Abstract
Periodontal ligament cell (PDLC) sheets have been shown to contribute to periodontal tissue regeneration. Dental follicle cells (DFCs), acknowledged as the precursor cells of PDLCs, have demonstrated stemness, embryonic features, heterogeneity, and pluripotency. Therefore, we hypothesized that DFC sheets might be more effective and suitable for periodontal tissue regeneration than PDLC sheets. In this study, we compared the biological characteristics of DFC sheets and PDLC sheets in vitro. To investigate the potential for periodontal tissue regeneration in vivo, complexes composed of two types of cell sheets combined with dentin matrix were implanted subcutaneously into nude mice for 6 weeks. Our results showed that, when forming cell sheets, DFCs secreted richer extracellular matrix than PDLCs. And compared to DFCs, DFC sheets expressed high levels of calcification-related genes, including alkaline phosphatase (alp), bone sialoprotein (bsp), osteopontin (opn), runt-related transcription factor (runx2), as well as the periodontal ligament-specific genes collagen III (col III) and periostin, while the gene expression of bsp, osteocalcin (ocn), and opn were greatly increased in PDLC sheets, when compared to PDLCs. col I expression did not change significantly. However, cementum protein 23 (cp-23) expression increased several fold in PDLC sheets compared to PDLCs but decreased in DFC sheets compared to DFCs. DFC and PDLC sheets were both positive for Collagen I (Col I), cementum attachment protein (CAP), ALP, BSP, OCN, and OPN protein expression, and Col I, ALP, BSP, and OPN expression were increased after cell sheets were formed. Furthermore, the levels of laminin and fibronectin were higher in DFCs and DFC sheets than that of PDLCs and PDLC sheets, respectively. In vivo, DFC and PDLC sheets could both regenerate periodontal tissue-like structures, but DFC sheets demonstrated stronger periodontal regeneration potential than PDLC sheets. Therefore, DFC sheets derived from discarded dental follicle tissue after tooth extraction may be more advantageous for clinical periodontal tissue regeneration in the future.
Keywords
Introduction
Periodontitis is one of the most widespread diseases and is characterized by chronic inflammation of the supporting tissue of the tooth, eventually leading to tooth loss. The ultimate goal of periodontal treatment is the regeneration of periodontal tissue (18). However, conventional methods of periodontal tissue engineering, such as guided tissue regeneration (GTR) or application of various growth factors and regeneration materials, only result in partial regeneration, depending on the periodontal defects of the patient (2, 28, 35). This is likely due to the unique structures of periodontal tissue, which include alveolar bone, cementum (hard tissue), and the connection between them—periodontal ligament (soft tissue). In addition, periodontitis leads to the destruction of periodontal tissue and loss of periodontal ligament cells, so it is often necessary to transplant cells into the periodontal defects (22). Therefore, suitable seed cells and an efficient regeneration system are crucial for the regeneration of the periodontium.
The dental follicle is an ectomesenchyme-derived loose connective tissue sac surrounding the enamel organ and the dental papilla of the developing tooth germ before eruption. Dental follicle cells (DFCs) are the progenitor cells of cementoblasts, periodontal ligament cells (PDLCs), and osteoblasts (11, 25, 43). Human DFCs, which have been isolated from wisdom teeth (25), are an embryonic-like stem cell population from dental germ that can be found in adults and express early neural progenitor markers and vascular endothelial growth factor receptors (3).
Structural and functional periodontal tissue regeneration is difficult to achieve via conventional methods. Cell sheets, which were developed by Okano et al. in the 1990s, maintain their naturally formed networks without the use of proteolytic enzyme treatment or mechanical methods often employed to destroy the extracellular matrix and surface proteins (6). Therefore, cell sheets provide a living microenvironment for seed cells, which increases the application efficiency and reduces the inflammatory reaction and immunologic response often induced by foreign materials. Presently, PDLC sheets have been used for periodontal tissue regeneration (5, 14, 15, 42). As the precursor cells of PDLCs, DFCs contain more stem cells and show stronger differentiation potential, which is beneficial for periodontal regeneration. As a result, DFC sheets may possess more potential to regenerate periodontal tissue that is structurally and functionally intact than PDLC sheets.
In this study, we explore the biological characteristics and capacities of the two kinds of cell sheets, and their potential for use in periodontal regeneration. An additional goal of this work was to determine if DFCs have potential as a seed cell, thus providing a more efficient way to regenerate periodontal tissue.
Materials and Methods
Human cell culture and animal experiments were approved by the ethics committee of Sichuan University. Informed consent of patients was obtained for all human studies.
Cell Culture
Human dental follicle was isolated from an embedded third molar after extraction. Human periodontal ligament was isolated from a first premolar extracted from an orthodontic patient. The dental follicle and periodontal ligament were carefully washed with phosphate-buffered saline (PBS) containing 100 units/ml penicillin and 100 mg/ml streptomycin, minced, and digested in a solution of 1% collagenase and 1% dispase (Sigma) sequentially for 40 min each at 37°C. Both single cells and digested tissues were suspended in 3 ml complete α-modified minimum essential medium (α-MEM) supplemented with 15% fetal bovine serum (FBS, Hyclone), 100 units/ml penicillin, and 100 mg/ml streptomycin. They were seeded into a 25-cm2 plastic flask (Corning) and incubated at 37°C in 5% CO2 in a humidified atmosphere. The culture medium was added to 2 ml after 24 h and changed every 2–3 days until cells were confluent.
Immunofluorescence Microscopy
To identify cell surface markers and characteristics, DFCs and PDLCs were seeded onto glass coverslips in a six-well plate (2 × 104 cells/well) and further cultured for 1 day. The cells were fixed with 4% polyoxymethylene for 10 min. Subsequent steps were performed according to the manufacturer's recommended protocol. Antibodies used included mouse monoclonal anti-Stro-1 at 10 μg/ml (R&D Systems, FWA10), mouse monoclonal anti-vimentin at 1 μg/ml (Thermo, OMA1-06001), and mouse monoclonal anti-cytokeratin-14 (CK-14, Abcam, ab49747) at a dilution of 1:800. The secondary antibody used was fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgM at a dilution of 1:200. The control group was incubated with PBS instead of primary antibody. All samples were examined under a fluorescence microscope (Leica).
Flow Cytometry Analysis
To test cell surface cluster of differentiation (CD) antigens, DFCs and PDLCs were collected, washed twice with PBS containing 20% FBS, resuspended to a final concentration of 1 × 107 cells/ml and 100 μl was distributed into each tube. Subsequent steps were performed according to the manufacturer's recommended protocol. Antibodies used included FITC-conjugated mouse anti-human CD31 (BD Biosciences, 555445; 20 μl), phycoerythrin (PE)-conjugated mouse anti-human CD146 (BD Biosciences, 550315; 20 μl), mouse anti-human Stro-1 (R&D Systems, FWA10; 100 μg/ml). Stro-1 was visualized by adding goat anti-mouse IgM-carboxyfluorescein secondary (R&D Systems, F0118) and incubating for 30 min. Finally, the stained cell samples were analyzed by flow cytometry (BD Biosciences).
CCK-8 Cell Proliferation Assay
To detect cell proliferation, cells were seeded into 96-well plates (5 × 103 cells/well). The culture medium was discarded, and 110 μl fresh culture medium containing 10 μl of cell counting kit-8 (CCK-8) solution (Dojindo) was added to each well. After 2 h of incubation in the dark, the absorbance was measured at 490 nm via a plate reader (Thermo). Each experiment was performed using three wells per experimental condition, and the experiment lasted for seven continuous days.
Transmission Electron Microscope (TEM)
To observe cellular microstructure via transmission electron microscope, DFCs and PDLCs were collected, washed twice with PBS, and centrifuged to cell pellets (1,000 rpm, 5 min). They were then fixed with 3% glutaric dialdehyde (pH 7.2–7.4) for 30 min, 1% osmium tetroxide for 10 min, dehydrated with a concentration gradient of alcohol, and embedded with epoxide resin. The samples were sliced into ultramicrocut and observed under transmission electron microscope (Hitachi).
In Vitro Multidifferentiation of DFCs and PDLCs
DFCs and PDLCs were seeded into a six-well plate (1 × 105 cells/well) separately and further cultured for 1 day.
To induce adipogenic differentiation, the medium was replaced with complete α-MEM culture medium supplemented with 10% FBS, 2 mM insulin (Sigma), 0.5 mM isobutyl-methylxanthine (IBMX, Sigma), and 10 nM dexamethasone (Sigma) for 14 days. The medium was changed every 3 days. The cells were fixed with 4% polyoxymethylene for 10 min and stained with 0.3% Oil Red O (Sigma) solution to evaluate adipogenesis.
To induce osteogenic differentiation, the medium was replaced with complete α-MEM culture medium supplemented with 10% FBS, 10 mM β-glycerophosphate (Sigma), 10-8 M dexamethasone (Sigma), 50 μg/ml ascorbic acid, and 10 nM vitamin D3 (Sigma) for 14 days. The cells were fixed with 4% polyoxymethylene for 10 min, followed by Alizarin Red (Sigma) staining to assess mineral deposition.
To induce neurogenic differentiation, the medium was replaced with complete α-MEM culture medium supplemented with 10% FBS, 2% dimethyl sulfoxide (DMSO), 200 μM butylated hydroxyanisole (Sigma), 25 mM KCl, 2 mM valproic acid (Sigma), 10 μM forskolin (Sigma), 1 μM hydroxycortisone, 5 μg/ml insulin (Sigma), and 2 mM l-glutamine (Sigma). After 24 h of incubation, cells were fixed and immunostained with rabbit monoclonal anti-neurofilament-L (NF, Millipore, AB9568) at a dilution of 1:200. Immunostaining controls were incubated with PBS instead of primary antibody.
The Oligonucleotide Primer Sequences Utilized in the RT-PCR

Cell Sheet Culture
Cell sheets were cultured as described previously (45). Third passage DFCs and PDLCs were separately subcultured into six-well plates (5 × 104 cells/well). After 2 days, the medium was replaced with complete α-MEM culture medium containing 10% FBS and 50 μg/ml ascorbic acid 2-phosphate (AA, Sigma) and changed every 3 days until cell sheets formed.
Scanning Electron Microscope (SEM)
To observe cellular microstructure, cell sheets were harvested after 4 weeks of culture. They were washed with PBS three times, fixed with 2% glutaraldehyde at 4°C for 12 h, dehydrated through a graded ethanol series, dried in a critical-point dryer, coated with conductive material, and observed under a scanning electron microscope (FEI).
Western Blot
Proteins were extracted from cells or cell sheets. To detect the protein expression, Western blot analyses were performed with samples normalized for protein concentration. Membranes were probed with primary antibodies including mouse monoclonal anti-actin (actin, Abcam, ab3280, 1:2,000), mouse monoclonal anti-collagen I (Col I, Abcam, ab6308, 1:1,000), rabbit polyclonal anti-alkaline phosphatase (ALP, Abcam, ab95462, 1:500), rabbit polyclonal anti-bone sialoprotein (BSP, Abcam, ab52128, 1:500), mouse monoclonal anti-osteopontin (OPN, Abcam, ab8448, 1:1,000), rabbit polyclonal to anti-periostin (periostin, Abcam, ab14041, 1:500). Following several washes in Tris-buffered saline with Tween (0.2%), membranes were incubated with horseradish peroxidase-conjugated anti-rabbit or mouse secondary antibody (1:5,000).
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
The Oligonucleotide Primer Sequences Utilized in the qRT-PCR
Enzyme-Linked Immunosorbent Assay (ELISA)
The culture supernatant and total protein of cells or cell sheets were collected separately and centrifuged for 20 min at 3,000 rpm at 4°C. Sandwich ELISA plates (R&D System) were adopted to detect the production of laminin (LN) and fibronectin (FN) quantitatively. The specification curve was established at first. Fifty microliters of sample was added to each well and incubated for 30 min at 37°C, and the plates were carefully washed at least three times. Fifty microliters of enzyme was added to every well, except the blank well, and incubated for 30 min at 37°C and washed thoroughly. Fifty microliters of chromogen solution A and 50 μl chromogen solution B were then sequentially added to each well and protected from light for 15 min at 37°C. Finally, 50 μl stop solution was added to stop the reaction. With the blank well as zero, the absorbance was read at 450 nm via plate reader (Thermo).
In Vivo Transplantation of Cell Sheets with Treated Dentin Matrix Complexes
To evaluate the periodontal regeneration potential of cell sheets in vivo, the cell sheets and treated dentin matrix complexes were transplanted into the subcutaneous dorsa of the nude mouse.
Premolars extracted from orthodontic patients were used to prepare treated dentin matrix (TDM) as described previously (21). The outer treated dentin matrix ring was inactivated through high temperature and pressure.
The cell sheets were carefully uncovered after 4 weeks of culture, wrapped in at least three layers of treated dentin matrix, and placed in the inactivated treated dentin matrix ring (Fig. 1). These complexes were then cultured in complete α-MEM culture medium containing 10% FBS and 50 μg/ml ascorbic acid 2-phosphate at 37°C for 1 h to allow stable adhesion. Ten male, 8-week-old nude mice were purchased from the animal center of Sichuan University. General anesthesia was performed by intraperitoneal injection of 10% chloral hydrate during all surgical procedures. The cell sheets and treated dentin matrix complexes were implanted into the subcutaneous dorsa of the nude mouse, placed at the muscle surface and sutured carefully.
Scheme for cell sheet complexes combined with treated dentin matrix (TDM). Inactivated TDM was deactivated through high temperature and pressure. TDM was used to instead of the root surface, and inactivated TDM, which mainly consisted of hyaluronan (HA), was used to imitate the alveolar bone.
Histological Analysis
Cell sheets were harvested after 4 weeks of culture, washed three times in PBS, fixed with 4% polyoxymethylene at 4°C for 24 h, dehydrated through a graded ethanol series, embedded in paraffin, and sectioned at 4 μm prior to hematoxylin and eosin (H&E) and immunofluorescence staining.
The nude mice were sacrificed 6 weeks after surgery. The cell sheets and TDM complex were dissected out and conventionally fixed with 4% polyoxymethylene at 4°C for 24 h. The transplants were decalcified with 10% EDTA (pH 8.0), dehydrated through a graded ethanol series, and embedded in paraffin. The 5-μm paraffin sections were prepared for further histological analysis.
For histochemical analysis, H&E, Masson's trichrome, or immunofluorescence (IF)/immunohistochemical (IHC) staining was performed on the sections according to the manufacturer's protocol. For immunofluorescence/immunohistochemical staining, sections were incubated with the following primary antibodies: anti-ALP (Abcam, ab95462, 1:400), anti-BSP (Abcam, ab52128, 1:100), anti-periostin (Abcam, ab14041, 1:500), anti-cementum attachment protein (CAP, Santa Cruz, sc53947, 1:200), anti-Col I (Abcam, ab6308, 1:500), anti-osteocalcin (OCN; Abcam, ab13418, 1:100), anti-OPN (Abcam, ab8448, 1:100), anti-mitochondria (Millipore, MAB1273, 1:100). Secondary antibodies included FITC-conjugated goat anti-mouse or anti-rabbit IgG at a dilution of 1:200. The Envision™ Detection Kit (Gene Tech) was used for immunohistochemical staining. The control group was incubated with PBS instead of primary antibody. All samples were examined under a compound microscope (Leica).
Quality Control
All samples were done in triplicate, and the results were confirmed by three independent experiments to duplicate the experimental conditions.
Statistical Analysis
All data are expressed as mean (± SD). For statistical analysis, one-way analysis of variance (ANOVA) was performed, followed by the Student–Newman–Keuls posttest using SPSS l7.0 statistical software. A value of p < 0.05 was considered statistically significant.
Results
Characteristics of Human DFCs and PDLCs
DFCs and PDLCs both displayed the typical morphology of mesenchymal cells. Nearly all the cells exhibited high levels of vimentin staining but were negative for CK-14 (Fig. 2A). DFCs exhibited higher expression levels of mesenchymal stem cell surface markers, including Stro-1 and CD146. DFCs and PDLCs were also weakly positive for CD31 (6.1% and 5.3%, respectively) (Fig. 2B).
(A) Immunofluorescence staining for Stro-1, vimentin, and cytokeratin (CK)-14 for dental follicle cells (DFCs) and periodontal ligament cells (PDLCs). Scale bars: 100 μm. (B) Flow cytometry analysis of Stro-1, CD146, and CD31 expression in DFCs and PDLCs. (C) The proliferation curves of DFCs and PDLCs. (D) The ultrastructure of DFCs and PDLCs. A divided nucleolus and electron-dense particles (arrows) could be found in DFCs. PDLCs were rich in rough endoplasmic reticulum and ribosomes (arrows). (E) The relative gene expression of DFCs and PDLCs were examined by qRT-PCR. *p < 0.05.
The cell proliferation curves revealed an obvious difference between DFCs and PDLCs. After 2 days, both DFCs and PDLCs began to grow very quickly. In particular, the proliferation rate of DFCs was much higher than that of PDLCs. This trend persisted until the 7th day, when PDLCs proliferation reached a plateau, while DFCs continued to expand (Fig. 2C).
Cellular ultrastructure was observed by transmission electron microscope. For DFCs, the divided nucleolus and some distinctive electron-dense particles were observed, in addition to multiple lysosomes, ribosomes, rough-surfaced endoplasmic reticulum, and cytoplasmic processes contacting other cells. While in PDLCs, we observed a large nucleolus, rich mitochondria, rough-surfaced endoplasmic reticulum, and ribosomes (Fig. 2D).
The distinguishing markers of DFCs were more highly expressed than those of PDLCs. All of the stem cell-related genes, including nestin, notch-1, octamer-binding transcription factor 4 (oct4), and nanog, were more highly expressed in DFCs (Fig. 2E).
Both DFCs and PDLCs exhibited pluripotency. Following 14 days of adipogenesis inducing culture conditions, oil droplets were observed in DFCs and PDLCs via Oil Red O staining. In addition, following osteogenesis, mineral nodules were also observed in DFCs and PDLCs after Alizarin Red staining. Induction of neurogenesis lasted for 24 h, and DFCs and PDLCs gradually assumed a multipolar, neuronal morphology. Immunofluorescence staining for neurofilament, a marker for late-stage neurogenesis revealed high levels of staining after induction. Cell differentiation-specific genes were also expressed (Fig. 3).
Cellular multidifferentiation. Adipogenesis: Oil Red O staining, lipoprotein lipase (LPL), and peroxisome proliferator-activated receptor gamma (PPARγ) gene expression; Osteogenesis: Alizarin Red staining and alkaline phosphatase (ALP) and bone sialoprotein (BSP) gene expression; Neurogenesis: immunofluorescence staining of neurofilament and β-tubulin III gene expression. Scale bars: 100 μm.
Morphology of Cell Sheets
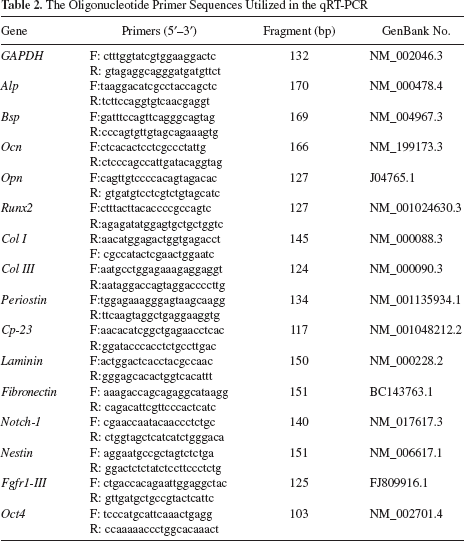
Cell sheets were formed after 10 days in culture. The cells became confluent and continued to grow until overlapping. Specifically, DFC sheets appeared thinner and softer than the PDLC sheets when observed with the naked eye. The appearances of cell sheets observed via scanning electron microscope were also very different. For instance, DFC sheets secreted rich extracellular matrix (ECM) and were prone to form a tight network of collagen fibers and less calcification, while PDLC sheets secreted cytoplasm-like mineralization and were prone to form a tenacious membrane. When the cell sheets were cut into slices and stained with H&E, the space between cells and ECM was lower in DFC sheets than PDLC sheets (Fig. 4).
Cell sheet morphology. (A) Gross observation: DFC sheets were thin and soft, while PDLC sheets were thick and tough. (B) Light microscope view. There was no significant difference between DFC sheets and PDLC sheets. (C) Scanning electron microscope views showed the cellular arrangement and extracellular matrix (ECM) of cell sheets. Black arrow, collagen fibers; white arrow, cytoplasmic-like mineralization; red arrow, extracellular matrix. (D) Histological observation showed the proportion of cells and ECM in cell sheets. Scale bars: 100 μm.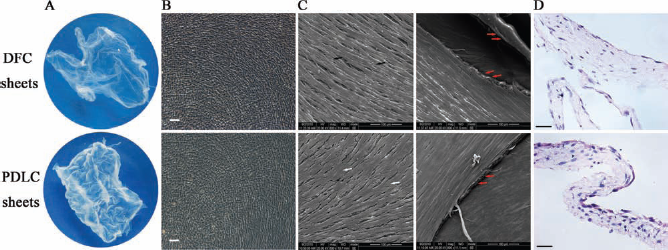
Protein and Gene Expression
We observed Col I, CAP, ALP, BSP, OCN, and OPN immunofluorescence in DFC and PDLC sheets. Furthermore, our Western blot results showed that Col I, ALP, BSP, and periostin expression were significantly increased in cell sheets when compared to cells but that the expression of OPN was nearly unchanged in DFC sheets (Fig. 5).
The expression of cell differentiation-related proteins in cell sheets. (A) immunofluorescence staining. Scale bars: 100 μm. (B) Western blot analysis. The ratio of relative intensity was the ratio of the gray value between the target protein and actin in the same sample. *p < 0.05. CAP, cementum attachment protein; OCN, osteocalcin; OPN, osteopontin.
Eleven genes related to cellular differentiation, adhesion, and migration were analyzed, including alp, bsp, ocn, opn, runt-related transcription factor 2 (runx2), col I, col III, periostin, cementum protein 23 (cp-23), laminin, and fibronectin. The relative gene expression profiles of DFCs and PDLCs were very different after forming cell sheets. For instance, compared to DFCs, DFC sheets exhibited high expression of calcification-related genes (alp, bsp, opn, runx2) and periodontal tissue-specific genes (col III, periostin), while the gene expression of bsp, ocn, and opn were greatly increased in PDLC sheets, when compared to PDLCs. Col I expression did not change significantly. However, cp-23 expression increased several fold in PDLC sheets compared to PDLCs but decreased in DFC sheets compared to DFCs. The two cell adhesion and migration-related genes were also expressed differently in sheets compared to cells. Laminin was decreased significantly, while fibronectin expression was slightly increased cell sheets (Fig. 6).
Cellular differentiation, adhesion, and migration-related genes were examined by qRT-PCR before and after forming cell sheets. *p < 0.05. Col I, collagen I; cp-23, cementum protein 23; runx2, runt-related transcription factor 2.
ELISA Analysis of Cellular Matrix
The synthesis and secretion of laminin and fibronectin were detected by ELISA kit. Overall, the production of laminin and fibronectin were higher in DFCs and DFC sheets than in PDLCs and PDLC sheets, both intracellularly and extracellularly. The production of laminin was the highest in DFCs, slightly increased in DFC sheets, but decreased in PDLC sheets. On the other hand, compared to DFCs, the production of fibronectin was decreased in the intracellular matrix of DFC sheets but increased significantly in the extracellular matrix. Fibronectin was decreased in PDLCs both intracellularly and extracellularly after forming cell sheets (Fig. 7).
ELISA analysis of laminin and fibronectin. (A) Laminin. (B) Fibronectin. *p < 0.05.
The Periodontal Regeneration Potential of Cell Sheets
Both DFC sheets and PDLC sheets formed periodontal tissue-like structures in the subcutaneous areas of nude mice. A layer of cementum-like structure was observed outside the dentin, with some fibers observed embedded within. The periodontal ligament was formed and weaved into a network, and there were very rich blood vessels within the ligament. This was demonstrated in DFC sheets after Masson trichrome staining. The cementum-like structure was stained red, but devoid of cells, while on the surface of the cementum lacunas were found, which might constitute the lacuna of cementoblasts. In the PDLC sheets group, a mineral matrix-like structure formed, which contained some cells outside the dentin (Fig. 8). With IHC staining, we found that the regenerated tissue in both kinds of cell sheets exhibited positive antibody staining for human mitochondria, thus confirming the human source of these cells. Col I and periostin, which are specific markers of periodontal ligament, were also expressed (Fig. 9).
The histological observation of regenerated tissue derived from DFC and PDLC sheets. (A–D) DFC sheet group. (E–H) PDLC sheet group. D, dentin; C, cementum; PL, periodontal ligament. Scale bars: 50 μm. Immunohistological staining against mitochondria, Col I and periostin in periodontal ligament-like structures derived from DFC and PDLC sheets. Scale bars: 50 μm.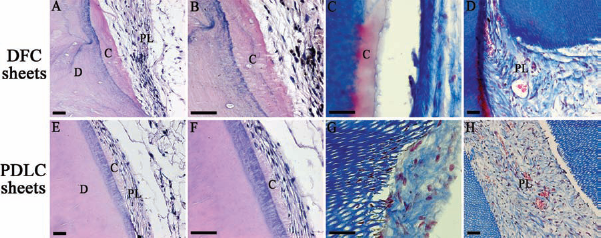

Discussion
The source of the seed cells for periodontal tissue regeneration is worthy of careful consideration (1, 13). Thus far, the potential seed cells under investigation for periodontal tissue regeneration have included PDLCs, DFCs, bone marrow mesenchymal stem cells (BMSCs), and adipose-derived stem cells (ASCs) (11, 24, 41, 46). However, Leucht et al. (20) found that the embryonic origin and homeobox (Hox) gene expression status distinguished neural crest-derived cells from mesoderm-derived skeletal progenitor cells and that both characteristics influenced the process of adult bone regeneration. Rawlinson et al. (33) found that adult bones acquired site-specific characteristics and also noted the persistent expression of site-specific markers associated with the development of the skeleton in both bone organ and isolated adult bone-derived cells. Therefore, whether the heterotopic seed cells could regenerate the normal structures in defect sites was debatable. Moreover, because the periodontal tissue consists of gingiva, alveola, cementum, and periodontal ligament, it is better to choose periodontal-derived source cells for periodontal regeneration.
DFCs and PDLCs are derived from different developmental stages of the periodontal tissue, so different characteristics may exist between them. Stro-1 and CD146 are surface markers of mesenchymal stem cells (MSCs) (4, 10, 17). In this study, DFCs showed a higher expression of Stro-1 and CD146, indicating that DFCs may contain more stem cells. Furthermore, the expression of the stemness-related genes (nestin, notch-1, oct4, nanog) also suggest the pluripotency of DFCs is superior to that of PDLCs. DFCs and PDLCs were also weakly positive for CD31, the surface marker of vascular endothelial cells, suggesting these cells have the potential for angiogenic differentiation (32). In addition, the ultrastructure of DFCs showed that these cells were in the active phase of differentiation. Meanwhile, it also suggested that PDLCs had the same ability to synthesize and secrete proteins as mature cells (30). Furthermore, DFCs also attained robust proliferative capacity, which may be an advantage of DFCs for transplantation applications. These results suggested that DFCs could potentially act as seed cells in periodontal regeneration.
Cell sheet technology use in periodontal regeneration has been a rapid and recent advancement (6, 14). Previous studies (15) showed that PDLC sheets could be used to achieve relatively complete periodontal tissue regeneration, including cementum, periodontal ligament, and alveolar bone in canines. However, as the precursor cells of PDLCs, DFCs could also differentiate into cementoblasts, PDLCs, and osteoblasts, as well as form supporting tissues of the tooth. In this study, we demonstrated that DFCs were more pluripotent than PDLCs. Therefore, DFC sheets might be more effective and suitable for periodontal tissue regeneration than PDLC sheets.
The greatest advantages of cell sheets include more effective utilization of seed cells, secretion of extracellular matrix, and preservation of cell surface receptors and junctional factors to achieve efficient reconstruction of tissue (6, 14). This study showed that, when cultured under the same conditions and period, DFC sheets were rich in extracellular matrix, which aids in the early adhesion of cell sheets and the formation of the new periodontal tissue attachments and is the key point for periodontal regeneration (38). Fibronectin, which is involved in cell adhesion, cell motility, opsonization, wound healing, and maintenance of cell shape (29), has already been applied in the clinical treatment of periodontal defects and to promote the formation of new attachments. In addition, laminin, the most abundant structural and biologically active component of basement membranes, has been found to modulate cell differentiation, morphology, and motility via its activity as a cell substrate adhesion protein (39). The higher expression levels of laminin and fibronectin in DFC sheets revealed that DFC sheets may be more easily attached to the tooth root and that these cells could move to the injured area to repair and regenerate the periodontal tissue, referred to as the new attachment of the periodontium. This constitutes the connection between tooth root and periodontal tissue and contained cementum, alveolar bone, and embedded periodontal ligament fibers (34). DFC sheets exhibited greater pluripotency than PDLC sheets, perhaps because DFCs are derived from embryonic developmental stages and retain embryonic characteristics. This may lead to the enhanced formation of new attachments.
Compared to the cells, DFC and PDLC sheets both showed a notably high expression of calcification-related genes. DFC sheets exhibited significantly higher expression of alp, runx2, and bsp genes, suggesting that these cells were in the early stage of osteoblast differentiation and the extracellular matrix had just begun to mineralize (27, 31, 36). But PDLC sheets showed an obvious increase in ocn and opn expression, which are involved in the formation of the main mineral matrix of mineralized tissue (31), including alveolar and cementum.
Periostin, also called osteoblast-specific factor 2 (12), was more highly expressed in DFC sheets. This gene is specifically expressed in periodontal tissue and is involved in periodontal tissue repair and regeneration (26). This suggests that DFC sheets have a stronger potential to regenerate periodontal tissues. Col I and Col III, the main collagen components of periodontal connective tissue (19), were also highly expressed in DFC sheets. Moreover, Norris et al. (26) found that periostin could regulate Collagen I fibrillogenesis and serve as an important mediator of the biomechanical properties of fibrous connective tissues. This suggests that DFC sheets have a greater potential to differentiate into periodontal connective tissue. However, compared to cells, cp-23, the specific marker of cementum formation, was increased several fold in PDLC sheets and decreased in DFC sheets. This could be due to the fact that the PDLC group included several differentiating cementoblasts that could secrete the cemental-related protein, but that DFCs need to differentiate and mature step by step.
In vivo, the cell sheets and treated dentin matrix complexes were constructed to simulate the periodontal microenvironment between root surface and alveolar bone in periodontal defects. Periodontal scaling and root planning was the effective treatment for periodontitis, but the hard tissues, including cementum and dentin, on the root surface were also removed (8). Previous studies found that TDM could release dentinogenic factors and promote cell attachment and growth through demineralization (9, 21), similar to the root surface after scaling and root planning. For this reason, TDM was used instead of the root surface, and inactivated TDM, which mainly consisted of Hyaluronan (HA), was used to imitate the alveolar bone. During periodontal regeneration, it was very difficult to completely regenerate both structure and function without the formation of a new periodontal attachment, which includes new cementum and embedding Sharpey's fibers (34). In vivo, DFCs appeared more pluripotent than PDLCs, especially in cementogenesis and new periodontal attachment. The Sharpey's fibers were embedded in the newly formed cementum and grew toward the outside to connect the cementum and alveolar bone. DFC sheets resulted in better periodontal regeneration, perhaps because the regeneration process was closer to the periodontal tissue development process, to some extent.
Previous work has shown that PDLCs contribute to periodontal regeneration (7, 37). In addition, Ding et al. (5) found that allogeneic PDLC therapy for periodontitis in swine did not trigger serious immune reactions, making allotransplantation of seed cells feasible. However, stem cells were less in the periodontal ligament than the dental follicle. DFCs were cultured from the discarded dental follicle after extraction of wisdom teeth or embedded teeth, and the chance of obtaining DFCs is less than for PDLCs. However, the relatively low immunogenic and tumorigenic nature of fetal stem cells makes them attractive candidates for transplantation (23, 40). DFCs were derived from the developmental phase of teeth and exhibited remarkable embryonic attributes either in vitro or in vivo (3), although this remains controversial (16). These advantages make DFCs more suitable for periodontal regeneration than PDLCs. Here we evaluated the periodontal regeneration potential of cell sheets transplanted into the subcutaneous space of nude mice. Further work must be done in large animal models of periodontal defects. The inflammation of periodontal tissue should also be taken into consideration when attempting to simulate the microenvironment of periodontitis. In addition, in the cell sheets, cells secreted the extracellular matrix, which also constituted the niche of cells. Therefore, the cell proliferation and differentiation must be regulated by the extracellular matrix. But how does the regulation work? And what are the differences between DFCs and PDLCs? These all need further study.
In sum, DFCs and PDLCs are derived from different development stages of periodontal tissue, with DFCs acting as the precursors of PDLCs. Therefore, DFCs exhibited more pluripotency, heterogeneity, and proliferative capacity than PDLCs. After forming cell sheets, DFC sheets were superior to PDLC sheets in cell differentiation and extracellular matrix secretion. In vivo, the periodontal tissue-like structure was observed both in the group transplanted with DFC sheets and that treated with PDLC sheets, but DFC sheets showed stronger periodontal regeneration potential than PDLC sheets. Therefore, DFC sheets may be more suitable and efficient for periodontal regeneration in situ and may also be suitable for bone regeneration (44).
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (30973348), National Basic Research Program of China (973 Program) (2006CB708505 and 2010CB944800), and Doctoral Fund of Ministry of Education of China (20070610064). The authors declare no conflicts of interest.
