Abstract
We have developed an autologous transplantation method using adipose tissue-derived multi-lineage progenitor cells (ADMPCs) as a method of periodontal tissue regeneration that can be adapted to severe periodontal disease. Our previous clinical study confirmed the safety of autologous transplantation of ADMPCs and demonstrated its usefulness in the treatment of severe periodontal disease. However, in the same clinical study, we found that the fibrin gel used as the scaffold material might have caused gingival recession and impaired tissue regeneration in some patients. Carbonate apatite has a high space-making capacity and has been approved in Japan for periodontal tissue regeneration. In this study, we selected carbonate apatite as a candidate scaffold material for ADMPCs and conducted an in vitro examination of its effect on the cellular function of ADMPCs. We further performed autologous ADMPC transplantation with carbonate apatite as the scaffold material in a model of one-wall bone defects in beagles and then analyzed the effect on periodontal tissue regeneration. The findings showed that carbonate apatite did not affect the cell morphology of ADMPCs and that it promoted proliferation. Moreover, no effect on secretor factor transcription was found. The results of the in vivo analysis confirmed the space-making capacity of carbonate apatite, and the acquisition of significant new attachment was observed in the group involving ADMPC transplantation with carbonate apatite compared with the group involving carbonate apatite application alone. Our results demonstrate the usefulness of carbonate apatite as a scaffold material for ADMPC transplantation.
Keywords
Introduction
Periodontal disease is caused by a bacterial biofilm formed on the root surface of the tooth, which leads to chronic inflammatory destruction of periodontal tissue consisting of alveolar bone, cementum, gingiva, and the periodontal ligament and is one of the leading causes of tooth loss in adults1,2. Although it is possible to control the progression of periodontal disease by removing the causative bacterial biofilm and calculus along with necrotic cementum as an initial treatment for periodontal disease, this conventional therapy alone does not lead to the regeneration of periodontal tissue destroyed by periodontal disease 3 . Therefore, after removing the cause, surgical periodontal regeneration therapies, such as the use of bone grafts and barrier membranes and localized administration of enamel matrix derivatives, platelet-derived growth factor, and basic fibroblast growth factor, are performed, which have resulted in consistent clinical effectiveness4–7. These therapies regenerate periodontal tissue by activating the inherent self-regeneration ability of periodontal tissue stem cells, such as those in the periodontal ligament. However, the number of stem cells in the body decreases with age, and in periodontal tissue, the ability of endogenous stem cells to proliferate and differentiate declines 8 . Furthermore, in periodontal tissue with severe periodontitis, the number of endogenous stem cells surrounding the affected tooth decreases with tissue destruction, and successful outcomes cannot be expected with this method of periodontal tissue regeneration. Therefore, a new treatment therapy involving the transplantation of mesenchymal stem cells collected from other tissues into the area of the periodontal tissue defect in severe periodontitis has been investigated9,10.
The efficacy of transplantation therapy using mesenchymal stem cells derived from bone marrow11,12, the dental pulp 13 , and the periodontal ligament14,15 has been reported. We conducted a study focusing on adipose tissue that can be harvested safely and effectively to supply stem cells. To isolate adipose tissue-derived stem cells (ADSCs), the method of Zuk et al. 16 is generally used; however, Okura et al. 17 defined the cells collected with ethylenediamine tetraacetate (EDTA) processing from the cells isolated using the same method as adipose tissue-derived multi-lineage progenitor cells (ADMPCs). Compared with ADSCs, ADMPCs exhibit a greater ability to differentiate into adipocytes, chondrocytes, and osteoblasts 18 , and therefore can be perceived as mesenchymal stem cells with higher purity. The periodontal tissue regeneration effect of autologous ADMPC transplantation has already been reported in nonclinical research using an experimental model of periodontal disease in beagle dogs 19 . We performed a clinical study to evaluate the safety and efficacy of autologous ADMPC transplantation in patients with severe periodontitis 20 . No serious adverse events were observed in any of the patients who underwent autologous ADMPC transplantation. Decreases in the periodontal pocket depth, acquisition of the clinical attachment level, and regeneration of the alveolar bone on X-ray image analysis were observed. The periodontal tissue regeneration effect of autologous ADMPC transplantation was confirmed even in cases where satisfactory effects could not be expected with conventional periodontal tissue regeneration therapy, such as those with one-wall and four-wall bone defects. Although this finding strongly suggests that autologous ADMPC transplantation might be effective as a method of therapeutic periodontal tissue regeneration for severe periodontal disease, in some severe cases, gingival recession and tissue collapse have occurred, and adequate space for tissue regeneration could not be ensured. In other words, lack of space for tissue regeneration may have limited the regenerative effects of ADMPCs on periodontal tissue regeneration. Therefore, we believe that for stable therapeutic effects in patients with severe periodontitis, concurrent use of a scaffold material with good space-making ability is needed for ADMPC transplantation. Moreover, to promote translational research that takes into consideration the practical application of this treatment method, candidates should be selected among scaffold materials that are already clinically applied in dentistry.
The periodontal tissue regeneration effect of placing a simple substance in the periodontal tissue has been reported in a previous study 21 . In this study, we selected carbonate apatite (CaAp) as the candidate scaffold material for ADMPC transplantation because its application in periodontal tissue has been approved in Japan. We conducted an in vitro examination of the effects of CaAp on the cellular functioning of ADMPCs and also performed an in vivo examination of the effects of ADMPC–CaAp transplantation on periodontal tissue regeneration using an experimental model of periodontitis in beagle dogs.
Materials and Methods
Cell Culture
Human ADMPCs supplied by Adipo Medical Technology Inc. (Osaka, Japan) were used for the experiment. The ADMPCs were maintained with expansion-medium (exp-med) consisting of Dulbecco’s modified Eagle medium–low glucose (DMEM-LG: Gibco, Grand Island, NY, USA), 40% MCDB-201 (Sigma–Aldrich, St. Louis, MO, USA), 10 µg/mL epidermal growth factor (Peprotech, Hamburg, Germany), 1 nM dexamethasone (Sigma–Aldrich), 100 nM ascorbic acid (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan), 5% fetal bovine serum (Nichirei Biosciences Inc., Tokyo, Japan), and 60 µg/mL kanamycin (FUJIFILM Wako Pure Chemical Corporation). The cells were cultured at 37 °C with 5% CO2 and 100% humidity on a 100-mm fibronectin-coated dish (Corning, Corning, NY, USA). Subculture was performed using 0.05% trypsin-EDTA (Thermo Fisher Scientific, Waltham, MA, USA), and ADMPCs were used for the experiment at passages 5–15. For the CaAp culture disk (CAD; GC, Tokyo, Japan) used in this study, ADMPCs were seeded and cultured on a 1-mm-thick and 4-mm-diameter CAD placed in a 96-well cell culture plate (Corning) or a 1-mm-thick and 11-mm-diameter CAD placed in a 48-well cell culture plate (Corning). Before the cells were seeded, the CADs were immersed for 7 days in exp-med.
Cell Morphology Analysis
The CAD was placed in the well base of a 48-well cell culture plate, and ADMPCs were seeded at a density of 2.5 × 104 cells/well and cultured in exp-med for 24 h. In the same manner, ADMPCs were directly seeded into a 48-well cell culture plate for use as a control group. The cells were fixed in phosphate-buffered saline (PBS) containing 4% paraformaldehyde (PFA; FUJIFILM Wako Pure Chemical Corporation), followed by treatment with PBS containing 0.25% Triton X-100 (MP Biomedicals, Tokyo, Japan). Thereafter, blocking was performed for 5 min in PBS containing 1% bovine serum albumin (Sigma–Aldrich). After being washed three times with PBS, the cells were left to react for 30 min at 4°C with PBS containing Hoechst33342 solution (1:500; Dojindo, Kumamoto, Japan) and rhodamine-phalloidin solution (1:100; Thermo Fisher Scientific). Subsequently, the cells were washed three times in PBS and examined using a Nikon ECLIPSE Ti-U fluorescence microscope (Nikon, Tokyo, Japan). After being cultured on the CAD and washed twice with PBS, the cells were fixed for 30 min at 4°C using 0.1 M phosphate buffer (PB: pH 7.4, FUJIFILM Wako Pure Chemical Corporation) containing 2% PFA (Nisshin EM, Tokyo, Japan) and 2% glutaraldehyde (EM Science, Hatfield, PA, USA). Next, they were changed to PB containing 2% glutaraldehyde and fixed overnight at 4°C. After being washed three times with PBS, the cells were fixed for 1 h at 4°C using PB containing 2% osmium tetroxide. This step was followed by dehydration of the samples three times, including once in 50% ethanol for 5 min at 4°C, once in 70% ethanol for 5 min at 4°C, once in 90% ethanol for 5 min at room temperature, and once in dehydrated ethanol for 5 min at room temperature. After dehydration, the samples were immersed in Quetol-812 epoxy resin (Nisshin EM) and polymerized for 48 h at 60°C to create resin blocks. Thin slices of 70 nm were made from the resin blocks using an ultramicrotome (Ultracut UCT, Leica, Vienna, Austria) and subjected to primary staining at room temperature for 15 min with 5% uranyl acetate (VI) (Tokai Denshi Microscopy analysis, Aichi, Japan). The sections were washed with distilled water, followed by secondary staining using a lead staining solution (Sigma–Aldrich) for 3 min at room temperature. The ultrathin-stained slices were observed under a transmission electron microscope (JEM-1400 Plus; JEOL, Tokyo, Japan).
Cell Proliferation Analysis
CADs were placed in the well base of a 96-well cell culture plate, and ADMPCs were seeded at a density of 2.5 × 103 cells/well and cultured for 2 days in exp-med. In the same manner, ADMPCs were directly seeded into a 96-well cell culture plate and used as a control. The viable cell count was evaluated as an indicator of the intracellular adenosine triphosphate (ATP) levels, and using the measurement obtained 6 h after seeding as the reference value, the cell proliferation rate was calculated after 12, 24, 36, and 48 h. The intracellular ATP level was determined with a luminescence assay performed using the CellTiter-Glo® 2.0 Cell Viability Assay Kit (Promega, Madison, WI, USA) and detected using the GloMax 96 Microplate Luminometer w/Dual Injectors (Promega).
Evaluation of Cell Death
CADs were placed in the well base of a 96-well cell culture plate, and ADMPCs were seeded at a density of 2.0 × 104 cells/well and cultured for 3 days in exp-med. In the same manner, ADMPCs were directly seeded onto a 96-well cell culture plate and used as a control. The culture supernatant was collected at 24, 48, and 72 h after seeding, and the number of dead cells was evaluated as an indicator of lactate dehydrogenase (LDH) in the supernatant. LDH was measured with a luminescence assay using an LDH-Glo™ Cytotoxicity Assay Kit (Promega) and detected with a GloMax 96 Microplate Luminometer w/Dual Injectors (Promega).
Reverse Transcription-Quantitative Polymerase Chain Reaction
Total RNA was extracted from the cultured cells using the Maxwell® RSC simplyRNA Cells Kit (Promega). With the extracted and purified total RNA as the template, reverse transcription was performed using a High Capacity RNA-to-cDNA™ Kit (Thermo Fisher Scientific) and cDNA was generated.
Analysis with quantitative polymerase chain reaction (qPCR) was performed using cDNA as the template, and specific reverse transcription-polymerase chain reaction (RT-PCR) primers for each gene were as follows (Takara Bio, Shiga, Japan, and FASMAC, Kanagawa, Japan): insulin-like growth factor binding protein-6 (IGFBP-6) (forward primer, 5′-AACCGCAGAGACCAACAGAG-3′; reverse primer, 5′-TTGGGCACGTAGAGTGTTTG-3′), hepatocyte growth factor (HGF) (forward primer, 5′-GAATTTGGCCATGAATTTGACCTC-3′; reverse primer, 5′-ATCATGGAACTCCAGGGCTGAC-3′), vascular endothelial growth factor (VEGF) A (forward primer, 5′-GAGCCTTGCCTTGCTGCTCTAC-3′; reverse primer, 5′-CACCAGGGTCTCGATTGGATG-3′), and hypoxanthine-guanine phosphoribosyl transferase (HPRT) (forward primer, 5′-GGCAGTATAATCCAAAGATGGTCAA-3′; reverse primer, 5′-GTCAAGGGCATATCCTACAACAAAC-3′). The PCR reaction was performed using the Fast SYBR® Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA) with the Step One Plus Real-time PCR System® (Applied Biosystems). The expression level of each gene was calculated as the absolute value for the specific gene with HPRT as the endogenous control gene.
Measurement of IGFBP-6, HGF, and VEGF Concentrations in the Culture Supernatant
ADMPCs were seeded onto 48-well cell culture plates at a density of 2.5 × 104 cells/well in the presence or absence of CAD, and quantitative analysis was performed using a Human IGFBP-6 ELISA-Kit (RayBiotech, Norcross, GA, USA) for the IGFBP-6 concentration, Quantikine ELISA Human HGF (R&D Systems, Minneapolis, MN, USA) for the HGF concentration, and Quantikine ELISA Human VEGF (R&D Systems) for the VEGF concentration in the culture supernatant.
Canine ADMPC Isolation
All animal experimental protocols were approved by the Institutional Animal Care and Use Committee of the Osaka University Graduate School of Dentistry. Canine ADMPCs were isolated from greater omentum tissue. When harvesting this tissue, an initial intramuscular injection of atropine sulfate (Mitsubishi Tanabe Pharma, Osaka, Japan) was administered at 0.04 mg/kg as premedication, followed by an intramuscular injection of a three-mixture solution to induce anesthesia. This solution included 0.4 mg/kg of butorphanol tartrate (Meiji Seika Pharma, Tokyo, Japan), 30 µg/kg of medetomidine hydrochloride (Nippon Zenyaku Kogyo, Fukushima, Japan), and 0.4 mg/kg of midazolam (Astellas Pharma, Tokyo, Japan). After confirming sedation, 6 to 8 mg/kg of propofol (Maruishi Pharmaceutical, Osaka, Japan) was injected intravenously to induce deep anesthesia, after which tracheal intubation was performed. While maintaining anesthesia using 1% to 5% isoflurane inhalation with an artificial ventilator combined with an intravenous drip infusion of propofol, following laparotomy, the peripheral aspect of the greater omentum was isolated by sequential ligation and dissection. Subsequently, multiple thin pieces of the greater omentum were excised. ADMPCs were isolated as per a previous report 19 , and the cells were cultured in accordance with the human ADMPC culture method.
Preparation of Alveolar Bone Defect and Transplantation of Autologous ADMPCs
We prepared two-wall bone defects in the distal side of the third premolar of beagle dogs aged 21 to 25 months and one-wall bone defects in the proximal side of the first molar of beagle dogs aged 55 to 73 months. We then examined the periodontal tissue regenerative effect of ADMPC–CaAp complex transplantation. Briefly, after inducing general anesthesia using the same method as for adipose tissue harvesting, the mandibular fourth premolars were extracted on both the left and right sides under local anesthesia using lidocaine hydrochloride (xylocaine for dentistry: Dentsply Sirona, Tokyo, Japan). At 12 weeks after the extractions, the operative field was disinfected using cotton balls soaked in 10% povidone–iodine solution (Kenei Pharmaceutical, Osaka, Japan) and 0.025% benzalkonium chloride (Kenei Pharmaceutical). Local anesthesia was induced using 1.8 mL of lidocaine hydrochloride, an incision was made in the gingival sulcus from the third premolar to the first molar, and a mucoperiosteal flap was raised. In the two-wall bone defect model, defects of 3 mm in buccolingual diameter, 4 mm in depth, and 5 mm in mesiodistal diameter were created in the distal part of the lower third premolar. The bone defects created on both the left and right sides were randomly allocated to the experimental and control sides. For the experimental side, ADMPCs and CaAp were transplanted, whereas ADMPCs and fibrin gel were transplanted on the control side (n = 5). The number of transplanted cells was set at 6.25 × 106 per defect for both the experimental and control sides. CaAp with a particle size of 0.3 to 0.6 mm was used. In the one-wall bone defect model, defects were created in a buccolingual direction with a depth of 6 mm and a mesiodistal diameter of 5 mm proximal to the mandibular first molar. Bone defects were created on the left and right sides; on one side, only CaAp was applied, whereas on the other side, the ADMPC–CaAp complex was applied (n = 3). The sham operation was performed in four locations on the left and right sides of two dogs. In the ADMPC–CaAp complex transplantation group, 1 × 107 ADMPCs were transplanted, and granules of 0.3–0.6 mm CaAp were used. After transplantation, the mucoperiosteal flap was repositioned, and simple Gore-Tex sutures® (W.L. Gore and Associates, Newark, DE, USA) were placed. Sham surgery was performed bilaterally on the two beagles in the control group. The sutures were removed 1 week after transplantation. The treatment site in the oral cavity was sprayed with chlorhexidine gluconate disinfectant solution (Maruishi Pharmaceutical) once a day for 2 weeks after transplantation. At 6 weeks after transplantation for the two-wall bone defect model and 12 weeks after transplantation for the one-wall bone defect model, the dogs were euthanized by sectioning the femoral artery and vein under deep anesthesia using propofol, and the mandible was harvested.
Micro-Computed Tomography Analysis
Computed tomography (CT) scans were taken using a three-dimensional (3D) micro X-ray CTR_mCT2 (Rigaku, Tokyo, Japan) and CT Skyscan 1276 (Bruker, Billerica, MI, USA). With the obtained two-dimensional images, 3D images were created using TRI/3D-BON 3D image analysis software (RATOC System Engineering, Tokyo, Japan). Quantitative analysis of the volume of X-ray nontransmissive hard tissue was performed using TRI/3D-BON 3D image analysis software and CT_An 3D image analysis software (Bruker). In each defect model, the notch created at the root surface of the defect base was used as a reference to define the area of the alveolar bone defect as the region of interest. Within the defined region of interest, anything with a CT value equivalent or higher than the CT value of the normal cancellous bone was defined as radiopaque material.
Histological Analysis
The mandible was immersed and fixed using 4% paraformaldehyde PBS (pH 7.4: FUJIFILM Wako Pure Chemical Corporation) at 4°C for 2 weeks. After fixation, the tissue was decalcified at room temperature for 2 months using 10% formic acid—sodium citrate decalcifying solution (900 mL/L distilled water, 100 mL/L formic acid; FUJIFILM Wako Pure Chemical Corporation), 29 g/L citric acid (FUJIFILM Wako Pure Chemical Corporation), and 18 g/L citric acid trisodium dihydrate (FUJIFILM Wako Pure Chemical Corporation). Thereafter, the sample was embedded in paraffin as per the standard method. To create tissue sections, the tissue block was sliced thinly at 150 µm intervals from the lingual side toward the buccal side. Hematoxylin and eosin staining was performed using Mayer’s hematoxylin (FUJIFILM Wako Pure Chemical Corporation) and 1% eosin Y solution (FUJIFILM Wako Pure Chemical Corporation). Azan staining was performed using a mordant (Muto Chemicals, Tokyo, Japan), Mallory azocarmine G solution (Muto Chemicals), 5% phosphotungstic acid aqueous solution (FUJIFILM Wako Pure Chemical Corporation), and Mallory aniline blue–orange G solution (Muto Chemicals). Newly formed fibrous attachment (new attachment) was quantified using tissue sections from the buccolingual central area and located 150 µm lingual or buccal to the central area. New attachment formed on the root surface was quantified as the ratio of the length of newly formed fibrous attachment on the exposed root surface to the length from the notch created in the base of the bone defect to the alveolar crest of the existing bone.
Immunohistochemistry
Similar to the histological analysis, paraffin sections were prepared and deparaffinized, and antigen retrieval was performed by heat treatment with Tris-EDTA buffer. The sections were immersed in 3% H2O2 (FUJIFILM Wako Pure Chemical Corporation) to remove endogenous peroxidase. After the sections were washed with PBS, they were blocked for 1 h at 25°C with PBS containing 1.5% bovine serum albumin (Sigma-Aldrich) (blocking solution). The primary antibody used was the anti-tartrate resistant acid phosphatase rabbit antibody (1:100, Abcam, Cambridge, UK cat; ab185716), and the secondary antibody used was the horseradish peroxidase-labeled anti-rabbit Envision antibody (DAKO, Denmark, Glostrup Kommune cat; K400), with each incubation carried out for 1 h at 25°C. Both the primary and secondary antibodies were diluted in the blocking solution. After the sections were washed with PBS, color was developed using 3,3’ Diaminobenzidine (DAKO), followed by nuclear staining at room temperature with Mayer’s hematoxylin.
Statistical Analysis
The experimental data are presented as mean ± standard deviation. To test for a significant difference, we used Student’s t test, and multiple group comparisons were performed with a one-way analysis of variance. Thereafter, post-hoc analysis was performed using the Tukey–Kramer test and P < 0.05 was considered a significant difference.
Results
Effect of CaAp on the Cellular Morphology of ADMPCs
First, we analyzed the cellular morphology of ADMPCs cultured on CADs. The control group was composed of ADMPCs cultured on cell culture plates. After the cytoskeleton was visualized with actin staining, the ADMPCs cultured on the CADs showed the same fusiform cell shape as the controls (Fig. 1A). In the CAD group, transmission electron microscopic observation revealed no pyknosis, fragmentation, or mitochondrial swelling. The Golgi apparatus was located near the nucleus and several granular endoplasmic reticula were observed; this appearance was similar to that of the control group (Fig. 1B).

Cell morphology of adipose tissue-derived multi-lineage progenitor cells (ADMPCs) cultured on a CAD: (A) actin and nucleus staining images of ADMPCs cultured on a CAD. ADMPCs were cultured on a CAD or tissue culture plates (Control) for 24 h followed by staining of the nucleus and actin with Hoechst33342 and rhodamine-phalloidin solution. (B) ADMPCs were cultured on a CAD or tissue culture plates for 3 days. Images of each organelle were captured by transmission electron microscopy. Black arrows indicate organelles (nucleus, mitochondria, and endoplasmic reticulum). Representative results from three experiments are shown. CAD: carbonate apatite culture disk.
Effect of CaAp on ADMPC Proliferation
After ADMPCs were seeded on the CAD and the cell proliferation rate was measured after 6 to 48 h, the CAD group had a significantly higher cell proliferation rate than the control group at 12 and 24 h after seeding (Fig. 2A). In contrast, no significant difference was observed between the CAD and control groups in terms of the number of dead cells in the 3-day culture as evaluated by measuring the LDH contained in the ADMPC culture supernatant 1 to 3 days after seeding (Fig. 2B).
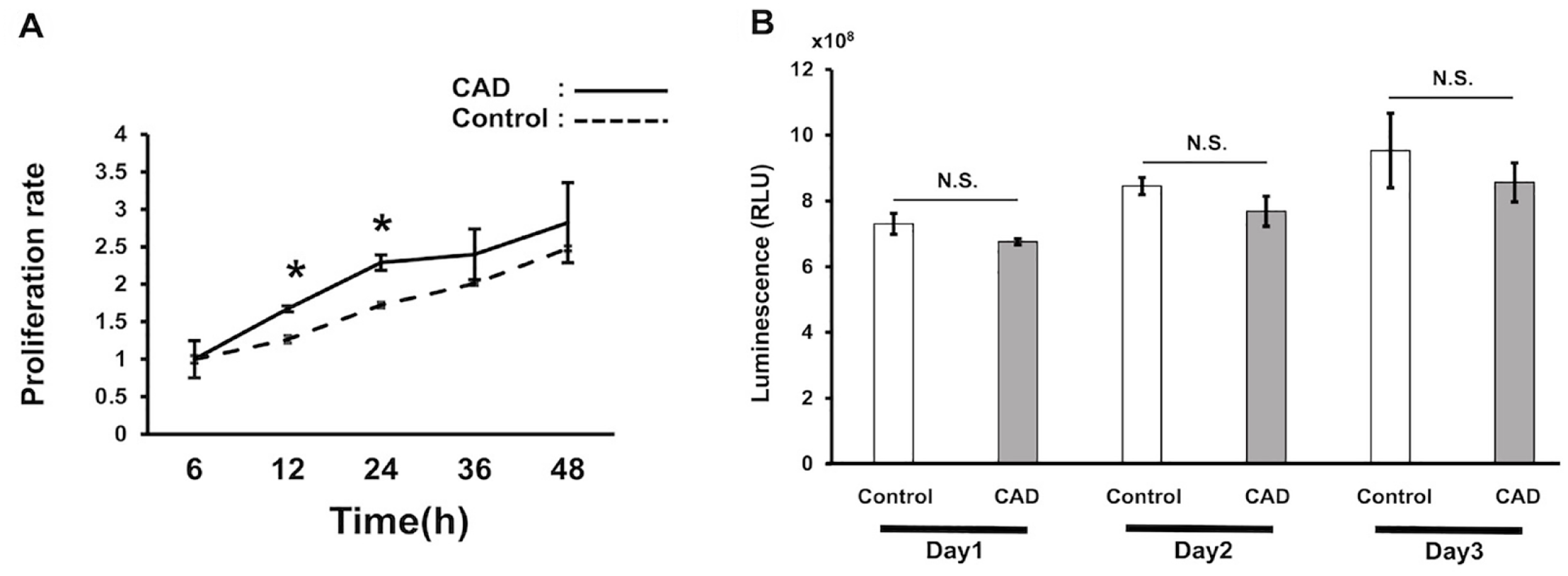
Proliferation and death of adipose tissue-derived multi-lineage progenitor cells (ADMPCs) cultured on a CAD: (A) proliferation of ADMPCs cultured on a CAD. 2.5 × 103 ADMPCs were seeded on a 96-well cell culture plate (Control) or on a 4-mm-diameter CAD placed in a 96-well cell culture plate (CAD) for 48 h and the intracellular ATP level was measured. The proliferation rate was calculated as a ratio to the value at 6 h. (B) Cell death of ADMPCs cultured on a CAD. 2.0 × 104 ADMPCs were seeded on a 96-well cell culture plate (Control) or on a 4-mm-diameter CAD placed in a 96-well cell culture plate (CAD) for 3 days. Lactate dehydrogenase levels in the supernatant at days 1, 2, and 3 were measured. Representative results from at least three experiments are shown. CAD: carbonate apatite culture disk; RLU: relative light unit; NS: not significant. *P < 0.05.
Analysis of Humoral Factor Production of ADMPCs Cultured on CaAp
When the expression of IGFBP-6, HGF, and VEGF was evaluated after the 3-day culture for both the control and CAD groups for all genes, the CAD group exhibited the same level of mRNA expression as the control group (Fig. 3A). Each factor contained in the culture supernatant was evaluated, and the CAD group showed the same level of IGFBP-6 expression as the control group, whereas HGF and VEGF concentrations in the culture supernatant were lower (Fig. 3B).

Humoral factor production from ADMPCs cultured on a CAD: (A) mRNA expression of IGFBP-6, HGF, and VEGF in ADMPCs cultured on a CAD. 2.5 × 104 ADMPCs were seeded on a 48-well cell culture plate (Control) or on an 11-mm-diameter CAD placed in a 96-well cell culture plate (CAD) for 72 h. mRNA expression of IGFBP-6, HGF, and VEGF were evaluated by RT-qPCR. Results were expressed as a ratio to HPRT mRNA expression. (B) IGFBP-6, HGF, and VEGF concentration in the supernatant of ADMPCs cultured on a CAD. 2.5 × 104 ADMPCs were seeded on a 48-well cell culture plate (Control) or on an 11-mm-diameter CAD placed in a 96-well cell culture plate (CAD) for 72 h and the concentration of each cytokine was quantified by enzyme-linked immunosorbent assay. Representative results from at least three experiments are shown. ADMPCs: adipose tissue-derived multi-lineage progenitor cells; CAD: carbonate apatite culture disk; IGFBP-6: insulin-like growth factor-binding protein-6; HGF: hepatocyte growth factor; VEGF: vascular endothelial growth factor; RT-qPCR: reverse transcription-quantitative polymerase chain reaction; HPRT: hypoxanthine-guanine phosphoribosyl transferase; N.S.: not significant. *P < 0.01.
Evaluation of CaAp as a Scaffold Material in Autologous Transplantation of ADMPCs Using a Two-Wall Alveolar Bone Defect Model
To evaluate the effectiveness of CaAp as a scaffold material of ADMPCs in vivo, autologous transplantation was performed on a two-wall bone defect model. On the control side, fibrin gel and ADMPCs were transplanted, and on the test side, CaAp and ADMPCs were transplanted. Six weeks after transplantation, the hard tissue formation in the bone defect was assessed using micro-CT analysis. The results revealed that while the control side had a depression in the bone defect on the buccal side, the test group exhibited radiopacities, including hydroxyapatite granules, filling the defect area (Fig. 4A, B). Subsequently, a quantitative evaluation of the volume of hard tissue in the created bone defects showed that the test sides had significantly higher volumes compared with the control sides (Fig. 4C).

Micro-computed tomography (micro-CT) analysis after ADMPC–CaAp transplantation into two-wall bone defects. Two-wall bone defects (3-mm buccolingual diameter × 4 mm depth × 5 mm mesiodistal diameter) were created on the distal side of the mandibular third premolar. ADMPCs were transplanted with fibrin gel (A) or CaAp (B). The mandibles were harvested 6 weeks after the transplantation, and micro-CT analysis was performed. Representative three-dimensional images are shown. The yellow area shows radiopaque material found within the fabricated bone defects. The volume of radiopaque material within the fabricated bone defects was quantified (C). CT: computed tomography; ADMPC–CaAp: adipose tissue-derived multi-lineage progenitor cell-carbonate apatite. *P < 0.05.
Effect of ADMPC–CaAp Transplantation on the Canine One-Wall Alveolar Bone Defect Model
To examine the effectiveness of ADMPC–CaAp transplantation on severe periodontitis, sham surgery was performed to create one-wall bone defects in beagles, with CaAp application alone and ADMPC–CaAp transplantation. After micro-CT analysis and histological analysis with Azan staining at 12 weeks post-transplantation, periodontal tissue regeneration was evaluated at the test site. The results of the micro-CT analysis revealed that in the sham surgery group, concave X-ray impermeability was observed at the bone defect site and that the height of the proximal end of the bone defect was reduced. In contrast, in the CaAp and ADMPC–CaAp groups, radiopacities were observed in most of the bone defects. Furthermore, no difference was observed in the volume of the radiopacities between the CaAp and ADMPC–CaAp groups (Fig. 5). Histological analysis of the sham surgery group revealed a concave shape in the center of the regenerated alveolar bone as per the micro-CT images (Fig. 6A). However, in the CaAp and ADMPC–CaAp groups, the bone defects were filled with the residual CaAp and new alveolar bone (Fig. 6C, F). Moreover, in both groups, multinucleated TRAP-positive cells were observed on the surface of the CaAp (Supplemental Fig. 1), indicating the gradual progression of absorption by osteoclasts. In addition, new bone formation together with collagen fibers, which were intensely stained by Azan staining, was found around the CaAp (Fig. 6E, H).
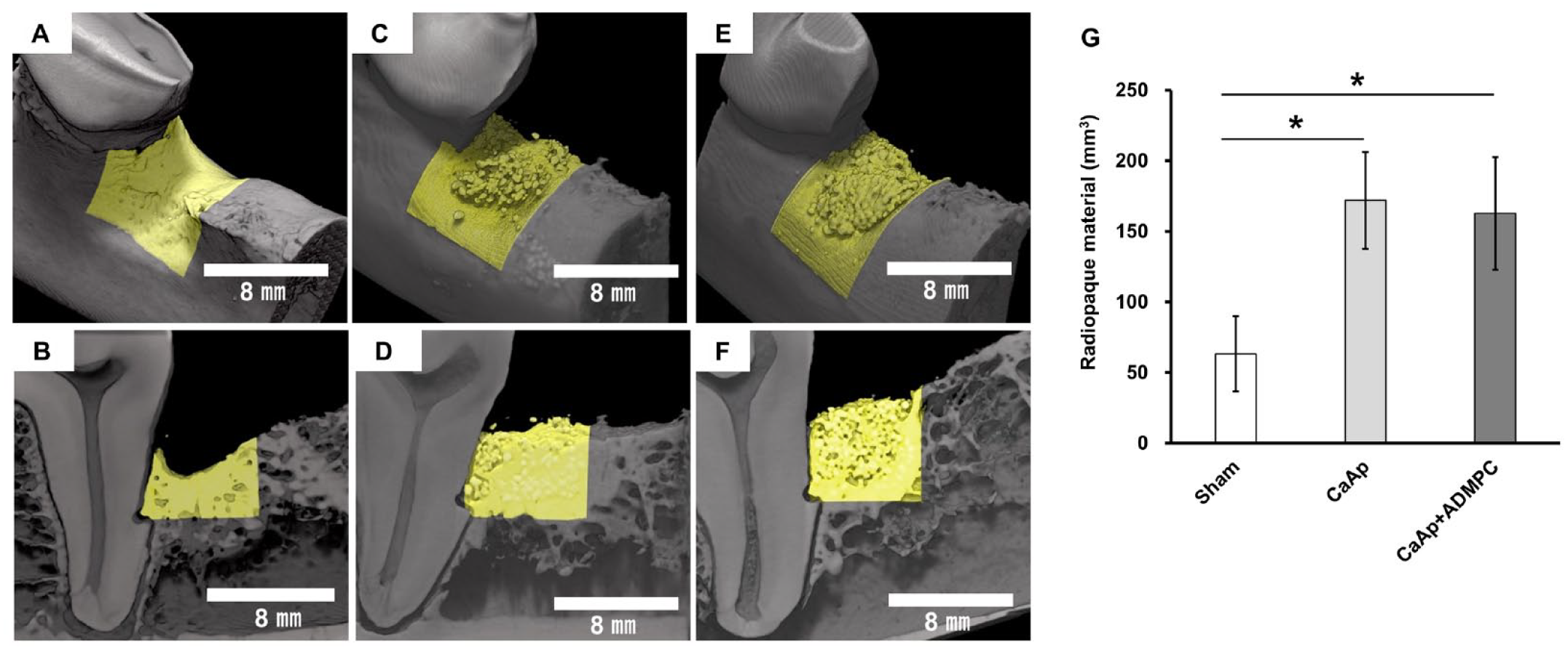
Micro-CT analysis after ADMPC–CaAp transplantation into one-wall bone defects. One-wall bone defects (6 mm depth × 5 mm mesiodistal diameter) were created on the mesial side of the mandibular first molar and a sham operation (A, B), application of CaAp only (C, D), and transplantation of ADMPC–CaAp (E, F) were performed. The mandibles were harvested 12 weeks after the operation, and micro-CT analysis was performed. Representative three-dimensional images (A, C, E) and two-dimensional sagittal images of the buccolingual central area (B, D, F) are shown. The yellow area shows radiopaque material found within the fabricated bone defects. The volume of radiopaque material within the fabricated bone defects was quantified (G). CT: computed tomography; ADMPC–CaAp: adipose tissue–derived multi-lineage progenitor cell-carbonate apatite. *P < 0.01.

Histological analysis after adipose tissue-derived multi-lineage progenitor cell-carbonate apatite (ADMPC–CaAp) transplantation. One-wall bone defects (6 mm depth × 5 mm mesiodistal diameter) were created on the mesial side of the mandibular first molar and a sham operation (A and B), application of CaAp only (C, D, and E) and transplantation of ADMPC–CaAp (F, G, and H) were performed. At 12 weeks after the operation, the mandibles were harvested and sagittal sections were prepared followed by staining with Azan. Representative sagittal images are shown. Higher magnification of A is shown in B, higher magnification of C is shown in D and E, and higher magnification of F is shown in G and H. D: dentin; P: periodontal ligament; NB: newly formed alveolar bone; C: CaAp, arrowhead: newly formed cementum.
More amount of hard tissue (newly formed cementum) showing deep staining associated with the implantation of vertical fibers (Sharpey’s fibers) formed on the root surface was detected in the ADMPC–CaAp compared to CaAp in the same individual (Fig. 6D, G). Quantitative analysis revealed that significant fibrous attachment was formed in the ADMPC–CaAp group compared with the CaAp group (Fig. 7).
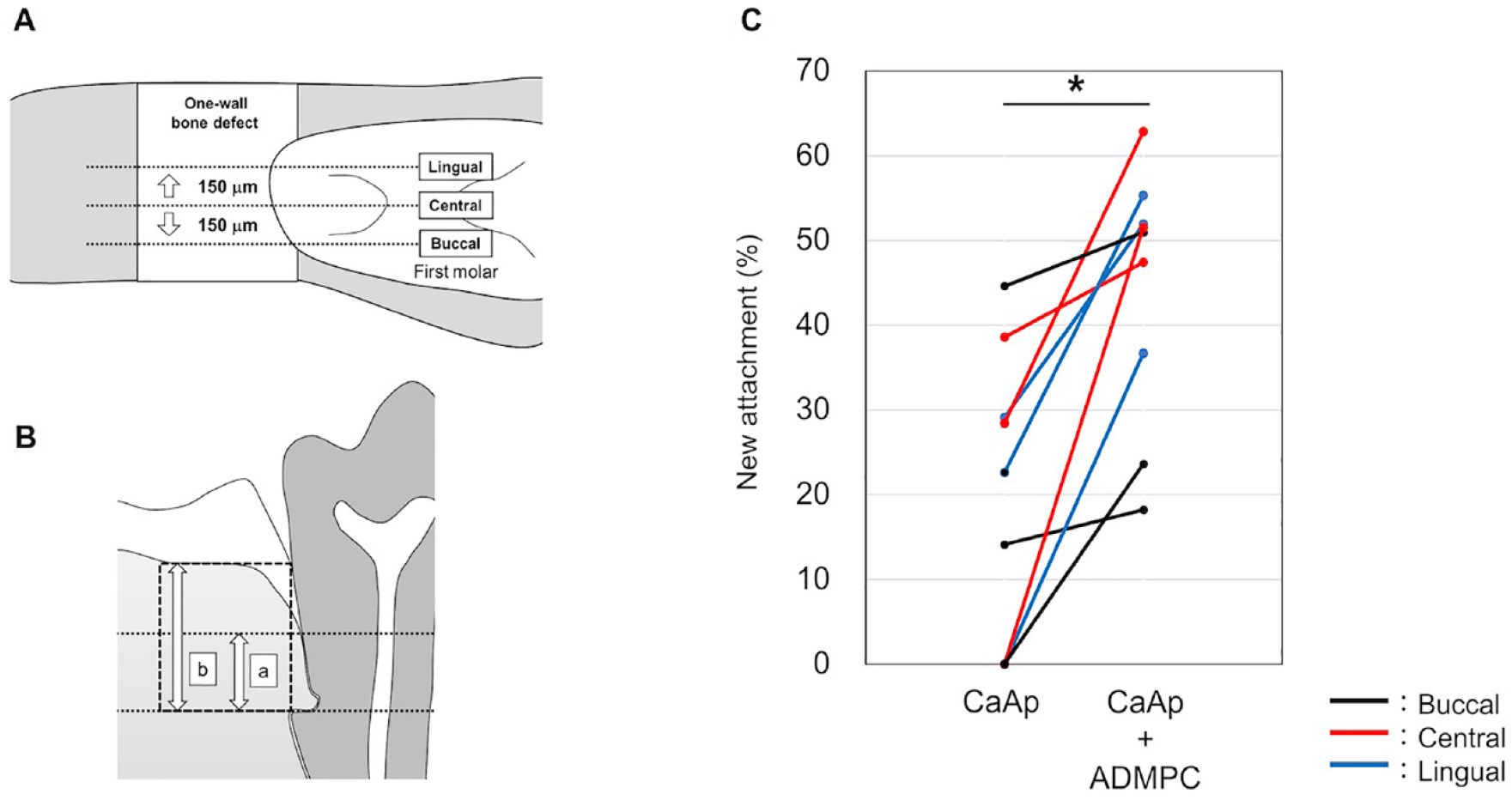
Quantitative analysis of new attachment formation after ADMPC–CaAp transplantation: (A) Azan-stained sagittal tissue sections were prepared from the mandibular bones 12 weeks after transplantation of CaAp or ADMPC–CaAp as shown in Fig. 6. The buccolingual central area and a location 150 µm lingual or buccal to the central area were included in the analysis. (B) Formation of new attachment was quantified as the ratio of the length of newly formed fibrous attachment on the exposed root surface (a) to the length from the notch created in the base of the bone defect to the alveolar crest of the existing bone (b). (C) The ratio of new attachment formed on the root surface was calculated and represented as corresponding values for the CaAp and ADMPC–CaAp sides of the same individual. Three locations (buccal, central, and lingual) from one transplanted site (n = 3) were analyzed. ADMPC–CaAp: adipose tissue–derived multi-lineage progenitor cell-carbonate apatite. *P < 0.01.
Discussion
The ultimate objective of periodontal treatment is to restore the original anatomical shape and function of the periodontal tissue lost due to periodontal disease. To achieve this objective, not only should the bacterial biofilm causing the periodontal disease be removed, but regenerative treatment of the periodontal tissue should also be undertaken. Periodontal tissue regeneration therapies already in clinical use can be effective for mild-to-moderate periodontal tissue defects. However, methods that can be applied to severe periodontal disease have not yet been established. We have undertaken research and developed a periodontal tissue regenerative treatment involving autologous ADMPC transplantation19,20. In this study, we aimed to advance translational research toward clinical applications and elucidate the effect of the concurrent use of CaAp on autologous ADMPC transplantation both in vitro and in vivo.
The effect of CaAp application alone on periodontal tissue regeneration in a model of three-wall bone defects in beagles has been reported 21 . In this study, we transplanted ADMPCs into two-wall bone defects using either fibrin gel or CaAp as the scaffold material and found that CaAp could maintain the tissue regeneration space. Even in one-wall bone defects, no concavity was observed in the gingival–periosteal flap defect site based on CT and histological analysis results after implantation, confirming the space-making ability of CaAp in severe periodontal tissue defects. Osteoclasts were observed around the CaAp; therefore, the transition to newly formed alveolar bone is thought to progress gradually.
The scaffold materials used in cell transplantation therapy should exhibit the following properties: no cytotoxicity toward transplanted cells; no impedance of cellular functions, such as adhesion, proliferation, and differentiation of transplanted cells; ability to create complexes for transplantation with a sufficient number of cells; and ability to be appropriately metabolized in the body after transplantation while maintaining mechanical properties as a scaffold material22,23. In this study, in vitro analysis demonstrated no difference in the cytoskeleton and anatomical morphology of the nuclei, mitochondria, and granular endoplasmic reticula between ADMPCs cultured on CaAp and the normal culture. In addition, ADMPCs on the CADs proliferated and humoral factors were produced. These results suggest that when ADMPCs are transplanted with CaAp into sites with a periodontal tissue defect, CaAp serves as a scaffold material to ensure space regeneration and provides an environment where ADMPCs can exhibit their cellular functions.
One molecular mechanism in autologous stem cell transplantation therapy is the repair effect, in which the transplanted stem cells contribute to tissue regeneration by directly differentiating into various cells that constitute the tissue. In a study in which bone marrow-derived mesenchymal stem cells labeled with green fluorescent protein were autologously transplanted into periodontal tissue defects in beagle dogs, it was demonstrated that at least part of the regenerated tissue originated from the transplanted cells 24 . In our previous study, we showed that ADMPCs have the ability to differentiate into hard tissue-forming cells and periodontal ligament cells 19 . Therefore, it is conceivable that in autologous transplantation of ADMPCs, tissue regeneration may be induced by the ADMPCs differentiating into periodontal tissue-constituting cells. However, the importance of the trophic effect, in which the transplanted stem cells secrete cytokines (growth factors), extracellular matrix, and even exosomes, thereby activating tissue stem cells and progenitor cells present around the transplantation site, has been emphasized in recent years25,26. As part of the mechanism underlying periodontal tissue regeneration via autologous ADMPC transplantation, we found that humoral factors secreted by ADMPCs activate the differentiation of periodontal ligament cells into cells that form hard tissue and that this molecular mechanism involves IGFBP-6 27 . In this study, we found that ADMPCs cultured on CADs produced levels of IGFBP-6 similar to those of a normal culture. This finding suggests that ADMPCs transplanted with CaAp can maintain their ability to regenerate periodontal tissue via a trophic effect. Although no difference was observed in the gene expression of HGF and VEGF, protein expression in the culture supernatant was found to decrease by approximately 30% to 50% for both molecules in the CAD culture compared with the normal culture. Because CaAp can absorb some proteins 28 , some cytokines produced by ADMPCs were possibly trapped in the CAD, thus decreasing the culture supernatant concentration. Humoral factors that secrete stem cells have been reported to contribute to stem cell proliferation by acting on the autocrine system 29 ; therefore, the part of the mechanism that enhanced the proliferation of ADMPCs cultured on the CAD might have consisted of ADMPC-derived humoral factors that remained on the CAD surface and acted on ADMPCs efficiently.
The mechanism of periodontal tissue regeneration using ADMPCs combined with CaAp could be further investigated based on the results of this study. ADMPCs transplanted in the periodontal tissue defect site together with CaAp tend to proliferate on the CaAp. By secreting humoral factors, a trophic effect that activates stem cells near the defect is triggered. Together with the osteoconduction capacity and space-making ability of CaAp, the tissue regenerative effect of ADMPCs could have induced the ideal regeneration of periodontal tissue. Examination of the optimal signaling factors used concurrently with these two materials is an important issue for future research. If the ideal combination of cells, scaffold materials, and signaling factors is elucidated, it could lead to the development of periodontal tissue regenerative treatment methods that could be applied to severe periodontal disease that is currently difficult to treat.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231198296 – Supplemental material for Periodontal Tissue Regeneration by Transplantation of Autologous Adipose Tissue-Derived Multi-Lineage Progenitor Cells With Carbonate Apatite
Supplemental material, sj-docx-1-cll-10.1177_09636897231198296 for Periodontal Tissue Regeneration by Transplantation of Autologous Adipose Tissue-Derived Multi-Lineage Progenitor Cells With Carbonate Apatite by Masahide Takedachi, Kohsuke Kawasaki, Keigo Sawada, Kazuma Sakura, Mari Murata, Junpei Shimomura, Kazuma Kawakami, Chiaki Morimoto, Koji Miki, Noboru Takeshita, Tomoaki Iwayama, Hanayuki Okura, Akifumi Matsuyama, Masahiro Saito, Masahiro Kitamura and Shinya Murakami in Cell Transplantation
Footnotes
Acknowledgements
Author Contributions
This study concept was designed by M.T., H.O., A.M., M.S., and S.M. Experiments were performed by M.T., Ko.K., Ke.S., Ka.S., M.M., J.S., Ka.K., C.M., K.M., N.T., and M.K. Data analysis and interpretation were performed by M.T., Ko.K., T.I., H.O., A.M., M.S., and S.M. The manuscript was written by M.T., Ko.K., and S.M. All authors have read and approved the manuscript.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Ethical Approval
The animal experiments performed in this study were approved by the Institutional Animal Care and Use Committee of the Osaka University Graduate School of Dentistry (approval number: 30-002-01).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Human ADMPCs provided by AMT were isolated from adipose tissue collected with informed consent at AMT.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.O. is a director, and A.M. is a Scientific Advisor of Adipo Medical Technology Inc. S.M. receives research funding from Adipo Medical Technology Inc. The remaining authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by AMED (grant no. JP21bk0104105) and Grants-in-Aids for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (grant no. 21H03118).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
