Abstract
Stamina-enhancing effects of human adipose derived stem cells (hADSCs) were investigated in young Sprague-Dawley rats. Ten-day-old male rats were transplanted intravenously (IV) or intracerebroventricularly (ICV) with hADSCs (1 × 106 cells/rat), and physical activity was measured by locomotor activity and rota-rod performance at post-natal day (PND) 14, 20, 30, and 40, as well as a forced swimming test at PND 41. hADSCs injection increased the moving time in locomotor activity, the latency in rota-rod performance, and the maximum swimming time. For the improvement of physical activity, ICV transplantation was superior to IV injection. In biochemical analyses, ICV transplantation of hADSCs markedly reduced serum creatine phosphokinase, lactate dehydrogenase, alanine transaminase, and muscular lipid peroxidation, the markers for muscular and hepatic injuries, despite the reduction in muscular glycogen and serum triglycerides as energy sources. Notably, hADSCs secreted brain-derived neurotrophic factor (BDNF) and nerve growth factor in vitro, and increased the level of BDNF in the brain and muscles in vivo. The results indicate that hADSCs enhance physical activity including stamina not only by attenuating tissue injury, but also by strengthening the muscles via production of BDNF.
Keywords
Introduction
Human adipose-derived stem cells (hADSCs) can be easily obtained from adipose tissue of humans and expanded in vitro for use in autologous cell therapy 1 . The widely-spread human adipose tissue provides an abundant source of hADSCs, which can be easily and safely harvested compared with other mesenchymal stem cells 2 . hADSCs can differentiate into various mesenchymal lineages and secrete various neurotrophins and growth factors, and has thus generated interest in cell therapy using ADSCs in various fields of study 3 .
Notably, enhanced brain activity elicited by transplanted hADSCs probably occurs as a result of their secretion of neurotrophic factors (NFs) and growth factors (GFs) including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial-derived neurotrophic factor (GDNF) 4 . In addition, it was reported that hADSCs produce high amounts of GFs such as vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), insulin-like growth factor (IGF), and leukemia inhibitory factor (LIF) 5 that are beneficial for tissue growth and body development 5 .
BDNF is a member of the family of NFs comprising of neurotrophin-3 (NT-3) and NT-4 6 . Initially identified as a growth factor supporting sensory neurons, BDNF has been recently recognized as a multifaceted trophic factor 7 . BDNF plays a critical role in the activity-dependent processes through the TrkB receptor, including synapse development and plasticity by strengthening excitatory (glutamatergic) synapse 8,9 . Acute application of BDNF induces a rapid Ca2+ influx into motor nerve terminals, and preferentially potentiates active synapses 10 . BDNF can also induce fast-twitch (Type II) to slow-twitch (Type I) transformation of muscle fibers by activating calcineurin-nuclear factor of activated T cells (NFATc1) via the TrkB receptor 11 . Furthermore, it promotes revascularization by local recruitment of TrkB+ endothelial cells 12 , and serves a key role in maintaining the population of muscle progenitors in adult muscle via regulation of satellite cell differentiation and skeletal muscle regeneration 13,14 .
In our previous studies, it was confirmed that hADSCs increased physical activity, improved cognitive function, and extended the lifespan and health span of 10-month old rats by intravenously (IV) injecting them once a month throughout their lives 15 , and that ADSCs improved cognitive and physical activities of ageing mice, in which intracerebroventricular (ICV) injection was superior to IV treatment, in parallel with the increase in NFs/GFs including BDNF and NGF 16 . As shown in aged animals, hADSCs also restored neurobehavioral activity of rats with neonatal hypoxic-ischemic encephalopathy, an animal model of cerebral palsy (CP) 17 . Such effects led us to investigate the effects of hADSCs on the physical activity and (energy-boosting) stamina in normal young animals. In the present study, we analyzed diverse physical activities, parameters of muscular injury, NFs as well as change in muscle types in post-natal day (PND) 45 rats after transplantation of hADSCs at PND 10.
Materials and Methods
Ethics Statement
Human adipose tissues were obtained from female healthy donors undergoing elective liposuction procedures after informed consent. All sample collection procedures from human beings and experimental protocols were approved by Institutional Review Board of K-Stem cell (KS-2015-01-001). All procedures in this study for animal experiments were conducted in accordance with the Institutional Animal Care and Use Committee of Laboratory Animal Research Center at Chungbuk National University (CBNUA-314-11-01).
Preparation of the hADSCs
The hADSCs were prepared as per detailed description in our previous study 15 –17 . The adipose tissues were digested with collagenase I, filtered through a 100-μm nylon sieve, and centrifuged at 470 g for 5 min. The pellet was resuspended in Dulbecco’s modified Eagle’s medium (Invitrogen, Grand Island, NY, USA) containing 0.2 mM ascorbic acid and 10% fetal bovine serum (FBS). The cell suspension was recentrifuged at 470 g for 5 min, and the cell pellet was collected. After overnight culture, nonadherent cells were removed by washing with phosphate-buffered saline (PBS). The cell medium was changed to keratinocyte-serum-free medium (SFM; Invitrogen) containing 0.2 mM ascorbic acid, 0.09 mM calcium, 5 ng/mL recombinant EGF, and 5% FBS. The cells were maintained for 4–5 days until confluent (passage 0). When the cells reached 90% confluence, they were subculture expanded in keratinocyte-SFM medium until passage 3. Karyotype analysis of the hADSCs was processed from GenDix (Seoul, Korea). The BDNF and NGF levels in the conditioned medium produced by 1 × 106 hADSCs were determined using enzyme-linked immunosorbent assay (ELISA). Human BDNF ELISA kit (ab99978, Abcam, Cambridge, UK) and human NGF ELISA kit (ab99986, Abcam) were used according to the manufacturer’s instructions.
Differentiation and Immunophenotype of the hADSCs
hADSCs were seeded in 12-well culture plates at a density of 2,500 cells/cm2 and expanded until cells reached 90% confluency. For adipogenic, osteogenic, and chondrogenic differentiation, hADSCs were stimulated for 28, 21 and 14 days in differentiation media (GIBCO, Grand Island, NY, USA) according to manufacturer’s instructions. Adipogenic, osteogenic, and chondrogenic differentiation were assessed by staining for Oil red O, Alizarin Red S and Safranin O, respectively.
Immunophenotype of hADSCs was carried out using flow cytometry for the following markers: SSEA-4 (MAB4304, Merck-Millipore, Darmstadt, Germany), CD34 (ab81289, Abcam), CD90 (#561970, BD Pharmingen, San Diego, CA, USA), CD105 (MCA1557, Bio-rad, Hercules, CA, USA) and HLA-DR (ab92511, Abcam). Samples were analyzed using a FACS Calibur-S System (BD Immunocytometry Systems, San Jose, CA, USA) and the obtained data were analyzed with FlowJo software (FlowJo, version 7.6.1).
Animals
Sprague-Dawley rats (PND 10) were purchased from Deahan-Biolink (Eumsung, Korea). The animals were divided into 3 groups: vehicle control (n = 10), IV transplanted with hADSCs (n = 10) and ICV transplanted with hADSCs (n = 10). The hADSCs were prepared as per detailed description in our previous study 15 . In the IV group, the hADSCs (1 × 106 cells/100 µL PBS/rat) were injected into the tail vein. In the ICV group, the hADSCs (1 × 106 cells/5 µL PBS/rat) were transplanted at the following coordinates: anterior/posterior +0.8 mm, right lateral 1.5 mm, and ventral 4 mm from the bregma. The rats received the cells only once at PND 10. The vehicle control group was IV injected with PBS. The rats were maintained at a constant temperature (23 ± 2°C), relative humidity of 55% ± 10%, and 12-hour light/dark cycle, and fed on standard rodent chow and purified water ad libitum.
Measurement of Physical Activity
Locomotor activity: Spontaneous activities and exploratory behaviors were evaluated using a video tracking system (Smart v2.5; Panlab Technology, Barcelona, Spain) connected to a CCTV (Samsung, Changwon, Korea) at PND 14, 20, 30, and 40. The rats were placed in a quiet chamber with dim light. And the times for each movement type, that is, resting, slow-moving and fast-moving times, were recorded for 5 min, and the ratio was analyzed.
Rota-rod performance: Motor balance and coordination were evaluated using a rota-rod test system (Panlab technology) at PND 14, 20, 30, and 40. Rats were placed on a rotating rod at a constant speed of 12 rpm, and the time taken for the rats to fall off the rod was recorded. The average latency was calculated from 3 consecutive measurements. The endpoint was set to 300 sec.
Forced swimming test: To analyze physical stamina, all rats were allowed to swim for 30 min for adaptation to a swimming pool with a constant water temperature of 25 ± 0.5°C for 4 days before the measurement. All rats were subjected to a weight-loaded forced swimming exercise at PND 41. The rats were loaded with a lead ring weighing 5% of their body weight attached to the tail, and then placed in the swimming pool. The test was performed by forcing animals to swim until exhaustion, which was determined by observing loss coordinated movements and failure to return to the water surface within 7 sec. This 7-sec cut off time was used as the criterion for the maximum swimming capacity of the animals.
Necropsy and Serum Biochemistry
After a 4-day recovery period following the maximum swimming time measurement at PND 41, the rats were euthanized under ether anesthesia at PDN 45. Blood was collected and centrifuged to obtain serum at 3,000 rpm for 20 min. The serum concentrations of muscle-injury parameters such as creatine phosphokinase (CPK), aspartate transaminase (AST), lactate dehydrogenase (LDH), alanine transaminase (ALT), blood urea nitrogen (BUN), creatinine, and triglycerides (TG) were determined using a blood chemistry analyzer (Hitachi 747; Hitachi Korea, Seoul, Korea).
The gastrocnemius and soleus muscles, liver, spleen, thymus, adrenal glands, testes, and epididymides, the organs susceptible or related to muscular energy metabolism and exhaustion, were removed and weighed.
Western Blot Analysis
The soleus muscle and brain of rats were excised quickly, freeze-clamped in liquid nitrogen, and stored at −80°C until use. For western blot analysis, frozen tissue samples were homogenized in 10 volumes (wt/vol) of RIPA buffer solution (Sigma-Aldrich, St. Louis, MO, USA). Proteins were quantified using a BCA protein assay kit (Pierce, Rockford, IL, USA). Proteins were denatured by heating for 5 min at 95°C in 0.5 M Tris-HCl buffer (pH 6.8) containing 10% SDS and 10% ammonium persulfate, separated by electrophoresis on 17.5% or 10% SDS-polyacrylamide gels, depending on protein size, and transferred to a nitrocellulose membrane in 25 mM Tris buffer containing 15% methanol, 1% SDS, and 192 mM glycine. After blocking for 1 hour with 5% skim milk in Tris-buffered saline-Tween (TBS-T) (pH 7.6), the membrane was incubated with anti-BDNF (1:500; rabbit polyclonal; Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 4°C. After washing with TBS-T, the membrane was incubated with a secondary goat anti-rabbit IgG conjugated with horseradish peroxidase (1:2,000; Santa Cruz Biotechnology) for 2 hours at room temperature. The membrane was then developed using an enhanced chemiluminescence solution (Pierce). The band densities were measured using ImageJ software (National Institutes of Health, Bethesda, MD, USA) and normalized to the density of β-actin.
Muscle Fiber Analysis
To distinguish between slow-twitch (slowly-contracting Type I) and fast-twitch (fastly-contracting Type II) muscle fibers in the rats, myofibrillar ATPase activity was analyzed by myosin-ATPase. That is, the soleus muscles were excised and cryosections were prepared in 8-μM thickness and processed for staining. Prepared tissue was incubated in reaction mixture containing 1 mM dithiothreitol (DTT) and 0.3 mg/mL ATP (pH 9.4) for 30 min at 37°C, washed in 1% CaCl2 3 times for 2 min each, and placed in 2% CoCl2 solution 2 times for 1 min each. Following this process, the tissue was washed very thoroughly at least 4 changes of distilled water, and placed in 1% ammonium sulfide solution for 30 sec for colorization. After the color had developed, the tissue was washed with distilled water, and mounted in glycerin jelly. The ratio of muscles stained light brown (slow-twitch muscles) and dark brown (fast-twitch muscles) was analyzed using ImageJ software (National Institutes of Health)
Muscle Glycogen Analysis
Samples of frozen tissue (30 mg) were hydrolyzed in 0.3 mL of 30% potassium hydroxide (KOH) solution in a boiling water bath for 30 min and intermittently shaken. After cooling to room temperature, 0.1 mL of 1 M Na2SO4 and 0.8 mL of ethanol were added, and the samples were boiled again for 5 min. The glycogen pellet was dissolved in 0.2 mL of water, and two additional ethanol precipitations were performed. The final pellet was dried and dissolved in 0.2 mL of 0.3 mg/mL amyloglucosidase in 0.2 M sodium acetate buffer (pH 4.8) and incubated for 3 hours at 40°C. The reaction mixture was added to 0.2 mL of the glucose assay solution containing 0.3 M triethanolamine-KOH (pH 7.5), 1 mM ATP, 0.9 mM β-NADP, and 5 µg G6PDH/mL. The absorbance at 340 nm was determined before and after addition of 1 µg of hexokinase. Glycogen content was expressed as μmole of glucosyl units/g.
Lipid Peroxidation Assay
Lipid peroxidation was measured by determining the formation of thiobarbituric acid-reactive substances (TBARS), that is, malondialdehyde (MDA), in the gastrocnemius muscles. Briefly, the gastrocnemius muscles of the rats were homogenized in 10 volumes of cold PBS and centrifuged at 3,000 rpm for 20 min at 4°C to obtain the supernatant. To induce lipid peroxidation, the muscle homogenate (475 μL) was mixed with 50 μM ferric chloride (25 μL) and incubated for 30 min at 37°C. SDS (500 µL of an 8.1% solution) and 1 mL of 20% acetic acid (pH 3.5) were added to the muscle homogenate and centrifuged. Aliquots of the clear supernatant were mixed with an equal volume of TBA solution (0.8% w/v) and heated in a glass tube capped with aluminum foil at 95°C for 30 min. Samples were cooled on ice, and 100 µL of each sample was pipetted into 96-well plates, and the absorbance was read at 532 nm with a microplate reader.
Statistical Analysis
Data are presented as mean+SEM. Statistical analyses were performed using the SAS program (version 6.12; SAS Institute Inc., Cary, USA; http://www.sas.com). The behavioral data, such as locomotor activity, rota-rod performance, and forced swimming time, were compared using Kruskal–Wallis nonparametric one-way analysis of variance (ANOVA), followed by a Mann–Whitney U-test when appropriate. Other data including the intensity of BDNF immunoreactivity were compared using Fisher’s exact probability test. P-values <0.05 were considered statistically significant.
Results
Characteristics of hADSCs
Before hADSCs were transplanted, they were analyzed the capapcity for diffirentiation (Fig. 1A). As shown Fig. 1B, the hADSCs were showed as spindle shaped plastic-adherent cells, which are negative for hematopoietic marker (CD34 and HLA-DR), positive for several stromal markers (SSEA-4, CD90 and CD105). In karyotyping analysis, they possessed normal 23 pairs of chromosomes without abnormal findings (Fig. 1C). To confirm releasing growh factors of hADSCs, BDNF and NGF were measued in their conditioned medium (Fig. 1D). High concentrations of NGF (78.5 ± 3.4 pg/106 cells) and BDNF (219.0 ± 33.7 pg/106 cells) were detected in the conditioned medium, but they were not detected in normal fresh medium.
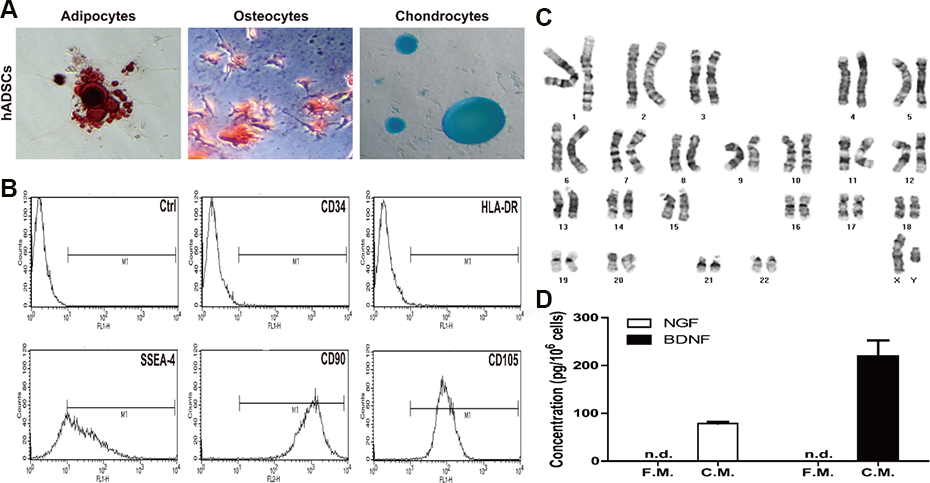
Characteristics of hADSC. (A) Capacity of differentiation of hADSCs. The cells were stained by Oil red O (adipogenic differentiation), Alizarin Red S (osteogenic differentiation), and Safranin O (chondrogenic differentiation). (B) Immunophenotype of hADSCs was carried out using flow cytometry for the following markers: SSEA-4, CD34, CD90, CD105, and HLA-DR. Karyotype of hADSC (C) and the concentration of NGF and BDNF released by hADSCs (D). F.M.; fresh medium, C.M.; conditioned medium, n.d.; not detected.
Effects of hADSCs on Physical Activity
To investigate the effect of hADSCs on physical activity of normal rats, locomotor activity was analyzed at PND 14, 20, 30, and 40 after hADSCs transplantation at PND 10. As shown in Fig. 2, for the moving time (slow moving + fast moving), there was no difference between the hADSCs-transplanted and vehicle control groups at PND 14 (Fig. 2A). Interestingly, however, the moving time increased in the hADSCs-transplanted groups in comparison with the vehicle control group from PND 20 to PND 40, in which ICV transplantation was more effective than IV injection.
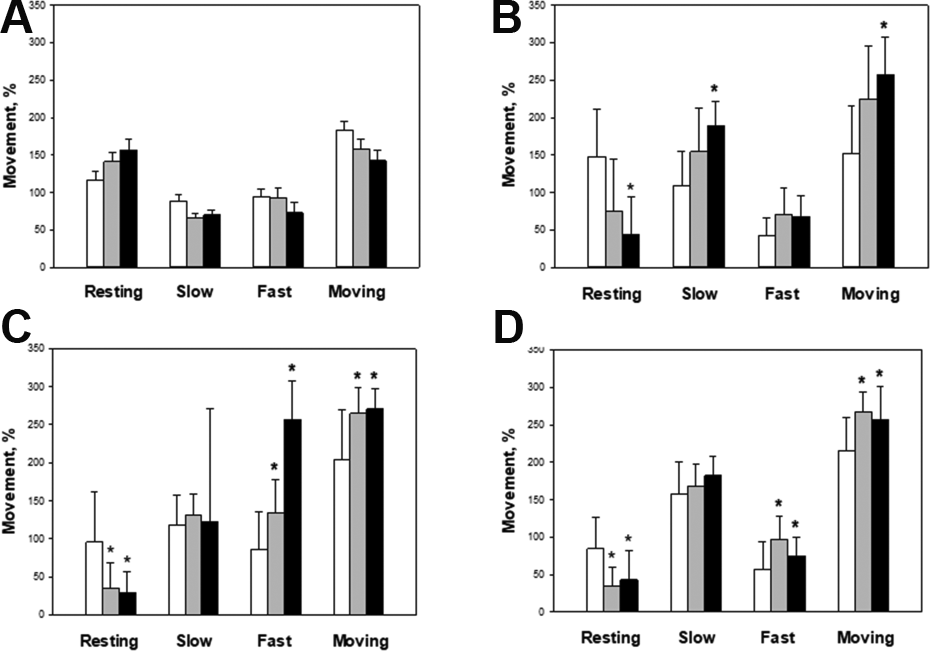
Locomotor activity of post-natal day (PND) 14 (A), 20 (B), 30 (C), and 40 (D) rats transplanted hADSCs on PND 10. The times for each movement type, i.e. resting, slow-moving and fast-moving, were recorded for 5 min, and the ratio was analyzed. White: control; Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
To assess motor balance and coordination, rota-rod test was performed. There were no differences in the latency time between the control and stem cell-transplanted groups from PND 14 to PND 30 (Fig. 3). In comparison, at PND 40 (Fig. 3D), the latency times of the stem cell-transplanted groups were longer than that of the control group: that is, 219.1 ± 28.4, 250.9 ± 26.1, and 300.2 ± 0.0 sec for the control, IV, and ICV, respectively. The time of ICV injection group was significantly longer than that of the control group.
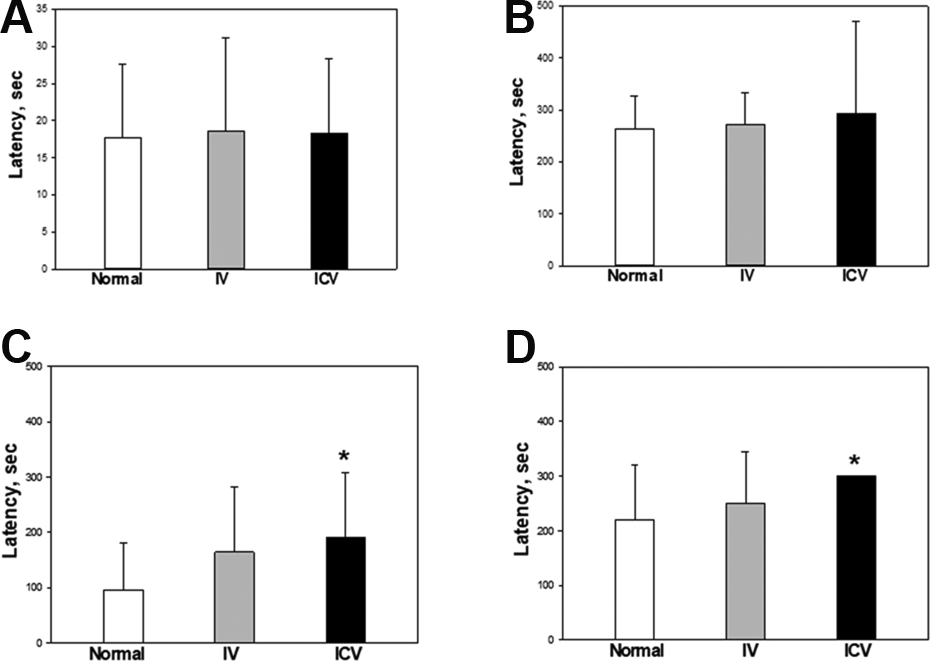
Rota-rod performance of post-natal day (PND) 14 (A), 20 (B), 30 (C) and 40 (D) rats transplanted hADSCs on PND 10. Rats were placed on rotating rod at a constant speed of 12 rpm. The endpoint was set up to 300 sec. White: control; Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
For the forced swimming test, the rats which were loaded with a lead ring weighing 5% of their body weight were placed in the water at room temperature to swim. The times to exhaustion of the control, IV, and ICV hADSCs transplantation groups were 736.2 ± 46.3, 976.3 ± 67.6, and 963.2 ± 46.5 sec, respectively (Fig. 4A). The duration was significantly increased by 32.6% and 30.8% following IV and ICV transplantation of hADSCs, respectively.
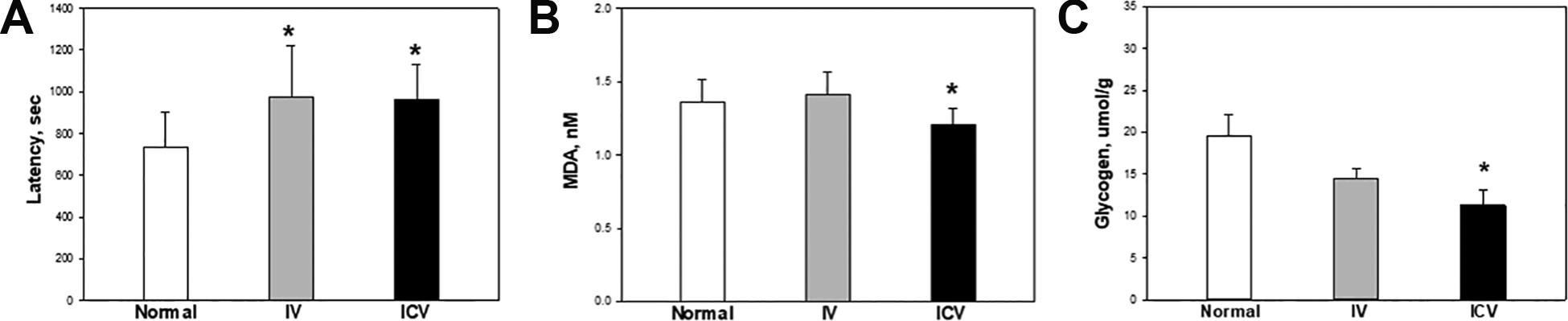
Endurance physical activity and damage markers in muscles. (A) The maximum swimming time was analyzed at post-natal day (PND) 41 after daily 30-min adaptaion for 4 days. The rats were loaded with a lead ring weighing 5% of their body weight attached to the tail, and then placed in a swimming pool. The cut off time was determined by failure to return to the water surface within 7 sec and used as a criterion of the maximum swimming capacity of the animals. The concentration of MDA (B) and glycogen (C) in the gastrocnemius muscles was measured after the forced swimming test at PND 45. White: control; Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
Levels of Muscular Damage Markers
To analyze muscular damage, TBARS (MDA) were analyzed (Fig. 4B). The TBARS content in the gastrocnemius muscles markedly increased after the forced swimming test. However, the increased lipid peroxidation was suppressed by ICV transplantation of hADSCs.
The levels of glycogen in the muscles of the control, IV, and ICV transplantation groups were 19.6 ± 2.4, 14.5 ± 1.2, and 12.1 ± 2.0 μmol/g, respectively (Fig. 4C). The glycogen concentrations were lower in stem cell-transplanted groups than those in vehicle group, and especially, the level of glycogen in the ICV transplantation group was significantly lower than that in the control group.
The blood damage markers of muscles such as CPK, LDH, TG, and AST were analyzed (Fig. 5). The serum CPK, LDH, TG, and AST levels in the ICV transplantation group decreased by 31.4%, 10.78%, 24.4%, and 11.9%, respectively, compared with the control group.
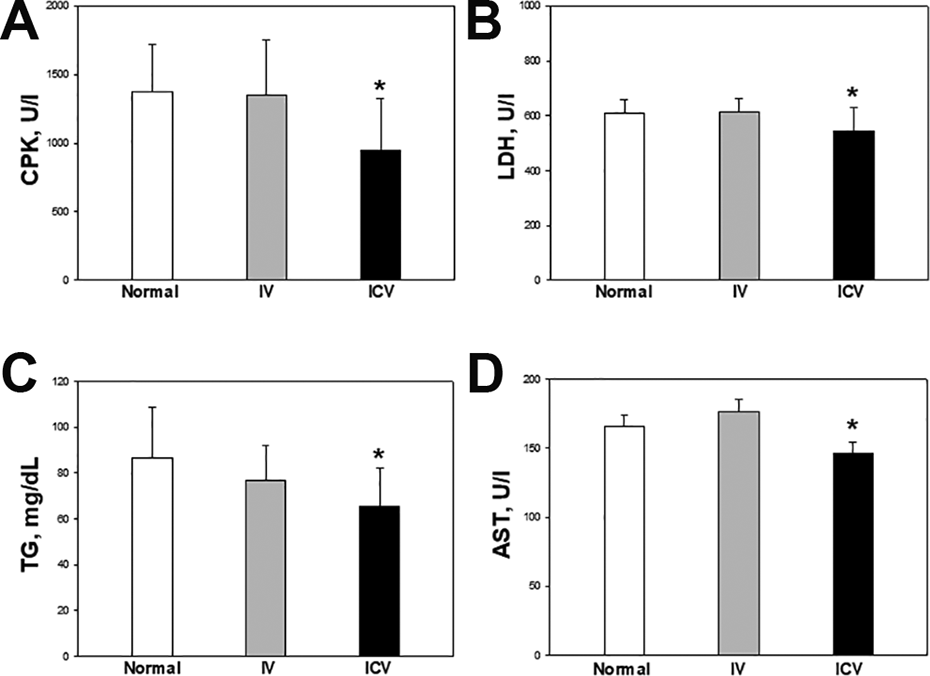
Muscle injury parameters in serum after endurance physical activity. Blood was collected and centrifuged to obtain serum at 3,000 rpm for 20 min from PND 45 rats. The (A) creatine phosphokinase (CPK), (B) lactate dehydrogenase (LDH), (C) triglyceride (TG) and (D) aspartate transaminase (AST) were used as muscle-injury parameters in serum. White: control: Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
Expression of BDNF
To determine whether the level of BDNF increased fllowing stem cell transplantation, western blotting was performed in the brain and muscles (Fig. 6). Transplantation of hADSCs increased the expression of brain and muscular BDNF in the stem cell transplanted groups compared with the control group. Especially, the effect of ICV injection of hADSCs on the expression of BDNF was superior to IV treatment.
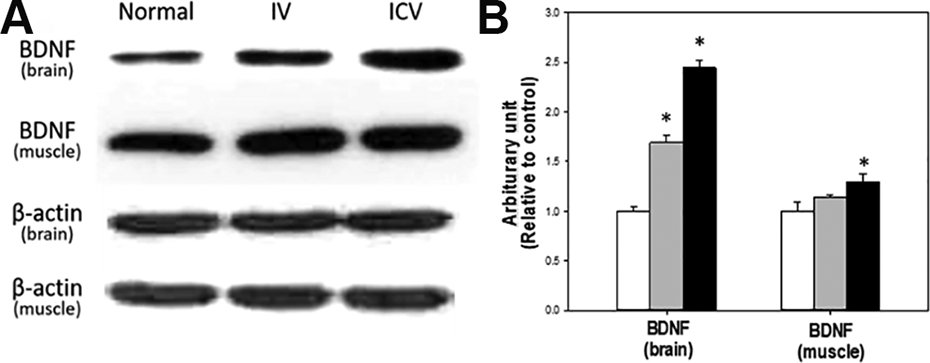
Expression level of BDNF in the brain and soleus mucles. Expression level was analyzed by western blot (A) and band densities were normalized to β-actin (B). White: control; Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
Myosin Heavy Chain (MyHC) Isoform Content
To determine whether transplantation of hADSCs affects the muscle fiber phenotypes, soleus muscles were stained for ATPase to analyze myosin type (Fig. 7). Percentages of slow-twitch fibers in soleus muscles of the control (44.6 ± 1.5), IV (71.6 ± 1.4), and ICV (81.6% ± 1.8%), indicated that the ratios of slow-twitch muslces in hADSCs-transplanted groups were significantly higher than that of control group.
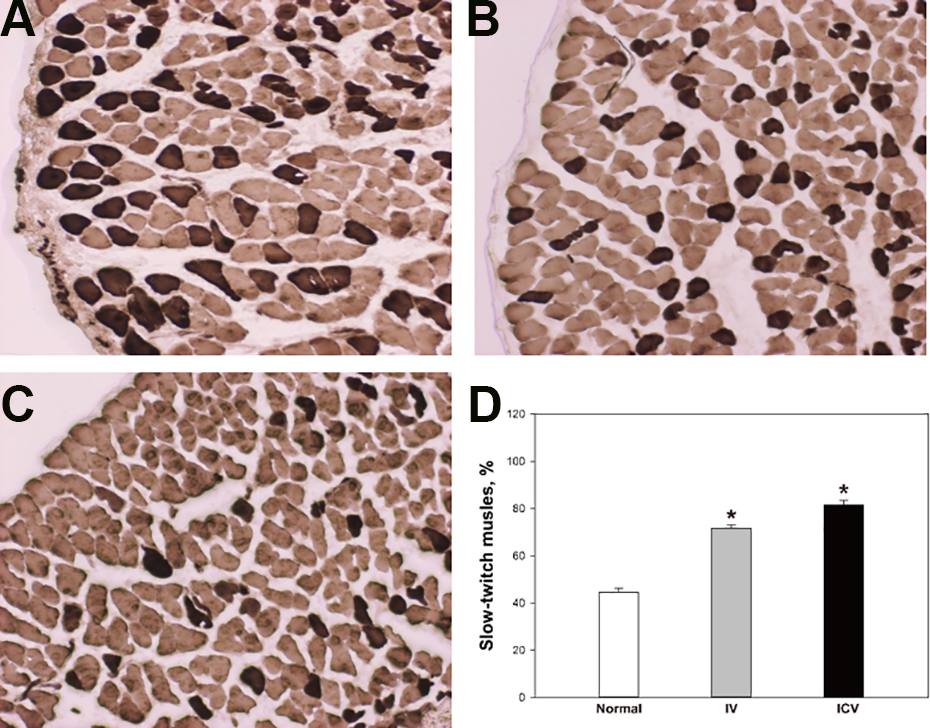
MyHC isoform content in soleus muscle. To analyze myosin type, soleus muscle was stained by ATPase stain (pH 9.4). (A) control, (B) IV, (C) ICV. (D) The percentage of slow-twitch fibers in soleus muscles. White: control; Gray: IV; Black: ICV. *Significantly different from control group (P < 0.05).
Adverse Effects of hADSCs Transplantation
To investigate the adverse effects of hADSCs transplantation, the rats were monitored to evaluate changes in body weights, clinical signs, and organ weights. There were no differences in body weight gains and clinical signs in all the control and hADSCs-treated animals (data not shown). As shown in Supplemental Table 1, there were no alterations in organ weights including gastrocnemius and soleus muscles governing and related to movement and fatigue among the control and hADSCs-treated groups.
Serum biochemical parameters of the rats treated with vehicle or hADSCs are depicted in Supplemental Table 2. There were no significant differences in the concentrations of BUN, creatinine, glucose, albumin, total cholesterols, total proteins, ALP, and ALT among groups. As referred to Fig. 5, muscular damage markers such as CPK, AST, LDH, and TG rather decreased following transplantation of hADSCs.
Discussion
Many researchers have focused on mesenchymal stem cell therapy because of its beneficial effectiveness. In particular, application of hADSCs in cellular therapy has been studied for neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS), muscular dystrophy, and CP 17 –19 . Also, hADSCs are one of the considerable sources of several NFs and GFs affecting biological functions in our body 20 –22 . Among NFs/GFs produced by hADSCs, it is well known that BDNF is likely to play the important role in physical activity 20 . Moreover, recent reports have shown that the widely-spread human adipose tissues provide abundant source of hADSCs, which can be easily and safely harvested as compared with other mesenchymal stem cells (MSCs) 2 .
In terms of adverse effects, there were neither considerable changes in body and organ weights including immune system, nor clinical signs of rats transplanted with hADSCs, in spite of no immunosuppressive therapy. It is well explained why MSCs are non-immunogenic and well tolerated in animals and humans due to negative expression of MHC II and releasing immunosuppressive cytokines 22 . Indeed, very few adverse events in clinical trials have been reported that can be related directly to the cell therapy 23 . The adverse events were rather related to the harvesting of adipose tissue, trauma associated with injection, or the nature of the underlying condition being treated 24 –26 .
Interestingly, in the present study, negligible number of hADSCs was observed in the brain or muscle tissues in normal rats 35 days after injection. Notably, we observed hADSCs 2 weeks in aged mice and 35 days in CP model rats after injection in previous studies 16,17 . Therefore, the fate of stem cells may be different according to the microenvironment in the normal and injured heterologous, allogeneic or autologous tissues. In spite of the rapid clearance of hADSCs, there were clear improvements in physical activity and stamina as well as muscle fiber change, which might be due to secretory factors from the stem cells.
Exercise leads to reactive oxygen species (ROS) generation in muscles, which induces lipid peroxidation, glutathione oxidation, and oxidative damage, as shown in the increase in TBARS 27,28 . Moreover, it has been well documented that exercise increases the activity of cytosolic enzymes in plasma such as LDH and CPK, blood parameters for muscular fatigue 29 –31 . Also, it was reported that excessive ROS increase triglycerides and AST in the serum 32 . In this study, CPK, LDH, AST, and TG increased during forced swimming were markedly decreased by ICV transplantation of hADSCs. Also, TBARS were reduced by ICV transplantation.
From the results of this study, it can be postulated that hADSCs have an anti-oxidative activity mediated by NFs/GFs. In the neurotrophin family, BDNF has been studied for its anti-oxidative cytoprotective effect 33 . Also, oxidative stress induces muscular fatigue after excessive exercise 34 . As a result of muscle contraction and metabolism, lactic acid is produced and accumulated in muscles by oxidizing enzymes such as NADH and lactate dehydrogenase 35 . Interestingly, BDNF decreased serum LDH activity and reduced infarct size in a myocardial infarction model 36 . Therefore, based on blood biochemistry, western blot, and TBARS analyses in this experiment, BDNF derived from hADSCs can be considered to exert a positive effect on reducing muscle fatigue and injury.
BDNF is related to locomotor activity 37 . Although its function is not clearly defined, BDNF expression increased in response to immobilization stress, but little is known about the physiological relevance 38 . Indeed, BDNF released from hADSCs increased physical activity in aged animals 15,16 and overexpression of BDNF led to an early improvement in locomotor function in spinal cord injury model 39 . BDNF acts as a muscle-derived neurotrophic factor for motoneurons, in which it rescued motoneurons from programmed or injury-induced cell death during development 40 . Furthermore, application of BDNF to the median gastrocnemius muscles significant increased electrical excitability of medial gastrocnemius motoneurons 41 . BDNF also plays an important role in the activity-dependent processes, including synapse development and plasticity 9,42,43 . Application of BDNF enhanced synaptic transmission by Ca2+ influx into the motor nerve terminals 44 . In similar mechanisms, it is believed that hADSCs increased the moving time in locomotor activity of rats via releasing BDNF.
On other hand, the latency to exhaustion of hADSCs-transplanted rats increased in rota-rod and forced swimming performances. Endurance exercise can trigger metabolic and structural remodeling in skeletal muscles, thus leading to changes in contractile properties and to increased angiogenesis in order to reduce muscle fatigue 45 . Also, fast-to-slow twitch transformation of muscles is mediated by the calcineurin-nuclear factor of activated T cells (NFATc1), and increased Ca2+ activates the calcineurin-NFATc1 cascade in skeletal muscles 11 . Increase in intracellular concentration of Ca2+ can lead calcineurin activation and NFATc1 nuclear import 46 . After nuclear import, it induces transformation of MyHC mRNA from a “fast” to a “slow” pattern with an increase in MyHC I mRNA and a decrease in MyHC IId mRNA 11 . With respect to BDNF, it can acutely elicit an increase in intracellular calcium concentration through the TrkB receptors and glutamatergic receptors, and induce Ca2+ transients in synapses 47 –49 . BDNF activates NFAT-dependent transcription via TrkB receptors 50 . It can activate the calcineurin-NFATc1 pathway and induce fast to slow transformation. Slow-twitch fibers (Type I) are more efficient at using oxygen to generate more ATP for continuous, extended muscle contractions over a long time and are highly resistant to fatigue 51 . As a result of physical activity increased by hADSCs, muscular glycogen levels were lowered by stem cell transplantation. Thus, it is thought that increased exercise in the endurance tests induced consumption of energy sources following hADSCs injection.
There are currently several available routes of stem cell transplantation for neurodegenerative diseases including the ICV and IV injection. However, the optimal route is unclear. For stroke patients, the intra-arterial route is a common preferred method after cerebral infarction 52 , since there may be a destruction of blood-brain barrier (BBB). By comparison, the intramuscular route is more effective in myopathy models 53 . In general, the IV route might be the most common and the leastly-invasive procedure 54 . After IV injection, however, most of the stem cells are distributed to other peripheral organs such as the liver, lung, and spleen, rapidly cleared, and unfortunately, only 1% of the injected cells are observed in the brain of healthy animals 55,56 . We confirmed that the transplantation of hADSCs enhanced physical activity, in which ICV transplantation was superior to IV injection. It is proposed that the ICV route delivers more cells, than the IV route, that reach cerebrospinal region and activate motoneurons governing skeletal muscles 57 . This may explain why ICV transplantation of hADSCs is more efficient in enhancing the physical activity, compared to systemic administration.
In the present study, we confirmed that transplantation of hADSCs to rats enhanced physical stamina. NFs/GFs, particularly BDNF, secreted from the cells might exerted multiple effects including antioxidant activity, glycogen metabolism, muscle type differentiation, and thereby enhanced physical activity. Although additional follow-up studies are required to clarify the exact action mechanisms, autologous and allogeneic hADSCs could be a good beneficial tool for the health care of aged or diseased people.
Supplemental Material
Supplemental Material, sj-docx-1-cll-10.1177_09636897211035409 - Stamina-Enhancing Effects of Human Adipose-Derived Stem Cells
Supplemental Material, sj-docx-1-cll-10.1177_09636897211035409 for Stamina-Enhancing Effects of Human Adipose-Derived Stem Cells by Eun-Jung Yoon, Hye Rim Seong, Jangbeen Kyung, Dajeong Kim, Sangryong Park, Ehn-Kyoung Choi, Yun-Bae Kim and Dongsun Park in Cell Transplantation
Footnotes
Authors Contribution
Eun-Jung Yoon, Hye Rim Seong: These two authors equally contributed to this work.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Ethical Approval
Ethical approval to report this research was approved by the Institutional Review Board of K-Stem cell, Seoul, Korea (KS-2015-01-001) and Institutional Animal Care and Use Committee of Laboratory Animal Research Center at Chungbuk National University, Cheongju, Korea (CBNUA-314-11-01).
Statement of Human and Animal Rights
All sample collection procedures from human beings and experimental protocols were approved by Institutional Review Board of K-Stem cell (KS-2015-01-001). All procedures in this study for animal experiments were conducted in accordance with the Institutional Animal Care and Use Committee of Laboratory Animal Research Center at Chungbuk National University (CBNUA-314-11-01).
Statement of Informed Consent
Human adipose tissues were obtained from male healthy donors undergoing elective liposuction procedures after informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
