Abstract
In vitro expanded beating cardiac myocytes derived from induced pluripotent stem cells (iPSC-CMs) are a promising source of therapy for cardiac regeneration. Meanwhile, the cell sheet method has been shown to potentially maximize survival, functionality, and integration of the transplanted cells into the heart. It is thus hypothesized that transplanted iPSC-CMs in a cell sheet manner may contribute to functional recovery via direct mechanical effects on the myocardial infarction (MI) heart. F344/NJcl-rnu/rnu rats were left coronary artery ligated (n = 30), followed by transplantation of Dsred-labeled iPSC-CM cell sheets of murine origin over the infarct heart surface. Effects of the treatment were assessed, including in vivo molecular/cellular evaluations using a synchrotron radiation scattering technique. Ejection fraction and activation recovery interval were significantly greater from day 3 onward after iPSC-CM transplantation compared to those after sham operation. A number of transplanted iPSC-CMs were present on the heart surface expressing cardiac myosin or connexin 43 over 2 weeks, assessed by immunoconfocal microscopy, while mitochondria in the transplanted iPSC-CMs gradually showed mature structure as assessed by electron microscopy. Of note, X-ray diffraction identified 1,0 and 1,1 equatorial reflections attributable to myosin and actin–myosin lattice planes typical of organized cardiac muscle fibers within the transplanted cell sheets at 4 weeks, suggesting cyclic systolic myosin mass transfer to actin filaments in the transplanted iPSC-CMs. Transplantation of iPSC-CM cell sheets into the heart yielded functional and electrical recovery with cyclic contraction of transplanted cells in the rat MI heart, indicating that this strategy may be a promising cardiac muscle replacement therapy.
Introduction
To increase the number of functional cardiomyocytes in the heart is a goal of regenerative therapy for advanced cardiac failure (14). It has been shown that induced pluripotent stem cells (iPSCs) differentiate into functional cardiomyocytes in vitro by specific culture regimens, suggesting that replacement of damaged cardiac tissue might be achieved by transplantation of iPSC-derived cardiomyocytes (iPSC-CMs) into the damaged area using an appropriate cell delivery method. Further, it has been shown that the cell sheet method, in which a scaffold-free sheet-shaped cultured cell cluster is placed on the surface of the heart, delivers a large number of the cells, while preserving the functionality of the cells and the myocardium, indicating that the cell sheet method may be an ideal delivery method of iPSC-CMs to replace the damaged cardiac area (8,9,15). In fact, we reported that transplantation of iPSC-CMs into the heart by the cell sheet method improves functional performance of the infarcted heart in pigs (5). However, in that study (5), synchronous contraction of the transplanted iPSC-CMs as “cardiac myocytes” that express the regular, cyclic actin–myosin cross-bridge motion, which is the aim of this treatment, was not demonstrated due to the limitations of current image analysis methods in vivo.
Shiba et al. reported that the fluorescent signal of calcium sensor, GCaMP3, which has been genetically encoded in the cells prior to transplantation into the heart, was useful for visualizing spontaneous contraction of the transplanted cells in the heart in vivo (16). However, the calcium sensor signal does not necessarily correlate with normal cyclic actin–myosin cross-bridge motion. On the other hand, third-generation synchrotron radiation (SPring-8, Hyogo, Japan) has been utilized to quantify actin–myosin cross-bridge dynamics in cardiac fibers of localized regions in vivo (12,17). We herein hypothesized that transplanted iPSC-CMs in a cell sheet manner may contribute to functional recovery via direct mechanical effects on myocardial infarction (MI) heart. We therefore explored functional and electrical integration of the transplanted iPSC-CMs in the acute MI rat heart using the latest imaging modality utilizing synchrotron radiation from a third-generation facility.
Materials and Methods
Studies were performed with the approval of the ethics committee of Osaka University Graduate School of Medicine and the Animal Experiment Review Committee of the Japan Synchrotron Radiation Research Institute. All animals used in this study received care in compliance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health Publication No. 85-23, revised 1996).
Cell Culture and Cell Sheet Generation
Germline-competent mouse iPSC line 256H18 was established by introducing only Oct3/4, Sox2, and Klf4 (without c-Myc), constitutively expressing red fluorescent protein (Dsred) (generously contributed by Professor S. Yamanaka, Kyoto University, Japan) (11). Maintenance of the 256H18 iPSCs and induction of cardiomyogenic differentiation was performed following the protocol established in Professor Yamanaka's and our laboratories, respectively (7,23). Briefly, the iPSCs were maintained on feeder layers of mitomycin C-treated mouse embryonic fibroblasts (Chemicon, Billerica, MA, USA). Embryoid bodies (EBs) were then generated under the presence of 6-bromoindirubin-3'-oxime (Calbiochem, Darmstadt, Germany) (23).
The EBs were plated on 12-well temperature-responsive culture dishes (CellSeed, Tokyo, Japan) at 37°C with the EB number adjusted to 20 per well; thereafter the EBs were differentiated in the serum-free medium with insulin–transferin–selenium-X (Invitrogen, Carlsbad, CA, USA). Subsequently, the dishes were removed to a refrigerator set at 20°C, while scaffold-free iPSC-CM cell sheets detached spontaneously from the dish surfaces (23). Cardiac troponin T positivity in this preparation assessed by immunohistolabeling was consistently 70-80% (7,23).
Generation of Acute Myocardial Infarction (MI) Model and Cell Sheet Transplantation
Female F344/NJcl-rnu/rnu rats 6 weeks of age (Crea, Tokyo, Japan) were anesthetized by inhalation of isoflurane (1.5%; Mylan Inc., Pittsburgh, PA, USA) and endotracheally intubated for mechanical ventilation. The left coronary artery was then permanently occluded under thoracotomy (3). Two weeks after the ligation, the cell sheets generated by iPSC-CMs or mitomycin C-treated mouse embryonic fibroblasts were simply placed on the surface of the left ventricle (LV, n=6 each) (6). Bupivacaine (1% in saline, 250 μl) was subcutaneously injected near the incision line to minimize the postoperative pain, and the rats were then recovered in a temperature-controlled individual cage.
Transthoracic Echocardiography Analysis
Transthoracic echocardiography was performed using a system equipped with a 12-MHz transducer and SONO S5500 (Agilent Technologies, Palo Alto, CA, USA) under isoflurane inhalation (1%). Diastolic/systolic dimensions (Dd/Ds) and ejection fraction (EF) of the LV were measured (n=6 each) (3).
Telemetry Study
A telemetry transmitter with two electrodes (Data Sciences International, St. Paul, MN, USA) was implanted prior to the treatment (day 14 post-MI) under inhalation of 1.5% isoflurane (n=4 each) (3). The electrocardiogram was then continuously monitored over 7 days. Ventricular premature contractions (VPCs) were detected, and the frequency of the VPCs was expressed as the number of VPCs per day divided by the daily cumulative heartbeats.
Electrical Potential Mapping Analysis
An electrical potential mapping study was performed under the repeated left thoracotomies in the same animal at days 2, 3, 4, 7, and 14 (n=4 each group). There was no repeated procedure-related morbidity or mortality. A 64 electrical potential mapping system (Alpha MED Scientific, Osaka, Japan) was directly placed on the surface of the LV via the thoracotomy under general anesthesia and endotracheal intubation with mechanical ventilation as above. The electrical potential was recorded as the calculated activation recovery interval (ARI) in the same animals at the indicated time points (n=4 each group).
Synchrotron Small-Angle Scattering Study
The fundamentals of synchrotron small-angle scattering techniques for the investigation of cross-bridge dynamics in the intact heart are presented in detail elsewhere (17). In brief, total thoracotomy was performed under general anesthesia and endotracheal intubation with mechanical ventilation as above at the synchrotron radiation facility SPring-8 (n = 4 in iPSC-CM-treated rats and in MI-only rats) as described in detail elsewhere (13). Cardiac catheterization was performed to allow continuous LV pressure–volume recordings simultaneous with all SAXS and arterial pressure recordings. Pressure volumetry was used to establish the timing of the cardiac cycle in all treatment periods and to permit assessment of actin–myosin contributions to global LV function. Heart rate (HR) was determined from the interval between end diastolic (ED) events in the pressure–volume loops. Hemodynamic data were recorded using CHART (v5.5.6; ADInstruments, Dunedin, New Zealand) at a sampling rate of 1,000/s.
A collimated quasimonochromatic beam with 0.08 nm wavelength (15 keV), dimensions 0.2 × 0.1 mm (horizontal × vertical), and beam flux ~1012 photons/s (ring current 90-100 mA) was focused on the surface myocardium at an oblique tangent (rat ~3 m from the detector). SAXS sequences (12 bit, 144 × 150 pixels) each lasting <2.1 s were collected at a sampling interval of 15 ms with the aid of an image intensifier (V5445P; Hamamatsu Photonics, Hamamatsu, Japan) and a fast charge-coupled device camera (C4880-80-24A; Hamamatsu Photonics). Patterns were then digitally recorded using HiPic32 software (v5.1.0; Hamamatsu Photonics). With rats in a supine position, X-ray diffraction profiles were recorded vertically through the iPSC-CM cell sheet grafts and infarcted myocardium of the anterior LV wall of the exposed in situ beating hearts (12). Periodically between diffraction recordings a laser aligned with the X-ray beam was used to determine the point of path trajectory at which the beam also passed through normal myocardium. Diffraction patterns obtained from in situ iPSC sheets were of lower intensity compared to normal myocardium (remote regions) and easily distinguished on the basis of established cardiac fiber intensity peak orientations (21).
Using custom software, the average radial line profile around the center of the spectrum was calculated using a three-point background curve-fitting process with manual definition of peak spectra limits. Background subtraction was then performed between user-defined inner and outer limits on either side of the 1,0 and 1,1 reflections. The integrated intensity of the 1,0 and 1,1 reflection intensities was then determined from the areas under the reflection peaks, defined as I1,0 and I1,1, respectively.
Enzyme-Linked Immunosorbent Assay (ELISA)
Culture supernatants of cell preparation were centrifuged to remove debris and cells. Content of vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) in the undiluted culture supernatants was determined with an ELISA kit (R&D Systems, Minneapolis, MN, USA).
Immunohistolabeling Analysis
Under anesthesia by 5% isoflurane inhalation, the heart was promptly excised, immersed in 4% paraformaldehyde (Wako Pure Chemical Industries, Osaka, Japan), cut transversely, and then frozen (n = 5 each group). Ten-micrometer cryosections were labeled with monoclonal anti-cardiac myosin (Molecular Probes, Eugene, OR, USA) or monoclonal anti-connexin (Cx) 43 (Millipore, Billerica, MA, USA) antibodies. The labeled sections were again labeled by the secondary antibodies (Alexa Fluor 488, or Alexa Fluor 594 phalloidin; Invitrogen), counterstained with 6-diamidino-2-phenylindole (DAPI; Invitrogen), and then assessed by immunoconfocal microscopy (FV1000D; Olympus, Tokyo, Japan)
Electron Microscopy Analysis
Sliced cardiac tissues were fixed with 1% glutaraldehyde (Nacalai Tesque, Inc., Kyoto, Japan) in 0.1 M phosphate buffer (pH 7.4; Wako Pure Chemical Industries) for 60 min at 4°C, washed and immersed overnight in PBS at 4°C, and fixed in 1% buffered osmium tetroxide (Wako Pure Chemical Industries), then dehydrated through graded ethanol and embedded in epoxy resin (Okenshoji Co., Ltd., Tokyo, Japan). Ultrathin sections (85 nm) were double-stained with uranyl acetate (Funakoshi Co., Ltd., Tokyo, Japan) and lead citrate (Nisshin EM Co., Tokyo, Japan) and were observed under electron microscopy (H-7100; Hitachi, Tokyo, Japan).
Statistical Analysis
All values were expressed as mean ± standard deviations. Contents of VEGF and HGF in the supernatant of the four different cultures were compared by one-way ANOVA followed by Bonferroni's test for individual significant difference. Frequency of the VPC, ARI, and LVEF were compared by two-way ANOVA followed by Bonferroni's tests for individual significant differences. A value of p < 0.05 was considered to be statistically significant.
Results
iPSC-Derived Cardiomyocytes as a Source of Potential Paracrine Effects
It has been shown that cell transplantation into the heart produces “paracrine effects” in which the transplanted cells release a variety of protective factors into the adjacent native cardiac tissue to enhance native regenerative process, such as neoangiogenic, antifibrotic, or antiapoptotic effects (2). Capacity to release protective factors, such as VEGF and HGF, which have been shown to be the most important factors in the paracrine effects, were investigated here in vitro. Supernatant of the culture dishes of iPSC-CMs and fibroblasts was collected to measure concentrations of VEGF and HGF by ELISA, showing that concentration of VEGF and HGF was not significantly different in the conditioned medium of the iPSC-CMs and the fibroblasts, suggesting potential paracrine effects of the iPSC-CM transplantation therapy.
Electrical Integrity and Functional Recovery After Cell Sheet Transplantation In Vivo
The cell sheet method has been shown to transplant abundant somatic tissue-derived cells into the heart, which can be integrated into the cardiac tissue with minimal damage to the transplanted cells and to the myocardium (9). However, functional integration of cell sheets generated by iPSC-CMs into the heart is poorly understood. We speculate that similarity of the phenotype and/or character of the iPSC-CMs to the native CMs might result in a better integration into the native cardiac tissue compared to somatic tissue-derived cells.
Scaffold-free cell sheets generated by Dsred-labeled iPSC-CMs of mouse origin were transplanted into the nude rat heart that had been subjected to permanent occlusion of the left coronary artery prior to the cell sheet transplantation. The cell sheets generated by Dsred-labeled fibroblasts were used as controls. Electrical integrity and arrhythmogenicity were assessed by daily Holter ECG monitoring and 64-channel electrical potential mapping. In addition, global cardiac function was serially assessed by transthoracic echocardiography.
Electrical potential mapping identified multiple ectopic excitations over the cell sheet transplanted area in the iPSC-CM group until day 2 (day 16 post-MI), which gradually disappeared from day 3 onward (day 17 post-MI) (Fig. 1A). In contrast, transplantation of the cell sheet derived from fibroblasts or MI-only rarely induced ectopic excitations over the study period. Ventricular premature contractions more frequently occurred post-iPSC-CM cell sheet transplantation than those post-sham operation (Fig. 1B). In addition, ARI was significantly less in the iPSC-CM group than the other groups from day 3 onward (day 17 post-MI) (Fig. 2A). Moreover, LVEF was significantly greater in the iPSC-CM group than the other groups between day 3 and day 14 (day 17 and day 28 post-MI) (Fig. 2B). These findings indicate that the transplanted cell sheets of iPSC-CMs generated functional and electrical integration into the acute MI rat heart more rapidly than those of fibroblasts.

(A) Representative image of electrical current over the induced pluripotent stem cell-derived cardiomyocyte (iPSC-CM)-transplanted heart surface in one cardiac cycle, visualized by 64-channel mapping system at day 2, day 3, and day 7 (n = 4 each). Multiple ectopic excitations, which were present at day 2 and day 3, were not detected at day 7. (B) Frequency of the ventricular premature contractions (VPCs) was calculated by continuous telemetric electrocardiogram monitoring (n = 4 each). It appeared slightly greater in the iPSC-CM-transplanted rats compared to the sham-operated rats, though there was no significant difference.
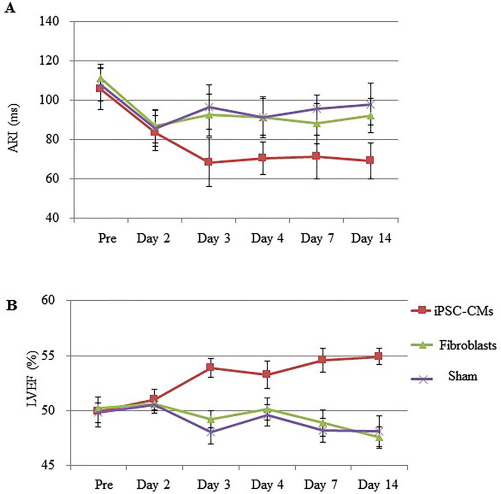
(A) Activation recovery interval (ARI), measured by daily 64-channel mapping studies, was significantly greater in the iPSC-CM group compared to the other groups from day 3 onward (n = 4 each). (B) Left ventricular ejection fraction (LVEF), measured by daily transthoracic echocardiography, was significantly greater in the iPSC-CM group compared to the other groups from day 3 onward (n = 6 each).
In Vivo Recording of Actin–Myosin Cross-Bridge Activity in the Transplanted iPSC-CMs in the Heart
While transplantation of iPSC-CMs into the rat infarcted heart was shown to induce functional and electrical recovery, mechanical or functional behavior of each transplanted iPSC-CM in the infarcted heart remains unclear. actin–myosin cross-bridge interactions in the transplanted iPSC-CMs in the rat infarcted heart were therefore investigated using fast synchrotron small-angle X-ray scattering.
At 4 weeks after the transplantation of iPSC-CM cell sheets on the surface of the infarcted heart (6 weeks post-MI), the rats were subjected to removal of the thoracic wall for the synchrotron study. X-ray diffraction profiles were recorded vertically through the iPSC-CM grafts and infarcted myocardium of the anterior LV wall of the in situ beating hearts. It was found that 1,0 and 1,1 equatorial reflections attributable to myosin and actin–myosin lattice planes typical of cardiac muscle fibers were detected within the iPSC-CM grafts, but not in the infarcted myocardium of the sham-treated rats (Fig. 3), albeit reflection intensity was generally much less than that obtained from the remote myocardium. Furthermore, in iPSC-CM hearts, cyclic changes in myosin masstransfer to actin with regular changes in myofilament lattice spacing were evident (Fig. 4), similar to that previously reported by us for in situ beating rat hearts (20). Importantly, the shift in myosin mass to actin of the iPSC-CMs was synchronous with LV pressure increase during the start of systole on a beat-by-beat basis (Fig. 5). The decrease in intensity ratio from ED through early systole was approximately linearly related to LV pressure development (Fig. 5, lower panel). Hence, significant cyclic systolic myosin head transfer to actin filaments, and therefore force-developing cross-bridges were detected within the grafts. Notably, implanted iPSC-CM sheets produced consistent reflections, but the same sheets fresh from culture did not reveal any reflections.
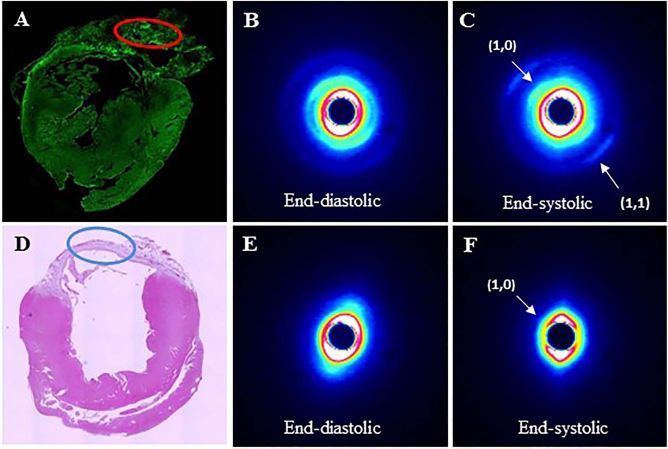
Small-angle X-ray scattering analysis of the red-circled area in the iPSC-CM-transplanted heart at day 14 (A) showed appearance of the 1,0 and 1,1 equatorial reflections at the end-systolic phase (B and C). In contrast, blue-circled scar tissue of the sham-operated heart (D) showed 1,0 reflections only at the end-systolic phase (E and F). n = 4 each.

Observed 1,0 reflection intensity change, calculated myosin interfilament spacing (d1,0), and myosin mass transfer index (equatorial intensity ratio I1,0/I1,1) over several consecutive cardiac cycles in an iPSC-CM sheet of a transplant heart. Regular loop-like relations between myosin mass transfer were evident.

Observed 1,0 reflection intensity change, calculated myosin interfilament spacing (d1,0), myosin mass transfer index (equatorial intensity ratio I1,0/I1,1), and simultaneously acquired LV pressure acquired over several consecutive cardiac cycles in an iPSC-CM sheet of a transplant heart. Significant 1,0 myosin reflections were only evident for part of the cardiac cycle due to heart movement. When significant actin–myosin reflections were evident the shift in myosin mass toward actin (decrease in intensity ratio) coincided with the rapid increase in LV pressure during systole, showing synchronized contraction of the iPSC-CMs in the sheet. Arrows indicate timing of end diastole.
Consistent with the findings in the synchrotron study, myosin and actin were well aligned in the cytoplasm of the Dsred-positive transplanted iPSC-CMs, which were present in the surface of the rat heart, assessed by immunohistolabeling (Fig. 6A). In contrast, Dsred-negative native CMs were rarely found in the border and infarct areas, which were assessed in the synchrotron study. These findings indicate that the in situ X-ray diffraction pattern originated from the transplanted iPSC-CMs, suggesting that regular actin–myosin cross-bridge motion had occurred in the transplanted iPSC-CMs in the rat heart.

Dsred-labeled transplanted iPSC-CMs expressed clear myosin-positive sarcomeres as shown in the representative confocal micrograph (A). The sarcomere of the transplanted iPSC-CMs consisted of myosin and sarcomeric actin (B). Distribution of the connexin 43 did not show the typical intercalated disks in the transplanted iPSC-CMs (C). Scale bar: 10 μm. The iPSC-CMs in vitro showed the cardiac myocyte-like sarcomeres with less dense mitochondrial structures (arrow) as shown in the representative electron micrograph (D). In vivo transplanted iPSC-CMs showed clear desmosome structure between the cells (arrow heads, E). Mitochondria of the transplanted iPSC-CMs in vivo gradually showed a dense structure at day 3 (E) and then at day 7 (F). n = 5 each.
Phenotypic and Morphological Fate of the Transplanted iPSC-CM in the Heart
Phenotype, morphology, and microstructure of the transplanted iPSC-CMs, which might be modulated following transplantation into the cardiac tissue, were then histologically analyzed by using immunoconfocal microscopy and electron microscopy. Dsred-positive transplanted iPSC-CMs showed myosin-positive sarcomeres at 14 days after transplantation (Fig. 6A, B). The sarcomeres consisted of myosin and sarcomeric actin. Although the transplanted iPSC-CMs expressed Cx43 at 14 days, the distribution of Cx43 was scarce and did not clearly show the typical intercalated disks between the ransplanted iPSC-CMs (Fig. 6C).
In vitro iPSC-CMs showed typical sarcomeric structures in cardiac myocytes with immature, less dense mitochondria (Fig. 6D). On the other hand, clear desmosomes were generated between the iPSC-CMs at 3 days after transplantation, while the mitochondria showed more mature structure compared to that prior to the transplantation. At 7 days, mitochondria showed mature structure, whereas the sarcomeric structure or the number of mitochondria was not as dense as those in the native CMs (Fig. 6E, F). This indicates that the transplanted iPSC-CMs by the cell sheet method might have established electrical/mechanical integrity with the native heart. In addition, maturity in the structure and functionality of the iPSC-CMs progressed after transplantation into the heart.
Discussion
We demonstrated here that the amount of VEGF and HGF released by iPSC-CMs was not significantly different from that of the same number of fibroblasts in vitro. Transplantation of the iPSC-CM cell sheets of mouse origin into the nude rat heart that was subjected to MI better preserved LV function and ARI, compared to that of the fibroblasts. Daily electrical mapping of the heart surface uncovered that transplantation of the iPSC-CMs induced multiple ectopic excitations over the cell sheet-implanted area for the initial 2 days, and subsequently ectopic excitations disappeared. In in vivo synchrotron radiation small-angle scattering studies, the transplanted iPSC-CMs displayed regularly contracting actin–myosin cross-bridge interactions, similar to that recorded in the native CMs of the remote myocardium of the same hearts. Immunohistologically, the transplanted iPSC-CMs, which were equipped with myosin-positive sarcomeres in the cytoplasm, formed Cx43 gap junction with the native cardiomycytes, while electron microscopically, the transplanted iPSC-CMs were equipped with immature sarcomeres and mitochondria, compared to the native cardiomyocytes.
The mechanisms underlying the global functional recovery by iPSC-CM transplantation include 1) that transplanted iPSC-CMs survived and showed synchronized contraction in vivo as proven by the diffraction analyses, 2) that transplanted iPSC-CMs were equipped with fully developed sarcomeres in vivo, and these cells might connect with the recipient myocardium, and 3) that there is functional integration of the transplanted iPSC-CMs into the native myocardium to produce direct mechanical contribution to cardiac function. The direct effects of the transplanted cells were studied chiefly by histological assessment of the excised cardiac tissues, which were examined for the presence and integration of the transplanted cells into the native cardiac tissue, but not the functionality of the transplanted cells. For the first time, in vivo scans dissected contractile motion of the transplanted cells by X-ray diffraction techniques using synchrotron radiation. Only X-ray diffraction using synchrotron radiation is able to detect rapid transient shifts in myosin mass most likely attributable to strong cross-bridge formation from relatively few muscle fibers within small regions (0.2 mm × 0.2 mm) (12,20). Myosin mass transfer in the first phase of systole in normal myocardial fibers is directly correlated with global LV force development (13). This feature has enabled us to investigate cardiomyocyte function within pinpoint regions of the graft, the infarct border zone, and infarct-remote myocardium in heart failure rats. In this study, contractile motion of the Dsred-labeled iPSC-CMs was clearly dissected from the native cardiomyocytes by the synchrotron study, as the cells were transplanted as a sheet form on the heart surface. In the absence of living native cardiomyocytes beneath the graft, it is reasonable to conclude that strong cross-bridge formation of the iPSC-CMs contributed to force development, albeit much reduced compared to remote myocardium. In addition, electrical and functional integration of the transplanted iPSC-CMs in the heart was revealed by electrical mapping and echocardiography, supplemented with histological studies, indicating that contraction of each transplanted iPSC-CMs was transferred into the native cardiac motion, at least in part, contributing to the functional recovery of MI heart.
Other possible mechanisms underlying the functional recovery by the transplantation of iPSC-CM cell sheets might include paracrine effects of the transplanted cells on the native heart (19). In fact, this study revealed that the known paracrine factors, such as VEGF or HGF, were released in vitro from the iPSC-CMs as from the fibroblasts, while fibroblasts were proven here not to be differentiated into functional cardiomyocytes after transplantation into the heart. Of note, functional recovery was produced in the iPSC-CM-transplanted hearts but not in the fibroblast-transplanted hearts, despite similar release of the paracrine factors between those two cell sources. Although these data would be insufficient to determine the magnitude of the paracrine effects by the iPSC-CM therapy, it is suggested that functional recovery by the iPSC-CM transplantation might be chiefly caused by mechanical effects of the transplanted cells, and partly by the paracrine effects. Further basic studies are warranted for magnitude and/or durability of paracrine effects by this treatment.
The transplanted graft was integrated into the cardiac tissue beneath the epicardial layer, in particular, the epicardial layer of the infarct and infarct border territory in this study. In contrast, it seemed that integration of the transplanted cells crossing the epicardium was less prominent in the remote myocardium from the infarct territory (data not shown). We thus consider that integration of the cell sheet was determined by the microenvironment of the native cardiac tissue and that ablation of the epicardium prior to the cell sheet transplantation may enhance the integration of the cell sheet graft (10). In addition, thorough immunohistolabeling and electron microscopy studies showed abundant expression of Cx43 in the graft but rarely identified the gap junction formation between the graft and the native tissue. We consider that presence of a few gap junctions might be enough to transfer electrical current from the native tissue to the grafted cells.
In this study, the transplanted iPSC-CMs showed an “immature” structure, which was not equipped with dense sarcomeric structure and mitochondrial arrangement, as shown in the immunohistological and electron microscopy analysis. This was also evident in the lower reflection intensities obtained from sheet-derived CMs compared to native remote myocardium. Importantly, this study indicates that maturity of iPSC-CMs may progress after transplantation into the heart, though quantitative analysis of the “maturity” of the transplanted cells is needed. The functionality of each transplanted iPSC-CM, in particular its contractility, is the most important factor in determining the therapeutic effects of this treatment. We also showed that the maturity of in vitro differentiated iPSC-CMs is variable according to the origin, the cell line, and the culture protocol, and therefore specific regimens used in culture protocols, such as mechanical stretching in the cell preparation, mimicking the in vivo environment, might be useful in enhancing the maturation in vitro (4,23). One can consider that cell–cell connections between the “immature” cardiomyocytes transplanted into the LV and the native LV cardiomyocytes is likely to produce electrical instability in the LV myocardium resulting in the induction of ventricular arrhythmias. In fact, multiple ectopic excitations appeared in the cell-transplanted area and gradually decreased. Possible causes of the prominent occurrence of premature ventricular contractions in the iPSC-CM-treated animals would include ectopic excitation directly produced by the transplanted and integrated “immature” iPSC-CMs, or macro/micro reentries generated by cell transplantation-induced myocardial heterogeneity (1). Although this study failed to dissect the exact cause of the premature contraction, we consider that heterogeneity in the native cardiac tissue would be the main cause, since frequency of the premature contractions was stable for 7 days despite the progressive electrical integration of the graft during this period as in Figure 1A. While it is unclear that maturation of the transplanted iPSC-CMs is associated with the progressive electrical stability in the LV myocardium in this study, more efficient culture protocols to enhance maturity of iPSC-CMs, and thereby maximize the therapeutic effects and minimize the arrhythmogenicity of this treatment, warrant further investigation.
A limitation of this study may be that we used different species for the iPSC origin and the recipient animal. Interspecies differences may influence functional and electrical integration of the transplanted cells into the recipient native heart. However, the mouse and rat share important proteins related to cell–cell connections and sarcomere structure in the heart (18). The data here therefore are unlikely to be affected by a use of such a xenotransplantation model. Another limitation could be the use of a single iPSC cell line. As it has been shown that the cell line is one of the important variable factors that determines the fundamental behavior of iPSCs and their derivatives (22), further studies are warranted to establish an efficient culture protocol to produce cells that have a consistent quality toward clinical application. In addition, quantitative study of the transplanted cell survival in the heart would provide further information regarding this treatment.
In conclusion, transplanted iPSC-CMs in a cell sheet form displayed regularly contracting actin–myosin cross-bridge interactions in the heart surface with synchronous contraction with the native cardiac tissue, contributing to global functional recovery from ischemic heart injury in the rat. High-flux synchrotron X-ray diffraction offers a promising potential as an in vivo cellular and molecular level modality for evaluating transplanted iPSC-CM function. Finally, it is important to develop culture protocols to enhance functionality of the iPSC-CMs for successful clinical application of this treatment.
Footnotes
Acknowledgments
This work was supported by the Research Center Network for Realization of Regenerative Medicine managed by the Centers for Clinical Application Research on Specific Disease/Organ and funded by the Japan Science and Technology Agency, and the International Synchrotron Access Programme (IA111) managed by the Australian Synchrotron and funded by the Australian Government. The authors declare no conflicts of interest.
