Abstract
Examination of the therapeutic efficacy of neural stem cells (NSCs) has recently become the focus of much investigation. In this study we present an insight of the effects of combined application with neurotrophin-3 (NT-3) and NSCs that derived from rat embryo spinal cord on delaying denervated skeletal muscular atrophy after tibial nerve was severed. NT-3 gene was amplified by PCR and subcloned into lentiviral vector pWPXL-MOD to construct a lentiviral expression vector pWPXL-MOD-NT-3. A positive clone expressing NT-3 (named NSCs-NT-3) was obtained and used for differentiation in vitro and transplantation. Sixty adult rats, whose tibial nerves were sectioned, were divided into two groups: one grafted with NSCs-NT-3 (experimental group, n = 30) and the other with NSCs transfected by pWPXL-MOD (control group, n = 30). The cell survival and differentiation, NT-3 gene expression, and effect of delaying denervated skeletal muscular atrophy were examined through immunohistostaining, RT-PCR, Western blot, electrophysiological analysis, and mean cross-sectional area (CSA) of gastrocnemius, respectively. The results show that the NT-3 gene, which is comprised of 777 bp, was cloned and significantly different expression were detected between NSCs and NSCs-NT-3 in vitro. Quantitative analysis of the choline acetyltransferase (ChAT) immunopositive cells revealed a significant increase in experimental group compared to the control group 4 weeks after implantation (p < 0.01). Twelve weeks after transplantation, the ChAT immunopositive cells were detected near the engrafted region only in experimental group. Furthermore, the effect in delaying denervated skeletal muscular atrophy is indicated in the EMG examination and mean CSA of gastrocnemius. These findings suggest that the neural stem cells expressing NT-3 endogenously would be a better graft candidate for the delay of denervated skeletal muscular atrophy.
Keywords
Introduction
Injuries to the peripheral nerve, such as brachial plexus, median nerve, or ulnar nerve, present a serious problem for society, affecting -~2.8% of all trauma cases and often leading to poor recovery of function and subsequent impaired quality of life for the patient despite significant advances in microsurgical techniques over the last three decades (1,2,30). The occurrence of target skeletal muscle atrophy due to long-term denervation still represents an important cause of poor clinical results after nerve reconstruction. Denervation of adult skeletal muscle results in a rapid decline of its functional capacity and progressive atrophy of the skeletal muscle fibers. In order to delay the denervated atrophy of skeletal muscles, NSCs were reported to restore skeletal muscle function, survive, and regenerate axons that grow along the peripheral nerve to form junctions with the denervated muscle fibers (14,37). Genetically modified NSCs have been shown to ameliorate experimental spinal muscular atrophy, providing neurotrophic support or partially replacing interrupted innervation between neural cells and skeletal muscles (10,11).
The neurotrophins are a family of proteins that regulate the survival, differentiation, and maintenance of neurons and some nonneural cells (23). One member of the family, neurotrophin-3 (NT-3), plays pivotal roles in the nervous system (33,36). NT-3 showed positive functions in promoting neuronal survival, migration, differentiation, neurite growth, and regeneration (7,9,16,19, 21,25,29,31,42).
However, to our knowledge, there is little information available in the literature about combined treatment of neural stem cells and NT-3 gene for the delay of denervated skeletal muscular atrophy in rat. We present the results of the combined therapy at the molecular, cellular, and histological level that may be of importance in understanding the theoretical mechanism and clinic application in peripheral nerve injury recovery.
Materials and Methods
Preparation and Culture of NSCs Derived From Rat Embryo Spinal Cord
Animal experiments were approved by local committee review and conducted according to China Public Health Service Policy on Humane Care and Use of Laboratory Animal. NSCs were collected as previously described (28), with slight modification. Rat NSCs were obtained from spinal cord of P14 Sprague-Dawley rat embryo. The cells were collected in Dulbecco's modified Eagle medium and F12 nutrient (DMEM/F12, Gibco, 1:1) with N2 supplement (Gibco, 1:100), B27 supplement (Gibco, 1:50), epidermal growth factor (EGF, R&D Systems, Inc., 20 ng/ml), basic fibroblast growth factor (bFGF, R&D Systems, Inc., 20 ng/ml) for 5–7 days. A half-volume of medium was replaced with fresh medium every 3 or 4 days.
Cloning of NT-3 Gene and Construction of Lentiviral Expression Vector pWPXL-MOD-NT-3
Rat NT-3 cDNA (RZPD, Berlin) was amplified by PCR using the forward primer GGCACTAGTTCATGT TCTTCCGATTTTTC and the reverse primer GGGAC GCGTCAAGGTGATGTCCATCTTGTTTT. The PCR product was digested and inserted into the lentiviral vector pWPXL-MOD (Telebio Biomedical, Shanghai, China) to construct a lentiviral expression vector pWPXL-MOD-NT-3 according to the manufacturer's instructions. The resulting vector, named pWPXL-MOD-NT-3, contains a rat NT-3 gene and green fluorescent protein (GFP) gene. After transfecting NSCs for 5 days, the positive clone of the expressing NT-3 gene, named NSC-NT-3, was screened via fluorescence and applied to the transplant and differentiation in vitro.
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
After the cells were replenished with PBS, total RNA was extracted with RNeasy RNA extraction kit (Axygen). Total RNA (2 μg) was reverse-transcribed with oligo(dT) primer and RNase reverse transcriptase for 1 h at 42°C (Gibco BRL). After treatment with RNase H, the synthesized cDNA was subjected to PCR amplification. PCR conditions were optimized and linear amplification range was determined for each primer by varying annealing temperature and cycle number. After amplification, PCR products were electrophoresed on a 1.5% agarose gel and visualized by ethidium bromide staining. As a control for the RNA and cDNA quality, amplification of a β-actin transcript was used. Samples lacking the amplified β-actin product were not further processed for PCR.
Western Blot
To verify NT-3 expression by the NSC-NT-3 producer cells, immunoblotting with polyclonal rabbit anti-NT-3 antibodies (Chemicon, diluted 1:2000) was carried out according to the procedure previously described (22). Briefly, the day before harvest, confluent cells grown on 24-well plates (Falcon) were washed with HBSS, and 500 μl of DMEM (serum free) was replenished in each well. The cells were cultured for an additional 24 h. The medium was then collected and mixed with an equal volume of 23 sample buffer (125 mM Tris, 4% SDS, 20% glycerol, 10% 2-mercaptoethanol, pH 6.8). The cells were gently scraped off and homogenized with a Teflon glass homogenizer in 5 vol of homogenization buffer [50 mM Tris (pH 7.5), 2 mM EDTA, 1 mM PMSF, 25 mM leupeptin, 1.0% aprotinin]. The homogenate was centrifuged at 13,000 × g for 15 min at 4°C. The supernatant was then mixed with an equal amount of 23 sample buffer. Samples containing medium or cell homogenates were loaded onto adjacent lanes and separated by 10% SDS-PAGE and then transferred onto polyvinylidene difluoride membranes (Millipore) and processed for Western blotting. The polyvinylidene difluoride membranes were first incubated with 5% nonfat dry milk in TTBS buffer (0.1% Tween 20, 150 mM NaCl, 50 mM Tris-HCl, pH 7.6) for 1 h followed by overnight incubation with primary antibodies for NT-3 (Chemicon, diluted 1:2000). The membranes were then rinsed three times with TTBS buffer for 10 min and incubated with secondary antibody HRP-conjugated goat anti-rabbit IgG (Sigma, diluted 1:4000) for 1 h. The immunoreactivity was visualized by chemiluminescence with ECL reagents (Pierce).
In Vitro Differentiation Procedure
Tissue culture-treated chamber slides (Nalge Nunc, Rocherster, NY) were coated with 0.01% poly-L-lysine (Sigma, St. Louis, MO) for 60 min at room temperature, then washed with deionized water and allowed to dry in the hood. NSCs transfected with pWPXL-MOD-NT-3 were seeded into the chamber slide at a concentration of 3000/cm2 with basic medium then maintained at 37°C in humidified 95% air and 5% CO2 in order to make them adherent. After 2 h, the basic medium was replaced by differentiation medium composed of DMEM/F12 + 20% FBS and then the cells were cultured under the same condition described above for 7 days.
Fluorescence Immunocytochemistry
Fluorescence immunocytochemistry was performed to evaluate whether the differentiated cells expressed special markers. Briefly, the culture medium was removed, and the cells on the culture slide were rinsed three times with PBS. The cultured cells were fixed with 4% paraformaldehyde for 30 min and then immersed in PBS for about 10 min, followed by exposure to 0.01% Triton X-100 at room temperature for 10 min.
For blocking nonspecific immune reaction, the cells were treated with 10% normal goat serum (Santa Cruz, CA) at room temperature for 30 min. Cells derived from NSCs-NT-3 were incubated with one of the following primary antibodies: mouse anti-ChAT (dilution 1:200, Chemicon; ChAT is an enzyme catalyzing the synthesis of acetylcholine; it is a marker for cholinergic neurons) monoclonal antibodies, mouse anti-glial fibrillary acid protein (GFAP; dilution 1:200, Chemicon; GFAP is an intermediate filament protein that is thought to be specific for glial cells and astrocytes) at 4°C overnight and washed three times with cold PBS for 3 min after observing fluorescence emitted from GFP. PBS was used instead of primary antibodies in negative contrast. Then, primary antibodies were washed and fluorescent secondary antibodies cy3-conjugated goat anti-mouse IgG (dilution 1:100; Sigma) was added for 2 h. The fluorescence emitted from the secondary antibody was observed through an appropriate filter using a fluorescence microscope (Leica DM IRB, Wetzlar, Germany) equipped with phase-contrast optics.
Preparation of Cells for Transplantation
On the day of surgery dishes containing 90% confluent cells were washed three times with Hanks' balanced salt solution (HBSS) for 3 min, trypsinized, gently triturated, counted, washed, pelleted (1500 rpm for 5 min), and resuspended in growth medium at a concentration of 105/μl. Cell viability was shown by trypan blue (Sigma) stain to be greater than 80–90% prior to surgery. The cells were maintained on ice during surgery.
Surgery
The animal study protocol was approved by local committee review and conducted according to China Public Health Service Policy on Humane Care and Use of Laboratory Animal. Sixty adult male Sprague-Dawley rats (120–150 g), divided into two groups [n = 30, grafted with NSCs-NT-3 (experimental group) and with NSCs infected by blank lentiviral vector pWPXL-MOD (n = 30, control group)], were anesthetized IP with sodium pentobarbital (50 mg/kg) during the surgical procedures. After skin incision, the sciatic nerve, tibial nerve, and peroneal nerve were exposed using a muscle splitting incision. With the aid of an operation microscope (SM-2000J, Shanghai Eder Ltd, Shanghai), the right tibial nerve was severed 1 cm above the gastrocnemius muscle. The tibial nerve was tightly ligated with surgical silk in two places, cut between the sutures, and a 0.4-cm section of the nerve was removed. Both proximal and distal nerve stumps were anchored in the surrounding tissue as far away from each other as possible. This procedure allows a permanent and complete denervation of the lower leg (4,40).
At the seventh day after the tibial nerve severed, suspension of medium and prepared NSCs-NT-3 (105/μl) were injected into tibial nerve just proximal to the nerve branch of the gastrocnemius using a microsyringe, 20 μl for each animal over 10 min and the needle was left in place for an additional 15 min before withdrawal with the aid of an operation microscope. Control rats (n = 30) received an equal volume (20 μl) of NSCs. Ligatures were placed proximal to the injection site to further secure the implanted cells and to prevent endogenous nerves from infiltrating the implanted nerve. The wound was subsequently closed in layers using 5-0 silk sutures. The rats were given food and water ad libitum. Animals in each group were sacrificed at 1 week ((n = 10, subgroup 1), 4 weeks ((n = 10, subgroup 2), and 12 weeks ((n = 10, subgroup 3) after surgery and tissue samples of tibial nerve and right gastrocnemius muscle were taken.
Electrophysiological Study
Electrophysiological recording was determined as reported by Mimura et al. (24), with minor modification. Briefly, 12 weeks after surgery, the rats were anesthetized IP with sodium pentobarbital (50 mg/kg) and the tibial nerves were exposed. Electric stimulation (duration of 0.2 ms, intensity of 3 mA, Keypoint® portable, Medtronic) was applied to the proximal side of implanted sites. The compound muscle action potential was recorded in the gastrocnemius with an active monopolar needle electrode 10 mm below the tibia tubercle and with a reference needle 20 mm from the active electrode. We calculated the area of the recorded muscle response (mV& × ms) and amplitude, which can be considered to reflect the amount of activated fibers (5).
Tissue Preparation
After electrophysiological study, animals were euthanized and perfused with 200 ml of normal saline followed by 500 ml of ice-cold 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). The distal tibial nerve containing the graft was harvested with the aid of an operation microscope and immersed in 0.1 M phosphate buffer (PB) at 4°C overnight followed by cryoprotection in 30% sucrose (in 0.1 M PB containing 0.5 mM Thimerosal) for 2 days. Tissue was then embedded in OCT compound (Fisher Scientific) and cut into 20-μm-thick longitudinal sections on a cryostat. Then gastrocnemius muscles were served and fixed for advanced examination.
Histological Examination
At the completion of electrophysiological study, the animals were euthanized. The gastrocnemius muscle in the right leg was removed. For advanced examination, right gastrocnemius muscles were fixed in 10% methanal. Transverse paraffin sections (7 μm) were taken from the midbelly of the muscle and air dried onto cleaned slides. Sections were stained with hematoxylin and eosin (H&E) staining. The sections were imaged digitally. The image of each muscle was covered with a grid of squares for random sampling of fibers. At least 50 squares were chosen, from which 500 fibers were measured per muscle (Panasonic, MV-CP410, Japan).
Immunohistochemistry
Fluorescence immunocytochemistry was performed to detect the differentiation of NSCs-NT-3 and NSCs in vivo. Tissue sections of tibial nerve were subjected to immunostaining with primary antibodies against nestin (dilution 1:200; Sigma) in subgroup 1 and ChAT (dilution 1:200; Chemicon) in subgroups 2 and 3 after observing fluorescence emitted from GFP. The immunore-active signals were observed by fluorescent secondary antibody cy3-conjugated goat anti-mouse IgG (dilution 1:100; Sigma). To avoid nonspecific immunostaining by anti-mouse secondary reagents, negative control staining was also performed using mouse IgG. Leica QWin Standard V3.1 System (Leica Microsystems Image Solution Ltd.) was used to count ChAT immunopositive cells.
Statistical Analysis
Data are expressed as mean ± SE. Comparisons between the experiment and the control group were performed using one-way ANOVA and Bonferroni pos hoc testing. A difference was considered statistically significant at p < 0.05.
Results
Characterization of the Cells Differentiating From NSCs-NT-3
The NSCs-NT-3 were cultivated with differentiation medium for 7 days. These cells differentiated from the NSCs-NT-3 and NSCs were fixed and proved to be ChAT immunopositive cells (Fig. 1A) and glial-like phenotype cells (GFAP immunopositive) (Fig. 1B) by using immunocytochemistry after fluorescence emitted from GFP was observed. No ChAT immunopositive cells were detected in NSCs group.
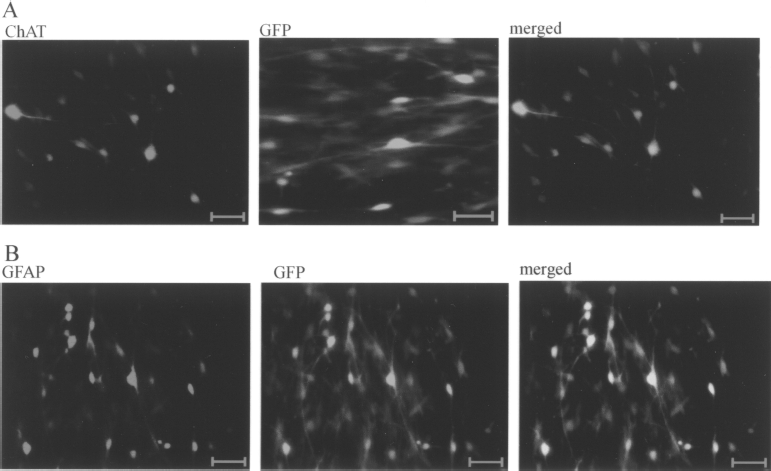
Fluorescence immunocytochemistry analysis of cells differentiating from NSCs-NT-3. (A) ChAT immunopositive cells emitting green fluorescence. (B) GFAP-positive cells emitting green fluorescence. Magnification: 200×. Scale bars: 100 μm.
Cloning of NT-3 Gene and Construction of Lentiviral Expression Vector
The NT-3 gene was cloned by PCR (Fig. 2). A full-length NT-3 gene comprises a 777-base pair nucleotide fragment. Sequencing indicated that the cloned fragment is accurate (data not shown). NT-3 gene was then inserted into pWPXL-MOD at the GFP site of the lentiviral expression vector pWPXL-MOD.

Cloning of NT-3 gene and construction of lentiviral expression vector. The NT-3 gene was cloned by PCR and agarose electrophoresis indicated a band of 777 bp.
Exotic NT-3 Effective Expression in NSCs
Total RNA was extracted from NSCs-NT-3 and NSCs after transfection for 24 h and analyzed by RT-PCR (Fig. 3A). The expression of the NT-3 in NSCs-NT-3 was further confirmed by Western blot (Fig. 3B).

Exotic NT-3 expression in NSCs. The expression of NT-3 was evaluated through RT-PCR (A) and Western blot (B). It is obvious that NT-3 has a high expression in NSCs-NT-3 at the transcriptional level. Protein expression was also further detected by Western blot experiment and was indicated an increase expression (B).
Detection of Grafted Region
One week posttransplantation, engrafted NSCs at the injection site labeled with NSCs marker nestin, as seen in Figure 4A. Four weeks after implantation, percentage of cells both ChAT immunopositive and emitting green fluorescence was 26.24 ± 5.64% and 9.29 ± 2.68%, respectively, in the experimental group and control group; in addition, there was high statistically significant difference (p < 0.01) (Fig. 4B, C). Twelve weeks posttransplantation, cells both ChAT immunopositive and green fluorescence were only detected in the experimental group and the percentage of these cells was 10.46 ± 0.65%; green fluorescent cells were not detected in control group (Fig. 4D). These results taken together showed NSCs-NT-3 and NSC can survive and differentiate into ChAT immunopositive cells in engrafted region but the former survived much longer and differentiated into much more ChAT immunopositive cells.
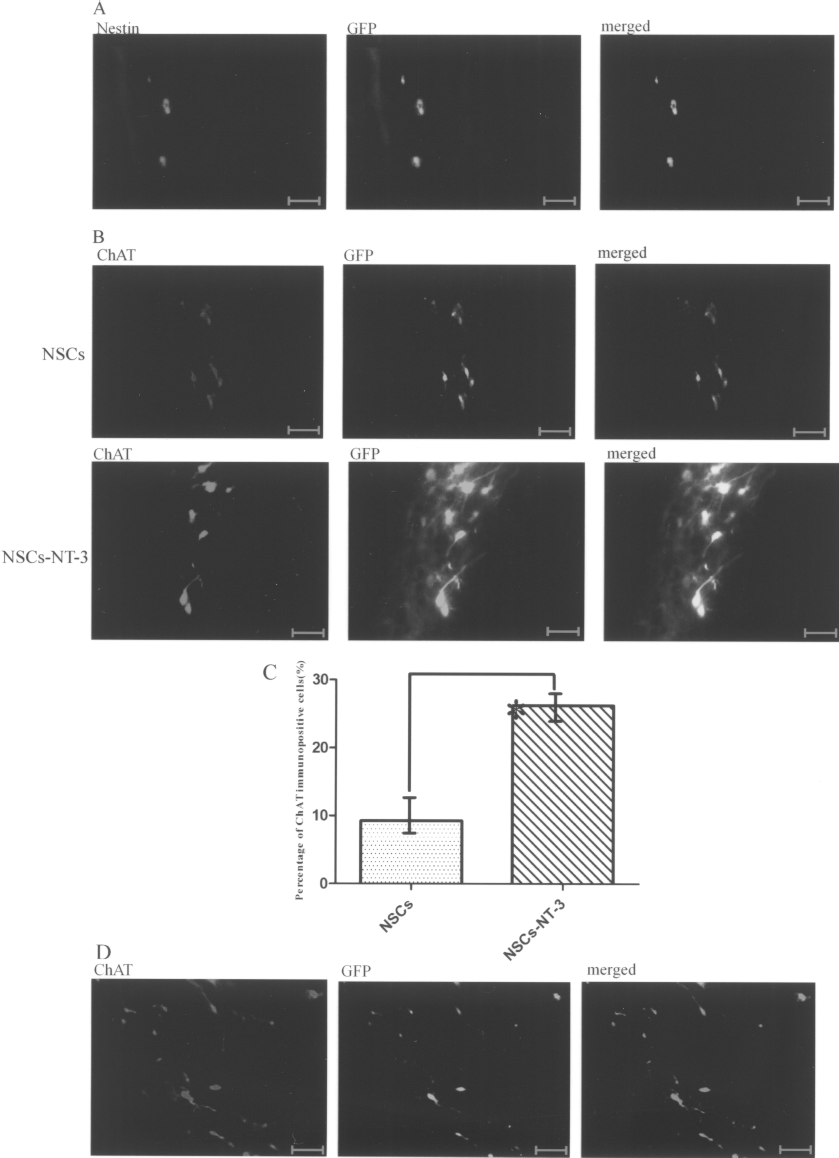
Detection of grafted region. Both nestin immunopositive and emitting green fluorescence cells were detected 1 week after transplantation (A, 200×). Significant differentiation difference between NSCs and NSCs-NT-3 was indicated through cholinergic neuron marker ChAT and green fluorescene (B, 400×) that the rate of ChAT immunopositive cells was sharply increased in NSCs-NT-3, in contrast to NSCs 4 weeks after grafting (C). The proportion of ChAT immunopositive cells was 26.24 ± 5.64% in NSCs-NT-3 while 9.29 ± 2.68% in NSCs by calculation (n = 10, mean ± SD; *p < 0.01). Twelve weeks after implantation, cells both ChAT immunopositive and emitting green fluorescence were only detected in the experimental group (D, 400×). Scale bars: 100 μm.
NSCs-NT-3 Is Propitious to Delay of Denerveted Skeletal Muscular Atrophy After Tibial Nerve Transaction in the Rat
Twelve weeks after NSCs were put into the tibial nerve, the compound muscle action potential (CMAP) was recorded in experimental animals (Fig. 5A). The impaired conduction latency and amplitude after transection were improved in two groups and there was significantly difference (p < 0.03 and p < 0.02, respectively) (Fig. 5B). The percentage of activated fibers was higher in the experimental group than the control group and it showed significant difference (p < 0.04) (Fig. 5B), as evidenced by the increases of whole response area under the curve.
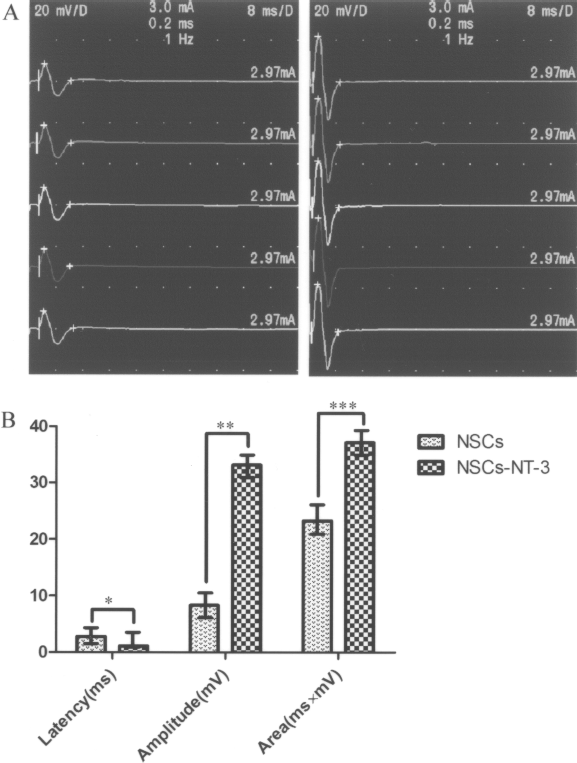
Electromyographic (EMG) analysis. The representative EMG recording waves were elicited in response to electrical stimulation of the tibial nerve proximal to the engrafted region in experimental and control groups 12 weeks after surgery (A). The stimulation intensity was 0.3 mA and the duration was 0.1 ms. The quantitative results of latency and compound muscle action potential determined by the calculation of (i) mean area (mV × ms) under the whole response curve and (ii) EMG amplitude are depicted (B). Latency, amplitude, and area differences were observed in two groups. *p < 0.03; **p < 0.02; ***p < 0.04.
The right gastrocnemius muscle was harvested completely. The results of H&E staining and muscle fiber areas from media gastrocnemius muscle are shown in Figure 6. These data suggested that NSCs-NT-3 are propitious to the delay of denervated skeletal muscular atrophy in rats.

Mean ± SE CSA for two groups 12 weeks following implantation (n = 10) was 882.54 ± 61.05 and 542.25 ± 22.17 μm2, respectively, in the experimental group and control group; furthermore, it showed highly statistically significant difference. *p < 0.01.
Discussion
Transection of an adult peripheral nerve causes detrimental changes to both muscle and nerve. Atrophic and degenerative changes within the denervated muscle impedes the reinnervation process, especially after long-term denervation. This suggests that prevention of the degenerative processes within the denervated muscle should promote reinnervation and be of clinical benefit in the treatment of nerve injury. Recently, NSCs transplantation was a potential therapy for regenerative medicine to replace the lost or damaged neural cells due to degenerative disease or trauma (20,41). Especially for the delay of denervated skeletal muscular atrophy, this approach is expected as a potential treatment that not only promotes neuroregeneration, but also rescues the denerveted skeletal muscles (11,37,40). Therefore, original preparation and genetic modification of the neural stem cell for transplantation were widely studied in the past years in order to explore better ways for the transplanted cell survival, differentiation, and functional recovery (6,8,19,42).
NSCs originated from embryonic spinal cord were selected as an interesting donation source for transplantation, because these NSCs have shown the ability of differentiation into neuron and glial cells. However, it is still difficult for us to induce a sufficient amount of NSCs into a special type of cell. Growth factors showed pivotal function in neuron development, so some investigations were reported utilizing the genetically modified NSCs to secret NGF or BDNF in order to affect surrounding cells differentiation (38). Neural stem cells infected by lentivirus with human NGF differentiated into neurons, astrocytes, and oligodendrocytes in vitro; some neurons even showed ChAT immunopositivity (13).
NT-3 is an attractive member of the neurotrophins, and plays a key role during brain development, especially in neural stem cells differentiation and migration, and it is involved in neurotransmitter regulation, neural peptide synthesis, neuron morphology, and synapses function (12,18,21,32). It has also been shown to promote neuronal survival, peripheral nerve regeneration, and maintain neurons and some nonneural cells (19,29, 34,35).
In present study, the effects of a combinatorial treatment of NT-3 gene and neural stem cells method were investigated. The NSCs stable expression of NT-3 showed motive differentiated into ChAT immunopositive cells in high proportion without the other cytokine induced. The data showed that endogenous NT-3 has distinctive roles in regulating the differentiation of NSCs, and is capable of driving neural stem cells differentiating into ChAT immunopositive cells. Potential challenge with this cell therapy includes developing certain techniques designed to promote better graft survival. Alternatively, enhancing graft-mediated secretion of neurotrophic factors, such as NT-3, could facilitate the effect of delaying atrophy. Therefore, transfecting a gene into neural stem cells may be an equally potent cell therapy.
Moreover, through electrophysiological analysis and histological examination, we found a distinct difference between NSCs-NT-3 and NSCs regarding the effect of delaying denervated skeletal muscular atrophy after tibial nerve was sectioned in the rat. We reasoned that NSCs-NT-3 can secret NT-3, which is not only a neurotrophic factor but also maybe a myotrophic factor persistently. Firstly, NT-3 can promote regeneration of injured tibial nerve, that is to say, it reduced time of denervation; furthermore, most important is the differentiation cells, whose survival was affected by NT-3, presumably partially replacing interrupted innervation between motorneurons and the denervated gastrocnemius muscle. Further studies are needed to clarify whether NT-3 has the myotrophic action on the denerveted muscle, although many investigations were reported that some growth factors have both neurotrophic action and myotrophic action; more importantly, they can be anterogradely transported within transected nerves (3,15,17,26, 27,39).
In conclusion, our in vitro and in vivo data clearly showed that the persistently endogenous expression of NT-3 enhanced the proportion of ChAT immunopositive cells, their survival, and effect to delay denervated skeletal muscular atrophy. The multiple merits of this method illustrate the ways to its extensive application to the NSCs transplantation. Our findings also have significant clinical implications. Results to date suggest that NT-3 may prove to be useful in clinical practice to assist in the delay of denervation atrophy after peripheral nerve injuries. It would be convenient to harness it to deliver NT-3 to both nerve and muscle at the same time with one administration.
Footnotes
Acknowledgment
This work was supported by National Science Foundation of China (30271308; 30672124).
