Abstract
Angiogenesis is a major obstacle for wound healing in patients with diabetic foot wounds. Mesenchymal stem cells (MSCs) have an important function in wound repair, and neurotrophin-3 (NT-3) can promote nerve regeneration and angiogenesis. We investigated the effect of NT-3 on accelerating wound healing in the diabetic foot by improving human bone marrow MSC (hMSC) activation. In vitro, NT-3 significantly promoted VEGF, NGF, and BDNF secretion in hMSCs. NT-3 improved activation of the hMSC conditioned medium, promoted human umbilical vein endothelial cell (HUVEC) proliferation and migration, and significantly improved the closure rate of HUVEC scratches. In addition, we produced nanofiber mesh biological tissue materials through the electrospinning technique using polylactic acid, mixed silk, and collagen. The hMSCs stimulated by NT-3 were implanted into the material. Compared with the control group, the NT-3-stimulated hMSCs in the biological tissue material significantly promoted angiogenesis in the feet of diabetic C57BL/6J mice and accelerated diabetic foot wound healing. These results suggest that NT-3 significantly promotes hMSC secretion of VEGF, NGF, and other vasoactive factors and that it accelerates wound healing by inducing angiogenesis through improved activation of vascular endothelial cells. The hMSCs stimulated by NT-3 can produce materials that accelerate wound healing in the diabetic foot and other ischemic ulcers.
Introduction
It is estimated that the number of patients with diabetes mellitus will increase to 439 million in 2030 (42); 15% of these patients will have diabetic foot disease, of which about 1% will lead to amputation (3). Thus, new methods that promote healing in diabetic foot disease are vitally important to reduce the morbidity and mortality from diabetes mellitus.
Patients with diabetic foot disease have wound healing disorders because they lack a normal wound healing mechanism (6, 19). However, little is known about the pathogenesis of wound healing disorders in diabetic patients. Current studies indicate that the main causes of diabetic foot disease are diabetic peripheral vasculopathy, peripheral neuropathy, and regional ischemic ulcer (16).
Presently, stem cell approaches that include endothelial progenitor cells (EPCs) and mesenchymal stem cells (MSCs) for treating diabetic foot disease are promising, and they have thus become a hot topic of research (1, 2, 8, 50). It is reported that growth factors, such as epidermal growth factor (EGF), insulin-like growth factor-1 (IGF-1), and basic fibroblast growth factor (bFGF-1), can promote proliferation in fibroblasts and thus can potentially be used to treat diabetic foot disease (7). Preliminary studies indicate that, though these factors directly promote neovascularization, the efficacy of one single factor in promoting vascularization for diabetic foot disease treatment will decline over time (27). Therefore, novel effective therapeutic strategies must be developed to promote angiogenesis in treating the diabetic foot disease ischemic ulcer.
MSCs are broadly applied to prevent human disease (15, 24). In the cardiovascular system, MSCs protect ischemic cardiac muscle through a paracrine mechanism (21) and various cytokines that promote angiogenesis (25). The nerve growth factor family, which includes nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and neurotrophin-3 (NT-3), has been shown to play a key role in cardiovascular development (9, 17). BDNF and NT-3 gene knockout mice exhibit severe heart malformation during development (9, 16, 22, 47). Moreover, NGF inhibits vascular endothelial cell apoptosis and promotes angiogenesis when it is applied to type 1 diabetes mellitus (46), and intramuscular NT-3 injection in ischemic limbs dramatically promotes muscular angiogenesis (13). Thus, these studies indicate that neurotrophic factors may be a promising therapeutic target for treating cardiovascular diseases.
In vivo, both the vasculature and the nerve system grow along the same route and influence each other (33). Thus, NGF may be a novel therapeutic target for promoting neovascularization (12). Based on our understanding of the primary pathogenic factors in diabetic foot disease, further studies are warranted on the efficacy of combining NGF and MSCs with applied biology engineering technology to promote neovascularization and accelerate diabetic foot wound healing.
Therefore, we used NT-3-stimulated human bone marrow MSCs (hMSCs) that were implanted into a novel biomaterial to treat mice with a diabetic foot. We found that NT-3 stimulation promoted MSC proliferation and migration. Furthermore, NT-3-activated hMSCs facilitated foot wound healing in diabetic mice. These results indicate that a combination of nerve growth factor and hMSCs may be a new method for treating diabetic foot disease.
Materials and Methods
Cell Culture and Experimental Groups
Green fluorescent protein (GFP)-transfected hMSCs from 18- to 40-year-old male donors were purchased from Cyagen Biosciences (Guangzhou, China), and human umbilical vein endothelial cells (HUVECs) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Medium that consisted of Minimum Essential Medium Eagle-Alpha Modification (α-MEM), l-glutamine (2 mM) (Hyclone Laboratories, Logan, UT, USA), penicillin (100 U/ml), streptomycin (100 mg/ml) (all from Sangon.Com, Shanghai, China), and 10% fetal bovine serum (FBS; Gibco, Gaithersburg, MD, USA) was used as the basic MSC medium. MSCs were used between passage 4 and passage 6 in vivo or in vitro experiments. During these experiments, cultures showed no evidence of crisis or senescence. MSC basic media that contained 5.6 and 25 mM glucose (Sigma, St. Louis, MO, USA) were used as low- and high-sugar glucose media (29), respectively. The cells in MSC high-glucose medium supplemented with 100 ng/ml NT-3 (R&D Systems, Minneapolis, MN, USA) composed the NT-3 group (NT-3); the cells with 5 μM triciribine (Kangchen Biotech, Shanghai, China), an V-Akt murine thymoma viral oncogene homolog 1 (Akt) inhibitor, added to the NT-3 group composed the Akt inhibitor group (Akt inhibitor). Neither NT-3 nor triciribine was added to the control group (control).
The MSCs were seeded into 24-well plates (Corning, NY, USA) at 1.5 × 105 cells/well and cultured in MSC high-glucose medium with NT-3 and/or triciribine. After 24 h, the MSCs were washed three times with phosphate-buffered saline (PBS; Beijing Zhongshan Golden Bridge Technology Co. Ltd., Beijing, China) after discarding original medium and new MSC high-glucose medium was added. The cells were continuously cultured for 48 h, and the supernatant was extracted [conditioned medium (CM)]; these cells are referred to as MSC CM, NT-3 CM, and Akt inhibitor CM, respectively. The conditioned media were filtered with a 0.22-μm filter (Corning) and stored at −70°C until use.
Roswell Park Memorial Institute (RPMI)-1640 medium (Hyclone Laboratories) with 10% FBS was the basic HUVEC medium. Media that contained 5.6 and 30 mM glucose were used as the HUVEC low- and high-glucose media, respectively. The HUVEC experimental groups are referenced similarly to the CM groups.
MTT Assay
The 3-(4,5-dimethylthiazol-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was used to detect hMSC and HUVEC proliferation. The cell were cultured in 96-well plates (Corning) at 8 × 103 cells/well. The cells were cultured with the appropriate cytokines or conditioned medium (1:5 dilution) and incubated at 37°C and 5% CO2 for 48 h. The MTT assay was performed by adding 20 μl MTT (5 mg/ml, Sigma) to each well for 4 h at 37°C. The supernatant was removed, 150 μl dimethyl sulfoxide/well (DMSO, Sigma) was added, and the samples were shaken for 10 min. The absorbance value (OD) of each well was measured using an Emax microplate reader (Molecular Devices, Sunnyvale, CA, USA) at 492 nm.
Cell Scratch and Transwell Chamber Experiments
The cell scratch experiment was performed as follows (2, 26, 30). HUVECs or hMSCs were seeded at 1.5 × 106 cells/well in six-well plates (Corning) and cultured with 0.1% FBS in MSC or HUVEC high-glucose medium for 24 h. A central scratch was created by scraping cells away with a p1000 pipette tip. The cells were cultured with the appropriate cytokines or conditioned medium (1:5 dilution) as well as 2 mM hydroxyurea (Sigma) and incubated at 37°C in 5% CO2 for 24 h. The cells were fixed with 4% paraformaldehyde (Sigma), and the HUVEC scratches were stained with 0.5% crystal violet (Sigma). The scratches were recorded with an OLYMPUS BX50 microscope (Tokyo, Japan), and the cell scratch area at 0 and 24 h were measured using IPP software (Media Cybernetics, Rockville, MD, USA).
The transwell chamber experiment was performed as follows (44). HUVECs were cultured with 0.1% FBS high-glucose medium for 24 h, and the cells were placed in 25 mm, 8.0 μm transwell cell chambers (Corning) in the upper chamber at 5,000 cells/well. The MSC CM, NT-3 CM, or Akt inhibitor CM (diluted 1:5 in serum-free medium) was added to the lower chamber. The cells were incubated at 37°C in 5% CO2 for 6 h. Cells that migrated to the other side of the membrane were fixed with 4% paraformaldehyde and stained with 0.5% crystal violet.
Detection of Cytokines in the Conditioned Medium
The levels of vascular endothelial growth factor (VEGF), NGF, and BDNF protein in the conditioned culture supernatants were determined using enzyme-linked immuno-assays (R&D Systems) according to the manufacturer's instructions.
Growth of hMSCs in the Biological Material
The biological material was composed of the following components by weight: 80% polylactic acid (PLA, Sigma), 10% silk fibroin (Sigma), and 10% collagen (Sigma). Using hexafluoroisopropanol (HFIP, Sigma) as a solvent, polymer solutions of 5 wt.% were prepared. The mixture was magnetically stirred at a temperature below 30°C in a closed system for 4 h to allow complete dissolution of the polymers. PLA sheets with 0.9–1.2 mm thickness were prepared by the electrospinning process with the following optimal process parameters: the solution was fed into a 10-ml syringe (BD, Franklin Lakes, NJ, USA), the applied electrical field strength was 22 kV/m, the diameter of spinneret hole was 0.4 mm, the tip collector distance was 9 cm, and the spinning speed was 0.5 ml/h. The PLA sheets were immersed repeatedly in the 0.1 M phosphate buffer solution (Zsbio.com, China) after drying for removing hexafluoroisopropanol (23, 39, 53). hMSCs were seeded in the biological tissue material at 8 × 103 cells/material; the cells were treated with 100 ng/ml NT-3 or 5 μM triciribine and incubated at 37°C in 5% CO2 for 48 h. The cells were counted using a Leica TCS SP5 laser scanning confocal microscope (Mannheim, Germany).
Preparation of Cell-Containing Biological Material
hMSCs were seeded in the biological tissue material at 2 × 104 cells/material. The cells were treated with NT-3 (100 ng /ml). After 48 h of incubation, the material was washed three times with PBS and then used in animal experiments.
Animal Experiments
Animal experimental ethical committee approval was obtained from the Third Military Medical University (Chongqing, China). Five- to 8-week-old male C57BL/6J mice were obtained from the Laboratory Animal Center in the Third Military Medical University. Streptozotocin (STZ, Sigma) was intraperitoneally injected at 40 mg/kg for 5 days. Diabetic mice were successfully generated when the fasting plasma glucose of the mice was more than 300 mg/dl after 4 weeks of experiments (38).
Four weeks after diabetes induction, the diabetic foot model was produced according to a previously described procedure (2). A full-thickness excisional wound (5 × 5 mm) was created after proximal femoral artery ligation. The wounds were covered with biological tissue material alone (material group) or the material containing 2 × 104 hMSCs. In separate experiments, wounds were covered with biological tissue material containing 2 × 104 hMSCs activated or not activated by NT-3. In separate experiments, the biological tissue material containing MSC CM or NT-3 CM was also applied onto ulcers. Ten animals were included in each group. Tegaderm™ transparent dressings [Minnesota Mining and Manufacturing (3M), Maplewood, MN, USA] were used. After surgery, the animals were maintained in individual cages with enough food and water in a temperature- and humidity-controlled environment.
Wound Healing Effects and Histology
After animal surgery, a Nikon D90 digital camera (Tokyo, Japan) was used to record images of the wounds on days 3, 7, 10, and 14, and the areas were calculated using IPP software (31). The wound areas were harvested for immunohistochemistry. Nine-micron-thick frozen sections were cut and processed for immunohistochemistry using an anti-cluster of differentiation 31 (CD31) primary antibody (Santa Cruz Biotechnology, Dallas, TX, USA), an IgG-tetrarhodamine isothiocyanate (TRITC) secondary antibody, conjugated goat anti-rabbit antibodies (Santa Cruz Biotechnology), and hematoxylin and eosin staining (H&E). The capillary densities of the wounds were assessed for histological quantitative analysis through light microscopic examination.
Statistics
Each experiment was repeated at least three times. The t test and variance analyses were performed using SPSS for Windows (Version 18.0, SPSS, Inc., Chicago, IL, USA). All of the experimental data are expressed as the means ± SE; all of the p values are two-tailed; and p < 0.05 was considered statistically significant.
Results
NT-3 Promotes MSC Proliferation and Migration
To explore the effect of NT-3 on MSC growth in a high-glucose medium, we used the MTT assay to examine hMSC proliferation. The OD value of NT-3-treated hMSCs was 140% of the control value, which suggests that NT-3 promotes hMSC proliferation and that the Akt inhibitor group significantly blocks NT-3-induced hMSC proliferation (Fig. 1A). Previous studies using RT-PCR and immunological assays showed that MSCs express the NT-3-specific receptor neurotrophic tyrosine kinase receptor type 3 (TrkC) and that TrkC expression increases in response to NT-3 stimulation (49, 54). Thus, under high-glucose conditions, NT-3 enhances MSC proliferation, and this effect can be blocked by an Akt inhibitor, which indicates that NT-3 upregulates hMSC proliferation through the Akt pathway. To determine if NT-3 stimulation affects hMSC motility, we performed an MSC scratch assay. The results indicate that the closure area for NT-3-treated hMSCs was 120% of the control area (Fig. 1B–F), which implies a role for NT-3 in promoting hMSC migration and cell scratch closure.
Neurotrophin-3 (NT-3) promotes mesenchymal stem cell (MSC) proliferation and scratch closure. (A) An MTT assay with the OD value (492 nm) for MSCs cultured over 48 h in each group. *p < 0.01 (n = 18) versus the control group of MSCs in a high-glucose medium (HG-control); #p < 0.01 (n = 18) versus the HG-Akt inhibitor group. The values are the mean ± SE. (B) The scratch closure rate in MSCs cultured for 24 h. *p < 0.01 (n = 10) versus the HG-control group; #p < 0.01 (n = 10) versus the HG-Akt inhibitor group. The values are the mean ± SE. (C–F) Scratch photos of green fluorescent protein (GFP) expressing MSCs cultured for 24 h. The yellow lines represent cell scratch area at 0 h, and the red lines represent cell scratch area at 24 h. MSCs in a low-glucose medium (LG-control; C), high-glucose medium (HG-control; D), NT-3-containing medium (HG-NT-3; E), and Akt inhibitor-containing medium (HG-Akt inhibitor; F) (original magnification: 40 ×; scale bar: 30 mm).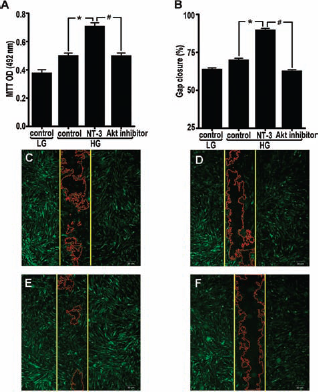
The Effect of Conditional Media on HUVEC Proliferation
To determine the effect of NT-3-stimulated hMSCs in conditioned medium (CM) on HUVEC proliferation, we performed the MTT assay under high-glucose conditions. We found that HUVEC proliferation with NT-3 in CM was 150% compared with the control medium. Moreover, HUVEC proliferation with the Akt inhibitor in CM was only 50% compared with the NT-3 CM group (Fig. 2A). These results suggest that NT-3 may activate secretion of specific factors in hMSCs that favor HUVEC growth and enhance vascular endothelial cell activity through the Akt pathway.
NT-3 upregulated VEGF, NGF, and BDNF protein secretion in MSCs and induced HUVEC proliferation. (A) An MTT assay with the OD value (492 nm) for human umbilical vein endothelial cells (HUVECs) from each group cultured for 48 h. *p < 0.01 (n = 18) versus MSC conditioned medium (CM) high glucose (HG-MSC CM); #p < 0.01 (n = 18) versus the HG-Akt inhibitor CM Group. The values are the mean ± SE. (B) The vascular endothelial growth factor (VEGF) concentration in conditioned media as determined by ELISA. *p < 0.01 (n = 10) versus the MSC CM group; #p < 0.01 (n = 10) versus Akt inhibitor CM Group. The values are the mean ± SE. (C) The nerve growth factor (NGF) concentration in conditioned media as determined by ELISA. *p < 0.01 (n = 10) versus the MSC CM group. The values are the mean ± SE. (D) The brain-derived neurotrophic factor (BDNF) concentration in conditioned media as determined by ELISA. *p < 0.01 (n = 10) versus the MSC CM. The values are the mean ± SE.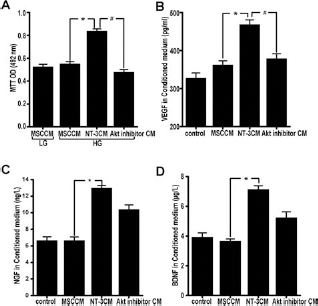
To verify our hypothesis, we used an ELISA to detect the VEGF, NGF, and BDNF levels in each CM. We found that the amount of VEGF in NT-3 CM was 160% compared with MSC CM, whereas the VEGF levels with the Akt inhibitor in CM were significantly reduced (Fig. 2B). Compared with the MSC CM group, the NGF and BDNF levels were 200% greater in the NT-3 CM group, whereas the amount of NGF and BDNF in the Akt inhibitor CM group was 60% compared with the NT-3 CM group (Fig. 2C, D). These results indicate that, in a high-glucose environment, NT-3 promotes the secretion of VEGF, BDNF, NGF, and other active vascular endothelial factors in MSCs, but the mechanisms may vary.
An NT-3-Stimulated Conditional Medium Promotes HUVEC Migration in a Scratch Experiment
Vascular endothelial cells are critical for angiogenesis. To determine the effect of NT-3-stimulated conditional medium on HUVEC movement, we performed a HUVEC scratch experiment (Fig. 3). After 24 h, the closure area for HUVECs in the NT-3 CM group was 200% compared with the MSC CM group; moreover, the Akt inhibitor inhibited HUVEC movement (Fig. 3A, C–F). Next, we performed a HUVEC migration assay in a transwell chamber. The number of migrating cells in the NT-3 CM group was 170% compared with the MSC CM group, and the number of migrating HUVECs in the Akt inhibitor CM group was significantly less than in the control medium (Fig. 3B, G–J). These results suggest that the active factors in the NT-3 CM group promote HUVEC migration as well as scratch closure and that the Akt inhibitor blocks this migratory effect.
NT-3-induced HUVEC migration and scratch closure in MSC-conditioned medium. (A) The scratch closure rate in HUVECs cultured for 24 h. *p < 0.01 (n = 10) versus high-glucose MSC CM group (HG-MSC CM); #p < 0.01 (n = 10) versus HG-Akt inhibitor CM group. The values are the mean ± SE. (B) The effect of the conditioned medium on HUVEC migration. *p < 0.05 (n = 10) versus high-glucose MSC CM group (HG-MSC CM); #p < 0.05 (n = 10) versus HG-Akt inhibitor CM group. The values are the mean ± SE. (C–F) Photographs of scratches in HUVECs cultured for 24 h. The yellow lines represent cell scratch area at 0 h, and the red lines represent cell scratch area at 24 h. HUVECs in low-glucose media (LG-control; C), high-glucose MSC CM (D), NT-3 CM (E), and Akt inhibitor medium (F) (original magnification: 40 ×; scale bar: 30 mm). (G–J) Migration of the HUVECs. HUVECs in a low-glucose medium (LG-control; G), high-glucose MSC CM (H), NT-3 CM (I), and Akt inhibitor CM (J) (original magnification: 100 ×; scale bar: 20 μm).
NT-3 Promotes MSC Proliferation in Biological Materials
A basic requirement for tissue engineering is that stem cells can grow in biological materials (4, 35) and that the biomaterial provides a microenvironment conducive for stem cell growth and differentiation (20). We observed that the number of hMSCs activated by NT-3 significantly increased as described above (Fig. 4); this indicates that the biological material is suitable for hMSC growth and that NT-3 promotes hMSC growth in biological tissue.
NT-3 promotes MSC proliferation in biological tissue materials. (A–C) GFP expressing MSCs cultured for 48 h in biological tissue material (A) and biological tissue material supplemented with NT-3 (B) or the Akt inhibitor (C) (control, NT-3, and Akt inhibitor). Scale bar: 200 μm. (D–F) Synthesized maps composed of biological materials with the control (D), NT-3 (E), and Akt inhibitor (F). Scale bar: 200 μm. (G) The biological materials for tissue engineering. (H) The diabetic foot wounds were bound by biological material and a transparent dressing bandage. (I) The cell count for MSCs cultured for 48 h in the biological material. *p < 0.01 (n = 20) compared with the control group; #p < 0.01 (n = 20) compared with the Akt inhibitor group. The values are the mean ± SE.
NT-3-Activated MSCs Accelerate Diabetic Foot Wound Healing
To determine if NT-3 activation of hMSCs (NT-3-MSC) promotes diabetic foot wound healing, we used a mouse model of a diabetic foot wound that was covered with a biological tissue material containing 2 × 104 NT-3-MSCs (treatment group) or MSCs (control group). The wound area was measured on days 3, 7, 10, and 14 after the operation (Fig. 5). We found that the NT-3-hMSC-treated group had significantly accelerated diabetic foot healing and that the healing rate for the hMSC control group was significantly slower (Fig. 5A, C). The healing rate of the biological tissue material group was the slowest (Fig. 5A, C). These data suggest that NT-3-activated hMSCs effectively promote diabetic foot healing.
The therapeutic effects of NT-3-stimulated MSCs on diabetic foot wounds in mice. (A) The wound-healing rates for the NT-3-stimulated MSC group (NT-3-MSC), MSC control group (MSC), and biological tissue material group (material). *p < 0.01 (n = 10) compared with the MSC group; #p < 0.01 (n = 10) compared with the material group. The values are the mean ± SE. (B) The wound healing rates for NT-3-stimulated MSCs in the conditioned medium group (NT-3 CM) and the MSC conditioned medium control group (MSC CM). *p < 0.01 (n = 10) compared with the MSC CM group. The values are the mean ± SE. (C) Typical photographs of wound healing for the above groups.
NT-3-Activated hMSCs Promote Vascular Regeneration
Angiogenesis is a key event in wound healing (6, 19). To determine the capacity of NT-3-activated hMSCs to pro mote diabetic foot angiogenesis, we collected skin around the wound on days 3, 7, 10, and 14 for H&E staining. The results showed that vascular density in the NT-3-MSC group was significantly higher compared with the MSC group (Fig. 6). A majority of MSCs in the NT-3-MSC group were transfected with GFP and also stained with rhodamine-labeled CD31 in vascular endothelial cells, and other hMSCs were located in the vascular and nervous periphery, which indicates that NT-3-MSC promotes angiogenesis. However, the hMSCs in the control group were distributed in sites distal to the vessel, and only a few of these cells were associated with the vascular endothelium (Fig. 7). Previous studies showed that MSCs participate in a wide variety of skin wound structure formations (40). We also found that NT-3-MSC promotes angiogenesis in diabetic foot not only because of their ability to differentiate into endothelial cells but also because NT-3 promotes secretion of specific factors in MSCs that promote vascular endothelial cell activity and induce peripheral capillary regeneration through paracrine secretion.
The NT-3-stimulated MSCs induce angiogenesis of diabetic foot wounds in mice. (A) The vascular density for the group treated with NT-3-stimulated MSCs (NT-3-MSC) and the MSC control group (MSC) on day 7. *p < 0.01 (n = 10) comparison with the MSC group. The values are the mean ± SE. (B) The vascular density of the group treated by NT-3-stimulated MSCs (NT-3-MSC) and the MSC control group (MSC) on day 14. *p < 0.01 (n = 10) comparison with the MSC group. The values are the mean ± SE. (C–F) Typical photographs for each group; vascular densities on days 7 and 14, respectively. (C, E) The NT-3-MSC group and (D, F) MSC group. Original magnification: 200 × (C–F), 400 × (photographs in the black box of C). Scale bar: 40 mm. The MSC distributions in the wounds treated with NT-3-stimulated MSCs (NT-3-MSC) and the control group (MSC) on day 7. (A—D) Typical photographs for the NT-3-MSC group. Merged photographs show that MSCs (green fluorescence) were distributed around the vascular CD31 (red fluorescence), and MSCs merged with vascular wall (arrow). (E—H) Typical photographs for the MSC group. Merged photographs show MSCs (green fluorescence) localized to the CD31-labeled vasculature (red fluorescence). Original magnification: 100 × (A, E), 400 × (B—D, F—H). Scale bar: 20 mm (A, E), 100 μm (B—D, F—H).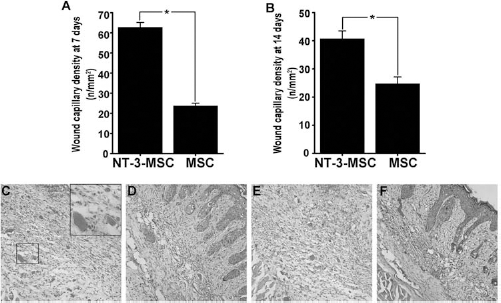

NT-3 CM in Diabetic Foot Wound Healing
To determine whether NT-3-activated hMSCs promote healing in diabetic foot through the paracrine system, we used the NT-3 CM group or MSC CM immersed biological tissue material to treat diabetic foot wound. The wound area was measured on days 3, 7, 10, and 14 after treatment. The wound treated with NT-3 CM-immersed material healed significantly faster than the MSC CM conrol group (Fig. 5B, C).
Discussion
The interactions between cell responses and inflammatory mediators can contribute to the wound healing process (32, 45), and angiogenesis is the key step in wound healing (6, 19). Based on diabetic foot pathological features, one efficacious way to promote wound healing is to initiate and effectively promote angiogenesis. MSCs are multipotent stem cells that can differentiate into a variety of cell types (49, 54). MSCs can secrete many factors that promote angiogenesis, such as IGF-1, bFGF, VEGF, and interleukin (IL)-6 (25). Recent studies implicate a critical role for neurotrophic factors, such as NGF and BDNF, in the cardiovascular system (9, 17), which provide a new therapeutic direction for treating the diabetic foot. Using tissue engineering techniques herein, we provide evidence that NT-3-activated hMSCs secrete VEGF and NGF through a paracrine system, which promotes angiogenesis in the diabetic foot and accelerates wound healing.
VEGF is closely associated with angiogenesis (18). NT-3 stimulates MSC differentiation into neurons by binding to its cognate receptor, TrkC, on the MSC surface (49, 54). In addition, NT-3 promotes nerve cell growth through the TrkC-rat sarcoma/V-raf-1 murine leukemia viral oncogene homolog 1-extracellular signal-regulated kinase 1/extracellular signal-regulated kinase 2-mitogen-activated protein kinase (TrkC-Ras/Raf-Erk1/Erk2-MAPK) or phosphatidylinositide 3-kinase (PI3K)-Akt signaling pathways (9, 36, 51). Although we did not examine its effects on MSC differentiation, the levels of NGF and BDNF in NT-3-stimulated hMSC with conditioned media were dramatically increased, which suggests that NT-3 promotes hMSC differentiation into neurons under high-glucose conditions. The PI3K/Akt signaling pathway is critical for cardiovascular system genesis and regeneration (43). NGF-bound TrkA promotes skin cell proliferation in blood vessels (34) and stimulates MSCs to generate VEGF and nitric oxide synthase (NOS) through the PI3K/Akt pathway, which promotes angiogenesis (48). Similarly, NT-3 may activate the PI3K-Akt signaling pathway that upregulates VEGF expression and promotes angiogenesis (9). Glioma cells can secrete high levels of chemotactic factors, including NT-3, which promote MSC migration to the tumor area and accelerate angiogenesis through binding to its cognate receptor, TrkC (5). Our studies have found that NT-3 could promote the closure of the MSC scratches through Akt signal pathway. In addition, the neurotrophin-3-induced Schwann cell migration depended on the Rho GTPases [Ras-related C3 botulinum toxin substrate 1 (Rac1) and cell division control protein 42 homolog (Cdc42)] and c-Jun N-terminal kinase (JNK) signal pathway, which are implicated in the formation of actin stress fibers, lamellipodia, and filopodia, respectively (52). However, NGF activates aorta endothelial cells and vascular smooth muscle cell migration by stimulating PI3K/Akt or ERK signal-dependent pathways (28, 37). This means that the reasons that NT-3, NGF, and other neurotrophic factor family promote cell migration are very complex, and we consider that this may involve a number of different mechanisms to produce the composite effect. Further studies are necessary to investigate other mechanisms through which NT-3 may activate MSCs to facilitate angiogenesis and wound healing in diabetic foot disease.
To verify whether NT-3-stimulated hMSCs activate HUVECs, we cultured HUVECs with NT-3 CM and found that NT-3 CM dramatically promotes HUVEC proliferation, migration, and scratch closure. These results imply that specific factors, such as VEGF, NGF, and BDNF, in the NT-3 CM activate vascular endothelial cells and may play an important role in promoting angiogenesis. Further studies are necessary to identify other factors in NT-3 CM that promote angiogenesis.
Based on studies from other groups and our group demonstrating that NT-3-activated MSCs play an important role in angiogenesis, we suggest that NT-3-activated MSCs can be used to treat diabetic foot disease. To this end, we used biological tissue material and NT-3-activated hMSCs to treat the diabetic foot in an animal model. We found that NT-3-activated hMSCs dramatically activate vascular endothelial cells, promote angiogenesis, and accelerate diabetic foot wound healing. We further found that certain NT-3-activated hMSCs were localized around blood vessels and are likely involved in vascular wall formation. NT-3-activated hMSCs can improve vascular endothelial cell activity in a concerted effort through the paracrine system to induce angiogenesis; however, we have not excluded other MSC mechanisms that promote wound healing (25).
Notably, NGF and NT-3 induce diabetic axonal growth and reverse diabetic neuropathy (10). Nerve fibers stimulate angiogenesis by releasing VEGF, substance P, and NO (9). NT-3-activated MSCs secrete VEGF, NGF, and other factors that promote nerve fiber growth around diabetic foot wounds (11, 14). New nerve fibers then secrete NGF, substance P, and other factors that activate vascular endothelial cells, which promote angiogenesis in a positive feedback loop (41). Further study is necessary to discern the mechanism underlying these factors' actions and determine their potential application in treating diabetic foot disease. Although NT-3-activated MSCs were used to treat diabetic foot wounds for only 14 days, we observed that NT-3-activated MSCs significantly promote angiogenesis and wound healing, which provides a promising strategy for diabetic foot disease treatment.
Conclusion
Our data presented herein show that NT-3 not only acts as a neurotrophic and angiogenic factor but also induces neovascularization and accelerates diabetic foot wound healing through the paracrine system. These findings suggest that application of stem cells in tissue engineering for treating diabetic foot ischemic ulcers is a promising approach.
Footnotes
Acknowledgments
This work was supported by the Joint Research Fund for Overseas Chinese Young Scholars (No. 310 28008), Natural Science Foundation Project of CQ CSTC (cstc 2011jjjq0016), the “twelve-five” key project of PLA (BWS11C 056), and the National Science Foundation of China (No. 3117 0935). The authors declare no conflicts of interest.
