Abstract
With favorable regenerative and immunotolerant profiles, patient-derived human mesenchymal stem cells (hMSCs) are increasingly considered in cell therapy. Derived from bone marrow (BM) and standardized with culture in fetal bovine serum (FBS), translation of hMSC-based approaches is impeded by protracted expansion times, risk of xenogenic response, and exposure to zoonoses. Here, human platelet lysate adherent to good manufacturing practices (GMP-hPL) provided a nonzoonotic adjuvant that enhanced the capacity of BM-hMSC to proliferate. The nurturing benefit of GMP-hPL was generalized to hMSC from adipose tissue evaluated as an alternative to bone marrow. Long-term culture in GMP-hPL maintained the multipotency of hMSC, while protecting against clonal chromosomal instability detected in the FBS milieu. Proteomic dissection identified TGF-β, VEGF, PDGF, FGF, and EGF as highly ranked effectors of hPL activity, revealing a paradigm of healing that underlies platelet lysate adjuvancy. Thus, GMP-adherent human platelet lysate accelerates hMSC proliferation with no chromosomal aberrancy, through an innate repair paradigm.
Introduction
Discovery of stem cell populations that exhibit proclivity to differentiate into specialized tissue types has provided the foundation for regenerative medicine (40). A case in point are human mesenchymal stem cells (hMSCs) that exhibit multipotency with a capacity for differentiation into cardiac, adipose, chondrocyte, or osteoblast lineages, and offer a patient-derived renewable source to support tissue restoration (7,23,38,39). With low levels of human leukocyte antigen, this immuno-privileged adult cytotype modulates rather than provokes immune response in the allogeneic state, distinguishing hMSC as favored candidates for tissue reconstitution (1,31). Experience to date has largely relied on bone marrow as the source for hMSC derivation (BM-hMSC) (24,29,53). As bone marrow is a heterogeneous tissue, composed of several stem cell pools, use of this resource necessitates large explants or protracted ex vivo expansion times, due to the small fraction of mononuclear cells conforming to the hMSC phenotype. To improve the efficiency of hMSC expansion alternative sources, such as adipose tissue, novel methods of culture are under investigation (10,27).
Initially, culture of hMSCs was established with fetal bovine serum (FBS), an adjuvant routinely used in cell culture (48). Although the overall risk of zoonoses associated with FBS culture is thought to be small, a call for greater scrutiny in stem cell banks for prions and zoonotic pathogens has been issued (14,16). Furthermore, recent reports have raised concern for the potential of immunogenic reaction at the time of transplantation due to xenogenic proteins transmitted from FBS to hMSCs during culture (47). Specific cases have documented the risk for anaphylactoid response after implantation of cells cultured in FBS, with reported diffuse urticaria and production of antibodies against FBS proteins after repeated stem cell treatment cycles (20,24,45,47). In addition, the culture environment has been noted to differentially regulate cell cycle transcripts of hMSC proliferation and influence lineage differentiation when compared to that of stem cells cultured with humanized culture adjuvants (19). The lot-to-lot variability seen with FBS creates an inherent heterogeneity in the derivation and propagation of hMSCs. As such, the use of FBS impedes adherence to regulatory requirements, hampering development of clinically applicable cell-based products (8,33).
To overcome potential hurdles associated with FBS-based culture, testing of nonzoonotic substitutes is warranted (8,33,41,46). Such substitutes should mimic naturally occurring microenvironments to optimally support hMSC proliferation while maintaining cell phenotype, multipotency, and chromosomal stability. In this regard, platelets provide a unique opportunity for stem cell culture as they consist of intracellular granules abundant in potent reparative and mitogenic substances (35). At the time of tissue injury, these bioactive factors are released into a resultant clot and exert extensive influence on tissue regeneration by triggering stromal cell proliferation, migration, and differentiation. Moreover, hMSC proliferation and chemotaxis in tissue homeostasis and restoration are a direct response to signals following injury (55). Ex vivo recapitulation of this nurturing paradigm would allow, in principle, the formulation of a milieu capable of supporting expansion of hMSCs for clinical translation.
Compared to standard FBS, good manufacturing practice (GMP)-adherent pooled human platelet lysate (GMP-hPL) provided here an adjuvant for accelerated expansion, maintenance of phenotype stability, and absence of clonal chromosomal aberrancy in BM-hMSC and adipose tissue hMSC (AT-hMSC) cultures. Proteomic dissection of this nonzoonotic milieu resolved signals that clustered within nonstochastic pathways of tissue repair and cell proliferation, identifying the molecular substrate underlying human platelet lysate support of efficient and safe hMSC procurement.
Materials and Methods
Isolation and Culture of hMSCs From Bone Marrow and Adipose Tissue
All patient-derived tissues were obtained in full compliance and approval of local institutional review boards (IRB). Bone marrow aspirates were obtained from iliac crest or sternum of 12 patients, washed with PBS, and centrifuged at 400 × g for 5 min. The pellet was treated with red blood cell lysis buffer for 5 min at 4°C, centrifuged, resuspended, and cultured in expansion media. Human adipose tissue was obtained under IRB guidelines from different anatomic compartments in 20 patients undergoing liposuction, appendectomy, bariatric, or hernia operations, minced with surgical scalpels for about 5 min, and incubated in 0.075% collagenase type I (Worthington Biochemical, Lakewood, NJ) for 90 min at 37°C. Digested tissue was centrifuged at 400 × g for 5 min with the pellet washed in PBS, passed through a 70-μm cell strainer (BD Biosciences, San Jose, CA), and incubated in red blood cell lysis buffer (154 mmol/L NH4Cl, 10 mmol/L KHCO3, 0.1 mmol/L EDTA). Expansion media consisted of DMEM with 10% fetal bovine serum or advanced MEM with 5% platelet lysate, 100 U/ml penicillin, 100 g/ml streptomycin, and 2 mM L-glutamine (Invitrogen, Carlsbad, CA). Cell yield was quantified with a hemocytometer.
GMP-Adherent Platelet Lysate Procurement
Standard blood donor criteria as determined by the Mayo Clinic Blood Bank were utilized for platelet harvest and lysate procurement. There was no selection bias, as all outdated platelets from 500 distinct donors were lysed and pooled into 50 lots containing between 8 and 12 donors (15). Laminar-flow cabinet sterility and air quality were maintained at International Organization for Standardization (ISO) 5 within an environment conforming to ISO 7 standards. Routine environmental monitoring was implemented for adherence to microbial, airborne particle, air pressure differential, temperature, and humidity standards. The manufacture using a freeze–thaw approach, composition, and release of platelet lysate was subject to quality assurance (maintenance of hMSC phenotype, viral and bacterial sterility, and product stability with full traceability of all material utilized) and independent oversight [certifying both environmental quality and validated standard operating procedures (SOP) for hPL manufacture]. Each generated batch fulfilled tracking criteria imposed by GMP standards, including quarantine during batch testing and release with demonstration of verified quality and purity characteristics (15).
Flow Cytometry of hMSCs
Isolated cells were collected and immunostained for analysis of cell surface expression using an Aria SE flow cytometer and Cell Quest Pro software (BD Biosciences). Cells were incubated with either CD34-fluorescein isothiocyanate (FITC), CD14, CD44, CD45, CD73, HLA-DR, and HLA-ABC (all from BD Biosciences), along with CD105, CD90 (eBioscience, San Diego, CA) and CD133 (Cell Signaling Technologies) (37).
Proliferation Assay
Bone marrow- and adipose tissue-derived hMSCs were initially plated at 5,000 cells/cm2 in 10% FBS or 5% GMP-hPL supplemented media (equivalent to platelet-derived media supplement; Mill Creek Life Sciences, Rochester, MN) and passaged for a total of five passages representing a total of 40–60 cell division events depending on the condition. Cumulative population doubling (CPD) was calculated using the formula: [log10 (NH) – log10(N1)]/log102, where NH is the harvested cell number and N1 is the plated cell number (8). Each passage was calculated and added to the population doubling of the previous passage to obtain the CPD. The generation time (i.e., the time between two cell doublings) was calculated as: [log102 × Δt]/[log10(NH) – log10 (N1)], where Δt is the time between passages (8).
Lineage Differentiation
Osteogenic and adipogenic differentiation was achieved with a Mesenchymal Stem Cell Osteogenic Stimulatory Kit and the Mesenchymal Stem Cell Adipogenic Stimulatory Supplements (Stem Cell Technologies), respectively. Chondrogenic differentiation was achieved by aggregating cells at a density of 2 × 104 cells/μl of induction medium DMEM high glucose, 4.5 g/L (Invitrogen), 100 U/ml penicillin, 100 μg/ml streptomycin, 4 mM L-glutamine, 1 mM sodium pyruvate, 0.17 mM ascorbic acid 2-phosphate, 0.1 μM dexamethasone, 1× ITS supplement, 0.35 mM L-proline (all from Sigma-Aldrich). Cell aggregates were then plated as droplets of 20 μl in a six-well culture dish and allowed to adhere for 2 h, followed by addition of 2 ml of induction medium. Aggregates were cultured for 3 weeks, before differentiation was assessed. Cardiogenic differentiation was achieved by subjecting cultured hMSC in a 5% platelet lysate condition with a cocktail of recombinant growth factors as previously described for a 5-day period (5,6). Following cocktail stimulation, cardiac progenitor cells were placed in a 1% platelet lysate milieu to achieve terminal myocardial differentiation (7,22).
Histology and Immunostaining
Prior to antibody incubation, cells were fixed in 3% paraformaldehyde and permeablized with 0.1% Triton X-100. The following antibodies were used for staining to address cell plasticity: Oct-4 (1:100, R&D Systems), SOX-2 (1:200, Cell Signaling Technologies), CD133, and CD90-FITC. Cardiac commitment was evaluated with: Mef2C (1:400, Cell Signaling Technologies), Nkx2.5 (1:150, Santa Cruz Biotechnology Inc.), Gata-4 (1:150, Santa Cruz Biotechnology Inc.), sarcomeric protein α-actinin (1:500, Sigma-Aldrich), and human-specific cardiac troponin-I (1:100, Abcam) (5). Histologic processing with Oil red-O was performed to assess for adipogenesis, Safranin-O (Sigma-Aldrich) for chondrogenesis, and Alizarin Red for osteogenesis. Images were obtained with LSM 510/LSM 700 laser scanning confocal or Apotome structured illumination microscopes (Carl Zeiss).
Chromosomal Analysis
Twenty metaphases cells from 20 distinct patient hMSC cultures underwent Q-banding using quinacrine (QFQ-banding), followed by G-banding using trypsin and Leishman's stain. Karyotyping was performed using the Applied Imaging CytoVision Karyotyping System. Spectral karyotyping analysis (SKY) was performed on metaphase chromosomal preparations subjected to single hybridization with fluorochrome-labeled DNA libraries specific for each of the 24 chromosomes. Results were visualized with a SpectraCube, Sagnac interferometer. SKY images correlated with conventional chromosome banding results and were confirmed with chromosome-specific painting prior to interpretation.
Proteomics
Dissection of platelet lysate composition was achieved using a human-specific label-based ELISA array (Ray-Biotech). Here, 100 μl of platelet lysate was dialyzed for 9 h in 500 ml of pH 8.0 PBS, with PBS exchanged at every 3-h interval, biotin-labeled, and dialyzed for an additional 9–12 h in pH 8.0 PBS. Array incubation with dialyzed biotin-labeled platelet lysate was performed by overnight incubation at 4°C followed by hybridization with GFP-labeled biotin-targeted secondary antibody. Proteomic array results were compiled and quantified using a GenePix 4000B fluorescence scanner and software. Quantified data were manually organized to correspond to Swiss-Prot, Genbank, and OMIM identification for entry into the Ingeniuty and GeneGo network analysis software suites (2,56). Protein expression was restricted to display data demonstrating values 10-fold greater than that of background in all three array replicates.
Network Analysis
Using Ingenuity Pathways Analysis and the MetaCore database, molecular interactions within the platelet lysate were examined to generate scoring and prioritization of networks/pathways according to the relevance within the initial list of root objects (17). Significance was based on overlap between imported datasets and canonical pathways as evaluated with hypergeometric distribution. The mean of this distribution was calculated as μ = Σ[n,r = 0]rP(r,n,R,N) = (nR/N) = nq; where N equals total number of MetaCore database nodes, R equals number of network objects corresponding to analyzed genes and proteins, n equals total number of nodes in each small network generated from imported dataset, r equals number of nodes with data in each small network generated from imported dataset, and q is the ratio of R/N. Dispersion was calculated σ2 = r2P(r,n,R,N) – μ2 = (nR(N – n)(N – R))/(N2(N – 1)) = nq(1 – q)(1 –((n – 1)/(N – 1)). Internetwork prioritization based on imported datasets was measured by z-score = (r – μ)/σ, ranking subnetworks according to saturation with root objects. Finally, confidence interval evaluation of network relevance to Gene Ontology biologic processes was measured as p-value = ((R!n!(N – R)!(N – n)!)/N!)Σ [min(n, R), i = max(r,R + n –N)]((1)/(i!(R – i)!(n – i)!(N – R – n + i)!)). Network interconnectivity analysis corresponding to established wound healing and cell proliferation signaling ontologies was probed to determine overrepresentation of subnetwork clusters. Both physical and functional interactions were represented within network objects. Network interconnectivity represented as (k) was plotted against the probability of achieving nonstochastic interaction (P(k)) to engender cluster coefficient plots. Identified over- or underrepresented nodes were probed versus canonical repair and proliferation pathways to determine significance through coordinate adherence in a Cartesian plot format (17).
Statistics
Data are presented as mean ± SD representing a 95% confidence interval (JMP 8; SAS Institute). Paired group analysis was performed using Student's t-test, and nonparametically validated using Wilcoxon signed rank test. A value of p < 0.05 was considered significant. Multiple comparison studies were validated by means of Bonferroni and Tukey's methods (JMP 8).
Results
Nonzoonotic Culture Enhances hMSC Growth Profile
Expired apheresis platelets from multiple donors were pooled into single lots to provide a nonzoonotic, GMP-hPL culture condition. Culture of BM-hMSC obtained from 6 individual patients was made in standard FBS at 10%, and compared to culture in GMP-hPL at 1%, 2.5%, or 5% (Fig. 1A, B). BM-hMSC cultured in the 5% GMP-hPL condition demonstrated significant increase in proliferation capacity versus lower GMP-hPL concentrations and FBS (Fig. 1A) over a 5 passage observation period. Generation time in early passages (P1–3) was quantified as 2.5 ± 0.5, 2.9 ± 0.6, 9.2, and 7.7 ± 2.5 days for 5% GMP-hPL, 2.5% GMP-hPL, 1% GMP-hPL, and FBS, respectively (Fig. 1B). In late passage (P5), generation times were quantified as 2.5 ± 0.4, 3.0 ± 0.6, 7.3, 5.0 ± 1.0 days for 5% GMP-hPL, 2.5% GMP-hPL, 1% GMP-hPL, and FBS, respectively (Fig. 1B).

Culture and derivation of hMSC from two different sources. (A) Bone marrow-derived hMSC (BM-hMSC) cultured in fetal bovine serum (FBS) and different concentrations (1%, 2.5%, and 5%) of platelet lysate (PL) with cumulative quantitation for 5 passages. (B) Time needed for two population doublings (Generation time) was calculated during early (1–3) and late (3–5) passage for BM-hMSC in the different PL and FBS culture conditions. (C) Equivalent volumes of bone marrow and homogenized adipose tissue underwent quantitative analysis demonstrating a 10-fold increase in total mononuclear cell yield from adipose tissue. (D) Analysis of mononuclear cell yield was quantified using a CD133+/CD45-/CD14-/CD34- surface antibody profile on FACS analysis. *p < 0.01 (A, B) where groups I and II are compared to group IV. *p < 0.01 (C).
To generalize the GMP-hPL based approach, adipose tissue hMSC was derived from different anatomic compartments and was obtained either in homogenized form from specimens after liposuction or in tissue bloc after surgical excision (Table 1). Equivalent volumes of freshly homogenized adipose tissue and bone marrow were evaluated for total mononuclear cell yield. Adipose tissue provided a 10-fold greater quantity of cells per volume in comparison to bone marrow (Fig. 1C). Enrichment of hMSC population was achieved by FACS analysis for cells depleted of the surface markers CD45, CD14, and CD34 and expressing CD133, previously reported to be a marker for freshly harvested multipotent MSC, lost upon adherence to plastic (Fig. 1D) (3,50). The population expressing CD133+/CD45-/CD14-/CD34- comprised 0.2 ± 0.1% of the total mononuclear cell fraction derived from adipose tissue, comparable to that observed in bone marrow. Adipose tissue derived hMSCs in FBS demonstrated a fivefold increase in growth (Fig. 2A) and enhanced cumulative population doubling (Fig. 2B) over BM-hMSCs (n = 6 patients for each group assessed over 5 passages). The 5% platelet lysate condition, efficient in cultivation of BM-hMSCs, was evaluated with adipose tissue-derived hMSCs (AT-hMSCs) to validate its broader utility. AT-hMSCs from six patients were cultured either in 10% FBS or 5% GMP-hPL demonstrating, over a 5-passage observation period, maintenance of platelet lysate proliferation advantage (Fig. 2C). This improvement translated into a decrease in generation times from 3.6 ± 0.5 in FBS to 1.7 ± 0.4 days in GMP-hPL at early passage (<10 population doublings) and 2.9 ± 0.4 in FBS to 2.0 ± 0.2 days in GMP-hPL at late passage (>30 population doublings) (Fig. 2D). Thus, GMP-hPL demonstrated a strong mitogenic influence on hMSCs to support their ex vivo cultivation, regardless of tissue of origin and throughout the culture period.
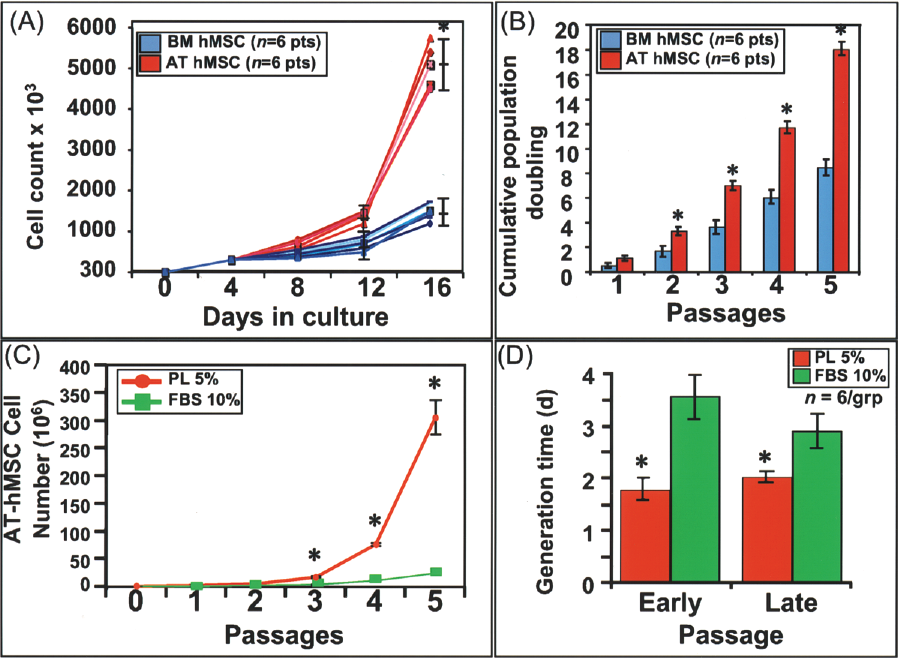
Nonzoonotic 5% platelet lysate (PL) condition supports proliferation of AT-hMSCs and BM-hMSCs. (A, B) Direct quantitation and calculated cumulative population doubling (CPD) in parallel cultures of bone marrow (BM) and adipose tissue-derived hMSCs (AT-hMSCs). (C) Culture of AT-hMSCs in FBS and 5% GMP-hPL for 5 passages with cumulative quantitation of cellular yield. (D) Generation time of AT-hMSCs quantified in the GMP-hPL and FBS condition at early (1–3) and late (3–5) passages. *p < 0.01 (A–C) and *p < 0.05 (D).
Consistency in Derivation of AT-MSCs Is Seen With Analysis of Lipoaspirated or Surgically Excised Adipose Tissue From Different Anatomic Compartments

Nonzoonotic Condition Maintains Phenotype Stability
Preservation of mesenchymal stem cell phenotype following protracted culture (12 passages) revealed a surface receptor profile devoid of CD14, CD34, CD45, and HLA-DR (class II) with positive expression of CD44, CD90, CD105, and HLA-ABC (class I) demonstrated >70 population doublings in six patient-derived hMSCs in either FBS or GMP-hPL conditions. This profile was noted to be stable throughout the culture period for FBS (Fig. 3A), and highly enriched in the GMP-hPL condition (Fig. 3B). Immunocytochemistry markers of stemness, such as Oct-4 and Sox2 previously shown to be expressed in a variety of somatic and stromal stem cells (26,28,50), were found to colocalize in CD133- or CD90-expressing cells, respectively, in both the GMP-hPL and FBS culture conditions (26) (Fig. 3C). GMP-hPL lot-to-lot variability was assessed in 50 distinct lots, identifying only limited variability in population doubling times (0.8 ± 0.2) (Fig. 3D). Furthermore, cells passaged in 50 different hPL lots revealed limited phenotype variability with robust maintenance of hallmark hMSC surface markers demonstrating on average 95 ± 5% expression for CD44, CD73, CD90, and CD105, as well as 85 ± 8% expression of HLA-ABC, and no expression of HLA-DR or 1 ± 1% expression of CD14 and CD45 (Fig. 3E). Moreover, late passage stem cells cultured in the GMP-hPL condition demonstrated maintained multipotency when subjected to differentiation. Specifically, the nonzoonotic condition supported adipogenesis, chondrogenesis, and osteogenesis, as monitored respectively by Oil-Red-O, Safrarin-O/Alcian blue, and Alizarin Red (Fig. 4A–C). Lineage commitment to the cardiac program (22), termed cardiopoiesis (5), was utillized on naive hMSCs (day 0) to induce nuclear translocation of early (Nkx2.5) and late (MEF2C) cardiac transcription factors (day 5) (7) (Fig. 4D). Immature organization of cardiac sarcomeric proteins, α-actinin and troponin-I, was detected 15 days after induction of cardiopoiesis but fully mature myocytes were not obtained in the 2D ex vivo culture conditions. Therefore, culture in GMP-hPL was devoid of lot-to-lot variability with robust cellular proliferation, and maintenance of phenotype stability or multipotency.
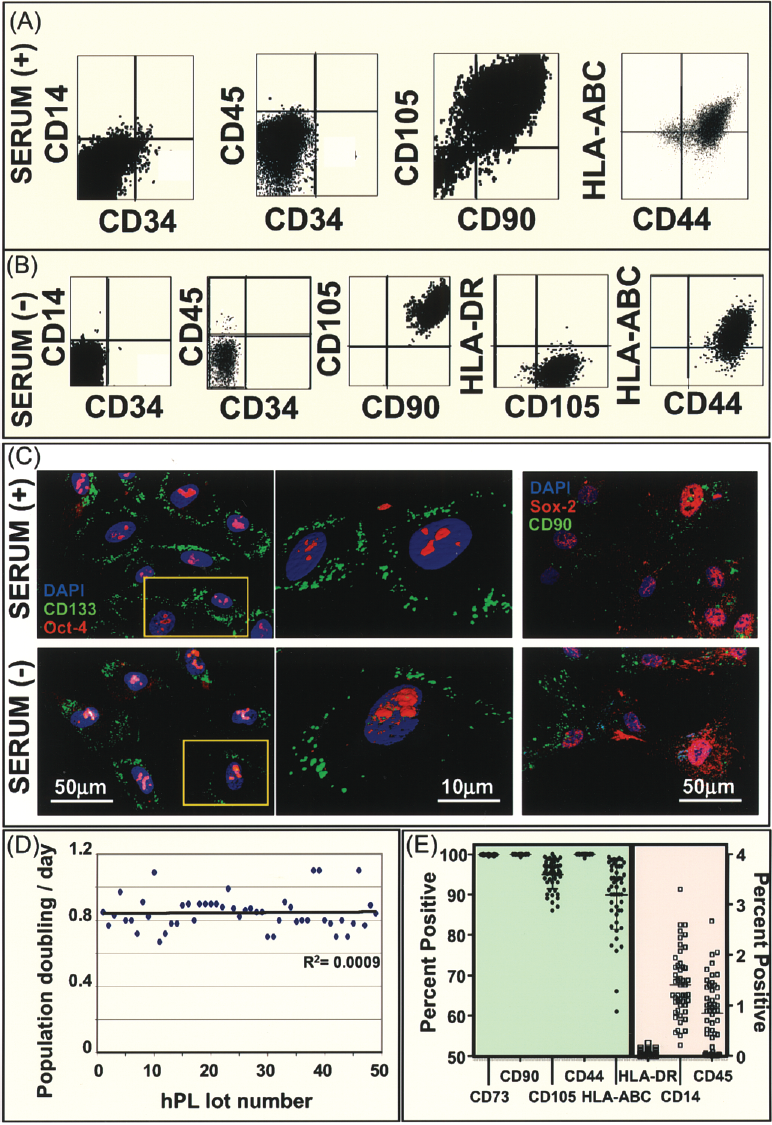
Long-term passage of hMSCs in nonzoonotic condition maintains phenotype. (A) hMSCs cultured with FBS [serum (+)] demonstrated expression a CD105+/CD90+/CD14-/CD34-/CD45- profile in greater than 95% of FACS-evaluated population at passage 12. (B) The nonzoonotic platelet lysate condition [serum (–)] maintained the hMSC marker profile CD105+/CD90+/CD14-/CD34-/CD45- with greater than 99% of the FACS evaluation population at passage 12. (C) Stem cells in both the FBS [serum (+)] and platelet lysate [serum (–)] condition maintained an immunohistochemical phenotype conforming to typical surface and stemness markers established in hMSCs. (D) hMSC population doubling times were assessed for 50 different lots of platelet lysate demonstrating limited variability. (E) Surface marker profiling of hMSCs cultured in 50 different platelet lysate lots revealed robust capacity for phenotype maintenance.

The serum-free condition maintains hMSC multipotency. hMSCs passaged 12 times in either FBS [serum (+)] or nonzoonotic [Serum (–)] condition continued to demonstrate capacity to undergo (A) adipogenesis with Oil-red-O, (B) condrogenesis with Safranin-O and Alcian blue, and (C) osteogenesis Alizarin Red. (D) Specialized differentiation was maintained in the nonzoonotic condition as hMSCs demonstrated capacity for sarcomerogenesis at passage 12 [early differentiation documented with nuclear translocation of cardiac transcription factors (D5) and myocyte formation determined by expression of sarcomeric proteins (D15)]. Bars labeled with measurements in each panel.
GMP-hPL Culture Is Associated with Chromosomal Stability
To address the impact of ex vivo expansion on risk for cell transformation, chromosomal stability of hMSCs was evaluated after long-term culture in GMP-hPL or FBS condition. At passage 12, 20 distinct patient-derived hMSCs were analyzed for 20 metaphases when grown in serum-free GMP-hPL condition with no clonal karyotypic derangements noted on traditional chromosomal smearing (Fig. 5A). In contrast, two of the patient-derived hMSCs evaluated at passage 12 in the FBS condition demonstrated cell transformation with clonal chromosomal aberrations including t(1:15) translocation for one patient (45 years old) and gain of a chromosome +21 with loss of –X for another patient (51 years old) (Fig. 5B). Of note, no chromosomal aberrations were noted in the same patients in nonzoonotic GMP-hPL culture (Fig. 5A and B are from the same two patients). High sensitivity spectral karyotyping (SKY) verified the stability of hMSCs grown in the nonzoonotic platelet lysate condition, as no clonal chromosomal aberrancies beyond normal and typical background changes were noted (Fig. 5C). Thus, hMSCs grown in the zoonotic FBS condition carry an apparent risk for transformation, a phenomenon not observed in hMSCs grown in serum-free GMP-hPL condition for extended periods of culture typical of production for clinical application.

Long-term passage in platelet lysate maintained chromosomal integrity. (A) The nonzoonotic platelet lysate condition [serum (–)] revealed no evidence of chromosomal aberrations after 12 passages. (B) hMSC demonstrated chromosomal rearrangement or trisomy after 12 passages in FBS [serum (+)] condition. Chromosomal smears in (A) and (B) are from hMSC-derived from the same patient cultured in either FBS or serum-free condition. (C) High sensitivity spectral karyotyping (SKY) revealed no clonal chromosomal rearrangements in hMSCs grown in nonzoonotic platelet lysate condition for 12 passages.
Proteomic Dissection of GMP-hPL Reveals Repair-Associated Signaling Cascades
To decode extracellular influence exerted by platelet lysate during expansion of hMSCs, GMP-hPL was subjected to high-throughput proteomic array analysis. Datasets were performed in triplicate and filtered based on background levels. Positive identification of proteins was made if expression exceeded negative controls by >10-fold in all three samples achieving a value of p < 0.001. Signaling pathways intrinsic to wound healing were uncovered via integration of GMP-hPL-expressed cytokines and growth factors into a network using unbiased bioinformatics algorithms (Fig. 6A). Additionally, nonarbitrary network evaluation with Ingenuity and GeneGo analysis programs ontologically ranked ordered wound repair and associated mechanisms at the top of biologic processes (Fig. 6B). Evaluation of highly connected hubs and established subnetworks provided candidate canonical intracellular pathways that are potentially activated within platelet lysate cultured hMSCs. The subnetwork topology conformed to a high degree clustering coefficient distribution, suggesting a nonstochastic architecture with hierarchical tendencies (Fig. 7A). Pathway prioritization identified, within the expressed hPL extracellular signaling molecules, namely CCL, FGF/EGF, TGF-β/BMP, and VEGF/PDGF as overrepresented (Fig. 7A). Cartesian plot analysis demonstrated a high correlation between highly connected extracellular proteins and injury repair (Fig. 7B). Further dissection of highly prioritized pathways provided identification of several potentially activated transcription factors, namely Nf-κb1, ETS1, RelA, c-Jun, SP1, STAT3, and STAT1, demonstrating high degree of correspondence to positive regulation of cell proliferation (Fig. 7C, D). Therefore, in silico bioinformatics evaluation of extracellular cues expressed within platelet lysate reveals candidate canonical pathways involved in wound repair and cell culture proliferation. Highly ranked extracellular signaling proteins expressed within GMP-hPL, as identified through proteomic and network analysis, were probed for 11 different GMP-hPL lots with ELISA analysis demonstrating conserved expression of TGF-β (125 ± 15 ng/ml), IGF-1 (130 ± 25 ng/ml), EGF (20 ± 3 ng/ml), PDGF (10 ± 3 ng/ml), VEGF (450 ± 130 pg/ml), and FGF (195 ± 100 pg/ml) (Fig. 7E). Thus, proteomic dissection of the platelet lysate here identified pathways central to healing as the molecular basis for the in vitro mitogenic effect and stability imposed on hMSCs.

GMP-hPL displays repair-associated extracellular signaling. (A) Integration of detected cytokines and growth factors (marked by red circle, see also Table 1) organized into a nonstochastic network. (B) Rank ordered evaluation of biologic processes using two distinct nonarbitrary network evaluation software programs (Ingenuity—orange, GeneGo—red, both—green) independently identified wound repair and associated mechanisms as a priori biological processes.
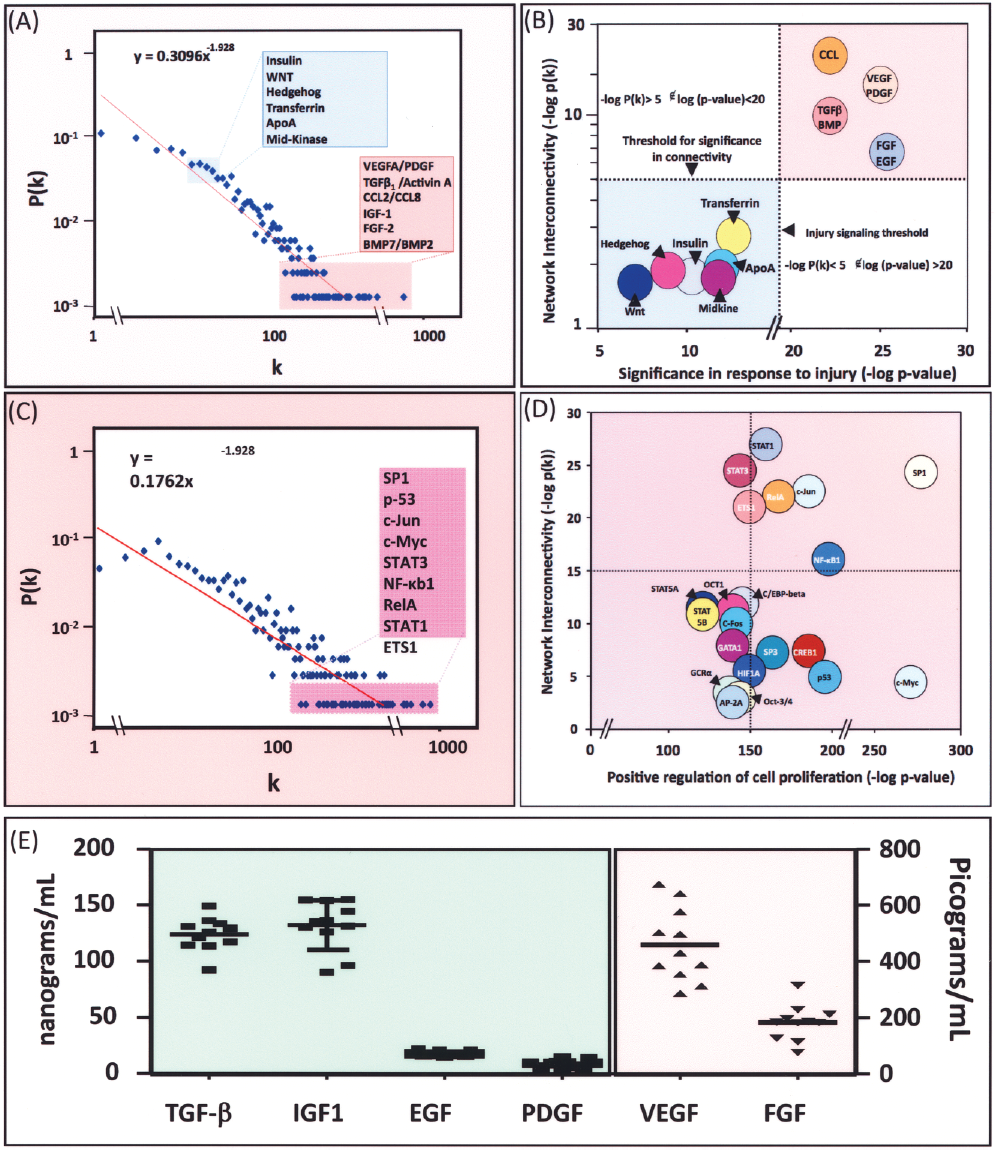
Ontologic nonarbitrary network evaluation associated extracellular network with the repair paradigm. (A) Degree and clustering coefficient distributions demonstrates nonstochastic architecture and hierarchical tendencies within the network. (B) Correlation of Cartesian plot analysis between network interconnectivity and injury repair signaling. (C) Degree and clustering coefficient distributions of downstream effectors identified overrepresented transcription factors within the extracellular network. (D) Cartesian plot analysis of all transcription factors with cell proliferation influence predicts the role of specific transcription factors as mediators of cell proliferation in the platelet lysate condition. (E) ELISA-based analysis of 10 different platelet lysate lots reveals conserved expression of highly ranked signaling molecules.
Discussion
This study identifies adipose tissue as an alternative body compartment to bone marrow for higher hMSC yield. Compared to FBS, this work further demonstrates human platelet lysate as a superior adjuvant to expedite derivation and proliferation of hMSCs regardless of source, while protecting against chromosomal aberrancy over protracted culture periods. Proteomic dissection of this nonzoonotic milieu identifies highly ranked signaling pathways, delineating the molecular basis for human platelet lysate-mediated mitogenic and stabilizing influence on the hMSC phenotype.
To date, expansion of hMSCs in FDA-approved clinical trials has been typically in the presence of FBS (24,25,29,32,49,51,53). The association of this standard practice with risk of virus and prion transfer along with zoonotic protein internalization and carryover has now been reported (13,14,46,47). In addition, the potential for deleterious outcome following cellular transplantation due to xenogenic protein-triggered immunogenicity or arrhythmogenesis has also been documented (11,16,47). Here, a donor-derived nonzoonotic platelet lysate, produced under good manufacturing practice (GMP) guidelines, was utilized to act as a substitute for FBS in the derivation and culture of clinical grade hMSCs. hMSCs cultured in this milieu demonstrated a growth profile exceeding that of FBS while preserving cell phenotype and multipotency when derived from either bone marrow or adipose tissue. The use of platelet lysate provided a humanized serum replacement adjuvant that conformed to all GMP guidelines to be utilized in the derivation and stable expansion of hMSCs for clinical applications.
Furthermore, despite evidence for genomic instability associated with the use of FBS, it has been argued that the risk for neoplastic change does not occur with passage numbers typical of clinical trials (42). However, recent evaluation of cells produced in cell therapy facilities for clinical trials revealed transient and donor-dependent recurring aneuploidy (48). Indeed, under zoonotic culture conditions, mesenchymal stem cells have been reported to undergo spontaneous clonal transformation at early or late passages with loss of phenotype and abnormal metabolism (18,42,43). In our hands, FBS cell culture for periods mimicking ex vivo expansion of clinical trials may carry a potential for clonal genomic instability, complicating therapeutic interventions. Such clonal chromosomal aberrancy was here not observed in hMSCs cultured in the platelet lysate condition with either standard or highly sensitive SKY karyotyping analysis, ensuring safety in cell transplantation.
The platelet lysate environment was conducive to an increase in cell proliferation. The rationale behind this cellular response to platelet lysate is thought to originate from mitogenic signals enriched within the clot that are released as platelets degranulate during tissue repair (35). Solid phase combinatorial ligand libraries have been utilized to capture and detect proteins of low abundance, and deplete high-abundance proteins such as albumin (9). Here the label-based approach, implemented for high-throughput proteomic analysis, provided a platform to dissect the proteome of the platelet lysate, uncovering distinct canonical pathways collectively conforming to the repair paradigm. Of the identified pathways, PDGF has been suggested as a major player in wound healing (52), and is the first growth factor to be approved for use in the treatment of nonhealing ulcers (36). This pathway along with VEGF receptor signaling has been recently shown critical in cell recruitment to sites of neovascularization in vivo (4). Members of the FGF family have broad impact on cytoprotection and survival under stress conditions. Furthermore, these factors have been shown to have mitogenic action in the healing wound as their direct application achieves tissue repair (52). The EGF receptor (EGFR) is dramatically upregulated after injury, and has been postulated to provide functional redundancy in the signaling paradigm of repair (21). The TGF-β superfamily has a broad spectrum of function with key roles in tissue differentiation homeostasis and repair (5,34,54). The CCL (e.g., monocyte chemoattractant proteins) family of chemokines has a well-established proinflammatory function during wound repair with chemoattractant activity on bone marrow cells (12,52), and capacity to induce cell proliferation. Indeed, a role for these signaling effectors on stem cell proliferation has been noted (4,30,38,44). Network interrogation of the extracellular factors identified within the platelet lysate predicted overrepresented signaling paradigms corresponding to wound repair and cell proliferation on subnetwork analysis. Further studies of the subnetworks through direct activation or inhibition of identified downstream targets are required to fully ascertain the specific mechanism by which platelet lysate regulates hMSC growth, maintenance, and chromosomal stability.
The present study thus establishes GMP-grade platelet lysate as a viable resource for the derivation and maintenance of hMSCs, from both bone marrow and adipose tissue. Replacement of FBS with GMP-hPL achieved superior hMSC growth, preserved cellular phenotype, and maintained chromosomal integrity. Proteomic dissection of the platelet lysate provided a comprehensive molecular cartography correlated with natural repair signaling. Therefore, this nonzoonotic condition establishes a model to diminish ex vivo hMSC expansion times and provides a GMP adherent resource amenable for safe therapeutic application.
Footnotes
Acknowledgments
We thank the Mayo Clinic Blood Bank for generous contribution of expired human platelets. This work was supported by the Mayo Clinic Discovery Translation Program, National Institutes of Health, Marriott Heart Disease Research Program, Cardio3 BioSciences and the Walloon Region, Mayo Clinic General Mills Clinician-Investigator Fellowship, and the Mayo Clinic Medical Scientist Training Program.
