Abstract
The ideal cells for tissue engineering should have the following characteristics: easy obtainment, safety, immune privilege, the capability of self-renewal, and multipotency. Adipose-derived stem cells (ADSCs) are a promising candidate. However, the immunogenicity of allogeneic mesenchymal stem cells limits their long-term benefits. In this study, we introduced human cytomegalovirus US2/US3 gene into the ADSCs to decrease the expression of MHC I protein of ADSCs and reduce the activation of T-cells of the recipient animals. Moreover, the biosafety and biological characteristics of ADSCs transfected with the US2/US3 genes (ADSCs-US2/US3) were similar to normal ADSCs. Then we took ADSCs-US2/US3 to construct a tissue-engineered bone for repairing bone defects in pigs and found that there were no great differences in repair effects or healing time between the allogeneic ADSCs-US2/US3 group and the autologous ADSC group. These results suggest that allogeneic ADSCs-US2/US3 have the advantages of biological safety, low immunogenicity, and effective osteogenesis. Such barely immunogenic ADSCs will be crucial for the success of future tissue-regenerative approaches.
Keywords
Introduction
The tissue-engineered bone provides great potential ways for repairing bone defects (7, 21, 29). As the cornerstone of tissue engineering, stem cells set off a global research boom (13, 24, 27) and had a profound impact on bone repair. Commonly used cells for bone defects are autologous stem cells, which were called “individualized treatment.” However, it is time-consuming for isolation and amplification before transplanting (23, 30), and the elderly and cancer patients may require cells from young, healthy donors. Even though the major histocompatibility complex (MHC) II antigens of stem cells were very low (18, 20, 22), the stem cells can express high levels of MHC I antigen and thus may be rejected on transplantation (10, 19).
The recognition by T-cell receptor can be avoided by decreasing the MHC expression of the allograft (6, 8, 28). It indicates that a feasible way to resolve the immunological rejection for allogeneic stem cells is through reducing the expression of MHC I antigen. Adipose-derived stem cells (ADSCs) have the advantages of extensive sources, great capacity of amplification, and capacity of multipotency (11) in vitro. In this study, ADSCs were transfected with the US2/US3 gene (ADSCs-US2/US3), which could decrease the expression of MHC I antigen (1, 14), to achieve the aim of immune escape. We also verified the biosafety, the biological characteristics, and the efficacy for bone defect repair of ADSCs-US2/US3.
Materials and Methods
Animals and Cells
Guizhou miniature pigs, New Zealand white rabbits, nude mice, and Sprague–Dawley rats were obtained from the Animal Center of Daping Hospital. Isolation and cultivation of ADSCs were performed according to the method described in literature (4). Briefly, subcutaneous adipose tissue of pigs or rabbits was minced and rinsed by phosphate-buffered saline (PBS). Type I collagenase (0.75 g/L) (Sigma, USA) was added for digestion in water bath of 37°C. One hour later, Dulbecco's modified Eagle's medium, nutrient mixture F12 (DMEM/F12), and 10% fetal bovine serum (FBS, Gibco, USA) were added to neutralize the enzyme. After centrifugation, the pellet was suspended in red blood cell lysis buffer and incubated for 10 min. After filtration and centrifugation, the cells were suspended in complete medium (DMEM/ F12 containing 10% FBS, 0.1% penicillin–streptomycin) and then plated at a density of 1 × 105 cells/cm2. After 24 h, the unattached cells were removed by washing with PBS.
The experiments were carried out in accordance with the guidelines of the current Chinese legislation for the use and care of laboratory animals. The protocols for animal care and manipulation used were approved by the Bioethics Committee of Third Military Medical University.
US2/US3 Gene Transfection and Expression Assay
The US2/US3 gene was reconstructed for expressing the vector pEGFP-N1-US2/US3, which carried enhanced green fluorescent protein (eGFP) and the neomycin resistance gene. The pEGFP-N1-US2/US3 and pEGFP-N1 were transfected into the cultured ADSCs using the Lipofectamine™ 2000 (Invitrogen, USA) according to the manufacturer's instructions. Following continuous selection in G418, the positive cell clones were picked and expanded. GFP-positive cells were counted by flow cytometry analysis.
Total RNA was prepared from the pig ADSCs-US2/ US3 by the RNA isolation mini kit (Qiagen, Valencia, CA, USA). For RT-PCR, the 20-μl RT buffer contains 1 μg total RNA, 0.5 μg Oligo dT, 1 mM dNTP mixture, 10 units AMV reverse transcriptase (Promega, USA), and 24 units RNase inhibitor (Promega). The mixture was incubated at 42°C for 1 h and 95°C for 5 min. The PCR was carried out in the appropriate buffer with 3 μg cDNA, 15 pmol of the relevant primers, 0.8 mM dNTP mixture, and 1.5 units Taq DNA polymerase. The following primers were used: US2 sense, 5′-ATGAACAATCTCTGGAAAGC-3′; a-sense, 5′-TCAGCACACGAAAAACCGC A-3′; US3 sense, 5′-AT GAAGCCGGTGTTGGTGCTCG-3′; a-sense, 5′-TCATGG TGGCATTG AGCAGC-3′; glyceraldehyde3-phosphate dehydrogenase (GAPDH) sense, 5′-GGCCATCACCATC TTCCAGGAG-3′; a-sense, 5′-GAAG GGGCAGAGA TGATGAC-3′. The PCR products were electrophoresed on agarose gel.
Characteristics of ADSCs-US2/US3
Test of proliferation capacity of pig ADSCs and pig ADSCs-US2/US3 by MTT assay was performed everyday. A week later, the growth curve of the cells was drawn. Another group of ADSCs-US2/US3 was detected by using the Annexin V-PI apoptosis detection kit at 24, 48, and 72 h after transfection, respectively, and the normal ADSCs acted as the control group.
Adipogenic and Osteogenic Differentiation Assays
For induction of adipogenic and osteogenic differentiation, pig ADSCs were cultured with specific induction media. Basal medium consisted of DMEM/F12, 1% gentamycin, and 10% FBS; adipogenic induction medium was supplemented with 0.5 mM isobutylmethylxanthine (Sigma-Aldrich), 0.1 μM dexamethasone, 0.1% insulin, and 0.1 mM indomethacin (BD Bioscience); osteogenic induction medium was supplemented with 0.1 μM dexamethasone, 0.05 mM ascorbate-2-phosphate, 10 mM β-glycerolphosphate (Sigma-Aldrich), and 1% insulin.
The adipogenic differentiation of pig ADSCs was assessed after 2 weeks. Cells were rinsed twice with PBS and fixed for 10 min with 4% formaldehyde, washed again with distilled water, and rinsed with 50% ethanol. For staining, 2% Oil Red O (Sigma-Aldrich) solution was added and incubated for 15 min. Afterwards, the cells were washed with 50% ethanol, followed by several changes with distilled water. Osteogenic differentiation was induced for 3 weeks and examined by ECM calcification using Von Kossa Stain Kit (American MasterTech Scientific, Inc., Lodi, CA, USA).
Immunoreactivity of ADSCs-US2/US3
MHC I Expression of ADSCs
The cultured pig ADSCs were divided into ADSCs-US2/US3 group, normal ADSC group, and ADSCs-EGFP group. The expression of molecular subtypes of MHC I [swine leukocyte antigen-1 (SLA-1) and SLA-6] were detected by RT-PCR. The following primers were used: SLA-1 sense, 5′-GGGGCAGGACTATTGGGATGA-3′; a-sense, 5′-CCCGTCTGGTCCCAAGTAGCA-3′; SLA-6 sense, 5′-ACCGCGATCAGTACAGTGGCTC-3′; a-sense, 5′-AG GGCCCAGCATCTTAGAGTGAC-3′; β-actin sense, 5′-CT GCGGCATCCACGAAACTAC-3′; a-sense, 5′-GGGCC GGACTCGTCGTACTC-3′. The cells were suspended in ice-cold lysis buffer. The protein concentration of the supernatant was determined by using the Bio-Rad protein assay after extraction. Then the proteins were separated by sodium dodecylsulfate-polyacrylarnide gel electrophoresis (SDS-PAGE) on 10% polyacrylamide gels and electrophoretically transferred to polyvinylidene difluoride (PVDF) membranes. Blocking was performed for 1 h at room temperature in Tris-buffered saline containing 0.1% Tween-20 (TBST) and 5% skimmed milk. The membranes were incubated for 1 h at room temperature with mouse anti-MHC-I antibody (ABR; 1:2,500) or β-actin (Sigma; 1:5000) in blocking buffer. Following careful washing with TBST, the second antibody (Sigma; 1:5,000) was applied for 2 h at room temperature. After washing five times in TBST, the proteins were visualized by using enhanced chemiluminescence.
Detection of Proliferation of T Lymphocyte and Splenic Lymphocyte in Peripheral Blood
New Zealand white rabbits were divided randomly into four groups (allogenic ADSCs-US2/US3 group, allogenic ADSC group, autologous ADSC group, and medium group), with 10 rabbits in each group. Corresponding cells (1 × 106 cells/ml) were injected into the auricular vein of the rabbits (2 ml/kg), respectively. Then five rabbits in each group were sacrificed 1 week after injection. Spleen lymphocytes were collected by trituration method. The proliferation of splenic lymphocytes was detected with MTT method. Each 0.5-ml anticoagulant blood was collected from the remaining rabbit's auricular vein at 2 h before injection and 1, 2, and 4 weeks after injection. CD3-phycoerythrin-cyanine 5 (PC5)/CD4-fluorescein isothiocyanate (FITC) double-staining positive cells, CD3-PC5/CD8-phycoerythrin (PE) double-staining positive cells, and CD3-FITC/CD25-PE double-staining positive cells in the peripheral blood were evaluated by flow cytometer.
Biosafety of ADSCs-US2/US3
Toxicity Test
Thirty-five adult nude mice were randomly assigned to seven groups. Each mouse in groups A, B, C, and D was injected with 0.4 ml pig ADSCs-US2/US3 (1 × 107 cells/ml) through the vena caudalis, and those in groups E and F were injected with 0.4 ml pig ADSCs-US2/ US3 (1 × 106 cells/ml). Group G was the negative control group, injecting medium only. Then injection was given once a week. After 4 weeks, these groups were observed to determine whether respiratory depression, shortness of breath, normal activities, rapid response, convulsions, paralysis, or death existed. At 24 h for group A, 48 h for group B, 72 h for group C, 1 week for group D, 3 weeks for group E, and 4 weeks for groups F and G, the mice were sacrificed at each time point to check whether they had peritoneal fluid or intra-abdominal adhesions. The heart, liver, kidney, and spleen were taken to prepare paraffin sections and were observed after hematoxylin and eosin (H&E) staining.
Tumorigenicity Test
Twenty-four adult nude mice were randomly assigned to four groups. Group A was subcutaneously injected with 0.3 ml pig ADSCs-US2/US3 (1 × 106 cells/ml) on the back and group B under the left armpit; group C was subcutaneously injected with 0.3 ml medium on the back and group D under the left armpit. No tumor was observed in these mice for a period of 12 weeks. Also, the sites were taken 12 weeks after sacrifice and were made paraffin sections, which were observed after H&E staining.
Teratology Test
Thirty-six Sprague–Dawley rats with 10-day pregnancy were randomly divided into six groups. They were injected with 0.4 ml pig ADSCs-US2/US3 through the vena caudalis. The concentrations of the fluid injected to groups A, B, and C were 1 × 106/ml, 0.5 × 107/ml, and 1 × 107/ml, respectively. Group D was injected with the normal allogeneic ADSCs (1 × 106/ml), and group E was only injected with the medium. The rats were then sacrificed on the 20th day of pregnancy to examine whether miscarriage, fetal death, or deformity existed.
Histocompatibility
Heating Test
Before injection, the basal rectal temperature of five healthy adult rabbits was measured twice a day for 3 days successively. Allogeneic rabbits ADSCs-US2/US3 (1 × 106/ml) were injected into the rabbits through the auricular veins with a dosage of 2 ml/kg. The body temperature was measured at 30 min and 8, 24, 48, and 72 h after injection.
Hemolysis Test
Fresh anticoagulant blood samples were drawn from the auricular veins of the healthy adult rabbits, which were equally divided into three groups: adding 2 ml culture medium to the negative control group, 2 ml normal allogeneic rabbits ADSCs (1 × 106/ml) to positive control group, and 2 ml allogeneic rabbits ADSCs-US2/US3 (1 × 106/ml) to the experimental group. The degree of hemolysis was determined with ELISA reader. The calculation formula was: the degree of hemolysis (%) = (absorbance of experimental group – absorbance of negative control group) / (absorbance of negative control group – absorbance of positive control group) x 100%.
Local Stimulation Test
The back of three rabbits was depilated by sodium thiosulfate. Three injection sites were selected on each side of the back, with a 3-cm interval between two of them. The three sites were injected subcutaneously with ethanol, 0.2 ml allogeneic rabbits ADSCs-US2/US3 (1 × 106 cells/ml), and 0.2 ml culture medium, respectively. Local reactions were observed at 6, 24, and 72 h and 1 week after injection.
Bone Transplantation
Acellular bone matrix (ACBM) was prepared with fresh bovine trabecular bones in a process according to the method described in literature (5). Immerge ACBM in cell suspension for 2 h, and then the ACBM was cultured in medium without serum at 37°C and 5% CO2 for 24 h.
The pigs were anesthetized by intramuscular injection of sodium pentobarbital (1 ml/kg). Then a 15-mm bone defect, which included the periosteum, was made in the middle segment of the ulna, and the radius was left intact for mechanical stability. The implanted materials were inserted into the bone defect and bound to the radius by thread without other fixation devices.
The pigs were divided into four groups according to different implanted repairing materials: group A (
Ulnar AP and LAT X-ray films were taken at 1, 2, 4, 8, and 12 weeks after operation. The repairing status of the defects was assessed by X-ray pictures. The assessment standard of repairing was classified into four classes: grade I (the swelling of the local tissue disappeared and the surrounding connective tissue began to fill the defects), grade II (the broken ends of the bone turned obscure and a small amount of callus began to form), grade III (new bone filled the defects and continuous callus began to form), and grade IV (bones with normal density filled the defects and marrow cavity was shaped). Single photon emission computed tomography (SPECT) imaging was carried out in groups A, B, and C at 2, 4, 8, and 12 weeks after operation. Time–radioactivity curve was drawn to assess the blood flow and semiquantitative analysis indicators of osteogenesis in every group at different time points. Histological experiments were made at 4 months after operation. The tissues taken from either side of the defects were decalcified consistently for 1 week with 20% EDTA microwave and medium wave. Paraffin-embedded sections were prepared to observe the osteogenesis after H&E staining.
Statistical Analysis
All the data were expressed as mean ± SD. Analysis was performed by using the Statistical Program for Social Science (SPSS) 13.0 for Windows. Analysis of variance followed by Student's
Results
Biological Characteristics of ADSCs-US2/US3
After G418 selection and amplification, about 80% of the cells were GFP positive (Fig. 1A, B). The ADSCs-US2/US3 passage for two generations were selected and compared with the fifth generation of the normal passage of ADSCs, and no significant difference was found in the morphology, growth curve, and apoptosis ratio (Fig. 1C, D). After 7 days of osteogenic induction, the ADSCs-US2/US3 morphology was mostly transformed from spindle into polygon and cube, and 20 days later, refractive particles were observed obviously and calcium deposition could be highlighted as black granules by Von Kossa stain (Fig. 2A, B). After 10 days of adipogenic induction, a small amount of cytoplasm vacuolization could be observed. Lipid droplets gathering could be found clearly 3 weeks later, and it showed red when stained by Oil Red O (Fig. 2C, D).

(A) The fluorescence-activated cell sorting (FACS) result of green fluorescent protein (GFP)-positive cells before G418 selection and (B) GFP-positive cells after G418 selection. (C) The growth curves of adipose derived stem cells (ADSCs) and ADSCs-US2/US3. (D) The apoptosis ratio of ADSCs and ADSCs-US2/US3. *

(A, B) ADSCs-US2/US3 can induce the differentiation of osteogenesis. 20d, refractive particle can be seen, and some areas are too deep in color that the cells cannot distinguish the boundaries. Arrows indicate the cells with calcium nodules formation (silver staining). (C, D) ADSCs-US2/US3 can induce the differentiation of lipoblasts. 14d, circles indicate stained lipid droplets (Oil Red O staining). Scale bar: 20 μm.
ADSCs Immunogenicity Reduced After US2/US3 Gene Transfection
In ADSC-US2/US3 group, the US2 and US3 mRNA levels increased significantly (Fig. 3A), and the mRNA expression of MHC I-related subtype genes (SLA-1 and SLA-6) significantly increased (Fig. 3B) at 3 weeks posttransfection. Meanwhile, the expression of MHC I protein decreased (Fig. 3C).

(A) RT-PCR for the mRNA expression of US2/US3. (B) RT-PCR for the mRNA expression of major histocompatibility complex (MHC) I-related subtype genes. (C) Western blotting for the expression of MHC I protein. SLA, swine leukocyte antigen; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
After cell injection, the proportion of CD3+CD4+ T lymphocytes in peripheral blood of recipient rabbits in each group progressively increased, but the allogeneic ADSC group (group B), compared with allogenic ADSCs-US2/ US3 group (group A), autologous ADSC group (group C), and the medium group (group D), increased more obviously in 2–4 weeks (Fig. 4A). The proportion of CD3+CD8+ T lymphocytes in group A and group B increased in 1–2 weeks and began to decrease in 4 weeks. However, compared with group A, group B increased significantly at different time points (Fig. 4B). The proportions of CD3+CD25+ T lymphocytes in groups A and B were increased progressively, but much more prominently in group B (Fig. 4C).

(A–C) The proportion of CD3+CD4+, CD3+CD8+, CD3+CD25+ T lymphocytes. (D) The proliferation of splenic T lymphocytes after transplantation of different cells. *
The proliferation of splenic lymphocyte in the allogeneic ADSCs-US2/US3 group (group A) was significantly lower than the normal allogeneic ADSC group (group B,
Stable Biosafety of ADSCs-US2/US3
The animals in ADSCs-US2/US3 group had no significant difference with the control group in toxicology test. Their daily activity and response remained normal. They had no shortness of breath, suppression, infection, convulsions, paralysis, or deaths. They had no weight loss, jaundice, or ascites. The liver, spleen, heart, and kidney tissue sections remained normal.
Tail vein injection of ADSCs-US2/US3 did not result in teratogenicity, abortion, or death. Meanwhile, there was no absence of visceral tissues or abnormal growth when dissecting the fetal animal. Bones developed, and the fontanelle was closed normally.
Heating test indicated that, after injection of ADSCs-US2/US3, the body temperature fluctuated in 1.5°C, and no significant heating effect was found in the recipient rabbits.
Hemolysis test found that experimental group hemolysis degrees were 3.65%, 4.56%, and 4.24%, respectively. In accordance with the standards, it was qualified with a hemolysis of <5%, thus allogeneic ADSCs-US2/US3 did not lead to hemolysis.
Regional irritation test found that no significant erythema or edema was observed both in the group injected with ADSCs-US2/US3 and the medium group at 6, 24, 48, and 72 h. Swelling was found in the alcohol group between 24 and 48 h, and the swelling reduced gradually at 72 h.
Effect of Allogeneic ADSCs-US2/US3 Constructing Tissue-Engineered Bone for Repair of Bone Defects
Preparation of ACBM (Fig. 5A) and ADSCs constructed the composite materials for bone defect (Fig. 5B). Comparing the regional blood flow of each group in 2, 4, 8, and 12 weeks after operation, the regional blood flow in the repair process in ADSCs-US2/US3 group was better than the other two groups (
Change of Regional Blood Flow (
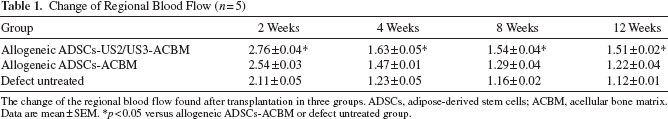
The change of the regional blood flow found after transplantation in three groups. ADSCs, adipose-derived stem cells; ACBM, acellular bone matrix. Data are mean ± SEM.
Quantity of Local Bone Formation With SPECT (T/NT) (

The quantity of the bone formation with single photon emission computed tomography (SPECT) found after transplantation in three groups. Data are mean ± SEM.
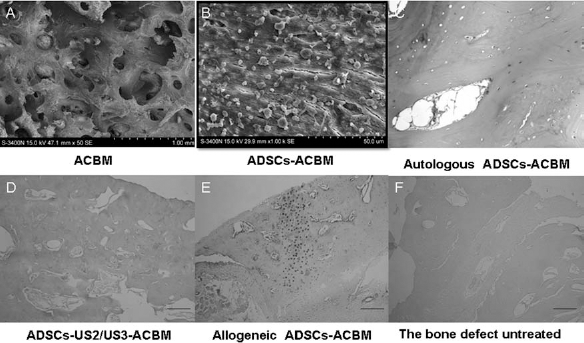
(A) Acellular bone matrix (ACBM) surface and pores without cells and debris. (B) Transmission electron microscopy (TEM) indicated ADSC adhesion in the gaps of ACBM. Sections of bone defects at 4 months after operation (H&E staining): the defects were mainly repaired by new bone (C); new bone filling the defected sites and the marrow cavity began to form (D); the defects were mainly repaired by new cartilage bone (E); the defects were mainly filling with fibrillar granulation tissue and connective tissue (F). Scale bar: 100 μm.

Results of X-ray check after operation. (A) Allogeneic ADSCs-US2/US3-ACBM, the grade of repair effect at 1 week (grade I), 4 weeks (grade II), 8 weeks (grade III), and 12 weeks (grade IV). (B) Allogeneic ADSCs-ACBM, the grade of repair effect at 1 week (grade I), 4 weeks (grade I), 8 weeks (grade II), and 12 weeks (grade III). (C) The bone defect untreated, the grade of repair effect at 1 week (grade I), 4 weeks (grade I), 8 weeks (grade I), and 12 weeks (grade II). (D) Autologous ADSCs-ACBM, the grade of repair effect at 1 week (grade I), 4 weeks (grade III), 8 weeks (grade III, IV), and 12 weeks (grade IV).
Discussion
Allogeneic ADSCs isolated from healthy, young donors are the nice candidates for clinical therapy. The present study decreased the immunogenicity of allogeneic stem cells by transfecting ADSCs with US2/US3 gene in a nonviral way. Biocompatibility and biosafety of ADSC-US2/US3 are the basic requirements. The study used a series of safety tests to evaluate the influence of ADSC-US2/US3 (3). The result of carcinogenic testing indicated that ADSCs transfected with US2/US3 gene by liposome had no significant carcinogenicity. In this study, we found that ADSCs-US2/US3 injection in mice at 10 days of pregnancy, which was the proper time to verify teratogenicity (25), did not cause obvious deformity on animal embryos, abortion, or death. In addition, the ADSCs-US2/US3 in other safety tests also showed no toxicity.
The self-renewal and the multipotency of ADSCs-US2/US3 are the prerequisite for tissue engineering. The results showed that transfection makes the proliferation of ADSCs decline, but with no statistical significance. In addition, continuous observation of the transfected cells under microscopy did not find obvious morphological difference. The results of osteogenic and adipogenic differentiation suggested that the transfection of US2/US3 will not change the multipotency of ADSCs. It means that the ADSCs-US2/US3 maintain the features of the stem cells.
MHC molecules of ADSCs are the most important target antigen generated by allogeneic graft rejection reaction. In this study, Western blotting results showed that the expression of MHC I protein in the pig ADSCs carrying US2/US3 genes decreased significantly, while the results of RNA reverse transcription indicated that the gene expression of SLA-1 and SLA-6 subtypes of MHC I increased significantly. This is mainly because the US2/ US3 protein can detain the host MHC I molecules in the cytosol before proteasome-mediated degradation instead of transporting to the cell surface (1, 14). MHC I expression at the transcriptional level is not suppressed, which causes a bulk aggregation of MHC I RNA in the endoplasmic reticulum (ER) and cytoplasm.
Recipient rejecting allografts is mainly the immune responses produced by the surface allogenic antigens, which is mediated by T-cell receptors (2, 5, 15, 16, 32). T lymphocytes can be divided into CD3+CD4+ and CD3+CD8+ subsets, both participating in the immunological rejection (17, 31). CD3+CD4+ is activated by indirect identification of the deciduous antigens of the allogenic cells or secretion of some exogenous antigens. The reaction usually appears at 2–4 weeks after transplantation (9). CD3+CD8+ is activated by direct identification of complete presentation of allogeneic MHC I on the cell surface; thus, a strong CTL-based acute rejection occurs usually at 1–2 weeks after transplantation. CD3+CD25+, as an indicator of T-cell activation, reflects the activation status of allogeneic cell transplantation caused by the immunogenicity of T lymphocyte. In this study, we detected T-cell subsets in rabbits and found that the proportion of CD3+CD4+ T lymphocytes in the allogeneic ADSC group increased more significantly than the other groups at 2–4 weeks after transplantation. The proportions of CD3+CD8+ and CD3+CD25+ T lymphocytes at different time points in the allogeneic ADSC group increased more significantly than the allogeneic ADSCs-US2/US3 group. The proliferation of splenic lymphocytes, which represented the overall cellular and humoral immune response to foreign materials (12, 26), was significantly lower in the allogeneic ADSCs-US2/US3 group compared to the allogeneic ADSC group. The results indicated that ADSCs transfected by US2/US3 gene can effectively reduce the recipient's immune responses.
In the study, the SPECT results showed that the active phase for bone formation of allogeneic ADSCs-US2/ US3 was earlier than the allogeneic ADSC group. The results of X-plain film of bone defect restoration and histological examination after operation for 4 months showed that the allogeneic ADSCs-US2/US3 group was close to the autologous ADSC group. It showed good restoration, new bone filling the defected sites, and the callus beginning to generate after 8 weeks. After 12 weeks, bones with normal density filled the defected site, and the bone marrow cavity formed. Our previous study has shown that the allogeneic ADSCs-ACBM group had better repair effect than the ACBM group (5). However, the effect of the allogeneic ADSC group was not as good as the first two groups. Regretfully, cell tracking in vivo is still relatively difficult in pigs. In our experiments, the transplanted GFP-positive cells could not be detected in 12 weeks after transplantation for this low immunogenicity ADSCs-US2/US3 all died or the plasmid lost gradually as time went by and the cells no longer expressed GFP. From the present data, ADSCs transfected with the US2/US3 genes decreased the immunogenicity and achieved repair effects as well as the autologous ADSCs. We presume that immune status of cells impacts the therapeutic effects and the ADSCs death caused by immune rejection and inflammatory response is partly responsible at least.
Conclusion
To sum up, ADSCs carrying US2/US3 genes can decrease the expression of MHC I protein and maintain biosafety and the multipotent capacity, and allogeneic ADSCs-US2/US3 can obtain the similar repair effect of autologous ADSCs for bone defects. The reasons may be due to the reduction of ADSC immunogenicity and the graft rejection reaction. Drawing on the experience of virus to evade immune response might provide a new approach for the success of future tissue regenerative.
Footnotes
Acknowledgments
