Abstract
Hypoxia plays an important role in limiting the engraftment, survival, and function of intrahepatically transplanted islets. Mesenchymal stem cells (MSCs) were recently used in animal models of islet transplantation not only to reduce allograft rejection but also to promote revascularization. Among different possible origins, adipose tissue represents a novel and good source of MSCs. Moreover, the capability of adipose tissue-derived stem cells (ASCs) to improve islet graft revascularization was recently reported after hybrid transplantation in mice. Within this context, we have previously shown that hyaluronan esters of butyric and retinoic acids can significantly enhance the rescuing potential of human MSCs (hMSCs). Here we evaluated whether ex vivo preconditioning of human ASCs (hASCs) with a mixture of hyaluronic (HA), butyric (BU), and retinoic (RA) acids may result in optimization of graft revascularization after islet/stem cell intrahepatic cotransplantation in syngeneic diabetic rats. We demonstrated that hASCs exposed to the mixture of molecules are able to increase the secretion of vascular endothelial growth factor (VEGF) as well as the transcription of angiogenic genes, including VEGF, KDR (kinase insert domain receptor), and hepatocyte growth factor (HGF). Rats transplanted with islets cocultured with preconditioned hASCs exhibited a better glycemic control than rats transplanted with an equal volume of islets and control hASCs. Cotransplantation with preconditioned hASCs was also associated with enhanced islet revascularization in vivo, as highlighted by graft morphological analysis. The observed increase in islet graft revascularization and function suggests that our method of stem cell preconditioning may represent a novel strategy to remarkably improve the efficacy of islets-hMSCs cotransplantation.
Introduction
Islet transplantation represents a fascinating procedure that, at moment, can be considered as alternative to standard insulin treatment or pancreas transplantation only for selected categories of patients with type 1 diabetes mellitus (27). Despite the different improvements described in the Edmonton protocol (39), insulin independence after islet transplantation is indeed still limited in time and strictly dependent on a high transplanted islet volume (1). It is estimated that 60–80% of the transplanted islet mass is lost within hours to days in clinical intraportal islet transplantation (13, 19, 23), mainly as a consequence of the instant blood-mediated inflammatory reaction (IBMIR) (4).
Besides IBMIR, however, other factors have been described that lead to poor islet engraftment, and among those hypoxia is considered to play an important role (9, 23, 41).
Normally, pancreatic islets have a dense capillary network, which is responsible for the higher oxygen partial pressure in islets compared to surrounding exocrine tissue and other organs (6, 9). During the process of isolation and in vitro culture, however, the islet vasculature degenerates and the reestablishment of an adequate blood flow to transplanted islets depends on an angiogenesis process that requires several days (28). Consequently, during the culture period and in the first days following transplantation, hypoxic stress and lack of nutrients lead to a loss of a significant portion of β-cells (12, 16, 23). Moreover, even when completed, the process of islet graft revascularization fails to recreate the same favorable condition of high oxygen tension for β-cells as in the native pancreatic gland (10). Consequently, besides other factors, inadequate blood supply is crucial not only to limit the islet transplant engraftment but also to impair long-term islet function (26).
Mesenchymal stem cells (MSCs) are able to improve the revascularization process by secretion of antiapoptotic and angiogenic cytokines (35) and by direct differentiation into endothelial cells (32, 40). Moreover, recently it has been reported that in animal models MSCs are able to enhance survival and function of islet graft not only by immune modulation mechanisms (5) but also by increasing islet revascularization (20). In detail, MSCs secrete cytokines like vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF), which are able both to protect endocrine and endothelial islet cells after isolation and to increase islet revascularization after transplant (8, 11, 14, 33).
Besides bone marrow and human cord blood, adipose tissue showed to be a high-throughput source for human MSCs (hMSCs) (46). Moreover, the capability of adipose tissue-derived stem cells (ASCs) to improve survival and function of transplanted islets was demonstrated after hybrid transplantation under the kidney capsule of mice (31).
Within this context, we have previously shown that hyaluronan esters of butyric and retinoic acids can significantly enhance the rescuing potential of hMSCs in both heart and kidney rat models of tissue damage (25, 44) and that these beneficial effects were mediated by the transcriptional action of hyaluronan grafted moieties intracellularly released by the mixed ester (44). Based on these findings, in the current study, we investigated whether ex vivo preconditioning of human ASCs (hASCs) with a mixture of hyaluronic (HA), butyric (BU), and retinoic (RA) acids may result in optimization of islet revascularization and function after stem cell cotransplantation in a syngeneic intrahepatic islet transplantation model in diabetic rats.
Materials and Methods
Isolation and Culture of hASCs
According to the policy approved by the local ethical committee, all tissue samples were obtained after informed consent.
Human subcutaneous adipose tissue samples were obtained from lipoaspiration/liposuction procedures. After washing, lipoaspirates were digested with 0.2% collagenase A type I solution (Sigma-Aldrich), under gentle agitation for 45 min at 37°C, and centrifuged at 2000 rpm for 10 min to separate the stromal vascular fraction (SVF) from adipocytes. If necessary, the MSC fraction was treated with red blood cell lysis buffer for 5 min at 37°C and then centrifuged again. The supernatant was discarded, and the cell pellet was resuspended and seeded in culture flasks in Dulbecco's modified Eagle's medium (DMEM)–low glucose (Lonza) supplemented with 20% heat-inactivated fetal bovine serum (FBS), 1% penicillin–streptomycin, and 2 mM l-glutamine and incubated at 37°C in a humidified atmosphere with 5% CO2. When the cultures were near confluence, the cells were detached by treatment with trypsin-EDTA 1x (Sigma-Aldrich), characterized, subcultured, and used at passage numbers 3–5.
Preconditioning of hASCs
Human ASCs were seeded at 8,000 cells/cm2 in DMEM/20% FBS, and after 24 h the medium was changed to DMEM/10% FBS containing HA (2 mg/ml), BU (5 mM), and RA (1 μM) (all chemicals were purchased from Sigma-Aldrich). Control cells were cultured in DMEM/10% FBS.
ELISA
To investigate whether the mixture of HA, BU, and RA may affect the production of angiogenic and anti-apoptotic factors, hASCs were cultured in DMEM/2% FBS in absence (control) or presence (preconditioned) of HA, BU, and RA, and supernatants were collected at defined time intervals of 1-2-3-6 days (n = 3). For each sample, cell proteins were extracted by trichloroacetic acid (TCA) precipitation/NaOH lysis, and the total amount was quantified by Bradford assay and used for normalization. VEGF concentration (pg/ml) was measured by enzyme immunoassay (Human VEGF ELISA Kit, Boster Biosciences Co., Ltd.). Data are expressed as pg of secreted factor per μg of protein at the time of harvest.
Gene Expression
Total RNA from control or preconditioned hASCs was extracted at 1-2-3 days using the RNeasy Micro Kit (QIAGEN), and 1 μg was reverse-transcribed into cDNA in a 21-μl reaction volume with SuperScriptTM III reverse transcriptase (Invitrogen). To assess gene expression, 2 μl of cDNA were used for real-time PCR performed with a Lightcycler system (Roche Applied Science) and with the SYBR Green I FastStart kit (Lightcycler® FastStart DNA MasterPLUS SYBR Green I), following the manufacturer's instructions.
Primers used (0.25 μM) were as follows: VEGF forward 5′-AGAAGGAGGAGGGCAGAATC-3′ and reverse 5′-ACACAGGATGGCTTGAAGATG-3′; KDR (kinase insert domain receptor) forward 5′-CTGCAAATTTGGA AACCTGTC-3′ and reverse 5′-GAGCTCTGGCTACT GGTGATG-3′; HGF forward 5′-ATTTGGCCATGAAT TTGACCT-3′ and reverse 5′-ACTCCAGGGCTGACA TTTGAT-3′; glyceraldehyde 3-phosphate dehydrogenase (GAPDH) forward 5′-CAGCCTCAAGATCATCAGCA-3′ and reverse 5′-TGTGGTCATGAGTCCTTCCA-3′.
Primers for β-actin (ACTB) and hypoxanthine phosphoribosyltransferase 1 (HPRT1) were purchased from OriGene Technologies, Inc.
Data were normalized using three housekeeping genes (ACTB, GAPDH, HPRT1) as an index of cDNA content after reverse transcription. Relative quantification of mRNA expression was calculated with the comparative Ct method using the “delta-delta method” for comparing relative expression results between treatments in real-time PCR (34).
Experimental Animals
Male Lewis rats were purchased from Charles River Laboratories, Inc. (Lecco, Italy). Rats weighing approximately 200 g were used as recipients, and those weighing 300–350 g were used as donors. The use of animals and the animal procedures of this study were approved by the Bioethics Committee of the University of Bologna.
Isolation of Rat Pancreatic Islets
Rats were anesthetized with isoflurane (Abbott Laboratories, Lake County, USA), and subsequently sacrificed by neck dislocation. Islet isolation and purification were performed according to a modified procedure described earlier (18). Briefly, after cannulation of the common bile duct, 10 ml of enzyme solution of collagenase NB8 (SERVA Electrophoresis, Heidelberg, Germany), at a concentration of 1.3 mg/ml in Hank's balanced salt solution (HBSS), were injected by retrograde perfusion of the pancreas. After organ procurement, enzymatic digestion took place for 12–15 min at 37°C in a water bath with two intermitted 1-min periods of vigorous shaking. Following a wash step with 10% FBS/HBSS and filtration through cheesecloth, islets were obtained by centrifugation in a discontinuous density gradient of Histopaque 1077 (Sigma-Aldrich) and HBSS, with subsequent hand-picking and microscope examination. The total number of islets in each diameter class was counted using an optical graticule. The number was then converted to the standard number of islet equivalents (IEQ). Islet viability was assessed using fluorescent staining with acridine orange and propidium iodide (Sigma-Aldrich). Islets with viability >90% were used for transplantation purposes.
Immediately after counting, isolated rat islets were incubated with hASCs that had been previously preconditioned for 5 days (as already described above). To facilitate the adhesion of hASCs to the islet surface, we used the procedure described by Johansson et al. (21). In brief, the total volume of isolated islets was mixed with control or preconditioned hASCs at a cell/islet ratio of 5,000/100 in islet medium RPMI-1640 (Lonza), supplemented with 10% FBS, 2 mM l-glutamine, 1% antibiotic–antimycotic solution. The cell suspensions were incubated at 37°C for 2 h in culture tubes and mixed gently twice per hour. Thereafter, the mixture of islets and hASCs was seeded in ultra-low attachment plates (Corning) and cultured for up to 3 days in a humidified incubator at 37°C, 5% CO2.
To detect the capability of hASCs to adhere to the islet surface, in a pilot test we cultured islets with hASCs transfected with green fluorescent protein (GFP). At the end of a 3-day coculture period, the eventual formation of composite hASCs–islets was evaluated by immunofluorescence microscopy.
For transplant purpose, at the end of the coculture period, islets and hASCs were hand-picked, washed three times with 10 ml of HBSS containing 2% Lewis rat serum, resuspended in transplant medium (HBSS/10% rat serum), and placed on ice until transplantation.
Cotransplantation of Islets and hASCs and Monitoring of Islet Graft Function
Diabetes was induced in rats by a single 70 mg/kg intraperitoneal injection of streptozotocin (Sigma-Aldrich). Only rats with blood glucose levels of more than 400 mg/dl for 3 consecutive days were used as recipients of islet grafts. The animals were anesthetized with xylazine–tiletamine–zolazepam (Zoletil®, Virbac, Carros, France) and placed on a delta-phase heating pad (2Biol, Besozzo, Italy). A suboptimal volume of 500 cultured islets (2,500 islet/kg) that in our experience allowed the reduction of blood glucose levels of transplanted rats without achieving reversal of diabetes and the cocultured control or preconditioned hASCs were transplanted in diabetic rats weighing approximately 200 g (n = 7 per group, control or preconditioned group, respectively). With the aim to favor graft detection after intraportal injection and to permit a comparative analysis between the two study groups, we transplanted islets only in the caudate lobe of the rat liver, as already described (24). Briefly, the cavity was accessed by midline incision. The portal branches to the left, middle, and right liver lobes were temporarily closed with microvascular clamps, after which islets of both study groups were slowly injected into the portal vein by a 26-gauge needle, in a volume of 300 μl of transplant medium, directing the islets to the caudate liver lobe. The clamps were released and the injection needle was removed. Bleeding was stopped with a cotton swap by gentle compression at the site of injection. After closing the abdomen with sutures, the recipient was allowed to recover. After transplantation, blood glucose levels were daily measured with a glucometer (A. Menarini, Firenze, Italy), using samples obtained from the tail vein. Animals exhibiting nonfasting blood glucose levels <300 mg/dl for at least 3 consecutive days were considered “with improved glycemic control,” while animals with levels <200 mg/dl after 3 consecutive days were considered “normoglycemic.” On the 15th posttransplantation day, intraperitoneal glucose tolerance test (IPGTT) was performed by injecting intraperitoneally normal saline containing 2 g glucose/kg body weight into overnight-fasting rats. The rat blood glucose levels were then measured at 30, 60, 90, and 120 min after injection.
Morphological and Immunohistochemical Examination
At the end of a 3-day coculture period, islet morphology was evaluated by direct microscopic observation using control and preconditioned hASCs transfected with GFP.
For histological examination islets were fixed in Bouin solution, embedded in 2% agarose in PBS, and then allowed to polymerize on ice. The enrobed islets were processed for paraffin embedding, and 4-μm sections were cut on a microtome (Leica RM2125RT). Sections were deparaffinized in xylene, rehydrated through decreasing concentrations of ethanol, and stained with hematoxylin and eosin (H&E). Deparaffinized sections were placed in TRIS/EDTA Buffer (pH 9.0) and heated for 1 min in a pressure cooker (110°C) for antigen retrieval. Subsequently, retrieved sections were blocked with 2% normal goat serum (NGS) in 1% bovine serum albumin (BSA) in PBS for 30 min at room temperature. The slides were then incubated with the anti-human mitochondria monoclonal antibody, clone 113-1 (1:50, Millipore) for 60 min at room temperature to evaluate the adhesion of the human MSCs to the islets. The islet sections were then stained for immunofluorescence by the mouse anti-insulin monoclonal antibody clone K36AC10 (1:500, Sigma-Aldrich) used at 4°C for overnight, followed by the secondary antibodies anti-mouse IgG (Fc specific) fluorescein isothiocyanate (FITC)-conjugated (1:250, Sigma-Aldrich) or Alexa Fluor® 555 (1:1,000, Invitrogen) in 1% BSA in PBS for 1 h at 37°C to evaluate β-cell viability. To detect intraislet endothelial cells, lectin Bandeiraea simplicifolia (BS-1) tetramethyl rhodamine isothiocyanate (TRITC)-conjugated (1:100, Sigma-Aldrich) was employed in 1% BSA in PBS for 45 min at 37°C, after a second blocking with normal mouse serum (NMS) for 30 min at room temperature.
To evaluate islet graft morphology and in particular their revascularization process, immediately after sacrifice the caudate liver lobes were harvested from the transplanted rats, fixed in formalin solution for 24 h, embedded in paraffin, and cut into 4-μm sections. Paraffin sections were stained for insulin and lectin BS-1 using the same products reported above. To evaluate the level of the islet graft revascularization, the number of insulin-positive cells and lectin BS-1-positive capillary segments were counted in seven pictures randomly selected for both study groups, and the obtained capillary/β-cell ratios were compared.
Furthermore, to evaluate the presence of hASCs after transplantation, sections were stained with the anti-human mitochondria monoclonal antibody plus the secondary goat anti-mouse Alexa Fluor® 488 (1:250, Invitrogen) in 1% BSA in PBS for 1 h at 37°C. Samples were coverslipped with ProLong antifade reagent with DAPI (Molecular Probes).
Slides were studied with a Leica DMI4000 B inverted fluorescence microscope and the image acquisition LAS AF software (Leica Microsystems).
Statistical Analysis
Data are presented as mean ± SD (standard deviation). Statistical comparison of two groups was carried out by two-sided unpaired Student's t test and log-rank test. Comparison of more than two groups was performed by one-way ANOVA, followed by Bonferroni post hoc test (GraphPad Prism). A value of p < 0.05 was assumed as the limit of significance (significance level: *p < 0.05, **p < 0.01, and ***p<0.001).
Results
Preconditioning of hASCs with a Mixture of HA, BU, and RA Enhances VEGF Secretion, and the Expression of Angiogenic Genes
ELISA revealed that control hASCs secreted consistent amounts of VEGF throughout the investigated time course. Noteworthy, culturing of hASCs in the presence of the preconditioning mixture resulted in a remarkable, time-dependent increase in VEGF secretion, starting as early as 24 h, and peaking after 6 days of exposure (Fig. 1A).

Enhanced VEGF secretion and angiogenic gene expression in preconditioned hASCs. Time-course analysis of vascular endothelial growth factor (VEGF) released by human adipose tissue-derived stem cells (hASCs) cultured for up to 6 days in the absence (white bar) or presence (black bar) of 2 mg/ml hyaluronic (HA), 5 mM butyric (BU), and 1 μM retinoic (RA) acids (n = 3) (A). VEGF (B), kinase insert domain receptor (KDR) (C), and hepatocyte growth factor (HGF) (D) gene expression was assessed by real-time PCR. Cells were cultured for up to 3 days in the absence or presence of HA, BU, and RA. The abundance of each mRNA in control cells was defined as 1, and the amounts of VEGF, KDR, or HGF mRNA from preconditioned cells were plotted relative to that value (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001.
Real-time PCR provided evidence that stem cell preconditioning increased the transcription of genes that have been shown to play a crucial role in the orchestration of angiogenic signals, including VEGF (Fig. 1B), KDR (Fig. 1C), encoding a major VEGF receptor, and HGF (Fig. 1D). VEGF expression peaked at 24 h after treatment and then decreased in spite of maintaining a level higher than in control cells. Downregulation of VEGF gene and the concomitant increased VEGF secretion suggest that an autocrine regulation is likely to occur.
Formation of Composite hASCs–Islets
Stem cell adhesion to the surface of cocultured islets was clearly detectable at microscopic examination using GFP-expressing hASCs. Approximately 50% of the islets presented hASCs on their surface at the end of the coculture period (Fig. 2A). The half part of the islets, however, appeared without hASCs coating their surface (Fig. 2B). The formation of composite hASCs–islets was confirmed by immunohistochemistry using the anti-human mitochondria monoclonal antibody (Fig. 2C). No difference in the formation frequency of composite hASCs–islets was evidenced between the two study groups.
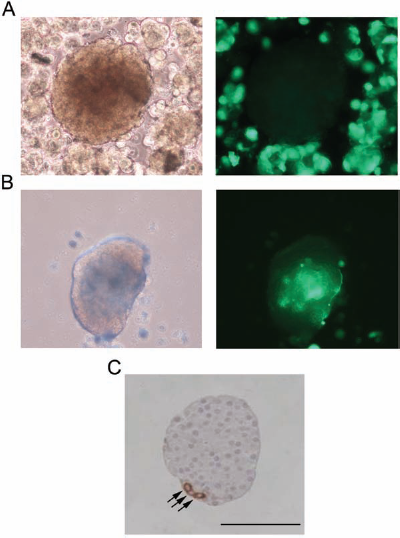
Formation of composite hASCs–islets. The half part of islets remained uncovered (A), while approximately 50% of islets appeared covered by green fluorescent protein (GFP)-expressing hASCs (B) without difference between the two study groups. Immunohistochemical detection of anti-human mitochondria-positive cells (arrows) in cultured rat pancreatic islets (C). Scale bar: 100 μm.
High Presence of Intraislet Endothelial Cells After Coculture with Preconditioned hASCs
The majority of islets from both study groups, except for the occasional presence of central necrosis in the core region of the larger ones, showed an intact morphology at immunohistochemical examination at the end of the 3-day coculture period. Interestingly, islets cocultured with preconditioned hASCs revealed a higher presence of intraislet endothelial cells, as compared to islets from control group (Fig. 3A, B).
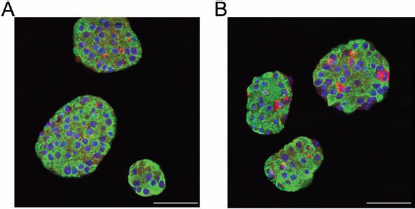
High expression of endothelial cells in islet cultured with preconditioned hASCs. Immunofluorescent staining of 3-day cultured islets in presence of hASCs (A) or preconditioned hASCs (B). β-Cells are stained with antibody anti-insulin (green), intraislet endothelial cells are stained with lectin Bandeiraea simplicifolia (BS-1) (red), and nuclei are stained with DAPI (blue). Scale bar: 50 μm.
Cotransplantation of Preconditioned hASCs Enhances Islet Graft Function
Diabetic rats transplanted with 500 syngeneic islets of preconditioned group demonstrated enhanced glycemic control, compared with rats of control group. In particular, significant differences (p < 0.05, p < 0.01, p < 0.001) in nonfasting glucose levels were registered between the rats of the two study groups at all the time points 1 week after transplantation (Fig. 4A). The percentage of recipient rats with achievement of improved blood glucose control (nonfasting blood glucose < 300 mg/dl) is reported in Figure 4B. A remarkable improvement in blood glucose control was registered in six of seven (85.7%) rats of preconditioned group, compared with three of seven (42.8%) rats of control group (p < 0.05). Moreover, two of the recipients of preconditioned group (28.5%) became normoglycemic (nonfasting glucose < 200 mg/dl), while none of the rats of the control group achieved normal glucose levels. The IPGTT performed at the 15th day after transplant detected significantly lower levels of glucose at 60 and 90 min (p < 0.01) in the rats transplanted with islets of the preconditioned group (Fig. 4C).

Improved islet graft function after cotransplantation with preconditioned hASCs. Diabetic rats from preconditioned group showed lower nonfasting glucose levels compared to rats of the control group at all the time points one week after transplant (A). Approximately 500 islets were sufficient to achieve a strong improvement of glycemic control (blood glucose levels < 300 mg/dl for at least 3 consecutive days) in 85.7% of animals receiving preconditioned cells, compared to 42.8% of control recipient rats (B). Intraperitoneal glucose tolerance test (IPGTT) performed in overnight fasting rats 15 days after transplantation revealed lower blood glucose levels after 60 and 90 min in rats of preconditioned group (C). *p < 0.05, **p < 0.01, ***p < 0.001 versus control group at respective time points.
Cotransplantation of Preconditioned hASCs Increases the Revascularization Process of Transplanted Islets
Islet transplantation only in the caudate liver lobe of the recipient rats allowed the detection of a high number of islet grafts at the histological examination and consequently permitted an accurate comparative immunohistochemical analysis of the graft vascularization between the two study groups (Fig. 5). As shown in Figure 5E, the capillary number per β-cell in the islet graft of preconditioned group was sensibly superior to that of control group (n = 7 sections examined per group) (0.117 ± 0.02 vs. 0.062 ± 0.02 capillary fragments per β-cell, p < 0.01). The presence of anti-human mitochondria-positive cells, located in proximity of the engrafted β-cells, indicated the viability of hASCs for up to 2 weeks after transplantation (Fig. 5F). This result clearly shows that a preconditioning strategy with natural molecules can improve the ability of hASCs to increase islet revascularization process after transplantation in an animal model.

Enhanced islet graft revascularization by preconditioned hASC cotransplantation. Two weeks after transplant, islets of preconditioned group showed intact β-cells detected by antibody anti-insulin (A) and a dense capillary network stained with BS-1 (B). Merged photograph of A and B (C). Merged photograph of control group islet (D). Scale bar: 100 μm. Analysing seven islet graft pictures for group, a higher capillary segments/β-cell ratio was detected in preconditioned versus control group (E) (**p < 0.01). Anti-human mitochondria-positive cells (green, arrows) revealed hASCs surrounding engrafted β-cells stained for insulin (red) (F). Scale bar: 25 μm.
Discussion
The employment of MSCs to improve the efficacy of pancreatic islet transplantation represents a new promising research approach (5, 20, 21, 33). Recently, the ability of ASCs to enhance pancreatic islet engraftment and functionality by inhibition of inflammatory host response and improvement of graft revascularization was demonstrated in an animal model of diabetes (31). An important element involved in the maintenance of a good function of the graft is the secretion of antiapoptotic and vasculogenic factors by MSCs (46) that exert a protective effect on islet cells during the culture period and increase the revascularization of transplanted islets (8, 20, 33). One of the most important cytokines produced by both mesenchymal stem cells and endothelial cells is VEGF, which has an antiapoptotic effect and exerts a fundamental role in vessel development. During the procedure of islet isolation and culture, not only a significant portion of β-cell mass but even intraislet endothelial cells, which are essential for the process of revascularization after transplant, get lost (7, 30). Therefore, the reduced production of VEGF, caused by necrosis of endocrine cells, impairs the already poor process of posttransplant revascularization (22, 26).
In this study, we showed that the regenerative action exerted by hASCs could be increased by preconditioning these cells with a mixture of natural molecules as hyaluronic, butyric, and retinoic acids. Specifically, the intrahepatic transplantation of a subtherapeutic volume of islets cultured for 72 h in the presence of preconditioned hASCs showed an enhanced revascularization and function, compared to islets cultured with control cells. The beneficial effects of this new strategy may result from the ability of the preconditioning mixture to increase the gene and protein expression of VEGF, as well as the transcription of KDR and HGF, which have been shown to be involved in autocrine/paracrine circuitries of angiogenic signaling amplification (2, 29).
The higher production of vasculogenic and antiapoptotic cytokines by preconditioned cells could also explain the higher presence of intraislet endothelial cells in islets evaluated after a 72-h coculture with preconditioned hASCs and the reduction of necrosis that is only occasionally observed in the core region of larger islets.
The in vivo studies indicate that the favorable prosurvival effect on intraislet endothelial cells, provided in vitro by stem cell preconditioning, leads to an improved vascular network after islet/preconditioned hASC cotransplantation in diabetic rats and that the animals subjected to this protocol could benefit from a remarkably enhanced glycemic control. This finding indicates that the currently described procedure not only preserves structural features of the islets and their inherent vascular network but can also impact on the molecular plight that controls the attainment of a proper islet function.
The use of adipose tissue as an alternative stem cell source is one of the most intriguing fields of recent interest. Fat can be used in autologous fashion and exhibits ideal characteristics, being readily available, inexpensive, and host compatible. It is also abundant and can be repeatedly harvested and cryopreserved for banking and subsequent use in one of the several therapeutic applications that may be envisioned from hMSCs. Moreover, hASCs have already been used in clinical settings in the treatment of several pathologies (15, 36, 43), and their efficacy and safety allow their clinical use in the field of islet transplantation.
It has been recently shown that lipoaspirates can be obtained after minimally invasive harvesting and processed with mild mechanical forces in the absence of any enzymatic digestion or growth factor additives, leading to a significant simplification and cost decrease in fat processing (42). Our preconditioning strategy may also be applied in principle to hASCs isolated from these nonenzymatic fat products, and studies are on the way to assess such a perspective.
Although the rescuing ability of MSCs has been a driving force behind initial studies examining their therapeutic effectiveness, their immunomodulatory properties are equally exciting in terms of exploring their potential implications in various disease models, including autoimmune diseases, as type 1 diabetes (3). An important immunological feature of MSCs is the inhibition of T-cell proliferation and dendritic cell differentiation, due to their low expression of costimulatory molecules and the absence of class II human leukocyte antigen (HLA). In addition, MSCs are able to synthesize trophic mediators involved in immunomodulation, and to induce T-cell anergy and regulatory T cells (Treg) (45). One of the great expectations linked to the use of MSCs may therefore reside in their ability to successfully engraft, evading an immune response and inducing peripheral host tolerance, allowing hMSC xenotransplantation (25, 37, 44, 45).
The results from our study show that preconditioning with natural molecules increases the protective effect of hMSCs on cultured islets, improving their posttransplant functionality. A period of islet culture may provide several advantages in the clinical setting of islet transplantation, like optimization of the quality of islet preparations, and leaves additional time for the selection and treatment of the recipient with suitable pretransplant strategies (38). Moreover the use of cultured islets may facilitate their shipment to remote transplant centers (17).
Further studies are in progress to evaluate the effects of hASC preconditioning using human islets, and to this aim, hybrid transplantations of hASCs and human islets in immunodeficient mice should be performed. Should these future studies confirm our promising results, then the employment of hASCs preconditioned with the currently described mixture may be considered as a useful strategy even in a clinical setting, especially when cultured islet transplantation is envisioned.
Footnotes
Acknowledgments
The authors thank Dr. W. Moritz for scientific discussion and Prof. G. Pasquinelli for the use of the fluorescence microscope and his precious support in immunohistochemical analysis. The authors further thank M. Pariali for her excellent technical advice. This research was supported by Regione Emilia Romagna, Programma di Ricerca Regione-Università 2007/2009, Area 1b “Medicina rigenerativa,” Italy; Ministero della Salute, Italy, Programma per la Ricerca Sanitaria: Attività di Ricerca sulle Cellule Staminali; Fondazione Fornasini, Poggio Renatico, Italy; Fondazione Cardinale Giacomo Lercaro, Bologna, Italy; Fondazione Luisa Fanti-Melloni, Bologna, Italy; Tavola Valdese, Rome, Italy. The authors declare no conflicts of interest.
