Abstract
Cell sheets could be used for bone regeneration without requiring a scaffold and can be easily produced from autologous mesenchymal stromal cells (MSCs). We compared the osteogenic potential of MSC-derived cell sheets in relation to culture time. Undifferentiated cell sheets (U-CS) and osteogenic differentiated cell sheets (O-CS) were generated using canine adipose-derived MSCs. Undifferentiated cells (UCs) were used as the control. Osteogenic differentiation was assessed by assaying alkaline phosphatase (ALP) activity. Expression of osteogenesis-related genes was evaluated by reverse transcription-polymerase chain reaction at 4, 7, 14, and 21 days after initiation of culture. The calcium content in cells was measured, and the cells were stained with Alizarin red S (ARS). The mRNA expression of transforming growth factor-β in U-CS and O-CS at day 4 was higher than that in UCs (p < 0.05). The level of bone morphogenetic protein 7 mRNA in O-CS increased significantly at day 4 and was significantly higher than that of U-CS at day 7. The mRNA level of runt-related transcription factor-2 in both sheet types increased significantly at 7 days of culture. The mRNA level of ALP in O-CS and U-CS increased significantly at day 7, and ALP activity was highest at days 7 and 14, respectively (p < 0.05). The mRNA level of osteocalcin in U-CS and O-CS increased significantly at day 21. O-CS and U-CS showed negative ARS staining but their calcium contents increased marginally at day 21. The O-CS cells started to aggregate at days 10–12, and only a partial sheet remained at day 21. The upregulation of expression of genes related to osteogenic differentiation, peak in ALP activity, and morphological changes in cell sheets suggest that the optimal time for application of O-CS and U-CS is between 7 and 10 days and after 14 days of culture, respectively.
Introduction
Mesenchymal stromal cells (MSCs) show great potential for therapeutic use in tissue engineering due to their immunoregulatory and paracrine effects as well as their ability to transdifferentiate (26,35). However, injection of a dissociated single-cell suspension of MSCs to deliver cells to target tissues has the disadvantage of the cells disappearing from the system soon after transplantation (21). To overcome this obstacle, Okano and coworkers developed a cell sheet technique to improve the therapeutic potential of MSCs (7). These cell sheets could be detached intact from the culture dish at lower temperatures (7,14), enabling the retention of abundant endogenous extracellular matrix (ECM) and proteins on the cell surface. In addition, an intact cell sheet structure ensures homeostasis of the cellular microenvironment (9,39) while delivering growth factors and cytokines over a prolonged period of time to promote tissue repair (21).
The cell sheet technique has been applied to repair several diseased organs such as large lesions of the esophagus, impaired periodontal ligaments, and various thin-layered tissues, including the cornea (23,25). Cell sheet transplantation has been used to repair damage in three-dimensional thickened tissues such as the heart muscle (33). Recently, functional recovery of the liver and pancreas was evaluated following cell sheet transplantation therapy (24,32). Osteogenically differentiated MSC sheets exhibit osteogenic potential in vitro (1,9,19,22,38). In addition, direct application of osteogenic cell sheets to damaged bone promoted bone repair (12,22,29,36), and osteogenic cell sheets implanted under subcutaneous tissues demonstrated in vivo bone formation (1,19). However, the optimal time needed to culture cell sheets for therapeutic applications as well as the requirement of osteogenic differentiation medium to culture MSCs for therapeutic use have not been elucidated. Therefore, in the present study we determined the culture time needed to ensure the highest osteoblastic activity of cells as well as high levels of matrix formation. We also compared the osteogenic potential of differentiated and undifferentiated cell sheets.
Materials and Methods
Isolation and Culture of Adipose Tissue-Derived (AD)-MSCs
Canine AD-MSCs were harvested according to the methods described in our previous article (30). In brief, adipose tissues were collected aseptically from the gluteal subcutaneous fat of 2-year-old beagle dogs (four females). All animal experimental procedures were approved by the Institutional Animal Care and Use Committee of Seoul National University (SNU-141210-1), Korea. Adipose tissue (approximately 1 g) was washed extensively with Dulbecco's phosphate-buffered saline (DPBS; Gibco, Grand Island, NY, USA) and then minced with scissors. The minced tissue was digested with 1 mg/ml collagenase type I (Sigma-Aldrich, St. Louis, MO, USA) for 2 h at 37°C. The samples were then washed with DPBS and centrifuged at 980 × g for 10 min. The resulting pellet of the stromal vascular fraction was resuspended by DPBS and filtered through a 100-μm nylon mesh, followed by overnight incubation in low-glucose Dulbecco's modified Eagle's medium (DMEM; HyClone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS; HyClone) at 37°C in a 5% CO2 humidified atmosphere. After 24 h, the unattached cells and residual nonadherent red blood cells were removed by washing with DPBS. The medium was changed at 48-h intervals until the cells became confluent. The cells were subcultured after they reached 90% confluence, and the cells at passage 3 were used for the experiments (13).
Cell Seeding and Harvesting
Cells were seeded in tissue culture polystyrene dishes and cultured in low-glucose DMEM supplemented with 10% FBS and 1% penicillin/streptomycin (HyClone) at 37°C in a 5% CO2 humidified atmosphere (basal medium). After the cells reached 60–70% confluence, the basal medium was changed to a different medium based on the desired experimental conditions. The undifferentiated cells (UCs) were cultured in basal medium until reaching 100% confluence. The MSCs for undifferentiated cell sheets (U-CS) were cultured in basal medium supplemented with 50 μg/ml L-ascorbic acid 2-phosphate (Asc; Sigma-Aldrich). The MSCs for osteogenic differentiated cell sheets (O-CS) were cultured in high-glucose DMEM supplemented with 10% FBS, antibiotics, 10–7 M dexamethasone (Dex; Sigma-Aldrich), and 50 μg/ml Asc. Both the cells and cell sheets were harvested at 4, 7, 14, and 21 days of culture.
Morphological Examination
ECM formation and morphological changes occurring in cells during culture were monitored under an inverted light microscope (model CKX41SF; Olympus, Tokyo, Japan), and images were captured with a digital camera (model E-330; Olympus).
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted using a Hybrid-R RNA Extraction Kit (GeneAll, Seoul, Republic of Korea), and the RNA concentrations were determined by measuring light absorbance at 260 nm using ImplenNanoPhotometer (model 1443; Implen GmbH, Munich, Germany). PrimeScript II First-strand cDNA Synthesis Kit (Takara, Otsu, Japan) was used to synthesize cDNA using 1,000 ng of total RNA as template, and 1 μl of cDNA was amplified by PCR using the T3000 Thermocycler (Whatman, Biometra, Biomedizinische Analtyik GmbH, Goettingen, Germany). The PCR mixture (25 μl) contained 12.5 μl of PCR Premix (EmeraldAmp™ PCR Master Mix; Takara), 0.2 μM forward primer and 0.2 μM reverse primer (Standard Oligo; Bioneer, Daejeon, Korea), 1 μl of cDNA, and 9.5 μl of distilled water. The primer sequences used for transforming growth factor-β (TGF-β), bone morphogenetic protein-7 (BMP7), runt-related transcription factor-2 (RUNX2), alkaline phosphatase (ALP), collagen1α-1 (COL1α-1), and osteocalcin (OCN) are shown in Table 1. The mRNA levels for each gene were normalized to that of the housekeeping gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The PCR conditions used were denaturation at 94°C (5 min), 27 to 37 cycles at 94°C (1 min), annealing at 50–60°C (1 min), extension at 72°C (1 min), and a final elongation at 72°C. The PCR products were analyzed by electrophoresis on a 2% agarose gel (LE agarose; Dongin, Seoul, Republic of Korea). Redsafe™ Nucleic Acid Staining Solution (iNtRON Biotechnology Inc., Korea) was used to stain the 2% agarose gel. The longitudinal sections of the visualized gel were analyzed using ImageJ (version 1.37; National Institutes of Health, Bethesda, MD, USA). All transcripts obtained from cultured cells were compared to those obtained from 4-day cultures of UCs.
Specific Primers for Reverse-Transcription Polymerase Chain Reaction With Amplicon Sizes and Optimal Annealing Temperatures

GAPDH, glyceraldehyde-3-phosphate dehydrogenase; TGF-β, transforming growth factor-β, BMP7, bone morphogenetic protein-7; RUNX2, runt-related transcription factor-2; ALP, alkaline phosphatase; COL1α-1, collagen1α-1; OCN, osteocalcin.
Alkaline Phosphatase (ALP) Activity Measurement
ALP activity in cells and cell sheets cultured in 100 × 20-mm cell culture dishes was measured using an ALP activity colorimetric assay kit (Biovision, San Francisco, CA, USA). Cells were washed with DPBS, detached using a cell scraper, and sonicated in 300 μl of assay buffer. After sonication, the solution was centrifuged at 13,000 × g for 3 min at 4°C, and 80 μl of the supernatant was added to 50 μl of p-nitrophenyl phosphate substrate in a 96-well plate followed by incubation for 1 h at room temperature (22–25°C) in the dark. Stop solution (20 μl) was added to the colored samples, and the absorbance of each well was measured at 415 nm with a microplate reader (model 680; Bio-Rad, Hercules, CA, USA).
Detection of Mineralization
In Alizarin red S (ARS; Sigma-Aldrich) staining, cells and cell sheets cultured in six-well plates were washed with DPBS twice and fixed in 4% paraformaldehyde (Wako, Tokyo, Japan) at room temperature for 10 min. The cells were then washed twice with distilled water, and 1 ml of 40 mM 2% ARS (pH 4.1–4.3) was added to each well. The plates were then incubated at room temperature for 20 min with gentle shaking. Any unincorporated dye was aspirated from the wells, and the wells were washed four times with distilled water (8).
The calcium content per well was measured using a calcium colorimetric assay kit (Biovision) based on the ortho-cresolphthalein complexone method. Cells and cell sheets cultured in six-well plates were washed in DPBS twice and incubated in 0.5 ml of 10% formic acid (Sigma-Aldrich) and 0.5 ml of 0.2% IGEPAL CA-630 (Sigma-Aldrich) containing 1 mM MgCl2 for 24 h at 4°C with gentle shaking (1). The chromogenic reagent (90 μl) and calcium assay buffer (60 μl) were added to 50 μl of each extract and mixed well. Samples were incubated for 10 min at room temperature and the optical density was measured at 540 nm using a microplate reader (13).
Statistical Analysis
Statistical analysis was performed using SPSS version 22.0 (IBM, Armonk, NY, USA). The Kruskal–Wallis test was used to analyze differences among the groups. The Mann–Whitney U-tests were carried out for the post hoc test. A value of p < 0.05 indicated a significant difference between the groups.
Results
Morphological Changes in Cell Sheets
UCs reached confluence at 4 days of culture (Fig. 1A), whereas U-CS and O-CS cells formed a sheet between 2 and 4 days of culture (Fig. 1B, D). However, the O-CS cells started to aggregate partially at days 10–12 (Fig. 1E), and some intact O-CS remained at 21 days of culture (Fig. 1F). On the other hand, the U-CS maintained an intact matrix form until day 21 (Fig. 1C).
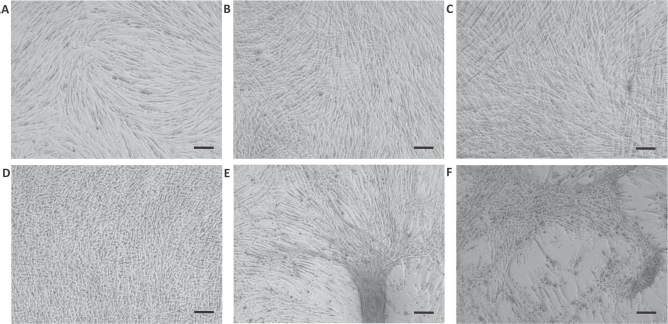
Morphology of canine adipose-derived mesenchymal stromal cells in different media. An inverted microscope was used; magnification: 100×; scale bars: 50 μm. (A) Undifferentiated cells (UCs) cultured in basal medium at 4 days. (B) Undifferentiated cell sheets (U-CS) cultured in basal medium containing 50 μg/ml L-ascorbic acid 2-phosphate (Asc) at 4 and (C) 21 days. The U-CS showed an intact cell matrix until 21 days. (D) Osteogenic differentiated cell sheets (O-CS) cultured in basal medium containing 50 μg/ml Asc and 10–7 M dexamethasone (Dex) formed a matrix after 4 days. (E) After 10 days, cells in the O-CS showed partial aggregation. (F) After 21 days, only partial matrix formation was observed in the O-CS.
Expression of Genes Related to Osteogenic Differentiation
TGF-β mRNA expression in U-CS and O-CS at 4 days of culture was significantly higher than that of UCs (p < 0.05) (Fig. 2A). The upregulation in TGF-β expression in O-CS and U-CS was maintained until days 7 and 14, respectively. BMP7 mRNA expression was significantly higher in O-CS after 4 days of culture than in UCs, and the highest expression was observed at day 7 (p < 0.05) (Fig. 2B). BMP7 expression was also upregulated in U-CS after 7 days of culture. After 14 days of culture, the difference was insignificant statistically in the BMP7 expression between O-CS and U-CS. There was a significant increase in RUNX2 mRNA expression at day 7 in both U-CS and O-CS compared to that in UC. RUNX2 expression was significantly higher in O-CS at days 14 and 21 than in U-CS (p < 0.05) (Fig. 2C). ALP mRNA expression in U-CS and O-CS was significantly upregulated after 7 days of culture compared to that in UCs (p < 0.05) (Fig. 2D). ALP expression in O-CS peaked at day 7, whereas that in U-CS increased gradually until day 21. Upregulation of COL1α-1 mRNA expression was observed in U-CS only at 14 days of culture (p < 0.05) (Fig. 2E). COL1α-1 expression of O-CS was upregulated from day 7. Significant upregulation of OCN expression was observed in U-CS at 4 days of culture compared to that in UCs (p < 0.05) (Fig. 2F). Significant upregulation of OCN expression was observed in both U-CS and O-CS at day 21.
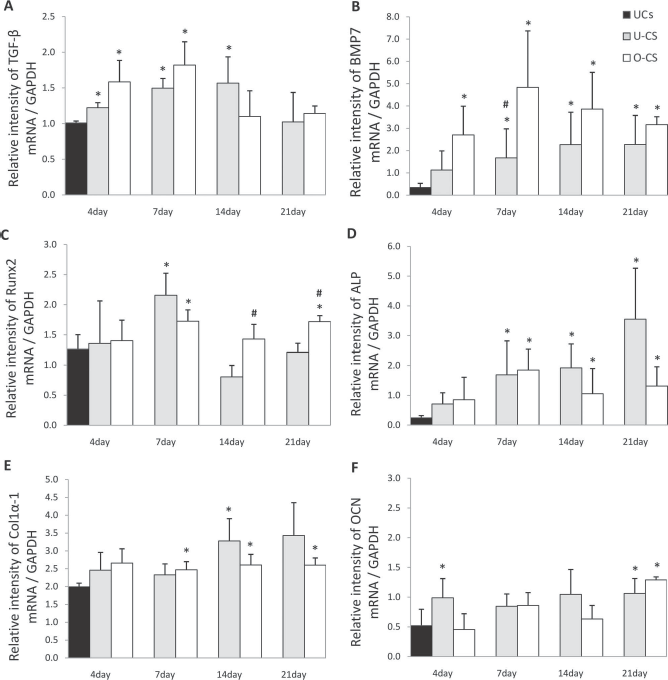
Osteogenic differentiation-associated gene expression profiles of undifferentiated cells (UCs), undifferentiated cell sheets (U-CS), and osteogenic differentiated cell sheets (O-CS) at 4, 7, 14, and 21 days. The effect of different media compositions on mRNA expression relative to GAPDH was assessed using semiquantitative reverse-transcription polymerase chain reaction (RT-PCR) for (A) transforming growth factor-β (TGF-β), (B) bone morphogenetic protein 7 (BMP7), (C) runt-related transcription factor 2 (RUNX2), (D) alkaline phosphatase (ALP), (E) collagen 1α-1 (COL1α-1), and (F) osteocalcin (OCN) mRNA expression at 4, 7, 14, and 21 days. Each bar represents the mean ± SD. $Statistically significant difference compared to UCs at 4 days (p < 0.05); #statistically significant difference between U-CS and O-CS at each day, respectively (p < 0.05).
ALP Activity
ALP activity was significantly higher in U-CS and O-CS at 7 and 14 days of culture compared to that in UCs at 4 days of culture (p < 0.05) (Fig. 3). ALP activity in U-CS at 14 days of culture was significantly higher than that in O-CS (p < 0.05).

Alkaline phosphatase (ALP) activity of undifferentiated cells (UCs), undifferentiated cell sheets (U-CS), and osteogenic differentiated cell sheets (O-CS) at 4, 7, 14, and 21 days. ALP activity of U-CS increased gradually from 4 days and peaked at 14 days. ALP activity of O-CS peaked at 7 days and decreased after 7 days. Each bar represents the mean ± SD. *Statistically significant difference between two groups (p < 0.05); #statistically significant difference between U-CS and O-CS at day 14 (p < 0.05).
ARS Staining and Calcium Contents
No ARS reactivity was observed at 21 days of culture in either U-CS or O-CS (Fig. 4B, C). Mineral deposition in both sheets was not detected until 14 days of culture (Fig. 4A). Small amounts of calcium were detected in O-CS and U-CS (0.23 ± 0.02 mg/dl and 0.74 ± 0.71 mg/dl, respectively), but not in UCs at 21 days of culture.

The calcium contents and Alizarin red S (ARS) staining at 21 days. Scale bars: 50 μm. (A) Calcium contents of undifferentiated cell sheets (U-CS) and osteogenic cell sheets (O-CS) increased marginally. (B) ARS stained negatively at day 21 in U-CS and (C) O-CS.
Discussion
The addition of 50 μg/ml of Asc to the culture medium stimulated the formation of cell sheets between 2 and 4 days of culture. Previous studies have shown that Asc supplementation to culture medium stimulated cell sheets to form multiple layers and secrete abundant ECM (9,38,40). Culturing cells in tissue culture polystyrene dishes preserves the structure of the ECM even after detaching cells using a cell scraper (1,9,12,19,22). Similar to some previous reports, we observed that the edges of the cell sheets started to fold away from the cell culture dish after 7–10 days of culture (9). Further, the cells in O-CS started to aggregate at 10–12 days, and only a partial matrix was observed by 21 days of culture. The cellular aggregation that occurs during osteogenic differentiation in vitro (10,37) could have contributed to the partial loss of the matrix observed in the present study. Therefore, the O-CS cannot be used for therapeutic application after 10–12 days of culture.
MSCs express many growth factors, including TGF-β (35). TGF-β1 promotes the proliferation of MSCs and regulates the formation and degradation of the ECM (5,28). TGF-β isoforms are stored in the ECM and interact with ECM proteins (6). The significant increase in TGF-β expression observed in both U-CS and O-CS at 4 days of culture compared to UCs was probably due to higher TGF-β secretion into the ECM from the MSCs.
BMP7 expression in O-CS was upregulated after 4 days of culture, demonstrating that osteogenic medium facilitates osteogenic differentiation. In addition, the upregulation of BMP7 in U-CS at 7 days of culture could be attributed to TGF-β-driven osteogenic differentiation. The coordination of RUNX2 and BMP/TGF-β signaling is critical for the early stages of bone formation. TGF-β and BMP7 signaling induce osteoblast differentiation through several pathways such as SMAD, MAPK, and Wnt, all of which converge at the RUNX2 gene to facilitate mesenchymal precursor cell differentiation (4,6,15,34). In the present study, RUNX2 expression in both U-CS and O-CS was upregulated at day 7, followed by upregulation of TGF-β and BMP7. RUNX2 regulates the commitment of mesenchymal cells to an osteoblast lineage and induces the expression of genes encoding bone matrix proteins such as COL1α-1 and ALP (15–17). The ALP mRNA expression and activity at the protein level as well as COL1α-1 expression were upregulated from 7 days of culture in O-CS.
The level of ALP activity increases during ECM maturation; the initiation of mineralization correlates with the peak in ALP activity, and mineralization correlates with a decrease in ALP activity (2,3,18,20,31). The peaks of ALP activity in O-CS and U-CS were observed at 7 and 14 days of culture, respectively. This suggests that the osteogenic ECM maturation of O-CS was earlier than that of U-CS. MSCs cultured in osteogenic medium without Dex have been reported to exhibit higher ALP activity and to mineralize, although after a delay of 1 week relative to Dex-treated cultures (11). Dex in osteogenic medium could accelerate RUNX2-related transcriptional changes by facilitating DNA binding of RUNX2 (15). In the present study, the use of O-CS medium resulted in faster differentiation compared to the use of U-CS medium.
After ECM maturation, mineralization occurs by deposition of calcium and phosphate (27). However, in the present study, the small amount of mineral deposition in both sheets was not sufficient to be detected by ARS staining. RUNX2 inhibits the expression of OCN during the maturation phase of osteoblasts (16). O-CS and U-CS, which progressed later than O-CS, might differentiate until the maturation phase, so that mineralization of neither sheet had yet occurred.
In the present study, ALP activity and the mRNA expression of RUNX2, ALP, and COL1α-1 indicated that U-CS and O-CS show osteogenic potential in the stage of ECM maturation. Considering the observed peak in ALP activity and morphological changes, the optimal times for application of O-CS and U-CS are between 7 and 10 days and after 14 days of culture, respectively.
Footnotes
Acknowledgments
This work was supported by the Research Institute for Veterinary Science, Seoul National University, and the National Research Foundation of Korea (NRF-2013R1A1A2004506). The authors declare no conflicts of interest.
