Abstract
Stroke is a result of cerebral ischemia that triggers a cascade of both physiological and biochemical events. No effective treatment is available for stroke; however, stem cells have the potential to rescue tissue from the effects of stroke. Adipose-derived stem cells (ADSCs) are an abundant source of adult stem cells; therefore, ADSC therapy can be considered as a future strategy for regenerative medicine. However, more research is required to improve the effectiveness of transplanted ADSCs as a treatment for stroke in the mouse stroke model. Ligustilide, isolated from the herb Angelica sinensis, exhibits a protective effect on neurons and inhibits inflammation. We also demonstrated that ligustilide treatment increases the expression levels of homing factors such as SDF-1 and CXCR4. In the present study, we evaluated the therapeutic effects of ADSC transplantation and ligustilide treatment in a mouse thromboembolic stroke model by behavioral tests, including beam walking, locomotor activity, and rotarod analysis. ADSCs pretreated with ligustilide were transplanted into the brains of stroke mice. The results showed that the therapeutic effect of ADSCs pretreated with ligustilide was better than that of ADSCs without ligustilide pretreatment. There was no difference between the recovery of mice treated by ADSC transplantation combined with subcutaneous ligustilide injection and that of mice treated only with ADSCs. The TUNEL assay showed fewer apoptotic cells in the brains of mice transplanted with ADSCs pretreated with ligustilide as well as in those without pretreatment. In summary, pretreatment of ADSCs with ligustilide improves the therapeutic efficacy of ADSC transplantation. The results of this study will help improve stem cell therapies being developed for future clinical applications.
Introduction
Stroke occurs in ~800,000 people in the US each year and was the second most common cause of death worldwide in 2011 according to the World Health Organization (19). Stroke is the result of failure of blood flow to the brain due to thrombosis or bleeding. This leads to infarction of the affected region resulting in long-term deterioration of health in stroke patients. Management of stroke patients accounts for 4% of the total healthcare costs worldwide, and there exists no effective therapy for stroke (4). In recent years, stem cell therapy and the field of regenerative medicine have provided new hope in the search for therapies against various diseases. Stem cell transplantation in stroke patients has since become an attractive focus of research for developing a promising therapy (1). Various types of stem cells are considered as potential candidates for cell replacement or promoting functional recovery after stroke (7). This includes embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and mesenchymal stem cells (MSCs) isolated from adult tissues. ESCs and iPSCs have a huge differentiation capacity, but ethical problems surrounding the use of ESCs and the high cost of iPSC production render them less favorable candidates for developing stroke therapies (18). MSCs can be isolated from various tissues such as bone marrow and adipose tissue. While bone marrow stem cells (BM-MSCs) have been studied for decades, isolation of BM-MSCs requires a painful procedure and results in low stem cell yield (22,34).
Adipose-derived stem cells (ADSCs) have emerged as one of the most preferred research targets because of their abundance as well as the accessibility of adipose tissue (5). While both BM-MSCs and ADSCs were shown to improve functional recovery after stroke, ADSCs have better proliferation and differentiation abilities compared to BM-MSCs (6,13,31). ADSCs have the ability to differentiate into multiple cell lineages, including osteocytes, chondrocytes, myocytes, adipocytes, and neuronal cells (33). The therapeutic effect of ADSCs in treating various forms of brain damage as well as neuronal diseases has been investigated in recent years (30). ADSCs are capable of reducing symptoms of brain diseases by providing protection to neurons both directly through cell replacement as well as indirectly through release of factors from their secretome (30). ADSCs transplanted into the brain express neurotrophic factors and protect neuronal cells from apoptosis (23). Furthermore, some factors secreted by ADSCs are capable of decreasing protein aggregates in brain tissue as well as within neurons (16). Although ADSC transplantation has shown great potential and therapeutic effects (30), researchers are still seeking further improvements to utilize ADSCs more efficiently and effectively.
Angelica sinensis is an herb indigenous to China and is the source of a well-known herbal medicine. The dried root of A. sinensis, commonly known as Danggui in Chinese medicine, is thought to have therapeutic benefits in several diseases (9,10,27). Additionally, Danggui and its extract have been shown to possess anti-inflammatory, antioxidant, and neuroprotective effects (12,27,29). Several chemical compounds have been identified and isolated from Danggui, including phthalides, organic acids, and polysaccharides (28). Ligustilide is a phthalide compound that was first identified in Ligusticum chuanxiong and is an active ingredient of the A. sinensis extract. Its metabolic mechanisms have been investigated previously (26). Furthermore, it is known to inhibit and attenuate inflammation in cell culture and animal models (11,17,32), provide neuroprotection against brain injury, and promote neurogenesis (17,21,25). Based on current evidence, we postulate that ligustilide has the potential to provide protection for brain cells in the poststroke state and increase speed of recovery. We also postulated that ADSC transplantation combined with ligustilide treatment may provide synergistic therapeutic effects. We further speculated that the possible synergism in combined treatment would likely improve recovery efficiency compared to current poststroke therapeutic methods.
In the current study, we investigated the potential for neuroprotection as well as the therapeutic effect of ADSCs on an acute thromboembolic stroke model. We induced a middle cerebral artery occlusion (MCAo) by injecting thrombin into the MCA in BALB/c mice. To minimize the physical burden on target mice, ADSCs were then transplanted intracerebroventricularly following induction of MCAo, as previously described by Ding and colleagues (3). The therapeutic effect of ADSCs was evaluated by monitoring recovery from stroke over a period of 14 days. We also evaluated the therapeutic effect of ligustilide when administered via subcutaneous injection as well as when used in combination with ADSCs by pretreating ADSCs with ligustilide for 24 h before ADSC transplantation. We combined different ligustilide and ADSC treatments and compared their effect with those of independent treatments. The recovery of mice was evaluated by observing various behavior parameters in the target mice, including coordination, balancing, and overall activity. The effectiveness of various ADSC and/or ligustilide treatments was evaluated by comparing the recovery of target mice receiving different treatments. We also evaluated the antiapoptotic effect of different treatments on brain tissue.
Materials and Methods
Study Methodology
The study was performed in accordance with the guidelines of the Institutional Review Board (IRB) of China Medical University and Hospital Research Ethics Committee and the TFDA, Ministry of Health and Welfare, Taiwan. The original protocol was approved by China Medical University and Hospital Research Ethics Committee (CMUH102-REC1-058). All donors gave written informed consent. Experimental protocols were also approved by the Institutional Animal Care and Use Committee of China Medical University (101-53-N).
Isolation and Characterization of ADSCs
ADSC isolation and characterization were performed as in the previous study by Harn et al. (8). In brief, human ADSCs were harvested from the subcutaneous fat of the abdominal wall during gynecological surgery (2–5 g). Adipose tissue samples were collected in Ca2+/Mg2+-free phosphate-buffered saline (PBS) (Gibco, Grand Island, NY, USA) and immediately transferred to the Stem Cell R&D Center of Gwo Xi Stem Cell Applied Technology Co., Ltd. The tissues were dissected into pieces of 1- to 2-mm3 size and dissociated with 0.1% collagenase I (Invitrogen-Gibco) for 60 min at 37°C. The dissociated cells were collected and cultured at 37°C in a humidified incubator supplied with 5% CO2 in keratinocyte-SFM (serum-free medium) (Gibco) containing bovine pituitary extract and epidermal growth factor (EGF) (Gibco), N-acetyl-L-cysteine (2 × 10–3 M; Sigma-Aldrich, St. Louis, MO, USA), L-ascorbic acid 2-phosphate sesquimagnesium salt hydrate (2 × 10–4 M; Sigma-Aldrich), and fetal bovine serum (FBS, 10%; Hyclone, Logan, UT, USA). The supernatant and debris were removed from culture after 2 days of incubation. The remaining ADSC culture was designated as passage zero. Surface markers were labeled by antibodies against CD14, CD29, CD44, CD45, human leukocyte antigen (HLA)-ABC (all from Dako, Carpinteria, CA, USA), CD34, CD49b, HLA-DR (all from Becton Dickinson, Franklin Lakes, NJ, USA) (1:50 dilutions). Cell samples were analyzed using a flow cytometer (LSR II; BD Biosciences, San Jose, CA, USA).
Culturing Adipose-Derived Stem Cells
ADSCs were cultured in keratinocyte-SFM medium containing bovine pituitary extract and EGF (Gibco) at 37°C in a humidified incubator supplied with 5% CO2. The culture medium was supplemented with I-acetyl-L-cysteine (2 × 10–3 M; Sigma-Aldrich), L-ascorbic acid 2-phosphate sesquimagnesium salt hydrate (2 × 10–4 M; Sigma-Aldrich), and FBS (10%; Hyclone).
Ligustilide Treatment and MTT Cell Viability Assay
Ligustilide (1 mg/μl) used in this study was synthesized by Pharmaron, Inc. (Beijing, China). Ligustilide was diluted to 100 μg/μl by mixing with 15% Kolliphor EL (Sigma-Aldrich), 15% filtered ethanol (Sigma-Aldrich), and phosphate-buffered saline (PBS). Further dilutions were prepared using PBS or culture medium. The MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay (Sigma-Aldrich) was performed by incubating ADSCs with the following concentrations of ligustilide for 24 and 48 h: 0.625, 1.25, 2.5, 5, 10, 20, and 40 μg/ml.
RT-PCR Analysis
ADSCs were treated with 0.625, 1.25, 2.5, and 5 μg/ml of ligustilide for 24 h, followed by RNA extraction using TRIzol (Life Technologies, Gaithersburg, MD, USA). Complementary DNA (cDNA) was reverse transcribed from extracted RNA using a SuperScript III Reverse Transcriptase Kit (Invitrogen, Carlsbad, CA, USA). Endpoint polymerase chain reaction (PCR) was performed using EX-taq polymerase (Takara Inc., Otsu, Japan) to determine gene expression of brain-derived neurotrophic factor (BDNF) (F: GAGCTGAGCGTGTGTGACAG, R: ACTGGGTAGTTCGGCACTGG), nuclear receptor related 1 (Nurr1) (F: CGACATTTCTGCCTTCTCC, R: GGTAAAGTGTCCAGGAAAAG), C-X-C motif receptor 4 (CXCR4) (F: GGCCCTCAAGACCACAGTCA, R: TTAGCTGGAGTGAAAACTTGAAG), stromal cell-derived factor 1 (SDF1αβ) (F: ATGAACGCCAAGGT CGTGGTC, R: CTTGTTTAAAGCTTTCTCCAGGTA CT), interleukin-6 (IL-6) (F: TGCCAGCCTGCTGACG AAGC, R: TCTGTGCCCAGTGGACAGGT), IL-8 (F: GC TGGCCGTGGCTCTCTTGG, R: TCCACAACCCTCT GCACCCA), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (F: GGAGCCAAACGGGTCATCAT CTC, R: GAGGGGCCATCCACAGTCTTCT). mRNA levels for each gene were compared to the expression levels of the housekeeping gene GAPDH.
Flow Cytometry Analysis
Flow cytometry analysis was performed on an LSR II flow cytometer (BD Biosciences). In order to characterize ADSCs and identify possible cell differentiation, dissociated ADSCs were stained with antibodies against CD44 (BD Biosciences) and CD105 (Invitrogen). Dissociated ADSCs cells were suspended in a 1:50 dilution of phycoerythrin-conjugated anti-human CD44 antibody (BD Pharmingen, San Jose, CA, USA) or a 1:50 dilution of phycoerythrin-conjugated anti-human CD105 antibody (Invitrogen).
Animal Treatment and Anesthetization
BALB/c mice were obtained from the Taiwan National Laboratory Animal Center (NLAC) (Taipei, Taiwan) and maintained in individually ventilated cages at the Laboratory Animal Service Center (LASC). The Institutional Animal Care and Use Committee of the China Medical University approved all animal treatments and experimental procedures. Mice were anesthetized 10 min before experiments by intraperitoneal (IP) administration of 10 mg/ml Zoletil (VIRBAC, Carros, France) in 0.04% xylazine (Health-Tech Pharmaceutical, Taoyuan, Taiwan) at a dose of 0.08 ml per 10 g bodyweight.
MCA Occlusion and Stroke Induction
Craniotomy was performed on 8-week-old mice with a bodyweight around 25 g, as previously described (20). Briefly, anesthetized mice were fixed on the stereotaxic instrument and microsurgery was performed carefully to expose the MCA by partial removal of the right parietal bone. Stroke was induced by carefully injecting 1 μl of thrombin (Sigma-Aldrich) into the MCA. During the injection of thrombin, blood flow inside of the MCA was recorded using a laser Doppler scan to confirm clot formation.
ADSC Transplantation
Within 2 h of stroke induction, ADSCs were transplanted into the brains of mice as described previously by Ding and colleagues (3). Briefly, 1 × 106 ADSCs were first stained with Hoechst (Invitrogen), which was followed by intracerebral (IC) injections of 18 μl of ADSCs (1 × 106 in saline; Taiwan Tech Co., Taoyuan, Taiwan) at three different locations.
Neuronal Behavior of Mice Before and After Stroke Induction
Neuron-dependent behavior of mice was evaluated by performing a locomotor activity test, a beam-walking balance test, and a rotarod coordination test. Locomotor activity of mice was monitored within an eight-channel locomotor activity box for 1 h. Horizontal and vertical movement and the distance traveled in 30 min were analyzed. The beam-walking balance test was performed by recording the time it took for the mice to cross an 80-cm beam and the rear foot slip frequency while crossing. Recording the duration for which the mice could stay stable on a rod rotating at 5 rpm constantly over a 3-min time period was used as the parameter for the rotarod coordination test. Data from at least three independent experiments were used for statistical analysis.
Brain Histology and TUNEL Assay for Apoptosis in Cortex
Anesthetized mice were perfused through the heart with PBS, followed by 4% paraformaldehyde (PFA; Sigma-Aldrich). The brains were harvested and incubated first in 4% PFA for 4 h. Samples were then incubated for 16 h in a 20% sucrose/PBS solution and again for 16 h in a 30% sucrose/PBS solution. Frozen sections were prepared from the brain samples collected. The terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay was carried out using an In Situ Apoptosis Detection Kit (Roche, Nutley, NJ, USA) according to the manufacturer's instructions. TUNEL-positive cells were identified using a fluorescence microscope (Zeiss, Oberkochen, Germany) at 450- to 500-nm excitation and 515- to 565-nm emission wavelengths. The number of positive cells within a defined area in the cortex was recorded and compared with those of the control mice. Hematoxylin and eosin (H&E) staining was performed by incubating tissue sections with each dye for 1 min and with a gentle water flow for 5 min in between.
Statistical Analysis
Results from all experiments were presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) and Tukey's post hoc test were performed to compare the means among different treatments using SigmaPlot (Systat Software, Inc., San Jose, CA, USA). A value of p < 0.05 was considered statistically significant.
Results
MTT Assay for Ligustilide-Treated ADSCs
We investigated the therapeutic effect of ligustilidetreated ADSCs in the mouse stroke model. To determine whether the ligustilide concentration interfered with cell growth, we performed the MTT assay. Viability of ADSCs was evaluated in different concentrations of ligustilide over 24 and 48 h (Fig. 1A, B). ADSCs showed significantly decreased viability after treatment with 40 μg/ml ligustilide for 24 h (Fig. 1A). ADSC viability also decreased at lower ligustilide concentrations of 10 and 20 μg/ml after incubation for 48 h (Fig. 1B). These results indicated that higher ligustilide concentrations have a significant negative effect on ADSC growth.

Effect of ligustilide treatment on the viability, morphology, and gene expression pattern of ADSCs. (A) Cell viability decreases at higher concentrations of ligustilide. Cell viability was evaluated by MTT assay after 24 h of treatment with various concentrations of ligustilide. (B) Cell viability decreased during prolonged incubation (48 h) with ligustilide at a concentration of 10 μg/ml and above. **p < 0.01 versus control (0 μg/ml ligustilide). (C) Expression of BDNF, Nurr1, CXCR4, and SDF1αβ was upregulated while expression of IL-6 and IL-8 was downregulated after ADSC treatment with increasing concentrations of ligustilide. GAPDH was used as the internal control. (D) CD44 and CD105 surface markers were present in both untreated and pretreated ADSCs. Flow cytometry analysis was performed based on data from three individually performed experiments. (E) Cell morphology was not affected by HK-002 (ligustilide) treatment. Cells treated with or without ligustilide for 24 h were observed using light microscopy.
Ligustilide Pretreatment of ADSCs Increased Expression Levels of Homing Factors Without Changing Cell Characteristics
Since higher ligustilide concentrations decreased the viability of ADSCs, we evaluated the effect of ligustilide concentrations below 5 μg/ml on the expression levels of differentiation, homing, and inflammation-related genes. Gene expression levels were evaluated by RT-PCR amplification of total mRNA extracted from ADSCs followed by gel electrophoresis. Expression levels of the neuronal differentiation-promoting and homing genes Nurr1, BDNF, CXCR4, and SDF1αβ as well as immunodifferentiation- and inflammation-promoting genes IL-6 and IL-8 (Fig. 1C) were evaluated. Our results suggested that there was an increase in the expression of four neuronal differentiation-promoting and homing genes (Nurr1, BDNF, CXCR4, SDF1αβ) for both 2.5 and 5 μg/ml ligustilide. At the same ligustilide concentrations, a decrease in the expression of the immunity- and inflammation-promoting IL-6 and IL-8 genes was observed. Taken together, the results from cell viability and gene expression studies suggest that 2.5 μg/ml ligustilide is the optimal concentration for treating ADSCs in order to prevent negative effects on cell growth while increasing the expression of neuronal differentiation-promoting genes and suppressing the immune response. In order to confirm that ligustilide pretreatment does not change ADSC characteristics, we further compared ADSC characteristics with or without pretreatment in flow cytometry analysis using CD44 and CD105 cell surface markers. Both markers were present in 99% of ADSCs regardless of whether or not cells were pretreated (Fig. 1D). We further examined the morphology of treated cells using bright-field microscopy. The morphology was no difference between untreated cells and cells treated with ligustilde for 24 h (Fig. 1E). The result suggested that cells exposed to 2.5 μg/ml ligustilide pretreatment did not significantly differ from those not pretreated.
Induction of Thromboembolic Stroke in Mice
We used the mouse thromboembolic stroke model to investigate the therapeutic effect of ADSCs and ligustilide pretreatment. Induction of stroke following thrombin injection into the MCA was monitored using laser Doppler flowmetry (Fig. 2B). Blood flow in the MCA decreased drastically after injection, indicating successful induction of stroke and subsequent MCA infarction. Mouse neuronal behavior was monitored for 1 day before thrombin injection. After stroke induction and ADSC treatment, changes in behavior were recorded until day 14. In addition, the direct effect of ligustilide on thromboembolic stroke was evaluated by injecting ligustilide subcutaneously poststroke in mice (Fig. 2A).

Schematic representation of the experimental design and identification of thrombosis. (A) Schematic of experimental design showing the timeline of behavioral monitoring, induction of thrombosis, cell transplantation, and ligustilide treatment. (B) Effect of thrombin injection was monitored by laser Doppler flowmetry; the arrow indicates injection and the drop in blood flow after thrombosis.
Subcutaneous Injection of Ligustilide Improves Poststroke Behavioral Recovery
Ligustilide is known to exhibit anti-inflammatory, antioxidant, and neuroprotective effects (12,27,29). To understand whether direct treatment with ligustilide in mice can improve poststroke recovery, we treated stroke-modeled mice with subcutaneous injections of 30 or 90 mg/kg body weight ligustilide. Neuronal behavior was then observed for 14 days after stroke (Fig. 3). During the 14-day poststroke period, stroke mice treated with both 30 and 90 mg/kg ligustilide showed a significant improvement in their coordination ability during rotarod analysis (Fig. 3A). The balancing ability (beam-walking analysis) of stroke mice treated with ligustilide improved within 3 days, and improvement continued until 14 days poststroke (Fig. 3B). Mice treated with 30 mg/kg ligustilide performed the best among all groups in the beam-walking analysis and locomotor activity test (Fig. 3C, D). Stroke mice injected with saline did not show any observable recovery in neuronal behavior over time, except an improvement in overall activity (Fig. 3).

Ligustilide treatment improved poststroke recovery. (A) Stroke induction significantly decreased coordination ability of mice when monitored on the rotarod device; there was a significant improvement in coordination ability following ligustilide treatment. The results at day 14 were quantified and compared as a bar graph. (B) Balancing ability of mice was impaired after thromboembolic stroke, and this ability was rescued by ligustilide treatment. Results at days 3 and 14 were compared. (C) Similar results were observed in the beam-walking test. The results at day 14 were compared. (D) Overall activity of mice was reduced after stroke and faster recovery occurred when they were treated with ligustilide, as determined by total traveling distance and total moving time within a locomotor chamber. Error bars represent mean ± SD, n ≥ 3. * p < 0.05 versus stroke + saline group.
ADSC Transplantation Combined with Ligustilide Injection Improves Poststroke Recovery
ADSCs have the ability to differentiate into neuronal cells. To investigate whether ADSCs have any therapeutic effect on MCA infarction, we transplanted ADSCs into the brains of stroke mice and observed their recovery. Furthermore, we compared the therapeutic effect of ADSC transplantation to ADSC transplantation combined with subcutaneous ligustilide injection (Fig. 4). The rotarod coordination test showed that ADSC transplantation with or without ligustilide cotreatment significantly improved poststroke motor coordination (Fig. 4A). Beam-walking analysis showed that the balancing ability of stroke mice transplanted with ADSCs was significantly higher than that of stroke mice injected with saline (Fig. 4B, C). The recovery of balancing ability started as early as day 3 poststroke, and mice treated with a combination of ADSC transplantation and 30 mg/kg ligustilide injection had better recovery during this early stage compared to treatment with higher doses of ligustilide (Fig. 4B). An improvement in locomotor activity of stroke mice was also observed following treatment with a combination of ADSC transplantation and ligustilide injections (Fig. 4D).
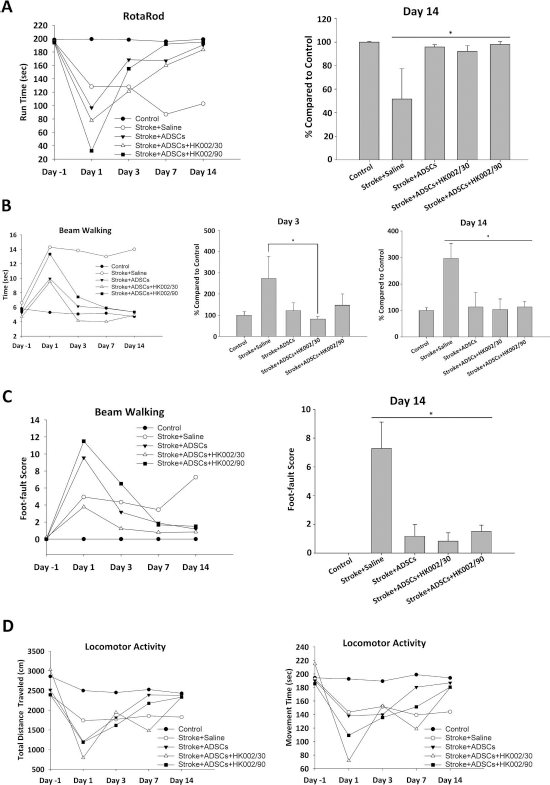
ADSC transplantation and ligustilide treatment accelerated poststroke recovery. (A) Coordination ability of mice showed faster recovery both in ADSC and ADSC + ligustilide treatment groups. The results at day 14 were quantified and compared. (B) Balancing ability of stroke mice showed better recovery when treated with ADSCs or ADSCs + ligustilide. Mice treated with ADSCs + 30 μg/ml ligustilide recovered faster (within 3 days) after treatment. Results from days 3 and 14 were compared in a bar graph. (C) Defects in balance after stroke induction were rescued by ADSC and ADSC + ligustilide treatments. The results at day 14 were compared. (D) Overall activity reduction poststroke recovered faster with ADSC or ADSC + ligustilide treatment. Error bars represent mean ± SD, n ≥ 3. *p < 0.05 versus stroke + saline group.
Pretreating ADSCs with Ligustilide Improves Therapeutic Effect of Cell Transplantation Therapy
Since both ligustilide and ADSC transplantation improved poststroke recovery independently as well as synergistically, we next investigated whether treating ADSCs with ligustilide before transplantation could improve the therapeutic effect of stem cell transplantation. ADSCs were treated with ligustilide (2.5 μg/ml) for 24 h prior to transplantation into stroke mice. Similar to the results of ADSC and ligustilide treatment, pretreating ADSCs with ligustilide improved neuronal behavior recovery of stroke mice (Fig. 5). The coordination ability (rotarod) of stroke mice improved significantly in mice transplanted with ADSCs as well as ligustilide-pretreated ADSCs, and there was no significant difference between the two groups (Fig. 5A). However, improvement of balancing ability (beam walking) was significantly higher at 3 days as well as 14 days poststroke when mice were transplanted with ligustilide-pretreated ADSCs (Fig. 5B, C). Similar results were obtained by the locomotor activity test (Fig. 5D).

Ligustilide pretreatment of ADSCs improved efficiency of short-term recovery. (A) Coordination ability of mice poststroke was rescued by both ADSC and pretreated ADSC transplantation. The results at day 14 were quantified and compared in a bar graph. (B) Pretreated ADSC transplantation improved balance ability more significantly at day 3 compared to that seen for the normal ADSC group. Results at days 3 and 14 were compared in a bar graph. (C) Defects in balance were rescued by both ADSC and pretreated ADSC transplantation. Results at day 14 were compared. (D) Reduction in overall activity was improved in both ADSC transplantation groups. Error bars represent mean ± SD, n ≥ 3. *p < 0.05 versus stroke + saline group.
Pretreated ADSC Transplantation Combined with Ligustilide Injection Improves Poststroke Recovery
Ligustilide treatment and ADSC transplantation together as well as transplantation of ligustilide-pretreated ADSCs yielded promising poststroke recovery. Therefore, we investigated whether combining pretreated ADSCs with ligustilide treatment could provide further improvement in poststroke recovery (Fig. 6). Our results showed that a combination of pretreated ADSCs and subcutaneous ligustilide injection had similar poststroke recovery as other treatment methods tested. Although transplantation of pretreated ADSCs combined with 30 mg/kg ligustilide injection showed a slight improvement in all behavior tests, this improvement was not statistically significant (Fig. 6).

ADSC or pretreated ADSC transplantation combined with ligustilide treatment improved poststroke behavior. (A) Loss of coordination ability poststroke was rescued by ADSC and ligustilide treatments individually as well as all of the combination treatments. (B, C) Defects in balance were improved by ADSC and ligustilide treatments individually as well as all of the combination treatments. (D) Rescue in reduction of overall activity was equal in all treatment groups. Error bars represent mean ± SD, n ≥ 3. *p < 0.05 versus stroke + saline group.
Ligustilide Treatment and ADSC Transplantation Protects Brain Cells From Apoptosis, While no Infarction Could Be Observed at the Endpoint
We demonstrated that ligustilide treatment, ADSC transplantation, and transplantation of ligustilide-pretreated ADSCs all improved poststroke neuronal behavior recovery. Finally, we investigated the apoptosis rate of these therapies in brain tissue and cells. Brains of stroke mice were harvested, preserved, and dissected after behavioral monitoring was complete. Brain tissue was then used for TUNEL staining to detect apoptosis (Fig. 7A, B). Apoptosis of cells within the cortex decreased significantly following treatment with any of the therapies tested in the present study (Fig. 7A, B). We further attempted to observe the infarct area (Fig. 7C). We were unable to identify any infarction in mice, including those without any treatment. This lack of infarction was possibly due to the fact that the brain was harvested at day 14, which served as the endpoint of experiments.
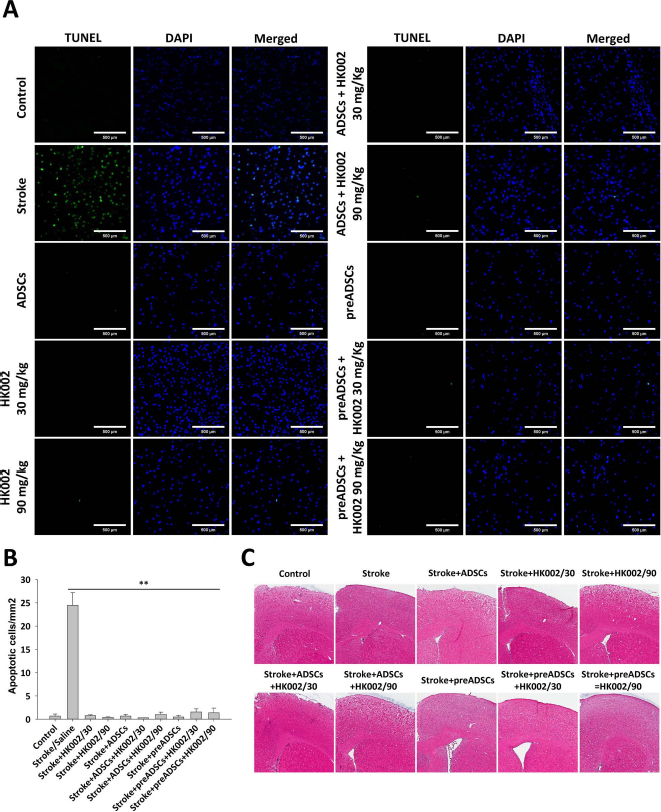
ADSC transplantation and ligustilide treatment significantly reduced apoptosis in the cortex of poststroke mice. (A) Apoptosis of cells was observed in large areas in the cortex of stroke mice and was significantly reduced with ADSC transplantation, ligustilide treatment, as well as the combination treatment groups. (B) Number of apoptotic cells from the TUNEL assay was quantified and displayed as a bar graph. Error bars represent mean ± SD, n ≥ 3. **p < 0.01 versus stroke/saline group. (C) H&E staining was performed on brain tissues from mice of all groups 1 month poststroke.
Discussion
Stroke is a common health problem worldwide, and no effective therapies exist for treating stroke. Furthermore, poststroke patient management imposes a heavy financial burden on the families as well as the healthcare system (4). Therefore, to improve the outcomes for stroke patients, it is important to develop new treatments, specifically stem cell therapy. Of the several kinds of stem cells, MSCs have attracted attention due to fewer ethical problems compared to ESCs and because they are less expensive to harvest than iPSCs (18). Although BM-MSCs have shown promise as a source of stem cells, ADSCs are the new focus for researchers since they are easily accessible (22,34).
In the present study, we examined ligustilide, a component of A. sinensis extract, for its neuroprotective ability. Through observations of neuronal behavior, we found that mice treated with subcutaneous ligustilide injections after stroke showed significant improvement in poststroke recovery (Fig. 3). A previous study showed that ligustilide ameliorates neuroinflammation and brain injury in rat models of ischemia by inhibition of toll-like receptor 4 (TLR4)/peroxiredoxin 6 signaling (17). Our study indicated that ligustilide reduced the viability of cells in culture at concentrations higher than 10 μg/ml after 48 h of incubation (Fig. 1). However, we also discovered that low concentrations of ligustilide (2.5 and 5 μg/ml) were capable of promoting neuronal development and had anti-inflammatory effects, as shown by gene expression patterns (Fig. 1). This suggested that prolonged incubation with high ligustilide concentrations may negatively affect cell function, but short-term incubation with low ligustilide concentrations is neuroprotective and can promote neurogenesis. Data on cell viability as determined by MTT assays were similar to that of a previous study (15).
In this study, we investigated the potential of ADSC transplantation on improving poststroke recovery. We demonstrated that ADSC transplantation significantly improved behavior in mice during the recovery process (Fig. 4). In addition, apoptosis of cortical neurons was also reduced with cell transplantation therapy (Fig. 7A, B). These results are in agreement with previous studies that have shown that ADSCs are capable of direct neuronal cell replacement and indirectly employ their secretome to promote neurogenesis and inhibit apoptosis (30).
Substances found in A. sinensis extract have anti-inflammatory and neuroprotective abilities (12,27). Ligustilide extracted from the plant is also known to promote neurogenesis (17,21,25). In this study, one of our aims was to examine whether pretreating ADSCs with ligustilide extract can promote neuroprotection and perhaps stimulate neurogenesis. We treated ADSCs with low concentrations of ligustilide to avoid cell toxicity from the drug and at the same time utilize its ability to promote neuronal gene expression (Fig. 1). We demonstrated that ligustilide-pretreated ADSCs have better poststroke recovery overall and more significant short-term recovery (3 days poststroke).
Several studies have indicated that stem cells are capable of homing to sites of ischemia through intravenous transplantation that is distant from the site of injury (2,14,24). Intravenous transplantation was beneficial to patients due to its less invasive nature; however, it was also noted that efficiency of such a method would need to be further optimized compared to intracerebral transplantation (14). In our study, stroke induction mouse model was performed with brain surgery; therefore, we chose to transplant stem cells via intracerebral injection in combination with this procedure to reduce burdens on target mice. It is necessary for future studies to investigate the advantage of intravenous transplantation and compare the efficiency of these two methods.
In summary, we have shown that ligustilide from A. sinensis and ADSC transplantation significantly improved poststroke recovery in a thromboembolic mouse stroke model. The therapeutic effect of each treatment was significant independently. The therapeutic effect of ADSCs was significantly improved when we combined stem cell transplantation and ligustilide treatment. Our findings suggest a highly potent stem cell therapy for stroke patients that can possibly reduce the time span for poststroke recovery. Our study also demonstrates the potential of A. sinensis and its extract in activating ADSCs and promoting neuroprotection and neurogenesis.
Footnotes
Acknowledgments
This study is supported in part by the Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW105-TDU-B-212-133019) and China Medical University Hospital (DMR-105-059) and China Medical University (10242643). The authors declare no conflicts of interest.
