Abstract
Transmyocardial revascularization (TMR) can improve refractory angina but does not consistently demonstrate an effect on myocardial function. Recent studies suggest a synergistic effect between TMR and exogenously supplied growth factors. We evaluated the clinical role of intramyocardial injection of autologous platelet-rich plasma (PRP) in conjunction with TMR. Twenty-five nonrevascularizable patients with class III/IV angina underwent minimally invasive sole therapy TMR during a 5-year period at a single institution. Group 1 (14 patients) underwent TMR alone while group 2 (11 patients) underwent TMR plus injection of PRP (Magellan plasma separator) between TMR channels. Blinded angina assessment and ejection fraction (EF) were measured preoperatively and at 6 months postoperatively. Baseline EF (57 ± 10% vs. 50 ± 7%), angina class (3.7 ± 0.5 vs. 3.7 ± 0.5), and the number of channels (48 ± 5 vs. 48 ± 4) were statistically similar in both groups. At 6 months, two class angina relief was similar in both groups (92% vs. 100%, p = 0.4); however, the TMR + PRP group had a lower average angina score (1.3 vs. 0.4, p = 0.07) and more were angina free (23% vs. 78%, p = 0.04) than the TMR-alone group. EF improved in the TMR + PRP group (–2.0% vs. +9.0%, p = 0.07) compared to the TMR-alone group. Two 30-day morbidities occurred in the TMR-alone group (atrial fibrillation and left pleural effusion) and one mortality occurred in the TMR + PRP group. Intramyocardial injection of autologous PRP combined with TMR may be more efficacious at relieving angina and improving myocardial function than TMR alone.
Keywords
Introduction
Transmyocardial revascularization (TMR) provides significant angina relief in patients with medically refractory, “no-option,” end-stage coronary artery disease (2). Decreased hospitalizations, less medication usage, and improved exercise times are also associated with TMR's overall improvement in patients' quality of life (5). However, sole TMR has not been shown to consistently improve myocardial function (7).
Recent studies have shown a possible synergistic effect with combined TMR and exogenous administration of growth factors in improving angina and possibly myocardial function (11). Clinically, autologous growth factors can be found in concentrated, centrifuged blood plasma and platelet separation in the form of platelet-rich plasma (PRP) (24). PRP has been used in orthopedic, dental, and sternal wound healing platforms (23), but not yet as a therapeutic option for treating myocardial ischemia.
The purpose of this study was to evaluate the clinical role of intramyocardial injection of autologous PRP combined with TMR in patients with medically refractory class III/IV angina.
Materials and Methods
From July 2002 to August 2007, 25 patients with medically refractory class III/IV angina who had no conventional revascularization options were referred to a single institution for sole therapy TMR. Patients were deemed inoperable for percutaneous coronary intervention or redo coronary artery bypass grafting if coronary arterial targets were measured as less than 1 mm in diameter. All patients underwent preoperative persantine Cardiolite nuclear medicine stress testing and had documented reversible ischemia in the anterior, lateral, or inferior walls of the left ventricle.
As previously described (25), holmium-YAG laser TMR (CardioGenesis Corp., Irvine, CA, USA) was performed using a minimally invasive 2–3-inch left anterolateral thoracotomy and a single 5-mm voice-activated robotic-controlled (AESOP, Computer Motion, Santa Barbara, CA, USA) thoracoscope. One millimeter transmural channels, 1 cm2 apart, were created in the distal two thirds of the left ventricle. All patients were intraoperatively monitored with radial and pulmonary arterial catheters. Nitroglycerin (15 μg/min) and lidocaine (2 mg/min) infusions were started 30 min prior to and given during the operative procedure. Patients were divided into two groups. Group 1 (14 patients) underwent sole therapy TMR alone. Group 2 (11 patients) underwent TMR plus intramyocardial injection of PRP.
Autologous PRP was obtained by removing two 60-ml syringes of whole blood from each group 2 patient 15 min prior to making an incision. The blood was anticoagulated with 8 ml per syringe of sodium citrate (Baxter Healthcare, Round Lake, IL, USA); 6 ml of PRP was isolated using a point-of-care Magellan cell separator (Arteriocyte Medical Systems, Cleveland, OH, USA) during 16 min of centrifugation. Using a tuberculin 27-gauge needle on a 1-ml syringe, 0.5–0.6 ml aliquots were injected intramyocardially in a perpendicular fashion between channels following the TMR procedure. Intraoperative transesophageal echocardiography showed no evidence of endoventricular bubble infiltration during PRP injection.
Baseline demographics were collected prospectively and Cardiolite nuclear medicine stress testing with computer-calculated ejection fraction (EF) was performed within 6 months preoperatively and at 6 months postoperatively. Study end-points at 6 months included a non-surgeon blinded angina assessment using Canadian Cardiovascular Society Class Angina Score, change in ejection fraction, hospital readmissions, and morbidity and mortality. Statistical analyses were performed using Wilcoxon Paired Rank Sum tests. This study was approved by the Institutional Review Board/New Procedures Committee at Peninsula Regional Medical Center, and detailed informed consent was obtained from each patient. Only primary health care providers practicing at the single institution were allowed access to confidential individual patient information. Others were provided with data summary only.
Results
Preoperative patient demographics were similar between TMR and TMR + PRP groups (Table 1), including average age (60 ± 11 vs. 65 ± 11 years, p = 0.3), baseline EF (57 ± 11% vs. 50 ± 7%, p = 0.1), and baseline angina class (3.7 ± 0.5 vs. 3.7 ± 0.5, p = 0.9). Operative characteristics, including the number of TMR channels (48 ± 5 vs. 49 ± 4, p = 0.9) and operative times (group 1 80.3 ± 24.6 vs. group 2 73.0 ± 13.0 min, p = 0.4), were also similar between groups. Tables 2, 3, 4, and 5 represent the blood characteristics of 6.0 cc of PRP obtained from 60 cc of whole blood (n = 9).
Patient Demographics

Data are shown as percent or median ± SD. All comparisons not significant (p > 0.1). TMR, transmyocardial revascularization; PRP, platelet-rich plasma; CCAS, Canadian Class Angina Score.
Whole Blood Characteristics of Platelet-Rich Plasma (PRP): Subcomponent Amounts

Whole Blood Characteristics of Platelet-Rich Plasma (PRP): Quantitative Cell Count Results

PLT, platelets; HCT, hematocrit; RBC, red blood cells; WBC, white blood cells; LYPH, lymphocytes; MID, cells that are rare cells in circulating blood that may include monocytes, eosinophils, basophils, blasts, and other precursor white cells; GRAN, granulocyte. K/μl: thousands per microliter; M/μl: millions per microliter.
Whole Blood Characteristics of Platelet-Rich Plasma: Percent Cell Count Results

PLT, platelets; HCT, hematocrit; RBC, red blood cells; WBC, white blood cells; LYPH, lymphocytes; MID, cells that are rare cells in circulating blood that may include monocytes, eosinophils, basophils, blasts, and other precursor white cells; GRAN, granulocyte.
Whole Blood Characteristics of Platelet-Rich Plasma: Percent Cell Count Results

PDGF, platelet-derived growth factor; TGF, transforming growth factor; VEGF, vascular endothelial growth factor; FGF, fibroblast growth factor; EGF, endothelial growth factor.
Six-month follow-up occurred in 13/14 patients in group 1 and 9/11 patients in group 2. Two patients (one from each group) were lost to follow-up after their initial 1-month postoperative appointment. Both of these patients had significant improvement in angina after their procedures. Follow-up in one group 2 patient could not be achieved because of a 30-day mortality. A comparison of angina scores at baseline and 6 months postoperatively for both groups are shown in Figure 1. When successful angina relief was defined as two class reduction in CCS angina score, both groups achieved significant improvement at 6 months (92% for TMR alone, 100% for TMR + PRP, p = 0.4), although absolute angina scores trended to be less in the TMR + PRP group (1.3 for group 1 vs. 0.4 for group 2, p = 0.07). At 6 months, however, 78% (7/9 patients) of the TMR + PRP group were angina free (CCSAS zero), compared to 23% of the TMR alone group (3/13 patients), as shown in Figure 2 (p = 0.04).

Canadian Cardiovascular Society Angina Scores 6 months after procedure (p = 0.07 at 6 months).

Angina improvement 6 months after procedure (p = 0.04 angina free).
Paired baseline and 6-month Cardiolite nuclear stress tests were performed to determine changes in EF, if any. We chose this method of testing because it was readily available in all study patients at the single institution. Regional myocardial perfusion in these Cardiolite nuclear stress tests was subjectively analyzed by 20 different cardiologists at the single institution. This subjective analysis was thought to have inherent bias between the examiners. Therefore, the changes in myocardial perfusion noted after TMR and/or TMR + PRP were not included in this preliminary study as a method of comparison. However, because the EF was an objective, computer-calculated value, the average numbers between the two groups were compared. Computer-calculated EF at 6 months, relative to the baseline EF, was improved in the TMR + PRP group (–2.0% vs. +9.0%, p = 0.07), compared to the TMR-alone group (Fig. 3).
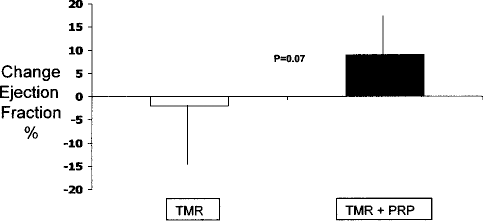
Change in ejection fraction 6 months following procedure (p = 0.07).
Overall operative mortality (30 day) in this series was 4% (1/25). The only mortality occurred in a TMR/PRP patient 11 days following discharge. The patient was a 60-year-old male with a baseline EF of 35% who had both congestive heart failure symptoms and class IV angina. He was discharged uneventfully on POD 1 with no arrhythmias, and reportedly doing well until POD 11 when he suddenly collapsed on a commode, and was found unresponsive, hypoxic, and in electromechanical dissociation. Family declined autopsy. Overall morbidity was 8% (2/25). Rapid atrial fibrillation occurred on postoperative day 3 in a TMR-alone patient, who was discharged on day 5 following medical conversion to normal sinus rhythm. A second patient who underwent TMR alone was readmitted on POD 14 with symptomatic left pleural effusion, and was discharged uneventfully 2 days following thoracentesis. No postoperative ventricular arrhythmias, bleeding, or transfusions were observed.
Discussion
Transmyocardial revascularization is a recommended surgical procedure (6) to treat patients with medically refractory class III/IV angina who are not candidates for conventional revascularization (4). Randomized prospective trials have demonstrated a significant improvement in angina, a reduction in cardiac medications and readmissions, and improved event-free survival when sole therapy TMR patients are compared to maximal medical therapy patients (2). These benefits have been shown to last as long as 5 years (1). However, TMR is not 100% effective. In approximately 25% of patients treated with sole therapy TMR, angina relief is not significantly improved at 1 year (5). Furthermore, complete angina amelioration was even less impressive in these studies, with only approximately one third becoming angina free (10). Finally, demonstration of improved myocardial perfusion and ejection fraction by TMR has been inconsistent.
Recent studies have evaluated the role of exogenously administered biologic substances, such as growth factors and stem cells, as new therapeutic options for medically refractory angina. Intramyocardial injection of specific growth factors, such as vascular endothelial growth factors (16) and fibroblastic growth factor, may provide angina improvement (17) and improve ventricular function (22) in inoperable patients. Similarly, intramyocardial injection of autologous bone marrow-derived mononuclear stem cells such as CD34+ (15) and AC133+ (20) stem cells have been shown to improve angina and myocardial perfusion in patients with refractory ischemia. Animal studies of TMR and gene therapy confirm these findings of improvement in contractility (12). This evidence suggests that isolated intramyocardial growth factor or stem cell injection therapy may be associated with angina relief and improved regional myocardial function and perfusion.
Animal models of myocardial ischemia can provide some insight into the importance of platelet-derived growth factors and stem cells in linking the mechanism of TMR and angiogenesis. Hughes and Lowe concluded that TMR-induced neovascularization is due to an upregulation of the angiogenic cascade secondary to the inflammatory response after laser treatment (13). Alturi and colleagues demonstrated that the acute healing phase of the laser injury involves an upregulation of injured myocytes, platelet activation with growth factor release from the thrombus that forms within the laser channel, and the recruitment of intrinsic myocardial stem cells (3). Lutter et al. suggested that administration of fibroblastic growth factor may enhance TMR's angiogenic response (18). Furthermore, Patel and colleagues have recently demonstrated enhanced stem cell retention when stems cells are injected into the border zone of the TMR channel, suggesting that the microenvironment created by TMR may be important for stem cell retention following myocardial ischemia (19).
Autologous platelet-rich plasma has been used in orthopedic, dental, and wound healing applications (8) with improved clinical outcomes. PRP is readily available from the centrifugation of whole blood and the concentration of plasma, and can be easily obtained within 15 min in the operating room. Qualitative assessment of PRP typically yields platelet counts that are approximately three to six times that of baseline from all the commercially available cell separators, including the Magellan point of care cell separator (9). Further analysis of the composition of PRP yields multiple angiogenic growth factors (21), including platelet-derived growth factor BB (PDGF-BB), transforming growth factor B (TGF-B1), vascular endothelial growth factor (VEGF), and epidermal growth factor (EGF) (24). PRP-derived growth factors may increase 18-fold after concentration with the cell separators (9), and may potentiate the differentiation of human mesenchymal stem cells (14). Finally, PRP has known clinical properties promoting hemostasis when combined with thrombin. Because PRP is autologous, readily available from a minimal amount of peripheral blood centrifugation, and contains many angiogenic factors, we hypothesized that PRP injection into chronically ischemic myocardium may provide a microenvironment rich in concentrated substances to enhance TMR's known angiogenic mechanism.
The results of our preliminary clinical study suggest that intramyocardial injection of autologous PRP, when combined with TMR, may provide superior angina relief and possibly improved ejection fraction in patients with inoperable class III/IV angina. When successful angina reduction is defined as two class angina improvement, 100% of TMR plus PRP patients had two class improvements at 6 months. Furthermore, angina was completely ameliorated in 78% of these patients. Additionally, EF improved 9% at 6 months in these “no-option” patients. These results suggest a possible synergistic clinical effect when TMR is combined with the biologic delivery of autologous PRP. However, the mechanism by which PRP injection, combined with TMR, improves angina relief and possibly global ejection fraction, remains unknown.
There are limitations to this study. First, this study is a simple comparison of two groups of patients with end-staged coronary artery disease and class III/IV angina undergoing sole TMR. Nonrandomization, limited patient accrual, and follow-up have obvious bias, despite preliminary results. Second, the quantification of angiogenic growth factors and peripherally circulating stem cells within the PRP concentrate was not evaluated in the present study. Further characterization of individual patient PRP may allow enhanced delivery of autologous regenerative and angiogenic substances. Third, the quantitative measurement of perfusion changes in regional myocardium was not obtained due to inherent difficulties in the clinical scenario, but new techniques of regional quantification of perfusion following TMR are currently being investigated at our institution. Finally, although we did not combine autologous PRP with exogenous thrombin, the hemostatic effects of PRP may have been seen in our patients having no transfusions, bleeding, or pleural effusions in the TMR + PRP group. Nevertheless, this pilot study warrants further investigation.
In conclusion, the results of this preliminary study suggest possible improvement in angina relief and overall EF in inoperable patients with class III/IV angina when PRP is combined with TMR. Autologous PRP is readily available and easily obtained in the operating room, and does not add significant operative time. This technique of autologous biologic delivery of PRP, when combined with TMR, may provide a novel therapeutic approach in patients with end-stage coronary artery disease. Larger randomized trials are necessary to determine the quantitative synergistic effect between TMR and PRP.
