Abstract
The administration of human adipose-derived stromal cells (hASCs) enhances skin wound healing. However, poor survival of hASCs that are administered to avascular wound regions may limit the therapeutic efficacy of the hASCs. The aim of this study was to determine whether the coadministration of platelet-rich plasma (PRP) and hASCs enhanced the skin wound-healing efficacy of hASCs. Skin regeneration was examined in skin wounds of athymic mice that were either untreated or treated with hASCs, PRP, or both hASCs and PRP. Coadministration of PRP and hASCs resulted in better skin regeneration than hASC administration alone in part by significantly improving the proliferation of administered hASCs by the angiogenic growth factor secretion of the hASCs and surrounding mouse host cells in the wound areas and by promoting neovascularization in the wound beds.
Keywords
Introduction
Several studies have demonstrated that the administration of adipose-derived stromal cells (ASCs) accelerates wound healing (14–16). ASCs can produce trilayered skin substitutes including epidermis, dermis, and hypodermis containing adipocytes (23). ASCs are advantageous in clinical applications because they are low risk, abundant, and relatively simple to isolate and have a rapid expansion rate (17). However, the poor survival of the cells administered to avascular wound areas may limit the skin regeneration efficacy of ASCs (12,22). ASCs that are placed in a harsh microenvironment that lacks any initial vasculature may be susceptible to apoptosis (21,27). Therefore, the administration of ASCs alone may not have a significant effect on skin wound healing.
To overcome these limitations, we hypothesized that the simultaneous administration of platelet-rich plasma (PRP) may enhance the skin repair efficacy of human ASC (hASC) therapy in treating wounds in mice. Degranulation of platelets in PRP by proteins such as thrombin causes platelets to release growth factors, including transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), epidermal growth factor, fibroblast growth factor 2 (FGF2), hepatocyte growth factor (HGF), and vascular endothelial growth factor (VEGF) (3,10). Because some of these growth factors are antiapoptotic and angiogenic, these growth factors may enhance the survival of hASCs administered to avascular wound regions and, subsequently, augment the dermal regeneration efficacy of the hASCs. However, the half-life of therapeutic growth factors in their free forms is generally too short for the growth factors to perform their biological functions when they are injected into the body (7,11,18). One feasible method to solve this problem and enhance the efficacy of growth factors is to release the growth factors locally over a sustained period in vivo. Previously, we have developed heparin-conjugated fibrin (HCF) (24), which can release heparin-binding growth factors (e.g., TGF-β, PDGF, VEGF, and FGF2) over a sustained period. Long-term delivery of FGF2 via HCF enhances angiogenesis and cell proliferation in the neodermis (5). In this study, HCF was used as a delivery system to release growth factors in PRP for a sustained period. To test the hypothesis, athymic mice were randomly assigned to one of four treatments as follows: (1) no treatment, (2) hASC administration, (3) PRP administration, and (4) coadministration of hASCs and PRP. The coadministration significantly improved the skin regeneration efficacy of hASCs in part by enhancing the survival, proliferation, and angiogenic growth factor secretion of the administered hASCs.
Materials and Methods
Isolation and Culture of hASCs
Human lipoaspirates were collected from patients using elective liposuction with informed consent according to the approved procedures. Liposuction tissue samples were washed extensively with phosphate-buffered saline (PBS, Sigma-Aldrich, St. Louis, MO, USA) and digested with 0.075% (w/v) collagenase type I (Sigma) at 37°C for 30 min. The samples were centrifuged at 1,200 rpm for 10 min to obtain the stromal–vascular fraction. The isolated stromal–vascular fraction was incubated overnight at 37°C with 5% CO2 in an expansion medium of minimum essential medium a (Gibco-BRL, Gaithersburg, MD, USA) that was supplemented with 10% (v/v) fetal bovine serum (FBS, Gibco-BRL), 100 units/ml of penicillin, and 100 μg/ml of streptomycin (Invitrogen, Carlsbad, CA, USA). After incubation, tissue culture flasks were washed to remove unattached cells. Cells were maintained at 37°C in 5% CO2 in the expansion medium for 7–10 days until confluence. hASCs at the third passage were used for the experiments.
Preparation of Platelet-Rich Plasma (PRP)
Whole blood (50 ml of whole blood/each person) was obtained from three healthy volunteers, two men and one woman, whose ages were 22, 26, and 31, after providing informed purpose and consent. After the first centrifugation step (1,500 rpm for 10 min), the plasma containing the platelets was separated from the red blood cells. After discarding the red blood cell fraction, the samples were centrifuged (3,000 rpm for 10 min) to separate platelet-poor plasma from platelet-rich plasma.
Preparation of HCF
HCF was prepared as previously reported (25). Briefly, heparin (molecular weight = 4,000–6,000 Da, Sigma) was covalently bonded to plasminogen-free fibrinogen (Sigma) using a procedure that employed standard carbodiimide chemistry. The carboxyl groups in heparin were activated using N-hydroxysuccinimide (0.04 mM; Sigma) and 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide hydrochloride (0.08 mM; Sigma) in a buffer solution (100 ml, pH 6) of 0.05 M 2-morpholinoethanesulfonic acid (Sigma). Fibrinogen (100 mg) was dissolved in PBS (20 ml, pH 7.4) and reacted with the activated carboxyl acid groups of heparin (60 mg). The product was then precipitated using a large excess of acetone and lyophilized. The resultant white powder was completely dissolved in PBS and dialyzed using a porous membrane bag (12,000–14,000 Da molecular weight cutoff, Spectrum Lab, Inc., Rancho Dominguez, CA, USA) at 4°C for 24 h to remove residual heparin. HCF was formed by mixing heparin-conjugated fibrinogen (40 mg/ml), normal fibrinogen (60 mg/ml), factor XIII, aprotinin (100 KIU/ml), calcium chloride (6 mg/ml), and thrombin (500 IU/mg; all from Sigma) in PBS. The hASCs and PRP were prepared for treatment by mixing with 100 μl of HCF.
In Vitro Study
hASCs were seeded on the culture plates and were allowed to attach overnight in 5% CO2 at 37°C using the expansion medium. They were then cultured in the expansion medium without FBS and with either 1% (v/v) PRP mixed with same volume of HCF or no PRP. After 24 h, proliferating cells were analyzed by immunofluorescent staining using antibodies against proliferating cell nuclear antigen (PCNA, Abcam, Cambridge, UK). More than 150 cells were analyzed for the immunocytochemical quantification.
Treatment of Skin Wounds
Six-week-old female athymic mice (20 g of body weight, Orient Bio, Inc., Sungnam, Korea) were anesthetized with xylazine (10 mg/kg) and ketamine (100 mg/kg), which were administered intraperitoneally. A square-shaped full-thickness wound (1.8 × 1.8 cm2) was created on the back of each mouse. Mice with skin wounds were randomly divided into four experimental groups (n = 6 per group). The wounds were treated with HCF (no treatment group, n = 6), hASCs (1.8 × 106 cells per defect), PRP (100 μl per defect), or hASCs (1.8 × 106 cells per defect) suspended in 100 μl of PRP (hASC + PRP group). The hASCs and PRP were mixed with HCF before the treatment. After gel formation, all groups were dressed with transparent Tegaderm™ (3M Health Care, St. Paul, MN, USA). Wound healing statuses were followed up to 16 days after the treatment. All animals received care according to the guidelines for the care and use of laboratory animals of Seoul National University. The study was approved by the Institutional Animal Care and Use Committee of Seoul National University (SNU-1006290-6).
Histology
The tissues of the wound regions were retrieved from athymic mice at 16 days after skin wound treatments. The tissues were fixed in 10% (v/v) buffered formaldehyde, dehydrated using a series of graded ethanol, embedded in paraffin, and were sliced into 4-μm-thick sections. Microscopic tissue regeneration was determined using Goldner's trichrome-stained tissue sections and a light microscope (KS400, Zeiss, Munich, Germany) (Fig. 1). The wound edge distance, intersubcutaneous distance, and interpanniculus carnosus distance (Fig. 1B) analyses (n = 6) were performed on digital images using the imaging software Image Pro Plus 6.0 (Media Cybernetics, Inc., Silver Spring, MD, USA).

Enhanced wound healing by the coadministration of human adipose-derived stromal cells (hASCs) and platelet-rich plasma (PRP) in skin wounds of athymic mice. (A) Goldner's trichrome-stained sections of wound healing regions at 16 days after the treatment. Scale bars: 5 mm. (B) Scheme of wound healing. (C) Wound edge distance (i.e., distance between the tips of the epithelium), intersubcutaneous distance (i.e., distance between the ends of subcutaneous tissue), and interpanniculus carnosus distance in wounds treated with various methods. ∗p < 0.05 compared to other groups.
Immunofluorescent Staining and Immunohistochemistry
The tissues of the wound regions that were retrieved 3 or 16 days after the treatment were embedded in an optimal cutting temperature compound (TISSUE-TEK® 4583, Sakura Finetek USA, Inc., Torrance, CA, USA), followed by freezing and slicing into 10-mm-thick sections at −22°C. Tissue sections were mounted in 4,6-diamidino-2-phenylindole (DAPI, Vector Laboratories, Burlingame, CA, USA). Antibodies against the human nucleus antigen (HNA, Chemicon, Temecula, CA, USA) and caspase 3 (Abcam) were used for immunofluorescent staining of tissue sections that were retrieved 3 days after the treatment to detect human cell survival and apoptosis, respectively. For tissue sections that were harvested 16 days after the treatment, immunohistochemistry was performed against involucrin (Abcam) and laminin (Abcam) to examine the regeneration of the epidermis and the basal layer, respectively. To assess the microvessel density, tissue sections were stained with antibodies against von Willebrand factor (vWF) (Jackson Immuno Research, West Grove, PA, USA). To perform the arteriole density analysis, sections were immunohistochemically stained with anti-smooth muscle (SM) α-actin antibodies (Abcam). A fluorescent microscope (IX71, Olympus, Tokyo, Japan) was used to count stained microvessels and arterioles. Four different images per slide from 20 random slides using 200x magnification were randomly analyzed for each group (n = 6 per group) for immunohistochemical quantification.
Western Blot Analysis
The samples (n = 3 per group) that were obtained from the mouse wound areas 16 days after the treatment were homogenized using a Dounce homogenizer (PowerGen 125, Fisher Scientific, Pittsburgh, PA, USA; 50 strokes, 4°C) in ice-cold lysis buffer (15 mM Tris–HCl, pH 8.0, 0.25 M sucrose, 15 mM NaCl, 1.5 mM MgCl2, 2.5 mM ethylenediaminetetraacetic acid, 1 mM ethylene glycol tetraacetic acid, 1 mM dithiothreitol, 2 mM NaPPi, 1 mg/ml of pepstatin A, 2.5 mg/ml of aprotinin, 5 mg/ml of leupeptin, 0.5 mM phenymethyl sulfonyl fluoride, 0.125 mM Na3VO4, 25 mM NaF, and 10 mM lactacystin; all from Sigma). Protein concentration was determined by the micro-bicinchoninic acid method of Pierce (Rockford, IL, USA). Western blot analysis was performed using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. After the proteins were transferred to an Immobilon-P membrane (Millipore Corp., Billerica, MA, USA), they were probed with antibodies against hVEGF, hHGF, hFGF2, mouse VEGF (mVEGF), mouse HGF (mHGF), mouse FGF2 (mFGF2), mhbeta-actin, and nerve/glial antigen 2 (NG2) (all from Abcam) and incubated with a horseradish peroxidase-conjugated secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 1 h at room temperature. The blots were developed using an enhanced chemiluminescence detection system (Amersham Bioscience, Piscataway, NJ, USA).
Statistical Analysis
Quantitative data are expressed as the mean ± standard deviation. Statistical analysis was performed by the analysis of variance using the ANOVA test followed by a Bonferroni test. A value of p < 0.05 was considered statistically significant.
Results
Effects of hASCs and PRP on Wound Healing
Goldner's trichrome staining of the midportion of the repair tissues at day 16 indicated that the hASC + PRP group showed the most extensive skin regeneration among all of the groups (Fig. 1A). Three different histological criteria were applied to quantitatively evaluate the wound healing as follows: wound edge distance, intersubcutaneous distance, and interpanniculus carnosus distance. Sixteen days after the treatment, these distances in the hASC + PRP group were significantly smaller than those in other groups (Fig. 1C).
Regeneration of Epidermis
The coadministration of hASCs and PRP promoted the regeneration of the epidermis and the basal layer in wound areas. Immunohistochemical staining of the wound skin that was retrieved 16 days after the coadministration of hASCs and PRP revealed significant staining for involucrin (epidermis) (Fig. 2A) and laminin (basal layer) (Fig. 2B) compared to that of the other groups.
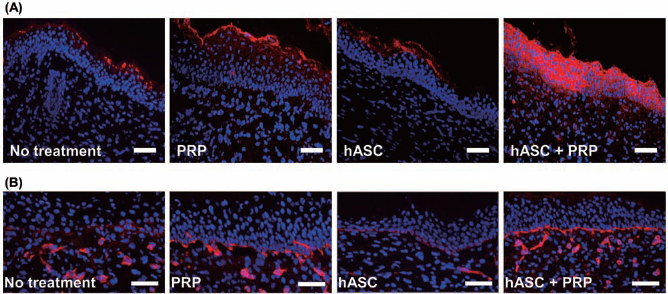
Regeneration of the epidermis and the basal layer. Immunohistochemical staining for (A) involucrin in the epidermis and (B) laminin in the basal layer in wound healing regions at 16 days posttreatment. Scale bars: 200 μm. Red staining indicates involucrin in (A) and laminin in (B). Blue indicates DAPI.
Enhancement of hASC Survival by PRP Administration
The coadministration of PRP and hASCs in the wound beds increased the survival of the hASCs. Immunofluorescent staining of the wound skin that was retrieved 3 days after treatment showed that HNA-positive cells (administered hASCs) were more abundant in hASC + PRP group compared to those in hASC group (p < 0.05) (Fig. 3). The proportion of caspase 3- and HNA-positive cells (apoptotic hASCs) compared to HNA-positive cells was significantly lower in hASC + PRP group than in hASC group (p < 0.05) (Fig. 3).

Apoptosis and survival of hASCs administered to avascular wound regions. (A) Immunohistochemical staining for caspase 3 and human nuclear antigen (HNA) in wound healing regions at 3 days posttreatments. Red indicates HNA, green indicates caspase 3, and blue indicates DAPI. HNA-positive cells indicate hASCs. The arrows indicate both caspase 3- and HNA-positive cells. Scale bars: 200 μm. (B) Ratio of HNA-positive cells to DAPI-positive cells, which indicates the ratio of hASCs to total cells (∗p < 0.05). (C) Ratio of caspase 3- and HNA-positive cells to HNA-positive cells, which indicates the ratio of apoptotic hASCs to total hASCs (∗p < 0.05).
Effect of PRP on Cell Proliferation
In vitro, PRP supplementation enhanced hASC proliferation. The immunofluorescent staining for PCNA and the quantification of proliferating cells (Fig. 4) showed that hASC proliferation was enhanced when cultured with PRP compared to no PRP supplementation (p < 0.01).
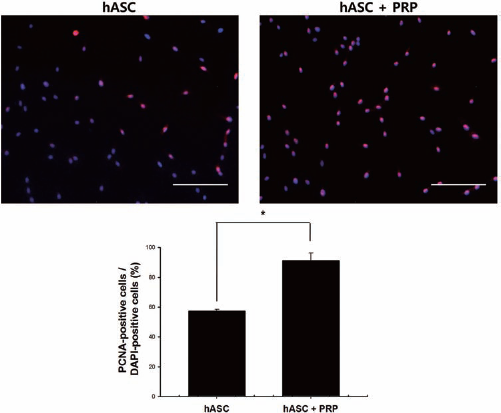
Effect of PRP on hASC proliferation in vitro. Immunohistochemical staining with anti-proliferating cell nuclear antigen (PCNA) antibodies of hASCs cultured for 24 h with or without PRP and quantification of proliferating cell ratio. Red (PCNA) and blue (DAPI) indicate proliferating cells and nucleus, respectively. Scale bars: 200 μm. ∗p < 0.01.
Angiogenic Factor Secretion and Angiogenesis
Coadministration of PRP and hASCs enhanced the secretion of angiogenic paracrine factors from locally administered hASCs and mouse host cells in the wound areas. Western blot analysis performed at 16 days after treatment demonstrated that the administration of PRP and/or hASCs enhanced human and mouse growth factor expression and even more when PRP and hASCs were coadministered (Figs. 5 and 6). Coadministration of PRP also enhanced hASC-mediated angiogenesis. The immunofluorescent staining for vWF and the quantification of microvessel density (Fig. 7A) showed that the coadministration of hASCs and PRP significantly enhanced (p < 0.05) the microvessel formation compared to no treatment, PRP, and hASC groups. The immunofluorescent staining for SM α-actin and the quantification of arteriole density (Fig. 7B) revealed that the coadministration of PRP and hASCs enhanced the arteriole formation (p < 0.05) in wounds compared to no treatment, PRP, and hASC groups. Western blot analysis showed that the coadministration enhanced pericyte migration in wound regions as indicated by NG2 expression (Fig. 8).

Effect of PRP administration on the secretion of angiogenic paracrine factors from administered hASCs. Human-specific angiogenic factor [human vascular endothelial growth factor (hVEGF), human fibroblast growth factor 2 (hFGF2), and human hepatocyte growth factor (hHGF)] expression in the wound healing area, which was evaluated by Western blot analysis and the quantification of the relative protein expressions (n = 3). #p < 0.05 compared to no treatment, ∗p < 0.05 compared to other groups.

Effect of PRP administration on the secretion of angiogenic paracrine factors from surrounding mouse tissues. Mouse-specific angiogenic factor (mVEGF, mFGF2, and mHGF) expression in the wound healing area, which was evaluated by Western blot analysis, and the quantification of the relative protein expressions (n = 3). #p < 0.05 compared to no treatment, ∗p < 0.05 compared to other groups.

Effect of PRP administration on angiogenesis. Immunohistochemical staining and quantification for (A) microvessels and (B) arterioles in wound healing regions at 16 days posttreatment. Scale bars: 200 μm. Green indicates von Willebrand factor (vWF) in (A) or smooth muscle (SM) α-actin in (B), and blue indicates DAPI.

Effect of PRP administration on angiogenesis. Western blot analysis and relative protein expression of NG2 (n = 3) to evaluate pericyte migration in the wound healing area. #p < 0.05 compared to no treatment, ∗p < 0.05 compared to other groups.
Discussion
Coadministration of PRP enhanced the wound healing efficacy of the administered hASCs. Our results show that the coadministration of PRP and hASCs significantly enhances skin regeneration compared to PRP or hASC administration alone (Figs. 1 and 2). hASCs promote tissue regeneration through the secretion of angiogenic growth factors and the promotion of angiogenesis (2,15,19,20,26). hASCs reconstitute nonvascular mesodermal and ectodermal cellular phenotypes (1,2). However, hASC administration alone may have limited efficacy due to the poor survival of hASCs in the avascular environment of wound regions (12). Coadministration of hASCs and PRP enhanced the skin regeneration efficacy in part by enhancing the survival (Fig. 3) of the administered hASCs, the proliferation of the hASCs (Fig. 4) in the wound areas, angiogenic factor secretion (Figs. 5 and 6) of the hASCs and the surrounding mouse tissues, and promoting angiogenesis (Fig. 7). The hASCs with an improved survival and proliferation in the wound beds produced larger amounts of growth factors for a longer period of time, which may induce more local mesenchymal and epithelial cells to migrate, divide, and produce extracellular matrices for wound healing (9,15).
PRP improved the survival of hASCs that were administered to wound beds (Fig. 3) by providing antiapoptotic and angiogenic growth factors and stimulating angiogenesis in the wound beds. PRP contains angiogenic growth factors, such as VEGF and PDGF (10), and stimulates angiogenesis in wound beds. Early vascularization in wound beds may prevent the exposure of hASCs to an avascular environment, thus sustaining the survival of the hASCs. VEGF in PRP is antiapoptotic (19) and may enhance not only the survival (Fig. 3), but also the proliferation (Fig. 4) of the hASCs that are administered to avascular wound regions with a harsh microenvironment.
PRP improved the angiogenic efficacy of administered hASCs by enhancing paracrine factor secretion from hASCs (Fig. 5). hASCs maintain long-term paracrine factor secretion due to the PRP-mediated enhanced survival and proliferation of the hASCs. The increased secretion of angiogenesis growth factors, such as HGF, VEGF, and FGF2, promoted angiogenesis in the wound beds (Fig. 7), contributed to the improved wound healing (Figs. 1 and 2). The persistence of hASCs near the vasculature stabilizes the generated vascular network in the wound regions (4), indicated by enhanced NG2 expression (Fig. 8).
The delivery of PRP using HCF as a delivery vehicle enhanced skin regeneration efficacy. In previous studies, sustained release of angiogenic growth factors such as FGF2 promoted neovascularization in ischemic tissues in animal models (8,13). Sustained and localized delivery of FGF2 using HCF enhances skin regeneration compared to the administration of FGF2 alone (5). Coadministration of hASCs and PRP using fibrin gel as a delivery vehicle did not show significant difference in wound healing compared to the administration of either hASC + platelet-poor plasma or PRP alone (6). Therefore, a long-lasting supply of angiogenic growth factors present in PRP by the use of HCF as a delivery vehicle may contribute to the improved wound healing (Figs. 1 and 2), which is, at least in part, attributed to enhanced neovascularization (Fig. 7) as well as the stabilization of newly formed vessels (Fig. 8).
The control of administered cells is one of the most basic and crucial factors in cell administration-mediated wound healing. Here, we demonstrated that coadministration of PRP using HCF as a delivery vehicle contributed to the survival of administered hASCs. Improved survival of hASCs enhanced the paracrine effect and angiogenesis to promote wound healing. This modality may be used to improve hASC therapy for wound healing.
Footnotes
Acknowledgments
This study was supported by a grant (2010-0020352) from the National Research Foundation of Korea and a grant (A100443) from the Korea Health 21 R&D Project, Ministry of Health and Welfare, Republic of Korea. The authors declare no conflicts of interest.
