Abstract
Culture of human islets before clinical transplantation or distribution for research purposes is standard practice. At the time the Edmonton protocol was introduced, clinical islet manufacturing did not include culture, and human serum albumin (HSA), instead of fetal bovine serum (FBS), was used during other steps of the process to avoid the introduction of xenogeneic material. When culture was subsequently introduced, HSA was also used for medium supplementation instead of FBS, which was typically used for research islet culture. The use of HSA as culture supplement was not evaluated before this implementation. We performed a retrospective analysis of 103 high-purity islet preparations (76 research preparations, all with FBS culture supplementation, and 27 clinical preparations, all with HSA supplementation) for oxygen consumption rate per DNA content (OCR/DNA; a measure of viability) and diabetes reversal rate in diabetic nude mice (a measure of potency). After 2-day culture, research preparations exhibited an average OCR/DNA 51% higher (p < 0.001) and an average diabetes reversal rate 54% higher (p < 0.05) than clinical preparations, despite 87% of the research islet preparations having been derived from research-grade pancreata that are considered of lower quality. In a prospective paired study on islets from eight research preparations, OCR/DNA was, on average, 27% higher with FBS supplementation than that with HSA supplementation (p < 0.05). We conclude that the quality of clinical islet preparations can be improved when culture is performed in media supplemented with serum instead of albumin.
Introduction
After the introduction of the Edmonton protocol, islet transplantation became a promising treatment option for selected type 1 diabetes patients (5, 7, 13, 15, 16, 20, 21, 24, 27, 46). There are stringent criteria for accepting a donor pancreas for clinical transplantation, and those organs that do not meet these criteria are generally used for research purposes. Research-grade pancreata are preserved in aqueous cold preservation solution alone, while clinical-grade pancreata are often preserved using the two-layer method to improve pancreas oxygenation, although its efficacy remains controversial (4, 9, 25, 36). There are also other differences in the islet manufacturing procedures that may affect the quality of islets, such as conditions during islet culture. Hence, there is a concern that research islets are not entirely representative of clinical islets in terms of viability, potency, or behavior in experimental pharmacological studies.
Originally, the Edmonton protocol required the infusion of islets in the recipient to occur within 4 h after isolation (45, 46). This narrow time window necessitated the advance relocation of prospective transplant recipients to the transplant center in anticipation of a transplant, and it limited safety and quality assessment of the islet product prior to transplantation. Islet culture for some time after isolation helps to address these logistical challenges. Furthermore, culture has been also shown to selectively deplete acinar tissue components and passenger leukocytes (28, 29), resulting in higher purity and reduced immunogenicity, although culture longer than 2 days may be required for these improvements to be substantial. Nonetheless, 2-day culture has emerged as the standard practice prior to clinical transplantation or islet distribution for research purposes (16, 20).
Although there is no consensus (14), supplementation of islet isolation, collection, and culture media with serum is generally considered better than supplementation with its albumin component alone. This is because serum contains other factors that are beneficial for islet viability (32) and also neutralizes endogenous pancreatic enzymes (6, 18, 43) or exogenous enzymes left over from the isolation process (6, 18). For this reason, a number of islet isolation centers have traditionally cultured research islets with fetal bovine serum (FBS). In contrast, when islet culture was introduced in clinical protocols, there could be no FBS supplementation to avoid the introduction of xenogeneic material into the clinical islet preparations. Instead, the use of human serum albumin (HSA) throughout the islet manufacturing process in the Edmonton protocol was simply extended by default to islet culture medium supplementation, and most American centers continue to this day to supplement clinical islet culture media with HSA. However, some centers have adopted the use of human sera in islet isolation and culture (18, 26). The Uppsala group has reported an islet isolation process using compatible human ABO serum that meets cGMP standards (18) and that photochemical treatment of human serum for pathogen inactivation is not detrimental to islet quality and potency following 4-day culture (47). The Geneva group has reported a lower level of apoptosis and a higher stimulated insulin secretion and insulin content using 10% human AB serum instead of 0.625% HSA (8). Moreover, recently, the Lille group showed higher viability, insulin content, and stimulation index of clinical-grade islets cultured for 5 days with 2.5% human AB serum when compared with 0.625% HSA (26).
With the exception of a preliminary report from the Geneva group with a small number of preparations (8) and a preliminary report of our data (33), direct quantitative comparisons of FBS and HSA in short-term (2-day) culture have not been documented. At our center, HSA medium supplementation is performed for clinical preparations, and FBS medium supplementation was traditionally performed for research preparations until the ICR center consortium requested in 2008 that human research islets be shipped in media supplemented with HSA. This enabled us to conduct such a quantitative comparison of FBS and HSA using both retrospective and prospective approaches. The analysis included postculture islet fractional viability assessed by oxygen consumption rate per DNA content (OCR/DNA) (11, 34, 37) and in vivo potency assessed by diabetes reversal (DR) rates in diabetic athymic nude mice. We also analyzed fluorescein diacetate/propidium iodide (FDA/PI) cell membrane integrity data.
Materials and Methods
Institutional Approvals
Research consent was available for all research preparations. U.S. Food and Drug Administration and Institutional Review Board approval was available for all quality assessments performed with clinical preparations. All animal protocols were approved by the Institutional Animal Care and Use Committee.
Islet Isolation
All islet isolations were performed in the University of Minnesota Molecular and Cellular Therapeutics GMP Facility as previously described (20, 21). Briefly, pancreata were distended with cold tissue dissociation enzyme solution through the pancreatic duct with a pressure-controlled pump system and digested using a modified Ricordi's semiautomated method (42). The digested tissue was then purified with a continuous iodixanol (OptiPrep, Axis-Shield, Oslo, Norway) density gradient in a COBE 2991 cell processor (Gambro BCT, Lakewood, CO). No FBS was used during the isolation process of any islets.
Islet Culture
Islets were cultured for 12–24 h at 37°C and thereafter at 22°C for a total period of 36–72 h with 5% CO2 before quality assessment and transplantation. Islet surface density was kept below 200 islet equivalents (IE)/ cm2, and islet culture volume density was 1,000 IE/ml. In the retrospective study, islets were cultured in CMRL supplemented medium (Mediatech, Manassas, VA) supplemented with 10 U/ml heparin and either 10% (v/v) FBS for research islets or 0.5% (m/v) HSA and 100 ng/ ml insulin-like growth factor 1 (IGF-1) (21, 39) for clinical islets. The concentration of HSA reflects the albumin concentration in 10% (v/v) FBS. The prospective paired study aimed to compare the effects of FBS and HSA on islet quality without the influence of any confounding factors; thus, IGF-1 was not added to the medium in either condition. Research islets were transported to a different room or facility for culture to eliminate the risk of xenogeneic cross-contamination of the clinical islets.
Islet Sampling
Except for the FDA/PI assay, islets were not hand-picked but sampled randomly to ensure that the tissue assessed was as representative as possible. The sampling method and sample size for all assays were the same for clinical and research preparations.
Islet Purity
Islet purity was visually assessed based on dithizone staining for both freshly isolated and cultured islets in increments of 5% from two samples of at least 100 islets each taken for islet counting as per standard practice (41). Only preparations of high purity were included in the analysis to minimize the effects of acinar tissue. The definition of high purity as 70% or higher is the same as that used by the Clinical Islet Transplantation Consortium (http://www.citisletstudy.org/).
Islet Fractional Viability: OCR/DNA and FDA/PI
Details on the OCR/DNA assay are provided elsewhere (11, 34, 37). Briefly, usually three (range, one to six) samples of cultured islets (generally 1,000–2,000 IE per sample) were placed in culture medium and sealed inside stainless steel chambers equipped with fiberoptic sensors (Instech Laboratories, Plymouth Meeting, PA) that measure the declining oxygen partial pressure in the well-stirred medium over time. The oxygen consumption rate (OCR) of each sample was calculated based on the rate of oxygen depletion from the chamber. Islets were then removed from the chambers with several washes, and the DNA quantity in each chamber was determined using the Quant-iT Picogreen dsDNA kit (Molecular Probes, Eugene, OR). The OCR was normalized by the DNA content to produce an OCR/DNA value for each sample, and the values were averaged across samples for each preparation. For FDA/PI, between 80 and 100 islets were hand-picked and incubated with 0.67 μM FDA and 4 μM PI, per standard protocols (31). Quadruplicate samples were read in a fluorometer at 485/530 nm (excitation/emission) for FDA and 530/645 nm for PI. Fractional viability was estimated from the ratio of the emitted intensities.
Diabetic Nude Mouse Bioassay
Following counts on day 2 postisolation, 2,000 IE were transplanted under the kidney capsule of diabetic athymic nude mice. Diabetes was induced with streptozotocin and defined as a pretransplant blood glucose (BG) level of >350 mg/dl. DR was defined as three consecutive BG measurements on different days being ≤200 mg/dl, which is the criterion used by the Clinical Islet Transplantation Consortium. By convention, DR times reported reflect the first day of the period during which the normoglycemia criterion was met. In most cases, one mouse received the IE dose examined in this study for each preparation. For the few preparations that had more than one mouse transplanted, the DR outcome (DR vs. no DR) was uniform, and the DR time representative of the preparation was calculated by averaging the DR times for the individual mice.
Study Design
The retrospective study included all high-purity islet preparations isolated at our center between 2000 and 2008 (prior to the ICR center consortium's requiring research islet preparations to be shipped in media supplemented with HSA) for which postculture data on OCR/DNA or DR rates in athymic nude mice were available. In total, we analyzed data from 103 islet preparations isolated from 58 research-grade and 45 clinical-grade pancreata. Figure 1 presents the flowchart of the islet isolation and culture process. Of the 45 isolations from pancreata accepted for clinical use (traditionally called “clinical” isolations rather than “clinical-grade” isolations), 18 had a low postpurification yield (IE per recipient body weight) and did not satisfy the release criteria. These isolations were considered failed, and the isolated islets became research islets to be cultured using FBS medium supplementation in exactly the same way as islets from research-grade pancreata. Together with the 58 research-grade islet preparations, 76 preparations in total were allocated for research (research islet preparations). The remaining 27 clinical islet isolations met the release criteria, and the isolated islets were allocated for transplantation (clinical islet preparations). These preparations were cultured in medium supplemented with HSA and IGF-1. For the prospective study, islets from 11 high-purity research preparations were cultured in two conditions to provide data for a paired comparison: with 10% (v/v) FBS supplementation and with 0.5% (m/v) HSA supplementation. IGF-1 was not added in either condition. Data with HSA and no IGF-1 supplementation from the prospective study were also retrospectively compared with data from the main retrospective study.

Retrospective study. Flowchart of research-grade and clinical-grade pancreata processed to research or clinical islet preparations. For further explanations, see text.
Statistical Analysis
Data are presented as arithmetic mean values ± standard deviation (SD). For the retrospective study, the two-sample, two-sided Student's t test was used to assess statistical significance for noncategorical variables. Equality of variances was assessed at the 95% level, and the appropriate version of the Student's t test was used (44). Categorical comparisons between groups were done using the version of the two-sided Fisher's exact test that calculates the p value from both asymmetric tails (1). Comparison of the DR time profiles was done with the log-rank test, using the GraphPad Prism (La Jolla, CA) software. For the prospective study, the paired two-sided Student's t test was used to assess statistical significance for noncategorical variables. Significance was assessed at the p = 0.05 level for all tests. Parts of the statistical analysis were performed with the assistance of the Administrative and Bioinformatics Coordinating Center for the ICR, City of Hope National Medical Center, Duarte, CA.
Results
Retrospective Study
Selection criteria are more stringent for clinical-grade than for research-grade pancreata. Key data on donor characteristics, pancreas preservation, and islet isolation outcomes are summarized in Table 1A. The donors of the clinical-grade pancreata were significantly younger and had a significantly higher body mass index than the donors of the research-grade pancreata. Most clinical-grade pancreata were preserved with the two-layer method; in contrast, most research-grade pancreata were preserved with the conventional method of immersion in a cold preservation solution. The average cold-ischemia time was 43% shorter for clinical-grade pancreata, as it was kept below 8 h for all but 2 of the 45 clinical-grade pancreata (the two exceptions were for failed clinical isolations performed for the Edmonton clinical trial that allowed longer cold-ischemia time). Digestion (phase I) time was similar for the two groups, and postpurification islet yield was 52% higher for clinical-grade pancreata (p < 0.001). Table 1B summarizes the same parameters divided based on actual use, namely, with the 18 failed clinical preparations lumped together with the 58 research-grade preparations form the research preparation group. Significance of statistical differences between the 76 research and 27 clinical islet preparations was generally similar to that between the 58 research-grade and the 45 clinical-grade preparations, as shown in Table 1A.
Retrospective Study: Selected Pancreas Donor, Pancreas, and Islet Isolation Parameters

Islet preparations are divided by intended use at the time of isolation. SD, standard deviation; IE, islet equivalent.
Retrospective Study: Selected Pancreas Donor, Pancreas, and Islet Isolation Parameters

Islet preparations are divided by actual use, that is, following reallocation of failed clinical preparations based on islet yield before entering culture. FBS, fetal bovine serum; HSA, human serum albumin; IGF-1, insulin-like growth factor 1; SD, standard deviation; IE, islet equivalent. The p values are for the two-sample two-sided Student's t test.
Due to the rather recent introduction of the assay, OCR/DNA data were not available for any preparation between 2000 and 2002. For clinical preparations, OCR/ DNA was first assessed following the approval by the U.S. Food and Drug Administration and the Institutional Review Board in early 2005. For this reason and because of limited islet availability with certain preparations, OCR/DNA data were only available for 50 of the 103 islet preparations examined. DR data were available for only for 75 of the 103 islet preparations due to limited islet availability with certain preparations or lack of DR outcomes (e.g., due to mouse death or lack of diabetes recurrence postnephrectomy). Table 2 summarizes the availability of quality assessment data for the retrospective study.
Retrospective Study: Numbers of Islet Preparations With Data Available for Assessment of OCR/DNA, Mouse DR, and FDA/PI

OCR/DNA, oxygen consumption rate per DNA content; DR, diabetes reversal; FDA/PI, fluorescein diacetate/propidium iodide; FBS, fetal bovine serum; HSA, human serum albumin; IGF-1, insulin-like growth factor 1.
The average purity was similar for research (n = 76) and clinical (n = 27) islet preparations, both preculture (84.6 ± 9.3% vs. 84.9 ± 8.2%, p = 0.88) and postculture (84.8 ± 7.9% vs. 86.3 ± 7.7%, p = 0.41).
OCR/DNA values ranged between 90 and 413 nmol/ min/mg DNA for research preparations and between 89 and 237 nmol/min/mg DNA for clinical preparations. Figure 2 shows the OCR/DNA distribution in groups, each comprising a range of 50 nmol/min/mg DNA (i.e., 50–100, 100–150, etc.). Figure 3 shows the average OCR/ DNA for the different islet preparation groups. Average OCR/DNA was 51% higher for the research preparations (205 ± 82 nmol/min/mg DNA, n = 38) compared with that for the clinical preparations (136 ± 44 nmol/min/mg DNA, n = 12), a difference that was highly statistically significant (p = 6.4 × 10−4). We also assessed the fraction of preparations with OCR/DNA greater than the 125–150 nmol/min/ mg DNA threshold, over which a substantial increase in DR rate in diabetic nude mice has been reported using either human (34) or rat (35) islets. This fraction was significantly higher for research than for clinical islet preparations both for >125 nmol/min/mg DNA [33 of 38 (87%) vs. 6 of 12 (50%), p = 0.016] and for >150 nmol/min/mg DNA [28 of 38 (74%) vs. 3 of 12 (25%), p = 0.0042].

Retrospective study. Fractional distribution of 38 research and 12 clinical islet preparations in groups based on OCR/DNA value, each group covering a range of 50 nmol/min/ mg DNA.

Retrospective study. Average OCR/DNA for research-grade, failed clinical, research (research-grade and failed clinical combined), and clinical islet preparation groups. Error bars denote SD. There was statistical significance (p < 0.05) between the clinical and each of the other groups.
The average OCR/DNA for failed clinical islet preparations (216 ± 26 nmol/min/mg DNA, n = 5) was similar (p = 0.50) to that for the research-grade islet preparations (203 ± 87 nmol/min/mg DNA, n = 33) and significantly higher than that for the clinical islet preparations (p = 0.0019). All five failed clinical preparations had an OCR/DNA value exceeding the 150 nmol/min/mg DNA threshold.
The islet fractional viability estimated from FDA/PI staining of islets cultured for 12–24 h was more than 90% for almost all preparations, and the average was almost identical for research clinical preparations [93.9 ± 4.4% (n = 57) vs. 93.9 ± 3.3% (n = 25), p = 0.98].
Figure 4 shows DR rates as a function of time for diabetic nude mice transplanted with 2,000 IE after 2 days of culture. DR rates were 54% higher for research than for clinical islet preparations [37 of 50 (74%) vs. 12 of 25 (48%), p = 0.039 for two-sided Fisher's exact test and p = 0.038 for log-rank test]. Failed clinical preparations had the highest DR rate [15 of 17 (88%)], which was 184% higher than the DR rate achieved with clinical preparations [12 of 25 (48%)], with the difference exhibiting high statistical significance (p = 0.0097 for two-sided Fisher's exact test and p = 0.0066 for log-rank test). There was no statistically significant difference (p = 0.17) relative to research-grade preparations [22 of 33 (67%)].
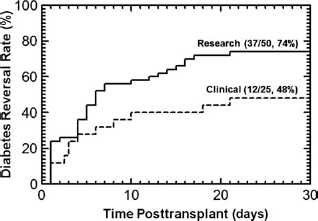
Retrospective study. Kaplan–Meier plot of the incidence of diabetes reversal (DR) in diabetic athymic nude mice upon transplantation with 2,000 IE from research and clinical islet preparations under the kidney capsule. DR rate for research islets was 54% higher compared with clinical islets (74% vs. 48%, p < 0.05).
Prospective Study
In the prospective study, islets from 11 research preparations were cultured in the absence of IGF-1 with either 10% FBS or 0.5% HSA supplementation, yielding 10 sets of paired data for OCR/DNA and 7 sets for DR in nude mice. Figure 5 shows the OCR/DNA paired data. OCR/DNA was higher for 9 of the 10 preparations, and by 34 ± 27% on average for islets cultured with FBS compared with those cultured with HSA, again with high statistical significance (p = 0.0080). The two conditions had concordant DR outcomes for six of seven preparations (five DR, one no DR). For the one discordant pair, only the islets cultured in FBS resulted in DR.

Prospective study. OCR/DNA for paired conditions cultured with FBS and HSA supplementation derived from 10 research preparations. For 9 of the 10 preparations, OCR/DNA was higher with FBS supplementation (p < 0.01).
Retrospective Analysis of IGF-1 Effects
The HSA arm of the paired prospective study was expanded by adding research preparations cultured with HSA but no FBS as part of another study, and the combined group was compared retrospectively with the clinical preparations. The only difference in the culture medium between these two groups was IGF-1 supplementation, which was absent for the research group but present for the clinical group. The average OCR/DNA was statistically significantly higher (p = 0.049) for preparations cultured in the absence of IGF-1 (184 ± 65 nmol/min/mg DNA, n = 12) than for preparations cultured in the presence of IGF-1 (136 ± 44 nmol/min/mg DNA, n = 12). Also, the DR rates were 56% higher for preparations cultured in the absence of IGF-1 [6 of 8 (75%)] than for preparations cultured in the presence of IGF-1 [12 of 25 (48%)], but this difference did not reach statistical significance (p = 0.24). A retrospective comparison of the same research preparations cultured with HSA with the regular research preparations cultured with FBS, both in the absence of IGF-1 supplementation, again showed that OCR/DNA was higher with FBS supplementation (205 ± 82 nmol/min/mg DNA, n = 38) than with HSA supplementation (184 ± 65 nmol/min/mg DNA, n = 12), but the difference was not statistically significant (p = 0.41). DR rates were also similar [37 of 50 (74%) for FBS vs. 6 of 8 (75%) for HSA].
Discussion
Islets prepared for clinical transplantation are distinguished from those designated for research by the care, effort, and expense involved in ensuring that donor attributes, pancreas procurement, and pancreas preservation, including cold-ischemia time, are optimal (Table 1A). Even after moving the failed clinical-grade islet preparations to the research islet preparation group, differences in body mass index, cold-ischemia time, and islet yield remained statistically significant (Table 1B). Hence, one would expect better outcomes for clinical as compared to research islet preparations. In this study, we found the opposite: research islet preparations had a 51% higher average fractional viability measured by OCR/DNA than clinical islet preparations (p < 0.001) (Fig. 3). Since average purity was high and similar for research and clinical preparations (85–86%) and OCR/DNA is similar in healthy endocrine and acinar tissue (unpublished data), this difference in OCR/DNA is expected to reflect not only the viability of the entire preparation but also that of the islet fraction. The results in the nude mouse bioassay were in accordance with the OCR/DNA data: DR rates were 54% higher (p < 0.05) (Fig. 4). It is noted that the islets used for mouse transplants were not handpicked but rather sampled from each preparation to ensure that the transplants are representative of the entire preparation. This practice can result in lower DR rates than those observed in other centers where islets are handpicked. This is expected as hand-picked islet samples do not contain as much acinar and damaged islet tissue, both of which can negatively affect engraftment and function (19, 22, 34, 35).
Fractional viability values based on FDA/PI were consistently above 90% and almost identical for research and clinical preparations. These values are much higher than those based on the OCR/DNA assay, which are about 45% for research and about 30% for clinical islet preparations, based on a 100% value corresponding to the highest OCR/DNA values observed in this and prior studies (11, 34). One explanation for the invariably high fractional viability values estimated with the FDA/PI assay is that the assay accounts only for cells with compromised membranes and not for cells in early apoptotic stages with intact membranes (11, 31, 34, 38, 41). Since the FDA/ PI assay apparently fails to reflect differences in islet quality that are identified by the OCR/DNA assay and the nude mouse bioassay, it is considered an inadequate tool for assessing islet viability (34, 38).
The retrospective study showed that research preparations have a higher quality than clinical preparations, and the prospective study provided evidence that this is related to differences in culture medium. Like other sera, FBS contains unidentified factors that promote islet viability (32). FBS deactivates some of the endogenous pancreatic enzymes and thus gives protection to islets after isolation from further digestion (43). Thermolysin, the predominant exogenous neutral protease used in islet isolation, can persist in islet preparations long after isolation, adversely affects islet functionality, and activates proapoptotic pathways in islets that otherwise show good quality in standard cell integrity, viability, and insulin release assays (6, 18). We found that 10% FBS or human AB serum deactivated thermolysin, whereas 0.5% HSA had a very limited inhibitory effect (unpublished data). In this respect, HSA serves only as a competitive inhibitor and, even at much higher concentrations (25%), exhibits no collagenase inhibition and only minimal endogenous protease inhibition (18, 43). In a separate paired study, we found an increase in OCR/ DNA in 13 of 16 islet preparations and an average increase in OCR/DNA by more than 10% when 10% AB serum was substituted for 0.5% HSA (unpublished data). This difference in OCR/DNA exhibited a trend towards statistical significance (0.05 < p < 0.10) but was smaller than the differences observed with FBS in this study. However, the observed increase of the average OCR/DNA from 136 to 149 nmol/min/mg DNA is very important in light of the 125–150 nmol/min/mg DNA threshold, over which a dramatic increase in graft success rate has been observed in mice with human (34) and rat (35) islets. Our results are in accordance with those by other groups comparing serum and HSA supplementation, as discussed in Introduction (8, 18, 26).
The relevance of our results is not limited to clinical transplants but also extends to research islet preparations. In 2008, the ICR center consortium requested that islet isolation centers participating in the research human islets shipping program use HSA in shipment medium as a means of standardizing shipping methods across participating centers. Our findings indicate that use of HSA instead of FBS for culturing and shipping research islets can adversely affect their viability and in vivo potency. These findings were communicated to the Integrated Islet Distribution Program (IIDP) that succeeded the ICR, which, in 2010, replaced HSA by FBS or human AB serum in the program.
The culture media used in our retrospective study did not differ only in the supplementation with FBS or HSA but also in the supplementation with IGF-1. IGF-1 has been used for islet culture in serum-free media for at least 30 years (10), but its use in media for research preparations is usually impeded by the high cost. At our center, IGF-1 is used for clinical islet culture at 100 ng/ml, a concentration at which mammalian islet cell survival under proapoptotic stress both in serum-free (3, 39) and serum-containing media (2, 23) is improved, β-cell activity is enriched, and possibly immunogenicity in human fetal pancreatic proislets is decreased (12). Also, serum deprivation-induced cell death in the rat insulinoma INS-1 cell line is reduced with IGF-1 (30) due to activation of a variety of antiapoptotic pathways (17). At our center, the use of IGF-1 in culture of clinical islets at our center has been associated with highly successful clinical outcomes (20, 21). Based on this evidence, it was anticipated that IGF-1 would limit the extent of islet cell apoptosis and that, at least, it would not adversely affect β-cell viability or potency in the clinical islet preparations. We directly tested the effect of IGF-1 by comparing the HSA arm of the prospective paired study (without IGF-1 supplementation) with the clinical islet preparations in the retrospective study also cultured with HSA (with IGF-1 supplementation). We found that islets cultured with IGF-1 had lower OCR/ DNA and DR rates, with the OCR/DNA difference being statistically significant (p < 0.05). This observation contrasts with original expectations but is in accordance with the smaller difference between FBS and HSA in the prospective paired study compared with that in the retrospective study. This outcome is also in contrast with a recently published preliminary report of a paired study with a similar IGF-1 concentration (76 ng/ml) in serum-free media that demonstrated small (<10%) improvements in islet recovery, OCR, and β-cell apoptosis (40). Further study on the effects of IGF-1 on islets during culture is warranted.
The observed differences in the viability and potency between research and clinical islet preparations apparently contrast with the better quality of pancreata allocated to clinical islet isolations and, hence, with the presumption of an advantage of clinical islets after isolation, putting the latter into question. To explore this aspect, we considered the failed clinical preparations that combined the presumed advantages of clinical-grade pancreata with those associated with culture in research media. Interestingly, these preparations exhibited the highest OCR/DNA (Fig. 3) and DR rates out of all groups in this study. With the caveat that the number of failed clinical preparations with assessment data was low (n = 5 for OCR/DNA and n = 17 for DR rates), the difference from clinical preparations was highly significant (p < 0.01) both for OCR/DNA and for DR results, and there was no statistically significant difference from research-grade preparations. These findings indicate that, in efforts to achieve high-quality islet preparations, culture medium supplementation is a more important factor than donor characteristics or pancreas procurement and preservation (which are similar for clinical and failed clinical preparations). The importance of medium supplementation is corroborated by the results of the paired comparison using research islets.
In summary, research islet preparations cultured in media supplemented with FBS were found to be of higher, rather than lower, viability and in vivo potency compared to clinical islets cultured in media supplemented with HSA, despite inferior donor selection criteria and pancreas preservation methods. This opens the possibility that postculture viability and in vivo potency of clinical islet preparations can be improved by culture medium modifications, including supplementation with a nonxenogeneic serum suitable for clinical islet use. Similarly, the quality of research islets can be improved by serum supplementation of culture and shipment media. Further studies on the effects of islet culture medium supplementation with IGF-1 are also warranted.
Footnotes
Acknowledgments
This study was supported by grants from the National Institutes of Health (U42 RR 016598-01 from the National Center for Research Resources; R01 DK56963 and R44 DK069865 from the National Institute for Diabetes, Digestive, and Kidney Diseases), the Juvenile Diabetes Research Foundation (#4-1999-841), the Iacocca Foundation, the Schott Foundation, and the Carol Olson Memorial Diabetes Research Fund. We thank LifeSource and other organ procurement organizations for their efforts in pancreas procurement; our animal care, islet isolation, project management, quality assessment, regulatory, and research teams, including Muhamad Abdulla, Brian Flanagan, Mel Graham, Jian Hao, Minna Honkanen-Scott, Jody Janecek, Lou Kidder, Jenna Kitzmann, Bob Konz, Jeramy Kulesa, Brita Kurmis, Christina Landwehr, Gopal Loganathan, Shinichi Matsumoto, Heather Nelson, Tetsuya Sakai, Kristen Stewart-Maynard, Josh Wilhelm, Laura Vonhof, Jean Witson, Hui-Jian Zhang, and several other valued colleagues; Mike Loughnane (Instechlabs) for helpful discussions on OCR equipment and measurements with islets; and Mary Knatterud for editing parts of the manuscript. The authors declare no conflict of interest.
