Abstract
Bone marrow released by microfracture or full-thickness cartilage defect can initiate the in situ cartilage repair. However, it can only repair small cartilage defects (<2 cm2). This study aimed to investigate whether autologous platelet-rich plasma (PRP) transplantation in collagen matrix can improve the in situ bone marrow-initiated cartilage repair. Full-thickness cartilage defects (diameter 4 mm, thickness 3 mm) in the patellar grooves of male New Zealand White rabbits were chosen as a model of in situ cartilage repair. They were treated with bilayer collagen scaffold (group II), PRP and bilayer collagen scaffold (group III), and untreated (group I), respectively (n = 11). The rabbits were sacrificed at 6 and 12 weeks after operation. The repaired tissues were processed for histology and for mechanical test. The results showed that at both 6 and 12 weeks, group III had the largest amounts of cartilage tissue, which restored a larger surface area of the cartilage defects. Moreover, group III had higher histological scores and more glycosaminoglycans (GAGs) content than those in the other two groups (p < 0.05). The Young's modulus of the repaired tissue in group II and group III was higher than that of group I (p < 0.05). Autologous PRP and bilayer collagen matrix stimulated the formation of cartilage tissues. The findings implicated that the combination of PRP with collagen matrix may repair larger cartilage defects that currently require complex autologous chondrocyte implantation (ACI) or osteochondral grafting.
Introduction
Articular cartilage has a very limited self-repair capability. When the defect size is larger than 2–4 mm in diameter, the damaged cartilage is rarely healed if left untreated (19). Unhealed articular cartilage lesions can induce mechanical instability of the joint and often lead to osteoarthritis (18). Current therapies include microfracture (9), abrasion, drilling (6), osteochondral grafting (16), and autologous chondrocyte implantation (ACI) (24). Generally, microfracture is used for treating small cartilage defects (<2 cm2) while larger defects (>2 cm2) are often treated by ACI and osteochondral grafting. However, ACI and osteochondral grafting are complex and costly.
Bone marrow released by microfracture or full-thickness cartilage defect can initiate the in situ cartilage repair with fibrin clot formation and progenitor cell migration from the bone marrow into the defective cartilage region (29). As a result, chondral resurfacing is enhanced thanks to the human body's self-healing capacity (3). However, the number of bone marrow stem cells (bMSCs) is limited. Moreover, bMSCs may leak from the hole because there is no stable scaffold to hold the cells. In addition, although the fibrin clot is formed, it is not always mechanically stable and cannot cover larger defects. Hence, addition of a scaffold may provide an optimal microenvironment for endogenous MSC infiltration, proliferation, and differentiation (4). A variety of scaffolds have been studied and used (5,13,20,27,30). Among them, collagen-based scaffold is the most promising biomaterial due to its excellent biocompatibility, biodegradability, and mechanical properties.
The quality of cartilage repair can be improved by growth factors, which are important for proliferation and differentiation of bMSCs and chondrocytes. Endogenous growth factors are often utilized because exogenous grow factors have not been approved for clinical cartilage repair. Platelet-rich plasma (PRP), which can be easily obtained from autologous blood, is a natural source of autologous growth factors. PRP has been used successfully in clinical practice (34). PRP contains many growth factors such as platelet-derived growth factor (PDGF), transforming growth factor (TGF-β1, TGF-β2), insulin-like growth factor (IGF-1, IGF-2), and vascular endothelial growth factor (VEGF) (15). It was proven effective for bone defect repair both in experimental and clinic studies (11,25,31,33,34). It was also demonstrated that PRP could stimulate chondrocyte proliferation and matrix biosynthesis in vitro (1). Moreover, it was reported that an injection of PRP and chondrocytes could induce an ectopic cartiage formation (32).
To explore a strategy of utilizing the body's self-healing capacity for larger cartilage defect repair, the present study hypothesized that the delivery of autologous PRP in collagen matrix can improve in situ bone marrow-initiated cartilage repair. Full-thickness cartilage defects were treated by collagen matrix with and without PRP. The quality and resurfacing area of cartilage defect repair were then investigated. The results might be useful to enhance the in situ articular cartilage repair of larger defects.
Materials and Methods
Fabrication of Bilayer Collagen Scaffolds
The biodegradable collagen scaffolds that were used in this study were comprised of two layers: a dense layer and a loose layer. The diameter of the scaffolds was 4 mm and the thickness was 3 mm. Insoluble type I collagen was isolated and purified from pig Achilles tendon using neutral salt and dilute acid extractions (21). Then collagen was dissolved in 0.5 M acetic acid (1.0 wt%). The collagen solution was added to a dish (thickness 4 mm) and frozen at −70°C. Then it was lyophilized in a freeze dryer (Heto Power Dry LL1500) and compressed mechanically. Collagen solution was added onto the compressed collagen matrix and was freeze-dried again. After that, the bilayer scaffold was ready for use. Scanning electron microscope (SEM) photographs revealed that the dense layer had microspores and the loose layer had macrospores. The average pore size in the loose and dense layer of the scaffold was measured as 100–200 and 10–50 μm, respectively. The relatively small pore size in the dense layer can efficiently prevent cells from leaking. All the pores around the unit area were measured by observation using internal standard (bar), and the average was calculated manually. The scaffold was cross-linked by severe dehydration (dehydrothermal cross-linking) (17). Then the scaffold was used for the following implantation.
Preparation of Platelet-Rich Plasma (PRP)
Prior to surgery, approximately 5 ml of venous blood of a rabbit was drawn into a sterile pipe containing sodium citrate as an anticoagulant for processing PRP. The platelets were enriched by a two-step centrifugation process. During the first centrifugation at 200 × g for 10 min in a Refrigerated Centrifuge 5415R (Eppendorf Corporation, German); the components of blood were separated into two phases: one phase was comprised of PRP and the other was comprised of erythrocytes and leukocytes. The samples of PRP underwent a second centrifugation at 560 × x g for 15 min that allowed the precipitation of the platelets, which thereafter were resuspended in 0.05 ml of plasma. Samples of PRP and whole blood were analyzed in an automatic counter (Sysmex F-820). PRP (0.05 ml) was loaded onto the scaffold immediately before the insertion into the cartilage defect.
Animal Model
Thirty-eight knee joints of male New Zealand White Rabbits (2–2.5 kg) were used in this study. Rabbits were maintained separately in stainless steel cages. The rabbits were anesthetized by an intramuscular injection. The knee joints were opened with a medial parapatellar approach. The patella was dislocated laterally and the surface of the femoropatellar groove was exposed. A full-thickness cylindrical cartilage defect of 4 mm in diameter and 3 mm in depth was created in the patellar groove using a stainless steel punch. The defects were either untreated (group I, n = 11) or treated with bilayer collagen matrix (group II, n = 11), and with PRP and bilayer collagen matrix (group III, n = 11). Five knee joints were used as normal control for biomechanical evaluation. The animals were returned to their cages and allowed to move freely without joint immobilization. At 6 and 12 weeks after transplantation, cartilage defect was evaluated histologically (n = 3 at each time point of each group). In addition, mechanical properties were evaluated at 12 weeks (n = 5).
All the animals were operated on according to the standard guidelines approved by Zhejiang University Ethics Committee.
Gross Morphology and Histology
At 6 and 12 weeks postsurgery, the rabbits were sacrificed by intravenous overdose of pentobarbital. The harvested samples were examined and photographed for evaluation. After gross examination, samples were fixed in 4% formalin, decalcified in 10% formic acid, then embedded in paraffin and cut to 7-μm-thick sections. Sections were stained with hematoxylin and eosin for the study of morphological evaluation and stained with Safranine-O for GAG distribution. According to the area of positive Safranine-O staining, the percentage of cartilage tissue in the area of defect was calculated. For the overall evaluation of the regenerated tissue in the defects, the repaired tissues were graded using the International Cartilage Repair Society (ICRS) Visual Histological Assessment Scale by one blinded observer according to the strategy of a previous study (14).
Mechanical Evaluation
Mechanical evaluation protocol was adopted from a previous study (10). Before testing, samples were placed in phosphate-buffered saline (PBS) at room temperature for 3–4 h to equilibrate. Compressive mechanical properties of the cartilage layer after a long-term (12 weeks, n = 5 samples) period in vivo were determined using a 2-mm-diameter cylindrical-shaped indenter to compressively load the cartilage tissue in Instron Testing machine (model 5543, Instron, Canton, MA) fitted with a 10 N maximum load cell. Native osteochondral samples were also evaluated (n = 5 plugs). The unconfined equilibrium modulus was determined by applying a step displacement (20% strain) and monitoring compressive force with time until equilibrium was reached. Thickness of the fully relaxed cartilage layer was tested to estimate strain for applied deformations. The crosshead speed used was approximately 0.06 mm/min. The ratio of equilibrium force to cross-sectional area was divided by the applied strain to calculate the equilibrium modulus (in MPa).
Statistical Analysis
To assess differences in histological scoring data and biomechanical data, one-way ANOVA, post hoc Student-Newman-Keuls (SNK) test was used. A value of p < 0.05 was considered significant.
Results
Platelet Concentration in the PRP
The average amount of PRP for transplantation was 0.05 ml after processing. The average platelet concentration in whole blood was 2.44 ± 0.46 × 105μl before processing, while it reached 18.25 + 1.21 × 105μl in the final PRP for transplantation. In other words, the platelet concentration increased more than sevenfold after processing.
Gross Morphology
The gross examinations of knee joints showed that there were no abrasions on the opposing articulating surfaces and no inflammation at both 6 and 12 weeks time points. The defects remained empty in small parts at 6 weeks after transplantation (Fig. 1A–C), while at 12 weeks the surface was smooth in all of the three groups (Fig. 1D–F).
Macrophotograph shows the defects in the three groups at 6 weeks (A, B, C) and 12 weeks (D, E, F) postsurgery. (A, D) are the control group (group I); (B, E) are the collagen group (group II); (C, F) are the PRP with collagen matrix group (group III). Scale bar: 5 mm.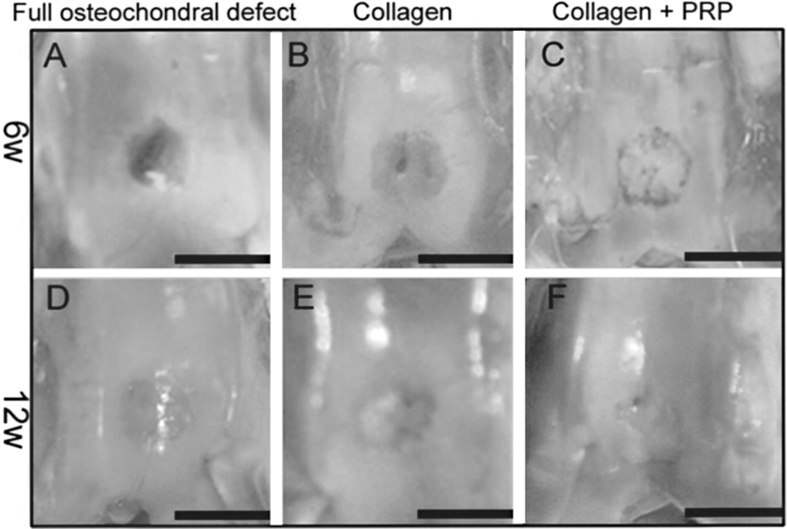
Histological Examination
At 6 weeks after transplantation, the defects in group I (n = 3) were filled with fibrous tissue. New bone tissues were abundant and bridged over the subchondral defect (Fig. 2A). In group II (n = 3) and group III (n = 3), the defects were repaired with mixture of fibrous tissue and cartilage-like tissue as shown by hematoxylin and eosin staining and Safranine-O staining. The amounts of chondrocyte-like cells and cartilage-like extracellular matrix in group III were more than those of group II as observed from hematoxylin and eosin staining (Fig. 2B, C) and Safranine-O staining (Fig. 2D–F). The subchondral space of both treated groups were filled with fibrocartilage-like tissues rather than bone tissues. It implicated that collagen scaffold may be able to protect space for cartilage formation by decreasing the ingrowths of new bone tissues.
Histological examination of the three groups at 6 weeks postsurgery. (A, D) are the control group; (B, E) are the collagen matrix group; (C, F) are the PRP with collagen matrix group. Hematoxylin and eosin staining (A, B, C); Safranine-O staining (D, E, F). Original magnification 40x. Scale bar: 500 μm.
At 12 weeks after transplantation, the defects in group I (n = 3) were almost filled with fibrous tissue and a little cartilage-like tissue at the margins of the defects. The subchondral space was filled with mature sponge bone (Fig. 3A, D). Group II had lager amounts of hyaline cartilage (Fig. 3B, E), whereas group III had the largest amounts of hyaline cartilage tissue (Fig. 3C, F). It was found that hyaline cartilage tissues were formed at the surrounding area of the defects while the fibrous tissues were located at the central part of defects. The subchondral bone was almost bridged over the defects in both treated groups.
Histological examination of the three groups at 12 weeks postsurgery. (A, D) are the control group; (B, E) are the collagen matrix group; (C, F) are the PRP with collagen matrix group. Hematoxylin and eosin staining (A–C); Safranine-O staining (D–F). Original magnification 40x. Scale bar: 500 μm.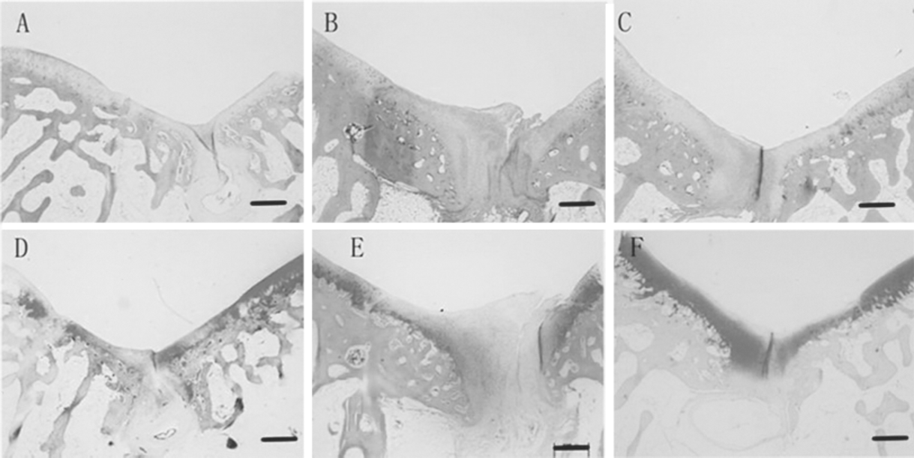
The percentage of cartilage tissue in the area of defect was calculated according to the area of positive Safranine-O staining. At 6 weeks the percentage of cartilage tissue in group III was 31.7 ± 7.07%, which was much higher than the 9.5 ± 2.12% in group I and 20 ± 7.07% in group II (p < 0.05) (Fig. 4A). At 12 weeks the percentages of cartilage tissue in the defects of group I, group II, and group III significantly increased to 27.5 ± 3.53%, 41.25 ± 7.07%, and 51.25 ± 3.53%, respectively (Fig. 4B). The differences between each two groups were statistically significant (p < 0.05). In addition, according to the Safranine-O staining, at 6 weeks the mean length of neocartilage in one side of the defects in group I, group II, and group III was 348 ± 293, 463 ± 16, and 863 ± 259 μm, respectively, while at 12 weeks the length increased to 467 ± 354, 750 ± 615, and 1102 ± 479 μm, respectively.
Comparison of the percentage of cartilage tissue in the area of defect of the three groups at 6 weeks (A) and 12 weeks (B) postsurgery. Values represent mean and SD.
The total ICRS histological scores of the specimens at 6 and 12 weeks revealed differences between the three groups (Fig. 5). The scores for the repaired tissue of group III were significantly higher than those for other two groups (p < 0.05). The histological scores of the specimens at 12 weeks were higher than those of specimens at 6 weeks.
Comparison of the total ICRS histological scores of three groups at 6 weeks (A) and 12 weeks (B) postsurgery (total scores are 18 points). Values represent mean and SD.
Biomechanical Evaluation
From the indentation test, the Young's modulus of repaired tissues from the three groups (12 weeks) were determined and compared with each other while the tissues from normal rabbit knee joint were used as normal control (Fig. 6). At 12 weeks, the compressive modulus of the repaired tissue in group II and group III samples showed greater improvement compared with specimens from group I. However, the modulus of repaired tissue in both group II and group III was inferior to that of normal cartilage (0.52 ± 0.01 Mpa) (p < 0.05). It was found that the difference between group II and group III has no statistical significance, which may be caused by the limitation of mechanical test. As the diameter of defect was 4 mm and the diameter of the indenter was 2 mm, the compressive test was carried out at the central area of those defects which were mainly fibrous tissue.
Biomechanical analysis of the repaired tissues at 12 weeks postsurgery. Values represent mean and SD (n = 5 in all groups). At 12 weeks postsurgery, there was a statistical significance between control group, collagen group, and PRP group (p < 0.05).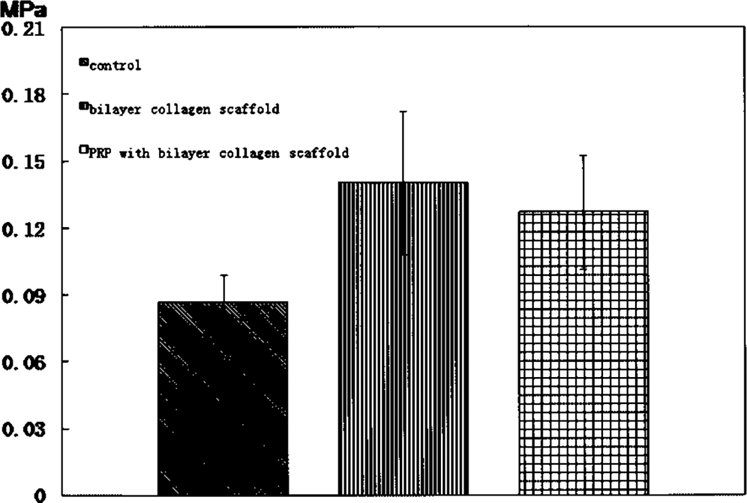
Discussion
The present study demonstrated that autologous PRP and collagen matrix stimulated the formation of cartilage tissues, which significantly improved the body's self-healing capacity for larger cartilage defect repair by providing scaffolds and growth factors for in situ bone marrow progenitor cells. It implicated that the combination of PRP with collagen matrix could repair larger cartilage defects that currently require complex autologous chondrocyte implantation or osteochondral grafting. Moreover, this study was the first to present that cartilage tissue formation was initiated at the surrounding area of the defects and crept to cover the central part of defects with time. It was different from the format of simultaneous or random cartilage tissue formation at the cartilage defect after ACI, which implicated that the cells or signals from the edge of native cartilage tissue played a crucial role in inducing the bone marrow stem cells for cartilage formation.
It is known that a large full-thickness cartilage defect (≥3 mm in diameter) in mature rabbit models could not heal spontaneously (22). In this study, the standard size of 4 mm diameter was chosen and all of the defects were not fully healed with cartilage tissues in all groups. It was found that at 6 and 12 weeks the repaired tissues treated with collagen were superior to those untreated defects regardless of histology observation or mechanical properties. At 6 weeks 20 ± 7.1% of the defect area in group II was filled with cartilage tissue, which was much higher than the 9.5 ± 2.1% in group I. At 12 weeks the percentage of group II increased to 41.25 ± 7.07%, which was nearly two times higher than the 27.5 ± 3.53% in group I. The finding was consistent with previous studies (8). Buma et al. (4) demonstrated that cross-linked collagen type I and type II scaffolds could improve cartilage repair without additional cells. In another study, it also demonstrated that hyaluronan-based polymersalone had the ability to enhance cartilage repair (28). It has been shown that the treatment of microfracture combined with a collagen membrane was exclusively superior to microfracture (2). The improvement could be explained that stem cells from bone marrow after microfracture could be captured and organized by the scaffolds (12).
PRP could enhance and initiate a more rapid and complete cartilage healing. The results showed that PRP exhibited its superiority at 6 and 12 weeks post-surgery. Histology and Safranine-O staining exhibited that group III had larger area of cartilage tissues. It implicated that PRP played a positive role in the process of cartilage repair. The growth factors released by PRP might induce cartilage regeneration in defects. It was worthy to note that PRP showed a more impressive effect at 6 weeks post-surgery than at 12 weeks. The short-lasting effect of PRP may explain the phenomenon. The growth factors and platelets in the PRP have limited half-life time and cannot achieve a long-term effect. It was also observed that the effect of PRP could persist for 4 weeks in bone defects (31). Hence, it is reasonable to increase the concentration of platelets for initiating the proliferation and migration of chondrocytes or bMSCs at the early stage, which is followed by a faster cartilage tissue formation. A manufacturing method to produce good quality and highly effective PRP should be investigated. Moreover, autologous chondrocytes or bMSCs implanting with PRP could be applied for even larger cartilage defect repair.
In this study we found that the quality of the repaired tissue at the edge was better than that at the center of defects. The cartilage tissue was formed at the surrounding area of the defects while the central area of the defects was almost filled with fibrous tissues. This suggests that the cells or signals from the edge of native cartilage tissue might play a crucial role in cartilage repair. This phenomenon might be partially caused by the migration of chondrocytes from nearby native cartilage and perichondrium into the scaffolds. Another reason was probably the signals from native cartilage, which induced the differentiation of bMSCs after microfracture. This was consistent with a previous in vitro coculture study (23). The chondrocytes may induce bMSCs to differentiate into cartilage lineage. These findings implicated the importance of differentiation factors when bMSCs were utilized for cartilage repair (7).
Interestingly, it was found that the neocartilage tissue was formed from the edge of native cartilage to the center of defects but never bridged up the defects. Moreover, the distance of neocartilage protrusion into the central area of defect was different in terms of time. It was longer at 12 weeks than at 6 weeks in each group. At 6 weeks the mean length of neocartilage in one side of the defects in group I, group II, and group III was 348 ± 293, 463 ± 16, and 863 ± 259 μm, respectively, while at 12 weeks the length increased to 467 ± 354, 750 ± 615, and 1102 ± 479 μm, respectively. It meant that in this rabbit cartilage defect model, bone marrow released from subchondral bone could repair 0.934-mm size defect, addition of collagen matrix could repair 1.5mm size defect, whereas the combination of PRP with collagen matrix could repair 2.2-mm size defect. These interesting findings could also give some hints on clinical cartilage repair. For the small cartilage defects of less than 2 cm2, microfracture is often chosen. When the defects are a little bigger than 2 cm2, microfracture with collagen matrix may be a cost-effective treatment. Furthermore, if the defects are even bigger, combination of PRP with collagen matrix can be considered. This novel and practical strategy is easy to be carried out by surgeons and accepted by patients.
One limitation of this study was that the center of defects in all of the three groups was almost filled with fibrous tissues. The mechanical test was carried on the central 2 mm of defects. Therefore, the results of mechanical test could not reflect the differences that were observed by histology. Another limitation was that only two time points (6 and 12 weeks postsurgery) were investigated. In previous study of osteochondral defects of rabbits, degenerative changes of the repaired tissue increased from 12 to 48 weeks (26). Hence, whether the neocartilage will degenerate still needs to be investigated. However, the aim of this study was mainly to compare the efficacy of different treatments for initiating cartilage repair.
In summary, autologous PRP and bilayer collagen matrix acted in stimulating the formation of cartilage tissues in articular cartilage defects. PRP can be easily obtained from autologous blood, and the operation procedure is simple and safe. It appeared that the combination of PRP with scaffold might have great potential in clinical application to improve articular cartilage self-repair capability.
Footnotes
Acknowledgments
This work was supported by NSFC grants (30600301, 30600670, U0672001), Zhejiang Province grants (R206016, 2006C14024, 2006C10384), MOE grant J20070258, and Foundation of Zhejiang Provincial Key Medical Discipline (Medical Tissue Engineering).
