Abstract
Conventional culture vessels are not designed for physiological oxygen (O2) delivery. Both hyperoxia and hypoxia—commonly observed when culturing cells in regular plasticware—have been linked to reduced cellular function and death. Pancreatic islets, used for the clinical treatment of diabetes, are especially sensitive to sub- and supraphysiological O2 concentrations. A result of current culture standards is that a high percentage of islet preparations are never transplanted because of cell death and loss of function in the 24–48 h postisolation. Here, we describe a new culture system designed to provide quasiphysiological oxygenation to islets in culture. The use of dishes where islets rest atop a perfluorocarbon (PFC)-based membrane, coupled with a careful adjustment of environmental O2 concentration to target the islet physiological pO2 range, resulted in dramatic gains in viability and function. These observations underline the importance of approximating culture conditions as closely as possible to those of the native microenvironment, and fill a widely acknowledged gap in our ability to preserve islet functionality in vitro. As stem cell-derived insulin-producing cells are likely to suffer from the same limitations as those observed in real islets, our findings are especially timely in the context of current efforts to define renewable sources for transplantation.
Introduction
To date, islet transplantation is the only successful form of cell therapy for type 1 diabetes. The progressive implementation of steroid-free immunosuppression protocols (49) and, more recently, T-cell-depleting interventions (3) have contributed to sustained graft function that now stands comparison with that reported for whole pancreas transplantation. However, the widespread clinical application of this therapy is limited by the shortage of donors (47), a problem that is compounded by the low efficiency of the process. Typical isolation procedures seldom recover more than 50% of the islets, of which up to 80% may succumb to instant blood-mediated inflammatory reaction (IBMIR) during the immediate posttransplantation period (2). Until such time a plentiful and/or renewable source of islets is defined, a priority in the field has been to make every single islet count. In this context, numerous chemical (11,12,15,25), gene-based (14,31,38), and protein transduction (33,43,45) interventions have led to measurable improvements in experimental models. However, there is a growing perception that we may need to go back to basics when it comes to preserving the viability of cultured islets.
One of the most blatant omissions in standard culture is the provision of adequate oxygenation. In this respect, the “one size fits all” approach followed for decades is in stark contrast with the zeal with which we ensure that other physiological parameters (temperature, pH, etc.) are just right. Devices with a unidirectional flow of air are known to impart an uneven diffusion of O2 throughout the tissue (36). While this may not be a reason of concern for some cell types (e.g., immortalized lines), in most other applications, this pattern of aeration invariably results in hypoxia- or hyperoxia-induced cell death (1,6,20). This is especially true of pancreatic islet β-cells. Although islets represent only a minute fraction of the pancreas (1–2%), they receive nearly 15% of the overall blood flow to the organ, using 25% of the pancreatic O2 supply (27). The destruction of their microvasculature upon isolation has been unequivocally linked to a dramatic reduction in viability (19).
Both for clinical and basic research purposes, β-cells are typically cultured at atmospheric O2 concentration (21%) in nonadherent dishes or flasks, with medium covering them up to ~1.5 mm above the bottom of the vessel. Under these conditions, islets are exposed to sharp gradients, ranging from overt hyperoxia to central anoxia (0 mmHg) (41). Only a small fraction of the islet mass receives physiological oxygenation, which ought to be around 40 mmHg (9). Considering that islets are exquisitely sensitive to both hypoxia (13,19,35,39) and oxidative stress (29,30,48), the observation that they die very quickly in vitro is hardly surprising (5,26). Seminal work conducted in the late 1970s showed that high O2 tensions were deleterious for pancreatic tissue (10). Later studies established that β-cells are sensitive to high O2-induced stress because of their naturally low expression of antioxidant enzymes (43,45,50). Another critical molecular event triggered by oxidative stress in the β-cells is the c-Jun N-terminal kinase (JNK)-dependent nuclear-cytoplasmic translocation of pancreatic and duodenal homeobox 1 (Pdx1), which leads to an overall downregulation of the glucose sensing/insulin secreting machinery (28). As for the JNK pathway, its activation in response to hyperoxia has been associated with nonapoptotic cell death (32). Taken together, these data indicate that supraphysiological O2 conditions (such as those affecting large parts of the islet in standard culture settings) are harmful for β-cell function and viability.
Low O2 tensions are equally detrimental. Insulin secretion is stalled below 7 mmHg in insulinomas (42). Low pO2 inhibits insulin release through a [Ca2+] decrease, which in turn reduces Na/K-ATPase activity (8). More importantly, hypoxic exposure results in cell death by activation of both the induced nitric oxide synthase (iNOS)– nitric oxide signaling cascade and JNK phosphorylation (34). In summary, subphysiological O2 conditions (such as those potentially affecting the islet core in regular culture settings) lead to β-cell loss of function and death.
The obvious conclusion from these studies is that current culture conditions are suboptimal for islet survival and function. We submit that a simple adjustment of the way islets are oxygenated in vitro may overcome the above limitations prior to transplantation. Here, we report that when human islets are cultured in a perfluorocarbon (PFC)-based system designed to optimize O2 diffusion, both parameters are dramatically enhanced. Our findings highlight the importance of mimicking the physiological microenvironment of islets to maintain β-cell viability in vitro, and present a straightforward culture system that could potentially become a new standard in islet culture for research and clinical applications.
Materials and Methods
Use of Human Tissue Material
Investigations with human tissues were preceded by University of Miami Institutional Review Board (IRB) approval and informed consent.
Human Islet Isolation
Human islet preparations were processed at the Diabetes Research Institute cGMP Core according to standard methods described elsewhere (24,46).
35-mm PFC/PDMS Dish Manufacture
We set out to build a mass-producible, disposable 35-mm-diameter prototype in which the inner and outer rings are held together by an interference fit, and the latter has three legs that elevate the membrane 1 mm off the surface to allow circulation of air from underneath (Fig. 1). Membranes were extruded by Specialty Silicone Fabricators (Paso Robles, CA, USA) according to our specifications. A two-part polydimethylsiloxane (PDMS) [RTV615 Part A (938.7 g); RTV615 Part B (312.9 g) (GE Silicones, Huntersville, NJ, USA)] and perfluorooctyltriethoxysilane (Alfa Aesar, Ward Hill, MA, USA) (408.9 g) were mixed to obtain a 0.012 in. (305 μm ± 12 μm) × 13 in. (33 cm) × 600 in. (1,524 cm) roll from which discs could subsequently be punched. Rapid prototyping was done with the Objet Eden 250 3D printing system (Stratasys, Minneapolis, MN, USA). Prototypes were manufactured with VeroWhite resin (Red Eye, Eden Prairie, MN, USA) to check fit and function. Advanced models were made by Computer Numerical Control (CNC)-assisted precision machining. Injection molding was done with Noryl HNA033 (Sabic, Pittsfield, MA, USA), an unfilled modified polyphenylene ether resin designed to withstand several autoclave cycles and γ-irradiation sterilization. The injection molding of the inner and outer rings of the 35-mm prototype was done at ProtoMold (Maple Plain, MN, USA). The assembly process was done at Biorep Technologies (Miami, FL, USA). For quality control, the assembled dish was placed on a tension meter jig, and tension was read with a Newman SR-Meter 1 (Stretch Devices, Inc., Philadelphia, PA, USA). Tension had to be within the 16–40 N/cm range.

PFC/PDMS-based device. (a) Diagram of the oxygen flow in standard (top) and perfluorocarbon (PFC; bottom) culture systems. Typical culture dishes exhibit a unidirectional flow of air from the top, whereas PFC/PDMS (polydimethylsiloxane) membrane-based devices allow for bidirectional oxygenation both from the top and the bottom, through a gas-permeable, liquid-impermeable membrane. (b) Diagram showing the design of the 35-mm PFC/PDMS culture dish. Inner and outer rings are held together by an interference fit. Once assembled, three feet elevate the dish off the surface to allow for bottom-up aeration through the PFC/PDMS membrane. (c) Photograph of a resin-based rapid PFC/PDMS dish prototype.
Diffusivity Studies
O2 spot sensors (PreSens GmBH, Regensberg, Germany) were affixed with ethyl cyanoacrylate (Krazy Glue, Columbus, OH, USA) to the center of the bottom surface of wells in a standard 24-well culture plate (VWR, Inc., Marietta, GA, USA). PDMS or PFC/PDMS membranes (3 mm) were subsequently fit into the wells atop the sensors, using quick-drying PDMS along the outer edges to ensure an air-tight seal. As a control, some sensor wells were filled with 603 μl of Hanks balanced salt solution (HBSS; Invitrogen, CellGro, Mediatech, Manassas, VA, USA). After equilibration at 38 mmHg O2 (5%), the entire sensor rigging was transferred to the upper stage of an incubator set to standard 95% room air/5% CO2 culture conditions. Taking into account vapor pressure differences, this translates to a pO2 of 142 mmHg. Care was taken not to agitate the plate to avoid convective disturbances. Additionally, the time of exposure to the temperature difference between the incubator and the room (approximately 12°C) was never greater than 5 s to minimize thermal effects. O2 partial pressure was measured in the system until the signal reached an equilibrium point at the new pO2 setting of 142 mmHg. Data points were recorded every 15 s for the length of the experiment. Our modeling assumed that (a) diffusion was 1D through the height of the measured compound, as the sides were treated as impermeable to O2; (b) effects due to diffusion through plastic and the edge seal were minimal; and (c) temperature shifts from moving the apparatus were negligible.
Theoretical Modeling
A retrospective analysis of islet size distributions from 184 human isolations was performed. The total volume of each count and the contribution of each size range to the total volume were determined as previously described (7). The majority of the tissue volume (77% pre-Ficoll and 80% postpurification in layer 1) fell in the range of 100–300 μm, with the largest percentage measuring between 150 and 200 μm (data not shown). This volume distribution was utilized for finite element modeling to maintain the largest tissue percentage [islet equivalents (IEQs) between 100 and 300 μm] at or near physiological pO2 while minimizing anoxia and hyperoxia.
The O2 consumption rate (OCR) of each preparation prior to plating was assessed using triplicate aliquots of 500 IEQs in a stirred chamber O2 measurement device (Instech Labs, Plymouth Harbor, PA, USA). 2D diffusion/reaction theoretical modeling was performed on permutations of control and experimental culture systems using COMSOL v.3.3 finite element analysis software (Comsol, Stockholm, Sweden).
Glucose-Stimulated Insulin Release (GSIR)
Glucose-stimulated insulin release was done by aliquoting 100 IEQs suspended in a Sephadex G10 slurry (GE Healthcare, Waukesha, WI, USA) within 10-ml microchromatography columns (Bio-Rad, Hercules, CA, USA). After an equilibration incubation in low-glucose (2.2 mM) modified Krebs buffer containing 0.1% (w/v) bovine serum albumin (BSA), 26 mM sodium bicarbonate, and 25 mM HEPES buffer (all from Sigma-Aldrich, St. Louis, MO, USA), sequential 1-h incubations were performed in low- (2.2 mM), high- (16.6 mM), and low-glucose (2.2 mM) buffers. Samples were collected for insulin analysis at the end of each hour following the preincubation. Insulin was quantified using the Mercodia Human Insulin ELISA (Winston Salem, NC, USA). The insulin data utilized were the difference in total insulin production per 100 IEQs between the high-glucose and first hour of low-glucose stimulation (Delta). In our experience, this value strongly correlates with full mass subrenal capsular transplant outcome in athymic nude mice.
O2 Consumption Rate (OCR)
The DNA-normalized index of OCR was calculated using the BD Oxygen Biosensor (BD Biosciences, San Jose, CA, USA) as previously described (17). Briefly, triplicate 200-μl aliquots of 500 IEQs each, suspended in either a modified Krebs buffer containing low (2.2 mM) or high (16.6 mM) glucose concentrations, were placed in individual wells of the BD Biosensor. Dry, medium-containing, and sodium sulfite (Sigma-Aldrich) controls were also run in triplicates. After equilibration at 37°C, fluorescence measurements were taken every 5 min for 16 h (ex: 485, em: 620). The early increase (between minutes 15–50) in fluorescence signal is indicative of the rate of O2 depletion in each well.
qRT-PCR
Total RNA was purified using miRNA Mirvana kit (Life Technologies, Grand Island, NY, USA). Random oligomers were used to generate cDNA with the High Capacity Reverse Transcription kit (Life Technologies). Relative expression of selected markers was calculated using TaqMan assays in Applied Biosystems thermal cyclers (both from Applied Biosystems, Foster City, CA, USA). The 7900HT model was used to run TaqMan Low Density Array (TLDA) predesigned cards (apoptosis). The ΔCt method for relative quantification was employed for all calculations.
Animal Studies
All animal procedures were approved by the University of Miami Institutional Animal Care and Use Committee. Female athymic nude mice (5-6 weeks old, Harlan Laboratories, Indianapolis, IN, USA) were housed in virus antibody-free rooms with free access to auto-claved water and food at the Division of Veterinary Resources. Personnel at the Translational Models Core of the Diabetes Research Institute performed all surgical procedures. A single streptozotocin (STZ; Sigma-Aldrich) injection induced selective destruction of islet β-cells and onset of diabetes. Animals were monitored, and insulin pellets were used if needed to maintain the overall health of the animals prior to transplantation. Under general anesthesia, the left kidney was externalized, and a small puncture made in its capsule. Islets were injected in a minimal volume of saline. The muscle/fascia was sutured with cat gut 4-0 (Ethicon, J&J, Somerville, NJ, USA), and the skin closed with surgical staples (Clay Adams Becton Dickinson, Sparks, MD, USA). Buprenorphin (Reckitt Benckiser, Slough, Berkshire, UK) was administered subcutaneously to alleviate postsurgical pain. After transplantation, recipients were followed up with blood glucose measurements (One Touch Ultra glucometer and strips from tail prick, Lifescan, Milpitas, CA, USA) to monitor graft function.
Statistical Analyses
The averaged results of in vitro assessments are expressed as the mean fold control ± standard deviation (SD) for the purpose of easily understood quantification. For statistical analysis, the raw data were utilized in nonparametric rank sum tests. When calculating the standard deviation of ratios, the following formula was used:
where R represents the ratio, X/Y, cv(X) is the variance of the numerator, and cv(Y) the variance of the denominator.
The raw data were utilized in the Mann-Whitney (rank sum) statistical test, assuming a non-Gaussian distribution. The Mann-Whitney test utilizes the parametric raw data, assigning a rank to each value independent of the group association. These ranks are then summed for each group, and the difference between the sums dictates the p value, where a larger difference correlates with lower p value. A value of p ≤ 0.05 was considered significant. Values of p ≤ 0.01 were considered highly significant.
Kaplan–Meier survival analysis with log-rank (Mantel– Cox) and Gehan–Breslow Wilcoxon tests for significance were performed on transplant data.
Results
A Device for Enhanced Oxygenation
We have designed a culture device in which cells and tissues can receive O2 both from the top (diffusing through the medium) and the bottom (across a perfluoro-hydrocarbon-silicone, or PDMS/PFC, membrane) (Fig. 1). Perfluorohydrocarbons, or PFCs, are inert compounds made of carbon-fluorine chains. Because of this molecular configuration, they can bind and transfer O2 with ease. The O2 solubility of PFCs is about 50 times higher than that of medium, which is superior to that of hemoglobin under certain conditions (4). Their O2 diffusivity is also considerably higher than that of water or medium (37). Computerized mathematical modeling, as well as direct in vitro measurements (17), shows that our system maximizes the volume of tissue exposed to physiological pO2.
O2 Diffusivity Through PFC/PDMS Exceeds That Measured on PDMS Alone Membranes
In order to address whether PFC/PDMS membranes had any advantage over those made of PDMS alone, we conducted diffusivity studies as indicated in Materials and Methods. The average diffusivity value calculated for the “PDMS alone” membrane was 3.9 × 10−5 ± 2.97 × 10−6 cm2/s (n = 4), which fell within the range of average values for silicone in the literature. In contrast, the average diffusivity calculated for the 20% (v/v) PFC/PDMS composition was 6.46 × 10−5 ± 4.51 × 10−6 cm2/s (n = 4), nearly 70% higher than that of the silicone (p < 0.01). Therefore, PFC/PDMS membranes have higher O2 diffusivity than those manufactured exclusively with PDMS. The advantage of adding PFC was further proven using a biological system (embryonic pancreatic buds) in vitro, as previously described (17).
Determination of the Human Islet Normoxic Range Using PFC/PDMS Dishes
Islets cultured at 21% O2 in regular dishes are known to receive suboptimal oxygenation (41). Due to the intrinsic limitations of conventional culture devices, mere changes in the incubator's O2 concentration still generate diffusion gradients that are detrimental for islet cell function and viability. PFC/PDMS dishes, in contrast, allow for a better targeting of a desired physiological range. Theoretical modeling of O2 diffusion through islets sized 150–200 μm (Fig. 2a) was done using COMSOL v.3.3 finite element analysis software. We modeled O2 diffusion through islets of different sizes plated in both regular plasticware and PFC/PDMS dishes. Environmental O2 concentrations spanned a range from 8% (low) to 21% (regular). Figure 2b shows the results of these calculations for four representative culture groups, namely, plasticware in atmospheric [O2], PFC/PDMS in atmospheric [O2], plasticware at low [O2], and PFC/PDMS at low [O2]. According to these models, a maximal volume of tissue at physiological O2 levels is reached when the islets are placed in PFC/PDMS dishes at an external pO2 of 63– 95 mmHg (approximately 8–12.5% O2), depending on the OCR. The ranges examined within the tissue were critically anoxic (<0.1 mmHg, no OCR), hypoxic (>0.1 mmHg <0.4 mmHg, OCR rapidly decreasing), physiological (>0.4 mmHg <100 mmHg), and hyperoxic (>100 mmHg). As shown in Figure 2c, the model indicates that islets cultured at reduced pO2 (95 mmHg, or 12.5%) on PFC/PDMS (“PFC low”) have the highest percentage of tissue volume (~75%) within the physiological O2 range. In this setting, there was almost no critical anoxia, and hyperoxia was undetectable. In contrast, all other groups had either a significant percentage of both anoxic and hypoxic regions [control plasticware, both at atmospheric (21%) and low (12.5%) O2 concentration] or a large hyperoxic portion of tissue [PFC/PDMS at atmospheric (21%) O2 concentration]. All modeling was done at a standard plating density of ~175–200 IEQs/cm2.
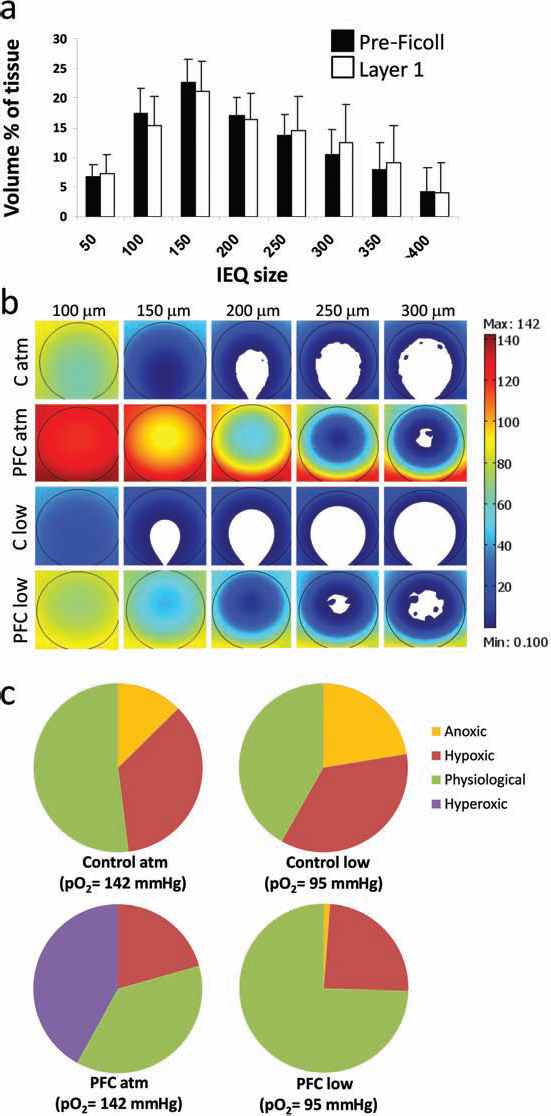
Theoretical oxygen diffusion modeling. (a) Retrospective analysis (n = 184) of the size distribution pattern in islet preparations. The largest percentage of islets (77% pre-Ficoll and 80% layer 1 postpurification) is in the 100–300 μm range, with the majority of them measuring between 150 and 200 μm. We used the latter figure for all subsequent finite element modeling calculations. (b) Theoretical modeling of all four groups: C atm [control dishes, atmospheric 21% (O2)], PFC atm [PFC/PDMS dishes, atmospheric 21% (O2)], C low [control dishes, 12.5% (O2)], and PFC low [PFC/PDMS dishes, 12.5% (O2)]. Islet sizes modeled are indicated in the top row (100–300 μm, in 50-μm increments). The color scale indicates pO2 levels from 0.1 mmHg (deep blue) to 142 mmHg (deep red). (c) Distribution of regions (anoxic, hypoxic, physiological, and hyperoxic; see definitions in the main text) in each one of the four groups. The “PFC low” group [PFC/PDMS dishes at 95 mmHg (12.5%); bottom right] exhibits the largest tissue volume percentage at physiological levels (74.5%) as well as the lowest percentages of hyperoxic and anoxic tissue volume (0% and 1.2%, respectively).
Targeting of a Physiological O2 Range Maximizes Islet Viability and Function
In order to test our theoretical calculations, we set up a series of experiments with isolated human islets. Individual preparations were analyzed according to several criteria, including β-cell fractional viability (24) and OCR index (16). Taken together, these criteria are generally predictive of islet cell function in humans, and help establish retrospectively whether a preparation is “good” or “bad.” The improvement observed in aliquots from low-quality preparations cultured in PFC/PDMS dishes (n = 5) was marginal compared to samples under control conditions. These preparations had very low OCR and glucose stimulation indices and were largely irresponsive regardless of the culture conditions (data not shown). However, eight samples were considered to be of fair/good quality according to the above parameters. Aliquots of these samples were plated in either regular dishes (control) or PFC/PDMS devices (PFC), both at normal (21%) and low (8–12.5%) O2 concentrations. As mentioned earlier, the latter was shown to be the optimal concentration range to target physiological islet oxygenation (~40 mmHg) using the PFC/PDMS system. Twenty-four hours after plating the aliquots in each of the four conditions, we analyzed islet loss (Fig. 3a), OCR index (Fig. 3b), and glucose-stimulated insulin release (GSIR) (Fig. 3c). Our data show that islets cultured in PFC platforms perform significantly better than those kept under regular conditions: When normalizing against the control group at 21% O2 (C atm =1), cell loss was 1.2±0.78 in the low O2 control group (C low), 0.91±0.13 in the PFC/PDMS group at 21% O2 (PFC atm), and only 0.53±0.45 in the PFC/PDMS group at low O2 (PFC low) (Fig. 3a). The difference in viability between C atm (regular conditions) and PFC low (maximal tissue volume at physiological pO2) was statistically significant (p < 0.01). Similarly, when using C atm as a normalizer (=1), OCR was 0.8±0.01 in C low, 1.09±0.05 in PFC atm, and 1.3±0.11 in PFC low (Fig. 3b). Once again, the difference in OCR between C atm (regular conditions) and PFC low (maximal tissue volume at physiological pO2) was statistically significant (p < 0.01). Finally, the GSIR delta normalized against the value obtained in C atm (=1) was 0.75 ± 0.321 in C low, 1.22 ± 0.25 in PFC atm, and 1.37 ± 0.17 in PFC low (Fig. 3c). These Delta values translate into relative GSIR indices of 1 (C atm), 0.68 ± 0.10 (C low), 1.91 ± 0.25 (PFC atm), and 1.94 ± 0.79 (PFC low). Statistical analyses of both metrics showed that the PFC low group was better than conventional culture (p = 0.05). In summary, survival and function were higher when adjusting the environmental O2 concentration to better target the islet physiological range.

Human isolated islets exhibit higher viability and function when cultured at physiological pO2 in PFC/PDMS dishes. (a) Overnight islet loss (-fold vs. control). C atm: Regular culture dishes, 21% (atmospheric) [O2]; C low: regular culture dishes, low [O2]. The latter percentage was adjusted within the 8–12.5% range based on the OCR of each individual preparation in order to target the maximal volume percentage at physiological pO2 within the tissue; PFC atm: PFC/PDMS dishes, 21% (atmospheric) [O2]; PFC low: PFC/PDMS dishes, low [O2] (see range above). n = 8 independent human islet preparations. The difference in viability between C atm (regular conditions) and PFC low (maximal tissue volume at physiological pO2) is statistically significant (p < 0.01). (b) Oxygen consumption rate (OCR) index (-fold vs. control). Groups are as above. n = 8 independent human islet preparations. The difference in OCR between C atm (regular conditions) and PFC low (maximal tissue volume at physiological pO2) is statistically significant (p < 0.01). (c) Glucose stimulated insulin release (Delta GSIR). Groups are as above. n = 5 independent human islet preparations. The difference in Delta between C atm (regular conditions) and PFC low (maximal tissue volume at physiological pO2) had a value of p = 0.05. Error bars, SD.
A Trend for Earlier Diabetes Reversal in PFC/PDMS-Cultured Islets
The marginal mass transplantation model in diabetic mice is particularly suited for analyzing the potential benefits of any given treatment (43). Transplantation of a small number of islets (500–1,000) under the kidney capsule leads to the reversal of hyperglycemia, but typically there is a measurable delay from the time of implantation. If an intervention is effective, time to reversal is reduced. This model allowed us to assess the effects of pretransplantation culture (24 h) of human islet preparations using PFC/PDMS devices.
Following isolation, human islets were allowed to recover overnight under regular conditions, and then separated in two aliquots: one was kept in standard dishes at 21% O2 and the other placed in PFC/PDMS dishes at 8–12.5% O2 (final concentration calculated based on the baseline OCR of each preparation). As mentioned earlier, the latter range sustains the maximal volume of the tissue at physiological pO2 when using PFC/PDMS dishes.
After 24 h, islets were aliquoted for transplantation (3 mice/group, 1,000 islets/animal) and in vitro studies. The OCR was consistently higher (1.85 vs. 1.43) in the PFC/PDMS group, which is in line with our previous results. This parameter is highly predictive of function after transplantation (17), with higher indices normally indicating faster diabetes reversal times upon transplantation in mice. Similarly, the Glucose Stimulated Insulin Release (GSIR) “delta” (high minus low 1) was on average twofold higher in the PFC group than in controls.
The outcome of transplantation experiments is represented in Figure 4. Based on in vitro metrics of viability, the preps were retrospectively grouped into either “rapid reversal” or “delayed/no reversal”. In the first group, there was a clear trend (p = 0.10) toward greater percentage of diabetes reversal (100% vs. 80%), defined as 5 consecutive days of blood glucose <200 mg/dl. Additionally, there was a trend toward earlier reversal times in the PFC group (5 days vs. 10.5 days) despite the small n (five experiments). The in vivo outcomes coupled with the in vitro assessments indicate that preparations with poor basal in vitro performance typically are not rescued by culture in the PFC/PDMS. Conversely, preparations with in vitro performance that correlates with rapid reversal in the full mass bioassay are improved both in their in vitro performance and in the marginal mass setting. Additional evidence was obtained from TaqMan-based low density array (TLDA) cards designed to quantitatively measure the expression of a panel of 94 genes involved in apoptosis, which has been already proven to be at the root of hypoxia-induced islet cell death (19,39). The analysis of samples representative of a “good” preparation (HP1915) and a “bad” one (HP1927) is presented in Figure 4b. Culture of HP1915 islets in PFC/PDMS dishes reduced the expression of proapoptotic genes by an entire order of magnitude versus controls (Fig. 4b, bottom panel). In contrast, the relative expression of these genes was comparable in both groups when using a bad preparation (HP1927, Fig. 4b, top panel). Absolute levels of expression were highly elevated in HP1927 versus HP1915 (data not shown), suggesting that, for the most part, these islets were irreversibly apoptotic. In summary, our studies demonstrate that (a) human islets exhibit enhanced survival and function when cultured in PFC/PDMS dishes; (b) this means of culture is translated in faster diabetes reversal times when islets are transplanted in immunodeficient diabetic mice; and (c) culture in PFC/PDMS dishes cannot improve the viability of poor quality islet preparations.

Human isolated islets exhibit higher viability and function when cultured at physiological pO2 in PFC/PDMS dishes. (a) In vivo test of function of human islet preparations cultured in either PFC/PDMS-low oxygen (PFC group) or regular conditions-atmospheric [O2] (control group). Kaplan-Meier analyses of the gain of normoglycemia versus time after transplantation of a marginal mass of islets into streptozotocin (STZ)-treated, diabetic mice are shown for the experiments using poor islet preparations (top) and those using good ones (bottom). (b) Poor islet preparations cannot be rescued by optimized oxygenation conditions. A TaqMan low density array (TLDA) qRT-PCR analysis of a panel of genes involved in apoptosis is presented to exemplify the differences between a good preparation (HP 1915) and a bad one (HP 1927). In the latter, culture in PFC/PDMS dishes and low oxygen prevents the upregulation of proapoptotic genes observed when placing an aliquot of the same preparation under control conditions [regular dishes, atmospheric (O2)]. In contrast, HP 1927, a bad preparation with marginal GSIR readings, showed no difference in apoptosis between regular and oxygen-optimized conditions. This suggests that islets may have had a substantial degree of apoptosis before the placement in the two culture conditions, which culture in the optimized one could not revert. BCL3, B-cell lymphoma 3; NFKB1, nuclear factor of κ light polypeptide gene enhancer in B-cells 1; BCL2A1, BCL2-related protein A1; BCL2L1, BCL2-like 1; BIK, BCL2-interacting killer; BNIP3L, BCL2/adenovirus E1B 19 kDa interacting protein 3-like; CASP6, caspase 6; DAPK1, death-associated protein kinase 1; HIP1, Huntington interacting protein 1; BIRC1, baculoviral inhibitory apoptosis protein (IAP) repeat-containing protein 1; NFKBIA, nuclear factor of κ light polypeptide gene enhancer in B-cells inhibitor, α; RELA, v-rel reticuloendotheliosis viral oncogene homolog A.
Discussion
A heightened sensitivity to O2 is rooted in the very ontogeny of β-cells. Beyond the primary role of O2 in respiration, its evolving tension levels in the embryonic pancreas are known to shape the outcome of differentiation by directly modulating not only developmental pathways such as Wnt/β-catenin and Notch but also the chromatin rearrangement of pancreatic-specific regulatory regions (18). A switch from “low” to “high” pO2 in the organ around the time of the onset of blood flow may trigger a wave of endocrine (and especially β-cell) differentiation (17). The molecular mechanism was first proposed to be hypoxia-inducible factor (HIF)-1α dependent by our team (17), a hypothesis later confirmed by others (21).
β-Cells are not any less sensitive to O2 during adulthood than they were during their development. As described in Introduction, they are exquisitely adapted to a rather narrow pO2 range, above or below which they will rapidly perish. It is paradoxical that, while we go to great lengths to preserve β-cell viability in vitro by making sure most environmental factors are on target, we still insist on the use of outdated culture platforms where oxygenation (arguably the most important one) is anything but physiological.
Most culture devices are variations on the common theme of a plastic container that holds cells or cellular aggregates bathed in medium. A design where air flows only from the top is bound to create diffusion gradients that are harmful for islets (36). Here, we present a novel means of culture in which air flows to the tissue in a bidirectional manner. This system is not necessarily designed to provide more O2, but rather to minimize the occurrence of gradients throughout three-dimensional aggregates. Indeed, mathematical modeling of O2 diffusion indicated that our devices best targeted the optimal physiological pO2 within islets when the environmental [O2] was adjusted to a lower-than-atmospheric 8-12.5%. This theoretical prediction was subsequently validated by key tests of viability and function, in which the above setting proved to be far superior to controls. The notion that the way of delivery is more important than the actual O2 concentration was established in our previous work on pancreatic stem cells (17), where we showed that a mere increase of the environmental pO2 in standard culture only contributed to the creation of even steeper gradients within the tissue, without any beneficial effect on survival or differentiation rate.
Papas and collaborators (41) have previously shown that human islets can be cultured at higher densities when plated on rubber membranes. Better O2 diffusion, they claimed, was sufficient to prevent high density-dependent hypoxia. However, we are the first ones to report that targeting a physiological oxygenation range in human islets results in better viability and function. Our system also differs from theirs in that an integral component of the membranes is PFC, which imparts higher O2 diffusivity than silicone alone.
A somewhat counterintuitive result is that the benefits of in vitro culture in PFC/PDMS platforms do not readily translate to better in vivo function. Although there is a clear trend toward faster diabetes reversal times versus islets cultured under control conditions, the difference is not significant. One obvious potential explanation is that a larger n might be necessary to achieve statistical significance, especially given the inherent variability between islet preparations. However, if taken at face value, our results could also be explained by the paradoxical (yet well-documented) effect that stressed islets may perform better in vivo than nonstressed ones. Hogan et al established that ischemic preconditioning improves islet recovery and function after cold preservation of the pancreas (23), a result that confirms findings in other transplantation models (22,40,44). Mounting evidence points to the hypothesis that the preactivation of stress pathways (provided that they do not reach a point of no return, which would be conducive to apoptosis) may result in better in vivo outcomes (23). If this hypothesis proved to be true, it is conceivable that the beneficial effects of PFC/PDMS culture in enhancing islet viability and function in vitro may be partially counteracted by a relative lack of preparedness of these islets to sustain the stress associated with transplantation. This intriguing hypothesis will be the basis of future work in our laboratory to explore if the benefits of PFC/PDMS culture and prestressing prior to transplantation can be compounded.
Another important observation is that islets of poor quality are already beyond recovery. The quality of islet preparations is dictated by numerous factors, ranging from donor characteristics to cold ischemia time and isolation-related stress. Our research indicates that physiological oxygenation can make a difference in islet preparations of moderate to high quality, which would otherwise suffer significant cell death and decreased biological activity postisolation. In a 2003 study, the mere addition of liquid PFC to the preservation solution used for pancreata prior to processing was enough to increase by fourfold the percentage of organs that ended up being used for transplantation (46). Still, more than half of the pancreata in the PFC group did not make the cut, in most instances due to poor overnight recovery in a setting (standard culture) where PFC was no longer available. The reported 2.5-fold increase in postisolation viability, as we observed with our system, could be decisive in making a borderline preparation transplantable, or a good one used for two patients instead of one. The fact that PDMS/PFC culture islets not only survive better but also exhibit highly improved biological activity reinforces the above perception. In a field where even modest increases in viability and function have justified the exploration of much more aggressive strategies (such as gene therapy), the conceptual simplicity of our system makes it particularly appealing from a clinical point of view. This is true not only of current islet transplantation trials but also of future stem cell-based therapies for type 1 diabetes. As the latter are readied to take up the baton in this new decade, we see already that a prerequisite to derive islets from stem cells might be to mimic the physiological oxygenation they need to survive in the first place. The successful application of our findings could lead to new standards not only for β-cell biology and differentiation studies but also for many other O2-sensitive culture applications.
Footnotes
Acknowledgments
This work was funded by the Wallace H. Coulter Center (WHCC), the Diabetes Research Institute Foundation (DRIF), and the National Institutes of Health/National Institute of Diabetes and Kidney Diseases (NIH/NIDDK) Phase I SBIR Grant 1R43DK083832. This work was also supported in part by NIH grants DK70460 and NIH U42 RR016603 and by the City of Hope (Duarte, California). We wish to acknowledge the contribution of the faculty and staff of the Preclinical Cell Processing and Translational Models Core at the Diabetes Research Institute (University of Miami). We also thank P. Latta (Converge, Inc.) and R. Pastori (University of Miami) for their critical review of the manuscript. C.F. researched the data and wrote the manuscript; S.C., S.A.-C., F.E., and A.B. researched the data; R.P., C.R., and L.I. contributed to the discussion; J.D.-B. wrote the manuscript. J.D.-B., C.R., L.I., C.F., and R.P. disclose their participation as stockholders in Ophysio, Inc., which is the sole licensee of the PFC/PDMS technology from the University of Miami. Dr. Juan Domínguez-Bendala is the guarantor of this work, had full access to all the data, and takes full responsibility for the integrity of data and the accuracy of data analysis.

