Abstract
Substantial damage to the bone marrow can be caused by exposure to radiation, which can then develop into severe thrombocytopenia. In this study, we investigated the in vivo impact of adipose-derived mesenchymal stem cells (ADSCs) on megakaryopoiesis and platelet recovery in irradiated mice. Radiation markedly reduced peripheral blood counts. Recovery of both platelets and WBCs was better in the ADSC-treated group compared with the saline group and the fibroblast group 21 days after irradiation. A significant increase in the total CFU and MK-CFU after irradiation was observed in the ADSC group compared with the saline group and the fibroblast group. Further, the proportion of CD41+ cells in the ADSC group was significantly higher than that in the saline group and the fibroblast group. ADSC treatment significantly improved the cellularity and decreased the apoptotic cells in the bone marrow while normal fibroblasts did not. Administration of ADSCs upregulated protein expression of phosphorylated Akt and Bcl-xL, whereas the expression of Bax, a protein related to apoptosis, was significantly lower in the ADSC group. In conclusion, this study suggests that ADSCs were capable of promoting platelet recovery, improving megakaryopoiesis, and inhibiting apoptosis of bone marrow cells in irradiated mice. The antiapoptotic effect of ADSCs is likely to be mediated via the PI3K/Akt pathway. These findings may provide a scientific basis for using ADSCs as a new therapy after irradiation.
Keywords
Introduction
Acute radiation syndrome (ARS), which affects multiple organ systems, is caused by exposure to high-dose radiation in a relatively short period of time (15). Because blood-forming cells are highly sensitive to radiation, substantial damage to bone marrow can be caused by exposure to only a few grays, having dramatic effects on circulating blood cells. Depending on the level of radiation exposure, severe neutropenia and thrombocytopenia can develop. Both conditions can be major life-threatening events in untreated individuals (5,35). However, much of the research into radiation-induced hematopoietic damage has focused on the amelioration of radiation-induced neutropenia, which has long been considered to be the pivotal predictor of survival after irradiation (8). However, recent studies have emphasized the prevalence of thrombocytopenia in irradiated individuals. Stickney et al. showed that the degree of thrombocytopenia was a better prognostic indicator of survival than neutrophil counts in rhesus monkeys (50). Yet little research focusing on developments to mitigate radiation-induced thrombocytopenia has been reported.
To date, growth factors, cell therapies, platelet concentrates, and fresh whole blood transfusions have been used to treat radiation-induced thrombocytopenia. TPO administration was highly effective in increasing platelet counts and preventing thrombocytopenia, but its ability to reduce mortality has not yet been demonstrated (39,42). Colony-stimulating factor (CSF) appears to have a modest but significant medicinal value after radiation (46). Although treatment with granulocyte-CSF (G-CSF) markedly reduced the radiation-associated lethality, the severity of platelet depletion was unchanged by this treatment (33). Furthermore, it is suggested that the maximal efficacy of these therapies relies on preirradiation administration (49), making it impractical for clinical use. In addition to growth factors, cell therapies are also being studied to address the effects of radiation on platelets (59). Ex vivo expanded megakaryocyte progenitors (MKPs) were confirmed to increase circulating platelets and megakaryocyte progenitors when administered to irradiated mice (36). However, the culturing, expansion, and cryopreservation of MKP products must be further developed and optimized for human use. Among all of the therapies available to patients experiencing radiation-induced thrombocytopenia, platelet transfusion is the most effective. Nevertheless, as a supportive therapy, platelet transfusion cannot substantially reconstitute the damaged hematopoietic properties of bone marrow. Therefore, there is an urgent need for easily administered and efficient therapies.
It has been demonstrated that adipose-derived mesenchymal stem cells (ADSCs) could support hematopoiesis in vitro and in vivo. ADSCs constitute a favorable positive regulator of hematopoiesis, evidenced by their ability to promote the proliferation and differentiation of hematopoietic cells (6,37). Radiation initiates the apoptosis of tissue cells. ADSCs are also well known for their ability to inhibit apoptosis by regulating the proteins involved in apoptosis (32). In addition, the quantity of stem cells in adipose tissue is relatively large, and obtaining them is less invasive (25). Isolated cells could be easily grown in culture, retaining their properties over many passages (16). With these advantages, ADSCs have shown considerable promise in supporting hematopoietic reconstitution in clinical settings. Nevertheless, the underlying mechanism associated with the hematopoiesis-supporting properties of ADSCs has not been well defined.
Activation of the PI3K/Akt pathway has long been believed to be involved in hematopoiesis and megakaryopoiesis. Studies have well demonstrated the role of this pathway as a key regulator of megakaryopoiesis. It was reported that the PI3K/Akt pathway contributed to the thrombopoietin-induced proliferation of primary megakaryocytes in vitro and in vivo (38). The PI3K/Akt pathway is activated by growth factors and cytokines, resulting in phosphorylation of Akt, which upregulates Bcl-xL, leading to inhibition of megakaryocyte apoptosis (19), and p-Akt was significantly more highly expressed in myeloproliferative neoplastic (MPN) megakaryocytes (26), suggesting that Akt is a therapeutic target in MPN (21).
Although previous studies indicated that ADSCs have hematopoiesis-supporting properties and are efficacious in improving platelet levels in ITP mice (56), the effects of ADSCs on radiation-induced thrombocytopenia and megakaryopoiesis remain unknown. In this study, we hypothesized that ADSC administration would mitigate thrombocytopenia after radiation exposure, and we investigated the in vivo in irradiated mice impact of ADSCs on megakaryopoiesis and platelet recovery and whether these ameliorative effects were mediated by activation of the PI3K/Akt pathway.
Materials and Methods
Radiation-Induced Thrombocytopenia in Mice
Male balb/c mice (6-8 weeks old) weighing approximately 20 g were obtained from the experimental animal center of the medical academy. All of the animal experimental procedures were performed under the approval of Peking University People's Hospital's Animal Experiment Ethical Committee. Mice were placed in isolated chambers, and then received whole-body irradiation with a single dose of 4 Gy (dose rate of 1.48 Gy/min) using a fixed HFY-YC Co-60γ irradiator (REVISS, UK). After irradiation, 15 mice immediately received caudal vein injections with suspended ADSCs (1 × 106 in 0.2 ml saline for each) and another 15 mice with equivalent saline. Ten mice that were not irradiated served as a control group. All of the mice from three groups were euthanized using CO2 21 days postirradiation. For each experiment, five mice from each group were analyzed.
Isolation, Culture, and Characterization of ADSCs
The procedures were performed as previously reported, with some modifications (23,53). Briefly, aseptically harvested rat adipose tissue that was extensively washed with phosphate-buffered saline (PBS) was finely minced and was digested for 60 min at 37°C with type I collagenase (Sigma-Aldrich, St. Louis, MO, USA). The cell suspension was then neutralized with Dulbecco's modified Eagle's medium (DMEM; Gibco, grand Island, NY, USA) containing 10% fetal bovine serum and 100 U/ml penicillin/streptomycin. After being passed through a 70-ml filter and being centrifuged at 1,500 rpm for 5 min, the solution pellet was resuspended. The cell suspension was plated in 75-ml cell culture flasks (Corning, Corning, NY, USA), maintained at 37°C in a humidified 5% CO2 atmosphere with the medium (DMEM containing 10% FBS) changed every 2 days. ADSCs were passaged at 80% confluence with 0.25% trypsin (Gibco) at 37°C, and the cells were used for analysis after three to five passages. The BJ Human foreskin fibroblast cell line was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The media and reagents for culture and passage were similar to those used for the ADSCs. To elucidate the specific efficacy of the ADSCs in thrombocytopenia, fibroblasts were employed as the cell control.
Flow Cytometry
ADSCs at passage three were digested, washed with PBS, and incubated with antibodies against rat CD31-PE (BD Biosciences, Franklin Lakes, NJ, USA), CD34-PE (BD Biosciences), CD41-PE (BD Biosciences), CD44-PE (AbD Serotec, Oxford, UK), CD45-FITC (BD Biosciences), and CD90-PE (BD Biosciences), as well as with Igg1-PE (BD Biosciences) and Igg1-FITC isotype (BD Biosciences) for 30 min (5 μl antibody in 100 μl cell suspension, dilution rate 1:20); they were washed and centrifuged at 1,500 rpm for 5 min. Cells (5 × 105/ml) were suspended with PBS and analyzed by flow cytometry. Five mice in each group were sacrificed 21 days postirradiation. Bone marrow cells extracted from their femurs were stained with CD41-PE (BD Biosciences) and then treated as above.
Peripheral Blood Cell Counts
Peripheral blood was obtained from retro-orbital bleeding. Bleeding was performed on days 7, 14, and 21 for platelet and WBC analysis. The blood was dripped directly after removal into tubes containing EDTA (Solarbio, Beijing, China). The analysis of peripheral blood parameters was conducted by a contract laboratory within 2 h of being obtained.
Murine Colony-Forming Unit-Megakaryocyte Assay
Bone marrow (BM) mononuclear cells (2 × 105) were incubated in methylcellulose (StemCell Technologies, Vancouver, Canada) at a final concentration of 2.7% supplemented with 30% FCS, 8% 2-mercaptoethanol (Solarbio), 0.03% L-glutamine (Solarbio), 10 U of GM-CSF (Sigma-Aldrich), and 10% ConA-SCM (Sigma-Aldrich). Then 0.2-ml cell cultures were dropped onto a coverslip in a 35-mm glass dish and were incubated at 37°C in a fully humidified atmosphere of 5% CO2. On the eighth day, the numbers of CFU-MK and CFU-mix were scored using an inverted microscope. A CFU-MK was defined as a cluster of three or more cells that were positive for acetylcholinesterase (AChE) staining.
Bone Marrow Smear
Five mice in each group were sacrificed 21 days postirradiation. Bone marrow cells were vigorously flushed from the whole bone marrow cavity, including metaphysis and diaphysis of femurs, and made into bone marrow smears, then stained with Giemsa (Solarbio). Mature megakaryocytes were counted under a microscope based on cell sizes and their features according to the literature using Giemsa-stained slides derived from five independent experiments (60,62).
Bone Marrow Histology
Five mice in each group were sacrificed 21 days postirradiation. Femur samples were decalcified and fixed in 40% neutral formaldehyde (Solarbio) then were embedded in paraffin (Solarbio). Paraffin-embedded samples were sectioned at a thickness of 5 μm and were stained with hematoxylin and eosin (H&E) (Solarbio). Then we evaluated the histopathology and acquired photos under a microscope. The numbers of bone marrow cells in each picture (400×) were counted using Quantity One (Bio-Rad, Hercules, CA, USA). TUNEL staining was performed with the In Situ Cell Death Detection Kit (Roche Molecular Biochemicals, South San Francisco, CA, USA), in accordance with the manufacturer's instructions.
Western Blot
To detect the expression of p-AKT, Bcl-xL, and Bax, total protein extracted from bone marrow cells underwent electrophoresis on sodium dodecyl sulfate polyacrylamide gels (Solarbio) and then were transferred to nitrocellulose membranes and blocked with skim milk (Difco, Corpus Christi, TX, USA) dissolved in Tris-buffered saline solution (Solarbio). Phospho-Akt antibody (Cell Signaling, Danvers, MA, USA), Bcl-xL antibody (Cell Signaling), and Bax antibody (Cell Signaling) were incubated overnight at 4°C (dilution rate was 1:500). After washing, the membrane was incubated with secondary antibody (Santa Cruz, Santa Cruz, CA, USA) (dilution rate was 1:2,000), and then was visualized with ECL detection systems. Cyclophosphamide (CTX; Sigma-Aldrich) (19,61) was used to induce apoptosis in the bone marrow cells as a positive control for Bcl-xL and Bax Western blots. CTX was given intraperitoneally at a dose of 100 mg/kg. Twelve hours later, the mice were sacrificed, and bone marrow cells were flushed out using PBS; the protein in the bone marrow cells was evaluated using Western blot. A specific Akt activator SC79 (Sigma-Aldrich) (18,51) was used to induce Akt phosphorylation in the bone marrow cells as a positive control for p-Akt Western blots. SC79 was given intraperitoneally at a concentration of 40 mg/kg of body weight. Two hours later, the mice were sacrificed, and bone marrow cells were flushed out using PBS; the protein in bone marrow cells was evaluated using Western blot.
Statistics
SPSS software was used for general statistical analysis. All data were presented as the mean ± SD. Significant differences in the numerical data between the groups were determined by one-way ANOVA, after which a comparison between the groups was performed using the LSD test. Values of
Results
ADSC Morphology and Phenotype Identification
ADSCs obtained from the inguinal adipose exhibited a fibroblast-like morphology (Fig. 1A); they had the ability to self-renew and adhere to plastic, and they extensively expanded in culture. Four to five hours after culture, a few cells were observed that were dispersedly adherent on the bottom of the dish. The cells began to grow rapidly after 3 days with long spindles, small volume, and no direction. Morphological observation under a light microscope fit the characteristics of ADSCs. After the third passage, FACS analysis was performed to verify the phenotype. Fluorescence-activated cell sorter analysis showed that our cultured cells were positive for CD29 (99.79%) and CD90 (97.82%), rather than CD31 (3.08%), CD34 (1.27%), CD41 (0.61%), or CD45 (1.04%), and this phenotype was maintained for passages (Fig. 1B).
Morphology and phenotype of ADSCs. (A) Micrographs of cultured ADSCs at passage three. Scale bar: 50 μm. (B). Surface antigens of ADSCs were analyzed by flow cytometry. Cultured ADSCs were positive for CD29 (99.79%) and CD90 (97.82%), rather than for CD31 (3.08%), CD34 (1.27%), CD41 (0.61%), or CD45 (1.04%).
In Vivo Effects of ADSCs on Peripheral Blood Cell Counts in Irradiated Mice
On day 0, the basal number of peripheral blood platelets in experimental mice was 878.00 ± 99.68 × 109/L, which decreased after irradiation. The platelet counts of the irradiated mice reached a nadir (238.60 ± 60.25 × 109/L) on day 7, but recovered gradually. There were no significant differences in platelet counts between the saline group and ADSC group on day 0 or day 7. Recovery was better in the ADSC-treated group than in the saline group, and the ADSC-treated animals had significantly higher platelet counts on day 14 (367.41 ± 40.25 vs. 299.10 ± 36.22 × 109/L, ADSCs enhanced the recovery of peripheral blood cell counts in radiated mice. (A) PLT counts from 7 days to 21 days postirradiation in four groups. (B) WBC counts from 7 days to 21 days postirradiation in four groups. *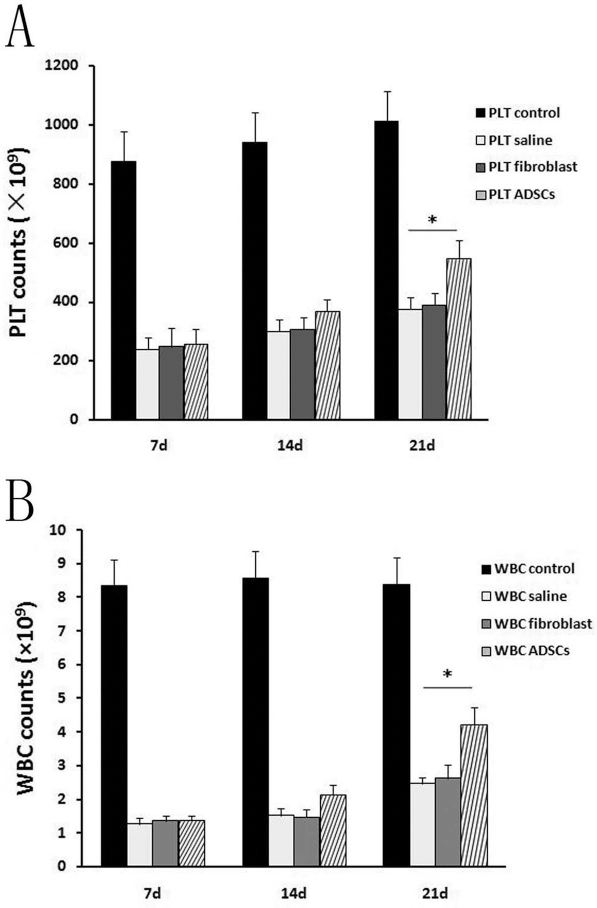
However, the RBC counts did not significantly change over the entire observation period.
Effects of ADSCs on Murine Bone Marrow Colony-Forming Unit (CFU) Formation
CFU formation assays were performed using the methylcellulose method. Consistent with peripheral blood cell counts, radiation markedly reduced total bone marrow CFUs (12.86 ± 2.15 vs. 56.30 ± 7.2 per 2 × 105 cells, ADSCs facilitated total bone marrow CFU formation and MK-CFU formation in irradiated mice. (A) ADSC administration significantly increased total bone marrow CFU formation 21 days postirradiation. (B) The number of bone marrow MK-CFUs significantly increased in the ADSC group 21 days postirradiation. *
ADSCs Specifically Protected Megakaryocytic Precursors From Irradiation
The analysis of Giemsa-stained cells extracted from the femurs of irradiated mice showed a strong reduction in megakaryocytes (0.88 ± 0.38 vs. 3.23 ± 0.89, ADSCs had positive effects on bone marrow megakaryocytes in irradiated mice. (A) Numbers of megakaryocytes counted on Giemsa-stained slides, derived from 10 random fields in four groups. (B) Flow cytometric analysis of CD41 (megakaryocytes) in bone marrow cells 21 days postirradiation. *
ADSCs Improved Cellularity and Reduced Apoptosis in Bone Marrow
We analyzed bone marrow sections in the three groups stained with hematoxylin and eosin to display bone marrow structure and cellularity or with TUNEL to reveal the presence of apoptotic cells. Hematoxylin and eosin staining revealed impaired hematopoiesis in irradiated mice, as evidenced by the low numbers of bone marrow cells (1,092 ± 357 vs. 1,812 ± 325 per field, ADSCs protected bone marrow from radiation-induced damage. (A) H&E and TUNEL staining of femur sections derived on day 21 from the four groups of mice. Images were obtained with 400× magnification. Scale bars: 200 μm. (B) Numbers of total bone marrow cells counted in 10 random fields in four groups. (C) Numbers of TUNEL-positive cells counted in 10 random fields in four groups. (D) ADSCs increased Bcl-xL protein expression in bone marrow cells. (E) ADSCs downregulated Bax protein expression in bone marrow cells after irradiation. *
ADSCs Exerted Radioprotective Effects via the PI3K/Akt Signaling Pathway
Bone marrow cells were extracted from the femurs of mice in the three groups. A Western blot was performed with antiphosphorylated Akt. The results showed increased expression of phosphorylated Akt in the bone marrow cells of the ADSC group mice compared with mice treated with saline alone (0.82 ± 0.06 vs. 0.19 ± 0.08, ADSC treatment increased the expression of p-Akt. (A) Representative images of p-Akt, Bcl-xL, and Bax expression detected by Western blot. (B) Quantification of p-Akt expression detected by Western blot. *
Discussion
In this study, we investigated the therapeutic efficacy of ADSCs in a murine model of radiation-induced thrombocytopenia. We reported that ADSCs effectively enhanced in vivo platelet recovery, facilitated megakaryopoiesis, and decreased the apoptotic bone marrow cells in irradiated mice. Our results further indicated that the antiapoptotic effect was likely to be mediated by activating the PI3K/Akt pathway.
Radiation can initiate a decrease in circulating blood cells, including platelet deficiency. It has been estimated that 70-80% of patients suffering from ARS had hemorrhage syndrome, in which antihemorrhage strategies were sorely needed to prolong patients' survival (52). Severe thrombocytopenia can also cause mortality postirradiation. Our study reported that 7 Gy of total body irradiation to mice could severely decrease circulating blood cells without recovery, and all of the animals died within 3 weeks. Mice receiving 4 Gy of total body irradiation also experienced a severe decrease in circulating platelets and leukocytes, with a nadir at 7 days postirradiation, and blood cells counts then began to recover gradually. However, different from platelets and leukocytes, circulating erythrocyte counts in irradiated mice did not vary over the entire observation period, likely due to the long half-life of erythrocytes. Bone marrow hematopoietic function had already recovered from irradiation before the depletion of circulating erythrocytes.
Platelets circulate for a relatively shorter time of approximately 10 days in peripheral blood (9); thus, thrombocytopenia appeared on day 7 after irradiation in our study. By the 21st day, the efficacy of the ADSC treatment was observed, so we selected that as the time point for the animal sacrifice and analysis. Although recent studies have suggested that radiation-induced thrombocytopenia plays an important role in radiation mortality, research focusing on the mitigation of radiation-induced thrombocytopenia has rarely been reported, and patients require transfusions of platelet concentrations repeatedly.
Mesenchymal stem cells (MSCs) have emerged as a new therapeutic candidate for the reconstituting of the hematopoietic microenvironment, by improving engraftment in stem cell transplantation. MSCs are the main cell type involved in tissue repair. When a tissue injury occurs, MSCs migrate to the location of the damage where they participate in tissue repair (55). It is widely accepted that the bone marrow is a primary residence to which systemically infused MSCs would home to. Although hMSCs mainly distribute in the lung and liver in the short term after intravenous infusion (1,13), for the long-term distribution, MSCs were shown to engraft mainly in the bone marrow (34). Studies have shown that systemically infused MSCs home to the bone marrow and remain there for 4-13 months postinfusion (1). Furthermore, MSCs preferably home to sites of injury and inflammation (45). They have chemotactic properties similar to other immune cells that respond to injury and sites of inflammation (45,47). Irradiation causes substantial damage and inflammatory reaction to the bone marrow, thus activating molecular pathways that increase the release of tissue chemokines, which attract MSCs to injured tissues (11). It has been reported that total body irradiation induced a 2.6-fold increase in the engraftment of MSCs to the bone marrow, which is a primary residence (11). Considering the fact that MSCs home to the bone marrow and that radiation-induced injury increases the engraftment of MSCs, it is reasonable to infer from the present study that homing and engraftment of MSCs to injured bone marrow may mediate the repair and regeneration in the bone marrow after irradiation.
As for the therapeutic mechanism of MSCs, previous studies tended to consider differentiation as the primary way that these cells promote tissue repair. However, with increasingly deeper insight, it has been accepted that differentiation and paracrine mechanisms cooperate in the promotion of tissue repair; paracrine mechanisms are thought to be even more crucial (55). ADSCs can secrete hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), epidermal growth factor, MMP family proteins, and many other cytokines that are used to repair injured tissue (4,40,52). Among the main cytokines ADSCs produce, VEGF and HGF have been proven to play a beneficial role in hematopoiesis. VEGF is indispensable for HSC survival, mediated by a cell-intrinsic autocrine mechanism (14). VEGF can maintain hematopoiesis and protect hematopoietic stem cells from hypoxia (44). VEGF can induce survival of hematopoietic progenitors through the activation of PI3K and Erk MAPK pathways (28). Another cytokine, HGF, which can be secreted by ADSCs, has been demonstrated to increase peripheral blood cell counts, maintain hematopoiesis, and induce survival after irradiation (29). Meanwhile, research has shown that ADSCs can express chemokine CXCL12, a critical regulator of hematopoiesis, to improve hematopoiesis in bone marrow (37).
MSCs can be isolated from bone marrow and less invasive tissues (such as adipose, gingiva, muscle, and umbilical cord) with similar functional effects (2). Among various mesenchymal stem cell lines, ADSCs appear to be a preferable source for cell-mediated therapy. ADSCs are highly self-renewing multipotent mesenchymal cells (63) that are abundantly available and can easily be harvested (30). Furthermore, ADSCs have been proven to have a profound impact on the improvement of hematopoiesis (6); moreover, ADSCs possess greater capacity to facilitate hematopoiesis and reconstitute the hematopoietic microenvironment than BMSCs in mice (37,41). In vitro coculture and progenitor assays showed that ADSCs generated significantly more granulocytes and progenitor cells from human hematopoietic stem cells (HSCs) than BMSCs (37). It was also revealed that ADSCs express the chemokine CXCL12, a critical regulator of hematopoiesis, at levels that are threefold higher than those with BMSCs (37). Moreover, in vivo cell homing assays indicated that ADSCs facilitated the homing of mouse HSCs to bone marrow better than BMSCs (37). Further study in human ADSCs demonstrated that ADSCs have a higher population doubling number than BMSCs in proliferation studies (41). In vitro coculture assays showed that ADSCs not only support human CD34+ peripheral blood stem cells (PBSCs) but also yield significantly more nonadherent hematic cells than BMSCs (41). In vitro progenitor assays revealed that ADSCs promote a higher frequency of early progenitors than BMSCs (41). Even more, ADSCs had a superior effect in improving bone marrow cellularity in an in vivo study (41). Therefore, ADSCs are one of the best candidate cell lines to use as vector cells to rescue radiation-induced thrombocytopenia.
Despite research reporting the hematopoiesis-supporting properties of ADSCs, few publications are available concerning the megakaryopoietic activity of ADSCs. Megakaryopoiesis involves the commitment of hematopoietic stem cells, proliferation and terminal differentiation of megakaryocytic progenitors, and maturation of megakaryocytes to produce functional platelets (7). Thrombopoietin (TPO) is the primary physiological and most potent cytokine for stimulating all stages of megakaryocytopoiesis and thrombopoiesis (20,24). In addition, the mimetics of TPO have been shown to interact with the transmembrane domain of the TPO receptor, initiating the activation cascades of several signaling transduction pathways, resulting in MK proliferation and maturation, followed by increased platelet production (10,22,48). Although studies have reported the hematopoiesis-supporting properties of ADSCs, few publications are available concerning the megakaryopoietic activity of ADSCs. The work of De Toni and colleagues revealed that CD34+ cells cocultured with ADSCs in vitro differentiated into myeloid and lymphoid lineages, including megakaryocytes (6). However, they did not perform further research in vivo or specifically verify the protective potential of ADSCs in megakaryopoiesis. In this study, we found that ADSC did not significantly alter megakaryocytopoiesis in the early stage (days 7 and 14). ADSCs start improving megakaryocytopoiesis from day 21. The effects of ADSCs in improving megakaryopoiesis after radiation were first confirmed by these cells' ability to increase CD41+ cells and to promote MK-CFU formation in the bone marrow of irradiated mice.
In this study, we showed that ADSC administration had positive effects on peripheral blood cell recovery in irradiated mice, which might have been partly due to the ability of ADSCs to promote antiapoptosis and to improve regeneration.
ADSCs have been well documented for their potential to reduce apoptosis in a broad spectrum of cells, including cerebral, renal, and myocardial tissue cells (31,54,57). The Bcl-2 (B-cell lymphocytic-leukemia proto-oncogene) family, which consists of two groups of proteins with distinct functions in cell fate signaling, is well known as a group of important regulators of apoptosis. Members of the Bcl-2 family are functionally classified into either proapoptotic or antiapoptotic members. Among these family members, Bcl-xL is one of the major members of the antiapoptotic Bcl-2 proteins discovered to date, while Bax is considered to play a crucial role in proapoptotic Bcl-2 proteins (27). However, their regulatory roles in ADSC-mediated therapeutic potency in irradiated mice have not ever been demonstrated. In our study, we for the first time confirmed that ADSC administration could markedly decrease apoptosis by increasing the protein expression of Bcl-xL and by downregulating that of Bax in the bone marrow hematopoietic tissue of irradiated mice.
PI3K pathway activation is often involved in the inhibition of apoptosis. Primary mouse hepatocytes were protected against high glucose-induced apoptosis by activating the PI3K/Akt pathway (17). Furthermore, recent evidence has indicated that inhibition of the PI3K/Akt pathway primed cancer cells for apoptosis by tipping the balance toward proapoptotic Bcl-2 proteins (12). The PI3K signaling also acts as a key regulator of hematopoiesis (43). Bartucci et al. showed that activation of the PI3K/Akt pathway participated in protecting megakaryocytes from cisplatin (3). Moreover, Ye et al. demonstrated that platelet-derived growth factor enhanced platelet recovery in a murine model of radiation-induced thrombocytopenia and reduced the apoptosis of megakaryocytes with subsequent activation of the PI3K/Akt pathway (58). However, whether this typical pathway participates in the therapeutic process of ADSCs in tissue repair was not well defined. In our study, we showed that ADSC administration significantly upregulated the expression of phosphorylated Akt, which could subsequently result in the mitigation of radiation-induced thrombocytopenia. Nevertheless, Akt plays a pivotal role in this complicated pathway, and the downstream signaling molecules specifically modulated by the therapeutic potential of ADSCs remain unknown; as such, further studies are needed.
In conclusion, we demonstrated that ADSCs were capable of promoting platelet recovery, improving megakaryopoiesis, and inhibiting the apoptosis of hematopoietic cells in irradiated mice, and this protective potential of ADSCs is likely to be mediated via the PI3K/Akt pathway, thus providing a novel potential therapeutic alternative for mitigating radiation-induced thrombocytopenia.
Footnotes
Acknowledgments
