Abstract
Mesenchymal stem cells (MSCs) are potential and optimal stem cells in clinical cell therapy, and fetal bovine serum (FBS) is widely used for expansion of MSCs, despite the risks of infectious disease transmission and immunological reaction of the xenogenic origin. This study was designed to compare human four blood group cord blood serum (CBS) with FBS in culturing human placenta-derived mesenchymal stem cells (hPDMSCs), which were derived from four blood group donors. The expansion medium included: 10% FBS, 10% A-CBS, 10% B-CBS, 10% O-CBS, and 10% AB-CBS. Cumulative population doubling, generation time, fold expansion rates and differentiation capacity, cell cycle, and immunophenotype were also assessed. The results showed that fold expansion rate and cumulative population doubling of hPDMSCs significantly increased during long-term MSC expansion in CBS medium, but the generation time decreased compared with FBS. CBS might be an effective supplement for stem cells expand rapidly ex vivo. Cell cycle and differentiation assays showed that most of the hPDMSCs expanding in the presence of CBS were in stationary phase, which was the characteristic of stem cells, and they retained their ability to differentiate into chondrogenic and endothelial cells. By comparing these four blood groups of CBS, we found that there was no significant difference among different blood groups in culturing hPDMSCs, which were isolated from different blood group donors. So CBS may be an optimal replacement to avoid the risks of FBS application in expansion of stem cell for clinical cell therapy and tissue engineering.
Keywords
Introduction
Mesenchymal stem cells (MSCs) exhibit multilineage differentiation potential towards diverse types of tissues including bone, cartilage, adipose, and endothelium (16, 30). Moreover, MSCs have the ability to support hematopoietic progenitor cells as feeder layer cells. At the same time, MSCs have a lower incidence of inducing immunological rejection because of their low immunogenicity (19,31). Today, regarding their clinical potential, human MSCs have been successfully used in the treatment of most diseases, such as the regeneration of damaged cardiovascular, metabolic disorders, bone marrow failure syndromes, and graft-versus-host disease (3,6, 13,29). It is necessary to develop a culture system ex vivo to expand stem cells for clinical application (11,23).
To date, fetal bovine serum (FBS) as the convenient supplement has been used for the isolation and cultivation of MSCs ex vivo or in vivo. However, FBS, as an animal serum, carries an intrinsic risk of bacterial or viral contamination as well as a potential risk of prion diseases such as bovine spongiform encephalitis (9). Moreover, xenogeneic proteins in FBS have been internalized by MSCs (7—30 mg/108 cells) and these proteins can trigger adverse responses such as an arthus-like immune response or anaphylactic reactions, requiring repeated administration in some patients (12,35,37,38). Recently, researchers have found several new culture media that have overcome these shortcomings, such as adult serum, platelet lysate, and cord blood serum (CBS).
During pregnancy, cord blood as a lifeline of nourishment is delivered from mother to fetus. With the nutrition in cord blood, the embryo evolves from the fertilized egg into a mature individual form. In CBS, the content of growth factors is higher than in adult blood serum, as some researchers have reported. For example, as shown in Table 1, the levels of erythropoietin (EPO), granulocyte colony-stimulating factor (G-CSF), granulocyte macrophage colony-stimulating factor (GM-CSF), and colony stimulating factor 1 (CSF-1) in CBS were higher than that in adult serum (18,21,32). The content of stem cell factor (SCF), interleukin-3 (IL-3), and IL-6 in cord blood serum was also higher (2,26,34,39). These factors can promote stem cells to grow. In cord blood, T and natural killer (NK) cells are naive and not primed for activation. At the same time, there is an activation growth factor absent in cord blood, which is present in adult serum (5). Therefore, in this study, human CBS was selected as the supplement in cell culturing ex vivo, compared with FBS. In blood transfusion, one of principles is that blood transfusion must be performed with the same ABO blood group between the recipient and the donor (ABO compatibility).
Comparison of Growth Factors

EPO, erythropoietin; G-CSF, granulocyte colony stimulating factor; GM-CSF, granulocyte-macrophage colony stimulating factors; CSF-1, colony-stimulating factor 1. In the previous studies, the levels of EPO, G-CSF, GM-CSF, and CSF-1 in cord blood serum were higher than in adult serum.
Placenta, a biological waste after delivery, has become an attractive source of MSCs for basic and clinical applications (10,14), and in our previous study we found human placenta-derived mesenchymal stem cells (hPDMSCs) had the same character as MSCs derived from human bone marrow (4,40,41).
In this study, the feasibility of using CBS as a replacement for FBS to culture hPDMSCs was evaluated. More importantly, first we compared the effect of ABO blood group by using four blood group CBSs to expand hPDMSCs, which were isolated from four blood group donors. Also, whether or not a distinct characteristic of hPDMSCs can be seen was assayed during cultured with CBS ex vivo.
Materials and Methods
Preparation of CBS and Isolation of hPDMSCs
Term cord blood samples (37—41 weeks gestation) from healthy donors (screened for infectious diseases, including HIV 1 and 2, hepatitis B and hepatitis C virus, and sexually transmitted diseases and without any complication during delivery) were obtained with informed consent approval according to procedures of the institutional review board. After blood group detection (Anti-A and Anti-B Bloodgroup Reagents, Brother Biotech, China), the blood (n = 20, 5 per blood group) was recovered in the absence of anticoagulants according to sterile principle and allowed to clot for 4—16 h at 4°C. The blood was centrifuged at 3,500 x g for 20 min at 20°C, the supernatant was collected, then centrifuged again at 1500 x g for 10 min at 20°C. Serum was sterilized by passing through a 0.22-mm filter (Millipore, USA), without bacterium contamination after culturing test in 7 days. Five samples of every blood group were pooled to avoid individual variations and stored at −20°C until used.
Term (37—41 weeks gestation, n = 12, 3 per blood group, according to the case) placentas from healthy donors were obtained by the same screening process above of cord blood, and informed consent approval according to procedures of the institutional review board after abdominal delivery. The deciduas were stripped and dissected into small pieces, washed several times with phosphate-buffered saline (PBS), and mechanically minced 1 mm3. The minced pieces were subsequently laid uniformly on petri dishes and cultured in Dulbecco's modified Eagle's medium-low glucose (DMEM, Invitrogen, USA), supplemented with 10% FBS (Bioind, Israel), 0.1 mM β-mercaptoethanol (Sigma, USA), 2 mM l-glutamine (Invitrogen, USA), 0.1 mM nonessential amino acids (MEM, Invitrogen, USA), 1% penicillin/streptomycin (Gibco, USA) maintained at 37°C in a humidified atmosphere with 5% CO2. Medium was replaced every 3—5 days. When cells were above 80% confluence, they were harvested and expanded with different culture conditions.
Preparation of Supplement and Media
hPDMSCs derived from four blood groups (A, B, O, AB; n = 3 each) were cultured under different culture conditions: DMEM-low glucose described previously supplemented with 10% different serum, including five groups: FBS (Bioind, Israel), A-CBS (A blood group CBS), B-CBS (B blood group CBS), AB-CBS (AB blood group CBS), O-CBS (O blood group CBS).
Proliferation Kinetics
hPDMSCs derived from four blood groups donors were respectively expanded using five different supplements. At each passage, 0.6 × 104 cells/cm2 were plated in a six-well plate (TPP, Switzerland) and cultured for 3—4 days, then digested with 0.25% trypsin. Cells (0.6 × 104 cells/cm2) were replated and 100 μl cell suspension was plated in a 96-well plate (TPP, Switzerland) overnight, then incubated with 50 μl/well MTT reagents (MTT cell proliferation and Cytotoxicity Detection, Keygen, China) for 4 h. The 150 μl dimethyl sulfoxide (DMSO) was added and the cell absorbance was analyzed, equipped with a 490 nm argon laser according to the manufacturer's instructions, which drew the growth curve of hPDMSCs. From passage 2, the fold expansion rate was determined by ratio of the absorbance of harvest cells to that of plated cells. The other way, hPDMSCs were plated at the density of 0.6 × 104 cells/cm2 on first passage. When cells were over 80% confluent, they were passaged at a dilution of 1:2. The cell expansion was discontinued if it did not reach a minimum confluence of 50% after 14 days. The population doubling was determined by using the formula (1): X = [log 10(NH) —log10(N1)]/log10(2), where N1 is the plated cell number and NH is the cell number at harvest.
The population doubling for each passage was calculated and then added the population doubling of the previous passages to determine the cumulative population doubling.
In addition, the generation time (average time between two cell doublings) of hPDMSCs within all medium conditions was calculated at each passage using the following formula (1): X = [log10(2)] x Δt/[log10 (NH) — log10(N1)], where N1 is the plated cell number, NH is the cell number at harvest, and Δt is the number of days for cells expansion at each passage.
Immunophenotype of hPDMSCs
The surface makers' expression of hPDMSCs before and after expansion were analyzed by flow cytometry at passage 0 (n = 3 per group) and passage 8 (n = 3 per group) for all culture conditions. After harvesting the adherent cells, a cell count was taken and approximately 50,000 cells were used for cell surface antigen expression studies. The harvested cells were washed using staining buffer containing 4% FBS in PBS and stained with the following phycoerythrin (PE)- or fluorescein isothiocyanate (FITC)-conjugated mouse anti-human antibodies: anti-CD34-PE, anti-CD105-PE, anti-CD31-FITC, anti-CD45-FITC, and anti-CD166-FITC (all from B&D, USA). These cells were acquired on a FACSC analyzed using a FACScan flow cytometer (20,36).
Cell Cycle of hPDMSCs
At passage 8, hPDMSCs were digested with 0.25% EDTA trypsin, washed with PBS, fixed in 70% ethanol, water bathed with RNase A, and stained with propidium iodide (PI) according to the manufacturer's instructions (cells cycle kit, Keygen, China). Then these cells were acquired using a FACScan flow cytometer.
Differentiation of hPDMSCs Assays
hPDMSCs were tested differentiation capacity at passage 8 after being expanded with all culture conditions.
Chondrogenic Differentiation
For chondrogenic differentiation, as described by Estes et al. (8), the media contained high-glucose DMEM (4.5 g/ml) supplemented with 10−7 mol/L dexamethasone (Sigma, USA), 10 ng/ml recombinant human transforming growth factor-β1 (R&D, Switzerland), 6.25 μg/ml insulin (Biodee, USA), 6.25 μg/ml transferin (Biodee, USA), 50 mg/ml vitamin C (Sigma, USA), 100 ng/ml insulin-like growth factor (Biovision, USA), and 5% FBS or 5% CBS, l-DMEM medium. Differentiation medium was replaced every 3 days. After 3 weeks, the differentiated cells were fixed with 4% formalin and immunocytochemistry was carried out using mouse anti-human collagen (1:100; Boster, China) according to the manufacturer's instructions.
Endothelial Cells Differentiation
For endothelial cell differentiation, as described by Fukuchi et al. (10), the media contained 50 ng/ml recombinant human vascular endothelial growth factor (rHuVEGF, Sigma, USA), 10 ng/ml basic fibroblast growth factor (rHubFGF, Sigma, USA), 0.1 mM β-mercaptoethanol (Sigma, USA), 2 mM l-glutamine (Invitrogen, USA), 0.1 mM nonessential amino acids (MEM), and 1% penicillin/streptomycin, and 5% FBS or CBS, l-DMEM medium. Differentiation medium was replaced every 3 days. At day 8, the differentiated cells were fixed with 4% formalin and immunocytochemistry was carried out using mouse anti-human vWF (1:100, Maxim, China) according to the manufacturer's instructions.
Statistical Analysis
All experiments were repeated three times. Results were expressed as mean ± SEM. Significance tests were performed using ANOVA and LSD post hoc with values of p < 0.05 considered significant. All other data were processed using SPSS.13 for Windows.
Result
Morphology of hPDMSCs
After being cultured for 3—7 days, a small amount of short- spindle cells crept from minced placenta pieces; subsequently more long-spindle cells and fewer cobble stone-like cells transformed and formed individual colonies (Fig. 1, A1). After 10—14 days, hPDMSCs were over 80—90% confluent and were passaged. The cells were fibroblast-like and formed colonies again (Fig. 1, A2).

The morphology of human placenta-derived mesenchymal stem cells (hPDMSCs) (100x magnification). (A1) From 3 to 14 days after inoculation, fibroblast-like cells were transferred from the placenta specimens. (A2) At passage 3, hPDMSCs presented the morphology of fibroblast-like cells. (B) Morphology of B-hPDMSCs after expanding with five culture conditions. At passage 6, cells of cord blood serum (CBS) groups appeared smaller, more spindle shaped, with fewer cytoplasmic processes and showed a distinct mesh-like growth pattern.
Morphology of hPDMSCs After Being Expanded with Different Supplement
hPDMSCs of all groups displayed a characteristic fibroblast-like morphology. However, hPDMSCs expanded with CBS appeared smaller, more spindle shaped, with fewer cytoplasmic processes and retained their spindle-shaped morphology for a longer time than those cells expanded with FBS and showed a distinct mesh-like growth pattern (Fig. 1B). From passage 6—7 hPDMSCs of the FBS group gradually lost their spindle-like shape and were more flattened, more spherical, and bigger, but from passage 10—11 hPDMSCs of the CBS groups had the same change. Morphology of hPDMSCs was not significantly different among the different CBS blood groups.
Proliferation Activity of hPDMSCs
The Fold Expansion Rates of hPDMSCs
At each passage, hPDMSCs were plated at the same density and cultured for the same time. The fold expansion rate was determined from passage 2.
As Figure 2A shows, all groups presented similar change tendency. Compared with FBS group, all CBS group cells expanded more passages (FBS: 10.25 ± 0.95, CBS: 13.75 ± 0.5; p < 0.05); the maximum of fold expansion rate of all CBS group cells was higher (e.g., A-hPDMSCs: A-CBS: 14.08 ± 1.174, B-CBS: 15.175 ± 0.431, O-CBS: 14.865 ± 0.87, AB-CBS: 14.24 ± 1.527, FBS: 9.16 ± 1.541) and the platform phase of the high fold expansion rate was longer. From passage 7 to expansion termination, in every passage the fold expansion rate of all CBS group cells was higher than that of the FBS group cells (p < 0.05) and there were no significant difference among different CBS groups (p > 0.05) (e.g., A-hPDMSCs: A-CBS: 11.495 ± 1.755, B-CBS: 12.905 ± 0.345, O-CBS: 11.05 ± 3.2, AB-CBS: 9.695 ± 2.155, FBS: 6.225 ± 1.005).

Comparison of the fold expansion rate of hPDMSCs. (A) Comparison of the fold expansion rate for all culture conditions. Cells cultured with CBS had a significantly higher expansion rate than fetal bovine serum (FBS), whereas there was no significant difference observed among different CBS blood groups. *p < 0.05, respectively compared with each CBS group. (B) Comparison of the fold expansion rates for hPDMSCs isolated from different blood group donors. Cultured with the same blood group serum supplement medium, there was no significant difference observed among different blood group hPDMSCs (p > 0.05, respectively compared with each group).
As shown in Figure 2B, while different blood group hPDMSCs were expanded in the same blood group serum supplement medium, the fold expansion rate of these hPDMSCs was not significantly different (p > 0.05).
Cumulative Population Doubling and Generation Time of hPDMSCs
On first passage hPDMSCs were plated at the density of 0.6 × 104 cells/mm2 and then they were passaged at a dilution of 1:2. The cell expansion was discontinued if cells did not reach a minimum confluence of 50% after 14 days. The cumulative population doubling and the generation time of each passage were determined by using the formula above.
The cumulative population doubling was calculated at passage 11. Figure 3A shows the cumulative population doubling of all CBS blood groups was significantly higher than that of FBS (p < 0.05), but we did not observe a significant difference among all CBS blood groups (p > 0.05) (e.g., A-hPDMSCs: A-CBS: 61.59 ± 3.412, B-CBS: 62.25 ± 3.735, O-CBS: 62.23 ± 3.155, AB-CBS: 61.1 ± 3.194, FBS: 49.27 ± 2.366).
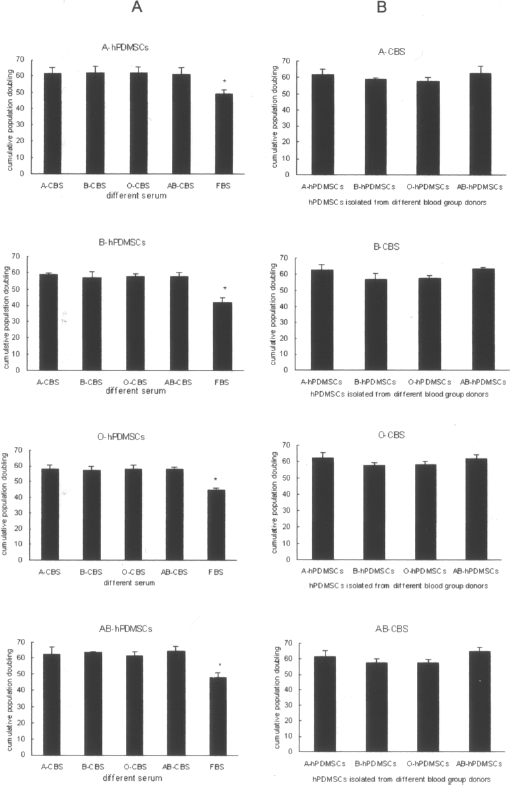
Comparison of the cumulative population doubling of hPDMSCs. (A) Comparison of cumulative population doubling for all culture conditions. At passage 11, cells cultured with CBS had a significantly higher cumulative population doubling than FBS, whereas there was no significant difference observed among the different CBS blood groups. *p < 0.05, respectively, compared with each CBS group. (B) Comparison of cumulative population doubling of hPDMSCs isolated from different blood group donors. Cultured with the same blood group serum supplement medium, there was no significant difference observed among different hPDMSC blood groups (p > 0.05, respectively, compared with each group).
Similarly, hPDMSCs derived from different blood group donors were cultured in the same blood group CBS supplement medium; the cumulative population doubling was not obviously different (Fig. 3B, p > 0.05).
We calculated the generation time at each passage. As observed from Figure 4A, the generation time of hPDMSCs prolonged gradually after maintaining a stable period. It revealed that the higher proliferation potential of MSCs began to reduce gradually after passages. Compared with the FBS group, all CBS group cells could be expanded for more passages (FBS: 11—13; CBS >15). At the same time, the shorter generation time phase was longer than that of the FBS group. For the different blood group hPDMSCs, from passage 7, 8, 9, 10, respectively, the generation time of all CBS groups was shorter than that of FBS (p < 0.05) and there was no significant difference among the different CBS blood groups (p > 0.05) (e.g., A-hPDMSCs, P8 A-CBS: 2.182 ± 0.2522, B-CBS: 1.965 ± 0.3291, O-CBS: 1.978 ± 0.1066, AB-CBS: 2.272 ± 0.0984, FBS: 3.178 ± 0.4621).

Comparison of the generation times of hPDMSCs. (A) Comparison of the generation time for all culture conditions. For the different blood group hPDMSCs, from passage 7, 8, 9, 10, respectively, the generation time of all CBS groups was shorter than that of FBS. *p < 0.05, and there were no significant difference among different CBS blood groups (p > 0.05). (B) Comparison of the generation time of hPDMSCs isolated from different blood group donors. Cultured with the same blood group serum supplement medium, there was no significant difference among four hPDMSCs blood groups (p > 0.05).
At the same time, as shown in Figure 4B, the generation time of hPDMSCs derived from four blood group donors was not significantly different throughout culturing in the same CBS blood group (p > 0.05).
Characterization of Surface Marker Expression of hPDMSCs
The flow cytometry analysis showed that hPDMSCs derived from different blood group donors displayed similar phenotypes with bone marrow-derived mesenchymal stem cells: they were stained positive for CD105 and CD166, but negative for CD31, CD34, and CD45 (Fig. 5A). There was no change in surface marker expression of hPDMSCs between passage 0 and passage 8 in culturing with CBS (p > 0.05) (Fig. 5B).

Characterization of surface marker expression of hPDMSCs. The result of B-hPDMSCs expanded with A-CBS supplement. (A) Phenotype of hPDMSCs at passage 8 by flow cytometry analysis demonstrated positive for CD105 and CD166, but negative for CD31, CD34, and CD45. (B) After culturing with A-CBS supplement, there was no changed in surface marker expression of B-hPDMSCs between passage 0 and passage 8 (p > 0.05).
Cell Cycle of hPDMSCs
Cell cycle analysis of hPDMSCs at passage 8 was examined by flow cytometry. Most of the hPDMSC groups were in G0/G1 phase, and the percentage of CBS groups was higher than that of the FBS group (p < 0.05) (Fig. 6).

Cell cycle of hPDMSCs for all culture conditions. (A) Cell cycle analysis of B-hPDMSCs at passage 8 were examined by flow cytometry. (B) Most of the hPDMSCs were in G0/G1 phase, and the percentage of CBS group cells was higher than FBS cells in G0/G1 phase (p < 0.05).
Differentiation Capacity of hPDMSCs
After being expanded with all culture conditions, the chondrogenic and endothelial differentiation potential of hPDMSCs was tested at passage 8. We demonstrated that FBS and CBS did not adversely affect the differentiation capacity of hPDMSCs from all donors. Chondrogenic differentiation was confirmed by type II collagen staining assays (Fig. 7A—E) and endothelial differentiation confirmed by vWF (Fig. 7F—J). We showed hPDMSC differentiation capacity was maintained throughout long-term culture with CBS.

Chondrocyte and endotheliocyte differentiation potential of B-hPDMSCs for all culture conditions (400x magnification). hPDMSCs were induced by chondrogenic differentiation medium. Immunocytochemistry was carried out using mouse anti-human collagen II, shown in (A—E, blue arrows). (A) B-hPDMSCs cultured in A-CBS; (B) B-hPDMSCs cultured in B-CBS; (C) B-hPDMSCs cultured in O-CBS; (D) B-hPDMSCs cultured in AB-CBS; (E) B-hPDMSCs cultured in FBS. B-hPDMSCs were induced by endothelial cells differentiation medium. Immunocytochemistry was carried out using mouse anti-human von Willebrand factor (vWF), shown in (F—J, green arrows). (F) B-hPDMSCs cultured in A-CBS; (G) B-hPDMSCs cultured in B-CBS; (H) B-hPDMSCs cultured in O-CBS; (I) B-hPDMSCs cultured in AB-CBS; (J) B-hPDMSCs cultured in FBS.
Because the trend was very same among all blood group hPDMSCs, only the data on characterization of morphology, surface marker expression, cell cycle, and differentiation capacity of B-hPDMSCs were shown.
Discussion
In the past two decades, stem cell transplantation has become the new treatment for many diseases. It has been demonstrated that MSCs, the multipotent and nonhematopoietic stem cells, were the ideal cells for cell therapy and tissue engineering by a number of clinical trials. However, in the clinic, almost all cell therapies require the number of cells to be over 1 × 108 (37). Therefore, clinicians have to face a bottleneck of cell insufficiency. Different studies have focused on the identification of effective and safe supplement for expansion of cells. Although serum-free medium could reduce the danger of FBS, the growth factors in serum are essential for cell proliferation. So it is necessary to find a replacement for FBS in stem cell expansion throughout long-term culturing ex vivo. These include autologous or allogeneic human serum, human plasma, CBS, and human platelet derivatives (7,15,17,20,22,24,25,27,28,33,36). Of note, CBS is safe serum medium that carries no risk of xenogeneic antigen and has lower immunogenicity. In addition, more distinct stimulating factors in CBS can contribute to the unique cellular characteristics of human MSCs; it is suggested that CBS may similarly provide a unique microenvironment for ex vivo expansion of MSCs (15,25,36).
Very recently, newly identified MSCs have been isolated from human placentas, which have the capacity to differentiate into osteoblasts, chondrocytes, adiporcytes, skeletal muscle cells, and neurons (4,40,41). Our previous studies have shown that the hPDMSC marker profile is similar to that of human bone marrow MSCs (40,41). Due to the easy accessibility and abundant cell numbers, hPDMSCs have become an attractive alternative source of progenitor cells for basic research in our laboratory.
In this study, we first selected four blood groups to evaluate the feasibility of using CBS as a replacement for FBS for culturing hPDMSCs and also compared the efficacy of blood group.
As shown, compared with FBS, hPDMSCs expanded with CBS appeared more primitive (smaller, more spindle shaped with fewer cytoplasmic processes) as the morphology of MSCs cultured with platelet lysate in Kocaoemer et al.'s report (20), and retained their spindle-shaped morphology during longer term culture. At the same time, a distinct mesh-like growth pattern showed that having more cell—cell junction to benefit to the intercellular interaction (Fig. 1). In our study, cultured in CBS ex vivo, both cumulative population doubling and fold expansion rate of cells increased, and the increased platform period of fold expansion rate was longer than that in FBS (Figs. 2A and 3A). In addition, cultured in CBS, the generation time of cells was shorter (Fig. 4A). These results revealed that cord blood serum contained abundant growth factors to maintain the proliferation potential of hPDMSCs over passages and to obtain large amounts of hPDMSCs ex vivo within a short time as others reported (15,25,36).
Cell cycle assays showed that most hPDMSCs expanded in the presence of CBS were in stationary phase, which is the characteristic of MSCs. No difference in hPDMSC surface markers (CD31, CD90, CD34, CD45, CD166, and CD105) was observed after cultured in CBS. hPDMSCs retained their ability to differentiate into chondrogenic and endothelial cells. Taken together, in our study, hPDMSCs maintained their stem cells character after being cultured in CBS ex vivo.
To determine if there is difference among four blood groups CBS on hPDMSCs' expansion capability ex vivo, we also did a comparison, both among culturing hPDMSCs derived from four blood group donors with the same blood group CBS, and among the same blood group hPDMSCs cultured with four CBS blood groups. There was no difference in cumulative population doubling, fold expansion rate, generation time, (Figs. 2B, 3B, 4B), cell cycle, surface markers, and differentiation capability. In cord blood, the content of immunologic active material was less than bone marrow and whole blood (5). Our next experimental design is about immunization of hPDMSCs expanded in the presence of CBS.
In our study, from one cord blood donation of 100 ml, a maximum of 40 ml serum can be obtained (i.e., supplementing medium with 10% CBS yields 400 ml of expansion medium). This expansion medium should be enough for a 1-week expansion period. And different CBS blood groups did not display a different effect on expansion of hPDMSCs. This breaks the limitations of CBS application among different blood groups. CBS is no risk to the donor (mother and fetus), had no donor attrition, and minimal risk of viral transmission; at the same time, it is immediate availability, has higher proliferative potential, and lower immunogenicity. Our method would allow us to obtain large amounts of CBS required for sufficient expansion of stem cells in short term at a clinical scale.
In conclusion, we showed that CBS would be an optimal replacement for FBS in expansion of stem cells for clinical cell therapy and tissue engineering. In addition, as CBS, placenta, an abundant tissue without suffering donors, would be a very potential and easily accessible source of stem cells for cell therapy in the future.
Footnotes
Acknowledgments
This study is funded by Innovation project grant #2006226031-211, which is supported by Science and Technology Department of Liaoning Province China. Special thanks to Professor. Haiqin Ren, and all donors for the donation of placenta and cord blood. The authors declare no conflicts of interest.
