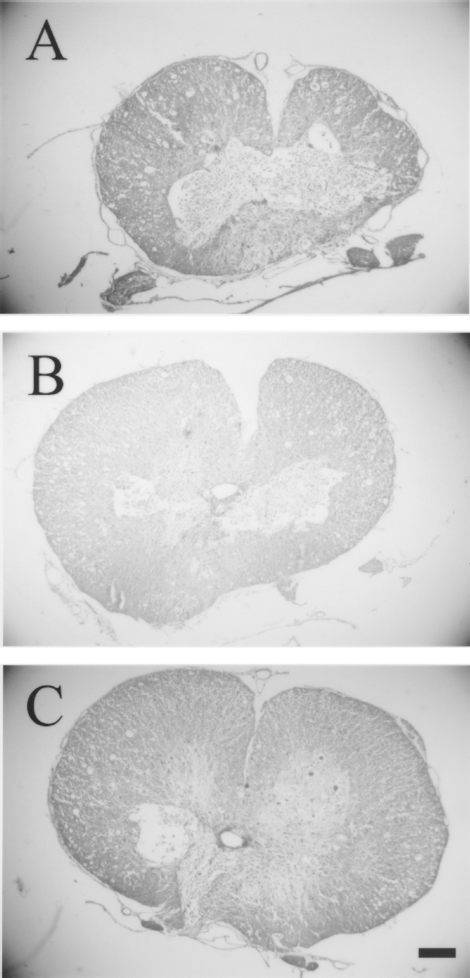
Transverse sections stained with HE through the lesion center T9. Contusion injuries in control rats (A) treated with saline and (B, C) treated with OECs and NT-3-OECs, respectively. Scale bar: 600 mm.
Then we labeled RSN and CSN after injections of HRP into the low thoracic and upper lumber region (Figs. 7 and 8). HRP retrograde staining result showed most labeled RSN were located in the ventral-lateral portion of the magnocellular nucleus of the RN. The number of HRP-labeled RSN and CSN were counted in all groups of animals (Fig. 9). On average, in normal animals, ~374 and ~2,524 neurons were labeled on both sides of RSN and CSN, respectively. After thoracic contusion, few HRP-labeled RSN or CSN were detected in the control group. With treatment of normal OECs, a few cells were retrogradely labeled. OECs transplantation may have provided a permissive environment that allowed a small percentage of axotomized neurons to regenerate into the caudal spinal cord (p < 0.01 vs. control group). The highest regeneration ratio was obtained in the NT-3-OECs group. Approximately 97 in RSN and 776 in CSN were labeled with HRP. These numbers were significantly higher than normal OECs group (p < 0.01).

Staining of midbrain after HRP retrograde tracing of RSN, 8 weeks after T9 contusion at comparable levels. (A) HRP staining of RSN from the rats following SCI treated with saline. (B, C) Rats treated with OECs and NT-3-OECs, respectively. Scale bar: 100 μm.

Staining of forebrain after HRP retrograde tracing of CSN, 8 weeks after T9 contusion at comparable levels. (A) HRP staining of RSN from the rats following SCI treated with saline. (B, C) Rats treated with OECs and NT-3-OECs, respectively. Scale bar: 100 μm.

Statistical analysis of HRP-labeled RSN (A) and CSN (B) from three experimental animal groups 8 weeks after transplantation. In rats transplanted with NT-3-OECs, there were significantly more RSN and CSN positive labeled than in those transplanted with wild-type OECs (p < 0.01). Significantly more (p < 0.01) RSN and CSN were positive labeled in rats transplanted with wild-type OECs than in those receiving saline. **p < 0.01 OECs versus control; ΔΔp < 0.01 NT-3-OECs versus OECs (factorial ANOVA). n = 4.
Behavioral Assessment
Figure 10 shows the behavioral results during the 8 weeks assessment period for each group of animals. All injured rats manifested complete hind limb paralysis immediately following injury. The BBB scores were in the range 0–2 in the control animals. Following transplantation of the OECs, hind limb functional recovery increased gradually; by 8 weeks after transplantation, all eight animals displayed BBB scores greater than that achieved by any of the eight controls (p < 0.01). Four of the eight experimental animals from the OECs group could support their body weight on the hind limbs; the other four animals had ankle, knee, and hip movements in one or both legs, but did not obviously bear weight. The NT-3-OECs group regained more functional recovery than the normal OECs group 8 weeks after transplantation (p < 0.01). Six of the eight rats treated with NT-3-OECs could walk in a coordinated manner.

Functional analysis of hindlimb movements following SCI by Inclined plane test (A) and BBB behavioral assessment (B). *p < 0.05, **p < 0.01, OECs versus control; Δp < 0.05, ΔΔp < 0.01, NT-3-OECs versus OECs (factorial ANOVA). n = 8.
Differences of IP score among the three groups were significant too. From 2 weeks postoperation, rats in the OECs group began to show more functional recovery than the control group (p < 0.05); and from 5 weeks after implantation, animals within the NT-3-OECs group had a higher score than the OECs group (p < 0.05).
Discussion
Combining neurotransplantation and gene therapy is perhaps one of the most powerful strategies to promote CNS repair. As implantation of OECs in the injured spinal cord has been reported to promote long-distance regeneration and functional recovery following SCI (14, 20), there is much interest in upgrading OECs to enhance the regenerative properties of these cells. Recently, researchers have begun to transfect OECs to express fluorescent markers for tracing experiments (26) or use transgenic animals expressing an exogenetic protein as a source for modified OECs (9). In the present study, OECs were genetically modified to overexpress exogenous neurotrophic factor and transplanted into contusion spinal cord. Functional and morphological research supports the conclusion that growth-promoting properties of OECs were significantly improved when genetically modified to secrete an increased amount of NT-3.
Ex vivo gene therapy is a valuable approach that achieves long-term and site-specific delivery of therapeutic agents in the CNS. Both retroviral and adenoviral vectors have been widely used for gene transfer. Retroviral vectors integrate with high efficiency and contain no viral genes so they can mediate long-term expression and avoid host cellular immune responses (24). In the present study, high levels of transgenic NT-3 mRNA could be detected even 2 months after implantation. The sustained high-level gene expression of NT-3 in OECs implants allows the possibility of manipulating the growth-promoting properties of OECs and microenvironment of the lesion during not only acute but chronic phase following injury. Retroviral vectors are limited by the viral LTR sequence, which may interfere with gene expression of various gene cassettes. No changes in morphology and function were detected after transduction of OECs by retroviral vectors in this study.
NT-3, a member of neurotrophin superfamily, is well known for its beneficial effects on survival of several neuronal cells, neuroprotection, and neurite outgrowth. Local delivery of NT-3 can counteract pathological events and induce a regenerative response after SCI. Supplement of exogenetic NT-3 might be a new approach to SCI repair. In the present study, we demonstrated that treatment with NT-3 gene-modified OECs could greatly rescue tissue necrosis and improve tissue sparing in adult rats after spinal cord contusion compared with normal OECs, which may directly relate to the locomotor capacity of spinal cord-injured rats (28). The results showed that treatment with NT-3 gene-modified OECs stimulated increased amount of regenerated corticospinal or rubrospinal axons after spinal cord injury compared with normal OECs. The HRP retrograde labeling studies clearly demonstrate that injured axons had indeed regenerated through the transection site. Numerous TMB-labeled neurons were detected in the NT-3-OECs group 8 weeks after SCI. However, in the normal OECs group, less than half TMB-labeled neurons were detected. NT-3 ex vivo gene delivery might enhance the growth-promoting properties of OECs after SCI in adult rats. Several reasons might be responsible for this phenomenon (25,30). Firstly, NT-3 elicited a chemotropic effect, directing the growth of axonal populations to regions of the highest growth factor concentration. Secondly, NT-3 increased the surviving ratio of RSN and CSN after axotomy, which result in an increase number of regenerated corticospinal or rubrospinal fibers. Moreover, a recent study also showed that cultured OECs express NT-3 receptor TrkC (16), so high levels of NT-3 secreting from NT-3-OECs may have trophic effects on themselves.
The locomotor functions of animals were evaluated using the inclined plane method of Rivlin and BBB scale. BBB scores differ from other locomotor scoring systems in several respects. First, the score is not a summation of component behaviors. Each BBB score re-quires fulfillment of a unique set of criteria. Second, the scores encompass many behavioral traits and represent a detailed characterization of rat locomotor function. Third, the scores are based on observations of rat recovery from SCI. The ordering of the scores assumes progressive recovery and that each recovery stage represents better locomotion than the preceding stage (2). By BBB scale and inclined plane method, we demonstrated a progressive recovery over time among the three groups. The functional recovery for the NT-3-OECs group achieved statistically significant improvements compared with the normal OECs group in these two tests. This result was in accordance with the morphological experiments. Significant improvements of locomotor function were also obtained in the NT-3-OECs and normal OECs groups compared with the control group.
In summary, the present study showed that growth-promoting properties of OECs were significantly improved when genetically modified to secrete increased level of NT-3. Genetic engineering of OECs opens new possibilities for their future clinical applications in SCI.
Footnotes
Acknowledgments
This work is supported by National Natural Science Foundation of China Grant 30400123 and 30570939 (to Y.Z.), and sponsored by Shanghai Rising-Star Program 05QMX1469 (to Y.Z.).
