Abstract
Platelet-rich plasma (PRP) is a matrix of fibrin and platelets that releases cytokines that are important in wound healing. PRP is produced from the patient's blood and therefore has less risk of allergic reaction and infection. We have obtained PRP with an enhanced white blood cell component (W-PRP) by optimizing the centrifugal separation of PRP from plasma. Here we show that injection of W-PRP into the auricle of nude mice gave greater tissue augmentation compared to PRP. Further augmentation occurred when bFGF was added to W-PRP, and there was a significant increase in the number of α-smooth muscle actin-positive cells in mice treated with W-PRP+bFGF. Our results suggest that W-PRP may have value in cosmetic surgery aimed at rejuvenation of wrinkled and sagging skin. W-PRP injection constitutes a new concept in cell transplantation, in which cells required for tissue regeneration are induced by cytokines released from the transplanted cells.
Introduction
Platelet-rich plasma (PRP) is made up of blood plasma fractions that contain many platelets. Activation and gelification of PRP results in release of platelet-derived growth factor (PDGF), transforming growth factor (TGR), vascular endothelial growth factor (VEGR), epidermal growth factor (EGF), keratinocyte growth factor (KGF), and fibroblast growth factor (FGF), which are cytokines involved in wound healing (6,12,15). Construction of a reticular matrix of fibrin and platelets is possible and may enable sustained release of these cytokines (20,23,31,33).
A method that uses the patient's own blood plasma has a low risk of allergic reaction and infection, making medical treatment with PRP particularly safe. The first uses of PRP were in promotion of bone formation of the mandibular bone (5,15–17,28,30), and subsequently PRP has been used for treatment of chronic ulcers (27), pressure ulcers (10,25), and tenosynovitis (19). Use of PRP as an in vivo adipocyte delivery system has been reported to favor cell survival (1) and PRP also improves cutaneous incisional wound healing (3,9). In recent years, PRP has been utilized in cosmetic surgery as a filler for wrinkles and sagging skin, with the aim of skin rejuvenation (6,13,32). An example of the wrinkle removal effect by injection of PRP into the lower eyelid is shown in Figure 1. However, while several fundamental studies of the effects of PRP have been performed for external applications such as ulcers, PRP injection into tissue has not been examined.
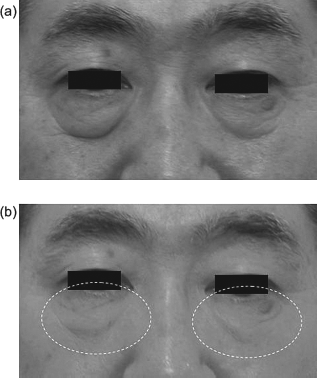
Wrinkle removal by W-PRP. Photographs of the lower eyelid (a) before and (b) 3 months after injection of autologous white blood cell-containing platelet-rich plasma (W-PRP). Wrinkle removal effects were observed after injection of W-PRP.
There are several problems regarding the use of PRP. First, since PRP is derived from individuals, variations in pharmaceutical preparations may occur and this is believed to be a cause of unreliable clinical effects. Second, the quality of PRP varies depending on the method of separation, and separation conditions appropriate for a given treatment have not been established. Furthermore, various anticoagulants and activation methods are in use (34), and accordingly PRP of varying quality is produced.
In this study, we were able to obtain PRP including an increased white blood cell component compared to general PRP by changing the centrifugation conditions. We refer to this material as white blood cell-containing platelet-rich plasma (W-PRP), and we show that W-PRP has improved effects on tissue augmentation compared to PRP. An investigation of the optimum separation conditions for W-PRP was undertaken. We also performed a histological investigation of tissues into which PRP or W-PRP was injected, and we examined if addition of bFGF to W-PRP increased the efficacy of the material.
Materials and Methods
Animals
Genetically immunodeficient mice (BALB/C-nu/nu, 8 weeks old, male) were purchased from SLC Japan Inc. (Osaka, Japan). All animals had free access to water and standard rodent chow. Animal experiments were performed according to the criteria of the Guidelines of the Committee on Animal Care and Use of Kyoto University.
Refining PRP and W-PRP
The optimum conditions were investigated for centrifugal separation of PRP and W-PRP for use in injections. Various methods have been reported for refinement of PRP. We used a PRP separation system from Cascade Medical Enterprises, LLC to enable refinement of PRP using one-time centrifugal separation. Blood samples (9 ml) were collected with trisodium citrate solution used as an anticoagulant. Several centrifugal separation conditions were used (Table 1), including 6 min at 2600 rpm as per protocol. A centrifuge with a 14.5-cm rotor radius (2410, Kubota, Japan) was used for centrifugal separation. After separation, 1 ml taken from the layer closest to the filter was retained and the supernatant (platelet-poor plasma: PPP) was discarded. The retained fluid was mixed to obtain the PRP. As indicated in Table 1, 6 min of centrifugal separation at 2600 rpm enables extraction of conventional PRP, and 10 min of centrifugal separation at 1800 rpm enables extraction of W-PRP, which is high in white blood cell content (i.e., met the objective of this study).
Relationships Between Conditions of Centrifugal Separation and Concentration of Platelets and White Blood Cells (WBCs)

Six minutes at 2600 rpm produces conventional platelet-rich plasma (PRP), which principally contains platelets. Ten minutes at 1800 rpm produces W-PRP (white blood cell-containing platelet-rich plasma), in which the concentration of white blood cells is higher (n = 6).
Separation was impossible.
Analysis of Quality of PRP and W-PRP
Platelets, white and red blood cells (WBCs and RBCs), fibrin monomer complex, bFGF, VEGF, and insulin growth factor-1 (IGF-1) were measured in the separated PRP, W-PRP, and PPP. bFGF and VEGF were measured using a human basic FGF and human VEGF Immunoassay Kit, respectively, from R&D Systems Inc. (MN, USA). IGF was measured by indirect radioimmunoassay (IRIA) using a test reagent from TFB Inc. (Tokyo, Japan). Gelification was also performed and the contents were hematoxylin and eosin (H&E) stained.
Injection of PRP and W-PRP Into the Auricle of Nude Mice
Next, to investigate the clinical effects of PRP, W-PRP, and PPP, these materials were injected into auricles of nude mice and histological examinations were conducted. We also investigated the effect of adding bFGF. For activation, a 10% solution of calcium chloride (CaCl2) was added at a level of 10% to PRP, W-PRP, PPP, and a saline control. The mice were anesthetized with pentobarbital (50 mg/kg, IP). PRP, W-PRP, PPP, W-PRP+bFGF, or saline (0.2 ml) was injected locally into the auricle of nude mice using a 30-gauge needle. bFGF was added to W-PRP at 1, 10, or 100 μg/ml. Histological examinations were performed 2 weeks after injection. In preliminary experiments using bFGF alone, bFGF+PPP, or bFGF+PRP, no tissue augmentation was found that was superior to that with W-PRP+ bFGF (data not shown).
Histological and Immunohistochemical Analysis
Mice ears were biopsied and processed for histology 2 weeks after injection of PRP, W-PRP, PPP, and saline control (n = 5–9). Samples were fixed in 10% formalin, routinely embedded in paraffin, and stained with H&E. Thickening in the region 5 mm from the tip of the auricle was compared with that of the maximally thickened part of each auricle. Sections were also stained with mouse monoclonal antibody against α-smooth muscle actin (anti-α-SMA, clone 1A4, from Dakocytomation Inc., Carpinteria, CA). To detect myofibroblasts, α-SMA was detected by first incubating dewaxed sections in 10% rabbit serum for 30 min, followed by mouse anti-human α-SMA antibody at 4°C overnight. The bound antibody was detected by biotinylated rabbit anti-mouse antibody and streptavidin-horse radish peroxidase (HRP)-diaminobenzidine (DAB). Negative controls were generated by incubating slides with immunoglobulin from the same species and at the same final concentration, but with no primary antibody. All the prepared sections were observed under an optical/fluorescence microscope (Biozero BZ-8000, Keyence Corporation, Osaka, Japan). The number of α-SMA-positive cells was measured in three different areas (400x magnifications) on the same section and the mean value was calculated.
Statistical Analysis
Data are shown as means ± SEM. Statistical evaluation was done by ANOVA, followed by a multiple comparison test with a Bonferroni adjustment using the StatView-J 4.5 software package (Abacus Concepts Inc., Berkeley, CA). A value of p < 0.05 was taken to indicate a significant difference.
Results
Centrifugal Conditions for PRP
The contents of platelets and WBCs in PRP vary depending on the conditions of centrifugal separation (Table 1). To examine the platelet-centered effects as considered conventionally, centrifugal separation conditions of 6 min at 2600 rpm were ideal, and to examine the effects of inclusion of WBC centrifugal conditions of 10 min at 1800 rpm were judged to be desirable. An investigation of both sets of conditions was undertaken. During centrifugal separation over 6 min at 2600 rpm, platelets were concentrated by about fivefold and WBCs fell to about 0.3 times the normal level. With 10 min at 1800 rpm, platelets were concentrated by about fivefold and WBCs were concentrated by about 1.0 times normal. Analysis of the contents of PRP, W-PRP, and PPP showed a high concentration of platelets in PRP as the principal finding, whereas in W-PRP a great many WBCs were present in addition to platelets. The amounts of bFGF and VEGF output were greater in W-PRP compared to PRP. PPP contained almost no platelets or WBCs (Table 2).
Analysis of the Contents of PRP, W-PRP, and PPP
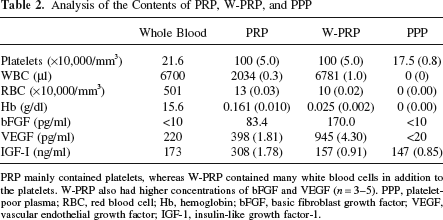
PRP mainly contained platelets, whereas W-PRP contained many white blood cells in addition to the platelets. W-PRP also had higher concentrations of bFGF and VEGF (n = 3–5). PPP, platelet-poor plasma; RBC, red blood cell; Hb, hemoglobin; bFGF, basic fibroblast growth factor; VEGF, vascular endothelial growth factor; IGF-1, insulin-like growth factor-1.
Injection of PRP Into Auricles
From external inspection, thickening of the auricle was observed in the PRP, W-PRP, and W-PRP+bFGF (10 μg/ml) groups. Auricles treated with W-PRP and W-PRP+ bFGF were not transparent to light because of the thickness of the auricle tissue. In contrast, those treated with PPP or saline showed no thickening and were transparent.
Histological Investigation
H&E staining of transverse sections of auricles conducted 2 weeks after local injection into the tips of the auricles showed thickening in the PRP, W-PRP, and W-PRP+bFGF (1, 10, & 100 μg/ml) groups. Conspicuous thickening of the skin and subcutaneous tissue was observed, and hair follicles also seemed to increase. Conversely, almost no thickening was observed in the PPP group or in control auricles that received saline injection (Fig. 2A). Examination of the maximally thickened part of each entire auricle, including a region that is not shown in the photograph (Fig. 2A), indicated superior auricle thickness in the W-PRP and W-PRP+ bFGF groups. Auricle thickening occurred in a concentration-dependent manner with bFGF addition (Fig. 2B), with the greatest tissue thickening in the W-PRP+bFGF (100 μg/ml) group.

Tissue augmentation using W-PRP. (A) H&E staining of mice ears 2 weeks after injection of (a) PRP, (b) W-PRP, (c) W-PRP+basic fibroblast growth factor (bFGF; 100 μg/ml), (d) platelet-poor plasma (PPP), and (e) saline (control). Treatment with PRP, W-PRP, and W-PRP+bFGF (100 μg/ml) gave thickening of the auricle, and this was particularly conspicuous in the skin and subcutaneous tissue. Hair follicles also seemed to increase. In contrast, almost no thickening was observed with PPP and saline. (B) Comparison of average values of the thickest part of the auricle, including the region not shown in (A). Superior thickening was observed with W-PRP and W-PRP supplemented with bFGF. Auricle thickening tended to increase in proportion to the amount of bFGF. *p < 0.01 versus control, #p < 0.01 versus others (n = 5–9).
α-SMA staining showed a tendency for an increase in the number of α-SMA-positive cells in the PRP and W-PRP groups compared with the PPP and control groups. A tendency for a greater increase in the number of α-SMA-positive cells was also observed in the W-PRP+bFGF (10 and 100 μg/ml) groups (Fig. 3).

Production of α-smooth muscle actin (α-SMA) using W-PRP. (A) Observations of α-SMA staining after treatment with (a) PRP, (b) W-PRP, (c) W-PRP+bFGF (10 μg/ml), (d) PPP, (e) saline (control). Compared to PPP and saline, more α-SMA-positive cells (arrows) occurred with PRP, W-PRP, and W-PRP+bFGF (100 μg/ml). (B) Graph showing the numbers of α-SMA-positive cells. An increase in the number of α-SMA-positive cells (400×) was observed with W-PRP supplemented with 10 or 100 μg/ml bFGF. *p < 0.05 versus other groups (n = 5–9).
Discussion
With the aging of society, greater emphasis is being placed on quality of life (QOL) and treatment for skin changes attendant with the aging process has gained greater attention. The safety of cosmetic surgery has also increased, with “no downtime” or “minimum downtime” procedures becoming standard. For these reasons, fillers such as hyaluronic acid are increasingly used in treatment of wrinkles and sagging skin, rather than surgery (which generates downtime). However, treatment with conventional fillers may be flawed: improvement is noticeable after completion of treatment, but long-term sustainability is poor and complete disappearance of the effect may occur within 3–6 months.
Treatment for wrinkles and sagging skin using autologous PRP is recognized as an effective way to compensate for this flaw without concern of infection, and in recent years this approach has been proposed in cosmetic medicine (6). However, fundamental research in this area has not been undertaken and its effectiveness is unknown. Furthermore, while conventional PRP treatment possesses the benefit of incurring almost no downtime, variations among individuals can occur in any treatment using an individual's own blood. We felt that this issue required further investigation.
Following injury, platelets work to stop bleeding and protect the wound, and an active ingredient released from the platelets is associated with healing of the wound. Activated and gelified PRP contains an abundance of platelets and releases cytokines such as PDGF, TGF, VEGF, EGF, KGF, and FGF, all of which are useful in wound healing. However, since variations can occur in platelet concentration and condition, and in the number of nucleated cells, depending on the method used to extract the PRP, we undertook an investigation of the conditions of centrifugal separation (34).
Anticoagulant citrate dextrose-A (ACD-A) fluid is widely used as an anticoagulant, and has generally been used for extraction of PRP with the assumption of blood transfusion. However, it has been suggested that the low pH might affect PDGF and TGF deleteriously. The pH of ACD-A is about 5.1–5.2, while the pH of sodium citrate buffer is about 6.3–6.5, making the sodium citrate anticoagulant preferable to ACD-A (11,12). For this reason, we used a sodium citrate buffer for anticoagulant. Given that time is important in clinical circumstances, we used a PRP separation system from Cascade Medical Enterprises Inc. to obtain PRP by one-time centrifugal separation.
The platelet recovery rate, platelet concentration rate, number of nucleated cells, and involvement of cytokines were examined for PRP obtained under different centrifugal separation conditions (Tables 1 and 2). We demonstrated that W-PRP produced by centrifugal separation over 10 min at 1800 rpm contained not only platelets but also many nucleated cells such as WBCs and macrophages, and was more effective in tissue augmentation (Fig. 2). These findings suggest the importance of both platelets and nucleated cells in this process, and show that inclusion of nucleated cells is more effective than conventional PRP containing platelets alone (14,21,26,35). The bFGF and VEGF concentrations were also higher in W-PRP because the increased platelet concentration, the interaction of platelets and WBCs, and the lower rotational speed led to greater cell protection (Table 2).
Proliferative factors in the wound healing process are generally produced by platelets and macrophages. At low concentrations cytokines cause cell migration, and at higher concentrations they promote cell proliferation. PRP contains many platelets and W-PRP contains an abundance of platelets and WBCs required for general wound healing. Therefore, we believe that W-PRP promotes the same sort of tissue regeneration as that in general wound healing. W-PRP treatment may serve as an artificial trigger of the wound healing process after the inflammatory stage by simultaneous injection of platelets and WBCs, both of which are thought to be important in the induction step. Furthermore, WBCs possess neovascular activity (7), and the WBCs in W-PRP may also have an important role in stimulating migration of peripheral and bloodstream macrophages to infusion sites. In general wound healing, macrophages are observed in great numbers in granulation tissues, and these macrophages are thought to stimulate migration of fibroblasts, which in turn produce collagen. Therefore, WBCs in W-PRP may induce macrophages that then stimulate migration and proliferation of fibroblasts.
The proposed mechanisms of action suggest that W-PRP injection constitutes a conceptually new method of cell transplantation, in which cells required for tissue regeneration are induced by cytokines released from the transplanted cells. This approach differs from the conventional concept of cell transplantation. Tissue regeneration and augmentation may be possible using this approach, with remedy of wrinkles since after injection the skin does not stiffen due to fibrosis because of promotion of wound healing. Shrinking of myofibroblasts within tissue may also be an effective remedy for sagging skin (8) (Fig. 3). Also, unlike the use of PRP in treatment of ulcers, a scaffold is thought to be necessary for tissue construction and augmentation (2), and a network formed from fibrin and platelet gel may be important as a model scaffold derived from in vivo material.
We observed that addition of bFGF to W-PRP caused further tissue augmentation in a bFGF concentration-dependent manner (Fig. 2). The most significant tissue thickening was observed for the W-PRP+bFGF (100 μg/ml) group. This bFGF concentration appears to be high, but W-PRP may be useful for sustained release of bFGF. Reports have indicated that bFGF is effective in skin tissue regeneration and wound healing, and bFGF has been used in external applications for treatment of ulcers and skin defect injuries in Japan (4,18). Local injection of bFGF preparations into wound sutures has been reported to give clean wound healing (22,29), and attempts have been made to improve the action of bFGF through applications using materials such as gelatin (20,24,31). Local injections of gelatin as a sustained-release agent to enable vascular formation for limbs affected by atherosclerosis obliterans (ASO) have been reported, with no side effects if appropriate quantities are used. However, partly due to the bovine spongiform encephalopathy (BSE) problem, the immediate clinical use of gelatin is unlikely. bFGF also increased the number of α-SMA-positive cells and we suggest that sagging skin can be further improved by adding bFGF to W-PRP. Moreover, bFGF also prevents fibrosis through efficient promotion of apoptosis.
Our results suggest that it is possible to remedy the occurrence of individual variations in the active components of PRP that occur with use of the patient's own blood in conventional PRP treatment. PRP can be created by maintaining platelets in as good a condition as possible after blood is collected, and with improvement of platelet concentration techniques, injection with W-PRP and W-PRP+bFGF will make treatment for wrinkles and sagging skin even more effective. However, investigation of the duration of the effects of this treatment will be necessary. In conclusion, we believe that platelets, a fibrin network, and WBCs are necessary for tissue augmentation after injection, making W-PRP more effective than conventional PRP for use in treatment for wrinkles and sagging skin. Myofibroblast cells are also believed to be involved in tissue thickening, and a further increase in thickening can be obtained by addition of bFGF to W-PRP.
Footnotes
Acknowledgments
This work was partly funded by Yoshikawa Hospital. The authors declare no conflicts of interest.
