Abstract
Fresh adipose-derived cells have been shown to be effective in the treatment of acute myocardial infarction (MI), but their role in the chronic setting is unknown. We sought to determine the long-term effect of the adipose derived-stromal vascular fraction (SVF) cell transplantation in a rat model of chronic MI. MI was induced in 82 rats by permanent coronary artery ligation and 5 weeks later rats were allocated to receive an intramyocardial injection of 107 GFP-expressing fresh SVF cells or culture media as control. Heart function and tissue metabolism were determined by echocardiography and 18F-FDG-microPET, respectively, and histological studies were performed for up to 3 months after transplantation. SVF induced a statistically significant long-lasting (3 months) improvement in cardiac function and tissue metabolism that was associated with increased revascularization and positive heart remodeling, with a significantly smaller infarct size, thicker infarct wall, lower scar fibrosis, and lower cardiac hypertrophy. Importantly, injected cells engrafted and were detected in the treated hearts for at least 3 months, directly contributing to the vasculature and myofibroblasts and at negligible levels to cardiomyocytes. Furthermore, SVF release of angiogenic (VEGF and HGF) and proinflammatory (MCP-1) cytokines, as well as TIMP1 and TIMP4, was demonstrated in vitro and in vivo, strongly suggesting that they have a trophic effect. These results show the potential of SVF to contribute to the regeneration of ischemic tissue and to provide a long-term functional benefit in a rat model of chronic MI, by both direct and indirect mechanisms.
Introduction
Myocardial infarction is a leading cause of mortality in Western countries (19). Large cardiomyocyte (CM) loss provoked by ischemia leads to impairment of cardiac contractility, loss of pump force, and subsequent risk of heart failure. Although new therapeutic strategies have contributed to improve survival and quality of life (3), regeneration of the myocardium remains an elusive goal which leaves organ transplantation the only alternative for chronic end-stage patients.
Over the last decade, the application of stem cells has opened new perspectives in the form of regenerative therapy whereby healthy cells can substitute the damaged tissue. A wide array of cell types has been tested in various animal models of MI, with bone marrow mononuclear cells and skeletal myoblasts reaching the stage of clinical trials (36). However, the results obtained with these two cell populations have shown only mild effects and, although long-term studies with larger cohorts of patients are required, new cell sources or approaches are sought in order to demonstrate the feasibility of this therapy.
Recent studies have demonstrated the capacity of stem/progenitor cells contained in the stromal vascular fraction (SVF) of the adipose tissue, not only in terms of their differentiation capacity (17), but also because they produce a wide array of growth factors and cytokines (8). Given that this cell population has been mainly tested in models of acute MI (43,44), the goal of the present study was to investigate the potential benefit of transplanting freshly isolated white adipose tissue-derived SVF cells in a rat model of chronic MI, focusing particularly on their long-term engraftment and differentiation capacity as well as their potential to restore the functional capacity of the heart.
Materials and Methods
Ethics Statement
All experiments were performed in accordance with the guidelines of the National Society for Medical Research and Institute of Laboratory Animal Resources. All animal procedures were approved by the University of Navarra Institutional Committee on Care and Use of Laboratory Animals.
Isolation and Characterization of Injected Cells
SVF cells were isolated from 4- to 8-week-old Sprague-Dawley eGFP rats as previously described (29), with slight modifications as follows. Briefly, rats were killed by cervical dislocation under CO2 anesthesia. Inguinal adipose depots were isolated, carefully minced and digested for 30 min at 37°C in DMEM-F12 (Invitrogen, Leiden, The Netherlands) containing 2% collagenase A (Roche, Madrid, Spain). After centrifugation of the pelleted SVF, the reaction was stopped by adding DMEM supplemented with 10% fetal calf serum to the sample, which was subsequently filtered through 100- and 40-μm filters. Cells were washed in PBS and incubated in lysis buffer in order to eliminate contaminating erithrocytes obtaining aproximatelly 1.15 × 106 cells/gr of white adipose tissue. Viability was shown to be around 98% as determined by trypan blue dye exclusion. Characterization of SVF was performed by fluorescence-activated cell sorting (FACS) analysis, staining cells with antibodies against major histocompatibility complex I and II [MHC I (RT1A) and MHC II (RT1B)], cluster of differentiation 11b (CD11b), CD31, CD44, CD45, CD73, CD90, and CD106 (all purchased from BD, Madrid, Spain). Expression of GFP was confirmed by FACS and direct visualization under a microscope equipped with epifluorescence, and ensured to be around 90% in all cases.
Experimental Model
A total of 82 female Sprague-Dawley rats (Harlan IBERICA S.L. Barcelona, Spain) underwent coronary artery ligation of the left coronary artery as previously described (9,18). Only those surviving animals (n = 57) in which LVEF decreased below 40% at 1 month post-MI were included in the study (n = 55). Heterogeneicity of the groups was excluded as no statistically significant differences were found in preimplantation EF values between SVF treated animals and the control group (Control: 29.87 ± 2.84%; SVF: 26.46 ± 2.26%; p < 0.29). A similar lack of significant variability intragroup was detected (Variances: Control: 80.48; SVF: 70.5; Levene's test for equality of variances: p < 0.678).
Five weeks post-MI, rats were reoperated by sternotomy and randomized to receive 107 cells per heart (total volume: 70 μl) of freshly isolated SVF from male GFP rats (n = 29) or media alone (n = 24), in two points of the infarct border region. No differences in early mortality after cell injection were detected in the cell-treated group versus the control group (only 1 out of 29 animals died during the injection versus none in the control group with 24 animals). All animals were daily immunosuppressed with cyclosporin A (20 mg/kg/day, IP, Sandimmun, Novartis) from 2 days pretransplantation until sacrifice at 1 and 3 days, 1, 2, and 4 weeks (2—4 animals/time point), and 3 months (10 animals).
Tissue Processing and Immunostaining
After sacrificed, the rat hearts were excised, fixed in 4% paraformaldehyde for 4 h at 4°C, and cut in three equal size blocks (apical, midventricular, and basal). Hearts were dehydrated in ethanol 70% (4°C o/n) and embedded in paraffin. For histological analysis 5-μm sections were performed. Cell detection was based on the presence of GFP-positive signals by inmunohistochemical methods [anti-GFP, Invitrogen, or chicken anti-GFP (Abcam, Cambridge, UK), diluted 1:500 in TBS] or immunofluoresecence. For immunofluorescence, a tyramide amplifying kit (Invitrogen) or a secondary antibody anti-chicken IgY coupled to 655 nm emitting quantum dots were used following manufacturer's instructions. Immunolabeling was performed with antibodies against α-smooth muscle actin (α-SMA) (diluted 1:1000 in TBS, Sigma, Madrid, Spain), cardiac troponin T (cTT) (diluted 1:100 in TBS; Labvision), ventricular myosin (diluted 1:2 in TBS; BioCytex), Ki67 (diluted 1:100 in PBS; Dako), caveolin-1 (diluted 1:100 in TBS, BD), connexin-43 (diluted 1:200 in TBS, Sigma), laminin (diluted 1:25 in TBS, Sigma), bFGF (diluted 1:100 in TBS, BD), HGF (diluted 1:100 in TBS, Assay Designs, Ann Harbor, MI, USA), active caspase 3 (diluted 1:100 in TBS, Cell Signalling, Beverly, MA, USA), MCP1 (diluted 1:100 in TBS, Abcam), VEGF (diluted 1:50 in TBS, Abcam), SM myosin (diluted 1: 400 in TBS, Abcam), TIMP1 (diluted 1:50, Abcam), TIMP4 (diluted 1:50, Abcam), MMP2 (diluted 1:100, Abcam), MMP9 (diluted 1:100, R&D), and MMP14 (diluted 1:100, Abcam). Secondary antibodies labeled with AlexaFluor-594 or AlexaFluor-488 were purchased from Molecular Probes (Invitrogen) if needed. EnVision™-HRP-conjugated system (Dako) was used as secondary reagent for immunohistochemistry. For confocal microscopy, a LSM 510 META (Carl Zeiss, Minneapolis, USA) microscope was used. For Sirius Red staining, sections were deparaffinized and immersed in 0.1% Fast Red (Sigma) in a saturated solution of picric acid, for 90 min, differentiated 2 min in HCl (Sigma) 0.01 N, dehydrated, and mounted in DPX. Hematoxylin-eosin staining was performed as detailed elsewhere. Briefly, sections where stained in Ehrlich's hematoxylin (Sigma) for 7 min, differentiated through water-HCl and Li2CO3 solutions, immersed in Eosin (Sigma) for 10 s, dehydrated, and mounted in DPX.
Electron Microscopy
For electron microscopy studies, hearts were fixed with 4% paraformaldehyde 0.5% glutaraldehyde. Tissues were cryoprotected in 25% sucrose for 48 h and cut in 100-μm sections. Sections were postfixed with 2% osmium, rinsed, dehydrated, and embedded in araldite (Durcupan, Fluka). Semithin sections (1.5 μm) were cut with a diamond knife and stained lightly with 1% toluidine blue. Later ultrathin (0.08 μ) sections were cut with a diamond knife, stained with lead citrate (Reynolds solution), and examined under a FEI Tecnai G2 Spirit transmision electron microscopy. Ultrathin sections were washed in phosphate buffer (PB), blocked in 0.3% bovine serum albumin-C (BSA, Aurion, Netherlands), and incubated in primary chicken anti-GFP antibody (Aves Labs; 1:200 in PB for 3 days at 4°C). Sections were washed in PB, blocked in 0.5% BSA and 0.1% fish gelatin (1 h, room temperature), and incubated in colloidal gold-conjugated anti-chicken secondary antibody (1:50 for 24 h). Sections were washed in PB and 2% sodium acetate. Silver enhancement was performed (as per Aurion instructions) and washed again in 2% sodium acetate. To stabilize, silver particles samples were immersed in 0.05% gold chloride (10 min at 4°C), washed in sodium thiosulfate then washed in PB, and postfixed in 2% glutaraldehyde (30 min). Sections were contrasted with 1% osmium and 7% glucose and embedded in araldite. Second, semithin 1.5-μm sections were prepared, selected at the light microscope level, and reembedded for ultrathin sectioning at 70 nm. Photomicrographs were obtained under a FEI microscope (Tecnai-Spirit) using a digital camera (Morada, Soft-imaging System).
Morphometric Analysis
Quantification of vascular density was performed in animals sacrificed 3 months posttransplantation. For capillary density (capillaries/mm2), serial sections 30 μm apart were stained with caveolin-1 and infarct border images were analyzed. Arteriolar density (arterioles/ mm2) and arteriolar area (μm2) were quantified in the same way after staining with anti-α-SMA-Cy3 in the following sections. Pictures were taken on a Nikon Eclipse E800 microscope equipped with epifluorescence optics and digital images were analyzed using imaging software (Jay Image), or on a Zeiss LSM 510 META laser confocal microscope. The degree of fibrosis was determined by quantification of collagen deposition stained by Sirius red staining of serial sections as previously described (6). Infarct size was assessed as the mean percent of infarcted area versus total LV area, ventricular infarct wall thickness as the average of repetitive wall thickness measurements per section, and fibrosis degree was measured in high-power photographs within the infarct as percent of collagen area (red) versus total tissue area (yellow). AnalySISR software was used for these analyses. Measurement of CM hypertrophy was performed on laminin-stained sections. High-power field images were acquired and CM cross sectional area was quantified using Image J software. A minimum of 100 CM per heart were analyzed.
Cell engraftment was quantified after GFP immunostaining. Positive cells were only counted if their nucleus were identifiable. Tissue sections were screened and GFP cells quantified from the first to the last section with engrafted cells. Percentage of surviving cells was referred to the quantity of injected cells (107 cells). Cell differentiation was measured in confocal laser microscopy images by counting total GFP-positive cells and those double-positive for GFP and lineage specific markers. GFP- and non-GFP-expressing adipocytes were counted by means of their characteristical morphology with AxioVision 4.6 (Zeiss, Madrid, Spain). Both differentiation and adipocytes are thus expressed as relative to the number of engrafted cells. Fifteen serial sections were analyzed, taking photographs of the whole left ventricle. Finally, cell proliferation and apoptosis were quantified by counting PCNA- or activated caspase-3-positive cells in serial sections double-stained for GFP and PCNA or GFP and activated caspase-3, respectively. All photographs were taken on a Zeiss LSM 510 META laser confocal microscope.
Echocardiographic Studies
Animals were slightly anesthetized prior to study with 2% isoflurane (Forane®. Lab. ABBOTT S.A., Madrid, Spain) in 100% O2 gas and placed in the supine or lateral position on a warming pad for transthoracic two-dimensional echocardiography, M-mode recordings, and Doppler ultrasound measurements as described (1). Echocardiography was performed using a Sonos 4500 ultrasound system (Philips) with a 12 MHz linear array transducer and Doppler measurement. Left ventricular remodeling was assessed by measuring end systolic and diastolic volumes and diameters, according to the American Society of Echocardiology. The left ventricular ejection fraction was determined in parasternal short axes (21) and diastolic function was assessed by measuring E and A waves of the mitral filling pattern by pulsed echo-Doppler technique in four-chamber apical views. Echocardiographic studies were performed in 10 animals/group at baseline (before infarct), before cell transplantation and at day 90 postransplant by the same investigator blinded to the group of treatment. Measurements were done in three cycles and the mean value was obtained.
18F-FDG PET Imaging Protocols, Image Reconstruction, and Semiquantitative Evaluation
Immediately before injection of cells and 90 days later, animals were subjected to PET analysis using the technique previously described by our group (27). Rats were anesthetized with 2% isoflurane in 100% O2 gas and after tail-vein injection of 18F-FDG (75 MBq in 100—200 μl) immediately awakened and placed back in the cage. Two hours after tracer injection, animals were anesthetized with isoflurane, placed prone on the PET scanner cradle, and kept during the overall study under continuous influx of the anesthetic. A static 60-min study (sinogram) was acquired in a Mosaic (Philips) small animal dedicated imaging tomograph. No transmission scan was performed. Scanner efficiency normalization, dead time, and decay corrections were applied during reconstruction. Images were reoriented for further processing and polar maps obtained using the specific cardiac imaging software package of the PET scanner. After reorientation of transaxial images into short- and long-axis slices polar maps were generated and divided into 17 different ROIs. Individual quantification of the 18F-FDG uptake in each of them was calculated. The total number of counts obtained for each of the ROIs was divided by its corresponding area to obtain counts per area unit. For each PET study, the maximal value of the 17 ROIs was considered as 100% and the remaining data transformed into percentage values. All further calculations and statistical analysis were performed on these sets of rescaled numerical data.
Cytokine Array
To obtain culture supernatants, freshly isolated rat SVF were cultured in DMEM/10% FBS at a density of 5 × 105 cells/cm2 until reach confluence (72 h), under normoxic or hypoxic (5% O2) conditions. Then, media was changed for DMEM/5% FBS and collected and frozen 48 h later for cytokine arrays or proliferation studies. Also, DMEM/5% FBS media was kept in the incubator during 48 h as control media. Cytokine levels were measured by a cytokine antibody array (Rat Array-I, Ray Biotech, Norkross, GA, USA) according to the manufacturer's instructions.
In Vitro Proliferation Studies
Rat smooth muscle cells (RAO Cell Applications, CA, USA) and murine endothelial cells (MS-I, ATCC, VA, USA) were plated in 24-well plates at a density of 2.5 × 103, 5 × 103 cells/well, repectively, and cultured in the presence of control media (NCM) or SVF conditioned media (CM) obtained under normoxic or hypoxic conditions (75% of the media). After 60 h, the number of viable cells was quantified by a luminescent cell viability assay (CellTiter-Glo™, Promega, USA) following manufacturer's protocol. Four independent experiments were performed and every cell type grown in CM or NCM was seeded in quadruplicate.
Gelatin Zymography
Serum samples prediluted 1:50 in saline were mixed with equal amounts of sodium dodecyl sulfate (SDS) sample buffer (Novex, Carlsbad, CA, USA) and electrophoresed on 10% SDS-polyacrylamide gels (Novex) containing 1 mg/ml gelatin as the protease substrate.
Following electrophoresis, gels were placed in 2.7% Triton X-100 for 1 h to remove SDS, and then incubated for 20 h at 37°C in developing buffer (50 mmol/L Tris base, 40 mmol/L HCl, 200 mmol/L NaCl, 5 mmol/L CaCl2, and 0.2% Briji 35; Novex). After incubation, gels were stained with gel code blue stain reagent (Pierce) for 1 h followed by destaining.
Statistical Analysis
Normal distribution was demonstrated by Shapiro-Wilk and Komogorov-Smirnov tests. All data are expressed as mean ± SD. Comparisons were performed using the paired or unpaired t-test, or ANOVA plus Tukey's HSD. Statistical analysis was performed with the SPSS 17.0 software and differences were considered statistically significant when p < 0.05.
Results
Cell Characterization
FACS analysis of SVF cells showed a heterogeneous population comprised of stromal, hematopoietic, and endothelial progenitor cells, among others, which proved positive for RT1A, low positive for RT1B, CD31, CD44, CD45, CD73, and CD90 and negative for CD11b and CD106 (Fig. 1).

Phenotypic characterization of SVF cells. SVF cells were stained with antibodies against RT1A, RT1B, CD11b, CD31, CD44, CD45, CD73, CD90, and CD106 (red line) or isotype controls (dashed lines).
SVF Cells Induce a Long-Lasting Improvement in Cardiac Function and Tissue Metabolism
The long-term functional efficacy of SVF cells was assessed by echocardiography. No significant differences at baseline were found between Control and SVF groups for any of the echocardiographic parameters. The results indicate that rats treated with SVF showed an improvement in cardiac function at 3 months, demonstrated not only by the increase in ejection fraction (from 26.46 ± 1.32% at preimplant to 38.25 ± 4.01% at 3 months; p < 0.01) and fractional shortening (FS) (10.16 ± 1.32% at preimplant to 16.3 ± 2.01% at 3 months; p = 0.006), but also by prevention of ventricle dilation as indicated by the limitation of the increase in the telesystolic diameter (ESD) and volume (ESV) (Table 1). By contrast, in the control group, no significant improvement in LVEF (from 29.87 ± 2.84% at preimplant to 28.1 ± 1.59% at 3 months; p = 0.63) and FS (from 12.3 ± 1.27% at preimplant to 11.34 ± 0.77% at 3 months; p = 0.586) was detected, while ESD and ESV deteriorated severely, indicating deleterious progression of the remodeling process (Table 1).
Echocardiography Studies

Values are mean (95% confidence interval). LVEF, left ventricular ejection fraction; SVF, stromal vascular fraction; FS, fractional shortening; EDD, end-diastolic diameter; ESD, end-systolic diameter; EDV, end-diastolic volume; ESV, end-systolic volume.
p < 0.01 (3 months vs. preimplant).
p < 0.05 (3 months vs. preimplant).
Consistent with the improvement in cardiac function, microPET studies revealed a significant increase in tissue metabolism within infarcted areas in animals treated with SVF-cells (from 50.6 ± 4.4% at preimplant to 60.6 ± 5.3% at 3 months; p < 0.05), whereas no significant changes were detected in control animals (from 45.3 ± 2.5% at preimplant to 42.7 ± 1.9% at 3 months) (Fig. 2).

MicroPET evaluation of tissue metabolism measured by 18F-FDG uptake. (A) 18F-FDG uptake was evaluated by microPET before and after 3 months of transplantation. Results are expressed as the percentage (mean ± SD) of 18F-FDG uptake in the infarcted area. *p < 0.05 (3 months vs. preimplant). (B) Representative images of tissue-polar map reconstructions, 3 months after SVF cells or medium injection.
Fate of Transplanted SVF Cells
Immunohistological detection of GFP throughout the experiment (1—3 days, 1—2 weeks, and 1—3 months) revealed no positive GFP signal in control hearts as expected (Fig. 3A—F). Conversely, the presence of SVF-derived GFP cells was demonstrated at all the time points analyzed in all cell-treated hearts (29/29) (Fig. 3G—L). Cells progressively changed their morphology from an initial fibroblastic appearance (Fig. 3G—J, asterisks) towards a more differentiated one (Fig. 3K, L, arrowheads). Apoptosis (Fig. 3M—R), although detected early after cell transplantation (1 day: 10.2 ± 2.2%; 3 days: 2.2 ± 0.5%, referred to total engrafted cells) disappeared after 1 week, whereas relatively high levels of proliferating GFP cells were detected within the area of injection during the first few days (1 day: 18.7 ± 2.8%; 3 days: 22.4 ± 1.2%, referred to total engrafted cells) and decreased afterwards (Fig. 3S—X). Quantification of cell engraftment indicated low retention of injected cells 1 day after transplantation (7.4 ± 1.1% engrafted cells) which decreased further at later time points (Fig. 3Y). Remarkably, cells could still be detected at 3 months (1.3 ± 0.5% of total implanted cells).

Engraftment of SVF cells. SVF engraftment was examined between 1—90 days posttransplantation. Representative images of animals injected with medium (A—F) or SVF (G—L) are shown. Cell engraftment was detected at all time-points by GFP-immunostaining. Undifferentiated cells were mainly identified at early time points (G—J, asterisks) whereas multiple GFP-derived phenotypes were present at 1 and 3 months (K, L, arrowheads). GFP+C3a+-apoptotic cells were detected up to 3 days (M, N, asterisks) but not thereafter (O—R). Engrafted proliferating GFP+Ki67+ cells (asterisks) were identified at different time points (S—X). Quantification (mean ± SD) of SVF engraftment (vs. total injected cells), apoptosis, and proliferation (vs. number of engrafted cells) (Y). Scale bars: 20 μm.
SVF Cells Positively Affect Vascularization Through Differentiation and Paracrine Mechanisms
Immunostaining demonstrated the potential of SVF cells to differentiate towards endothelial (caveolin-1+) (Fig. 4A—C) and mural cells (α-SMA+) (Fig. 4D—F) which was further confirmed by electron microscopy (TEM). Importantly, colloidal gold-stained endothelial cells formed functional vessels, as depicted by the presence of erythrocytes within the lumen (Fig. 4C). Mural GFP+α-SMA+ cells displayed pericytic morphology, surrounding newly formed capillaries (Fig. 4F). Interestingly, the percentage of cells that acquired a vascular phenotype increased over time to a total of 5.2 ± 1.8% and 9.4 ± 2.1% GFP-positive endothelial (Fig. 4G) and smooth muscle cells (Fig. 4H), respectively, at 3 months. Moreover, transplanted cells expressed the proangiogenic cytokines VEGF, HGF, and the proinflammatory cytokine MCP-1 (Fig. 4I—K, respectively, arrowheads).
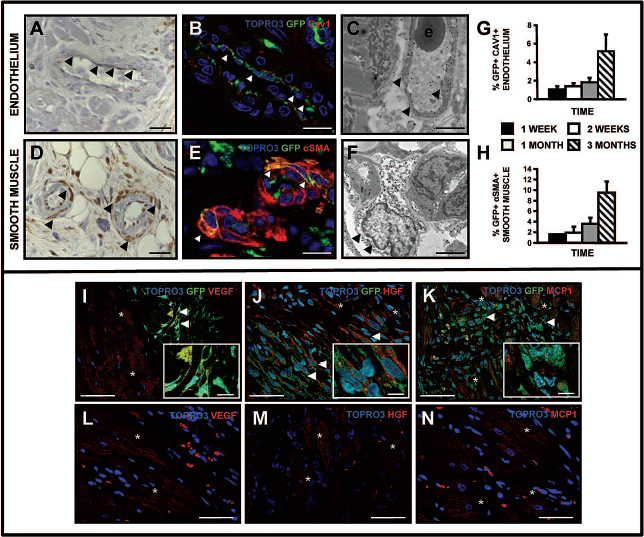
SVF cells differentiate towards vascular phenotypes in vivo. Endothelial and mural cell phenotypes were tested by immunohistochemistry (A, D) and double immunofluorescence for GFP and lineage-specific markers (B, E) was tested by. Representative pictures are shown (arrowheads). Transmission electron microscope (TEM) analysis confirmed the results, finding endothelial and pericytic GFP colloidal gold-positive cells (C, F, arrowheads; e: erythrocyte). Quantification of transplanted cell differentiation towards endothelium and smooth muscle was performed by counting the number of cells double-positive for GFP and lineage specific markers (caveolin-1 and α-SMA, respectively). Levels of endothelial cells (G) and pericytes (H) were expressed as a percentage of total number of engrafted cells. Results represent the mean ± SD. Expression of VEGF (I, arrowheads), HGF (J, arrowheads), and MCP1 (K, arrowheads) in transplanted cells and in host cells in the proximity of the transplant (I—K, asterisks) and in the peri-infarct area (L—N, asterisks). Scale bars: 15 μm (A, B, D, E), 2 μm (C, F), 25 μm (I—N), 8 μm (I—K, insets).
These factors were also secreted by host-derived cells in the proximity of the transplant (Fig. 4I—K, asterisks) and the peri-infarct zone (Fig. 4L—N), thus amplifying the paracrine effect induced by SVF cells. No positive cells were detected in the scar area or in the control groups (data not shown). This in vivo secretion of VEGF and MCP-1 was confirmed by cytokine array of the SVF-conditioned media (Fig. 5A), which also induced a significant proliferation of vascular cells (endothelial and smooth muscle cells) (Fig. 5B). Given the putative vasculogenic capacity of the transplanted SVF cells, we analyzed this aspect in more depth. As shown in Figure 6A, a higher vessel density was observed in the areas of engraftment, which again suggested the trophic effect of these cells. TEM of transplanted areas also revealed a high density of vessels (Fig. 6B, C) which, while functional, had an extremely thin wall, and marked them as newly formed/developing vessels. A significant increase in tissue vascularization was detected 3 months after transplant, both at the capillaries (small caliber-caveolin-1-positive vessels in control group: 683.9 ± 37.8 capillaries/mm2; SVF group: 883.9 ± 42.7 capillaries/ mm2; p < 0.05) (Fig. 6E—F) and larger vessels (α-SMA-positive vessels in control group: 46.3 ± 1.9 vessels/ mm2; SVF group: 76.4 ± 3.4 vessels/mm2; p < 0.01) (Fig. 6G—I). Thus, SVF transplantation not only directly contributes to form new functional blood vessels, but also stimulates a response that increases the vascularization of the peri-infarct through paracrine mechanisms.

Cytokine release pattern of the SVF conditioned media and in vitro proliferative effect. Angiogenic and inflammatory cytokine levels were analyzed utilizing arrays specific for rat (RayBio-Rat-I). To obtain culture supernatants, freshly isolated rat SVF were cultured in DMEM/10% FBS at a density of 5 × 105 cells/cm2 until reach confluence (72 h), under normoxic or hypoxic (5% O2) conditions. Then, media was changed for DMEM-5% FBS and collected and frozen 48 h later. Frames highlight factors that are not present in control arrays (incubated with DMEM/5%FBS) (A). The vasculogenic potential of the SVF secreted factors was confirmed in vitro by culturing the endothelial (MS-I) and smooth muscle cell (RAO) lines for 60 h in the presence of nonconditioned media (NCM) or SVF-conditioned media obtained under hypoxic or normoxic conditions and viable cell number quantified. Representative pictures show the cell density after treatment. The graphs represent the mean fold increase (vs. NCM) ± SD, quantified in four independent experiments, each performed in quadruplicates (**p < 0.01) (B). Scale bars: 150 μm.
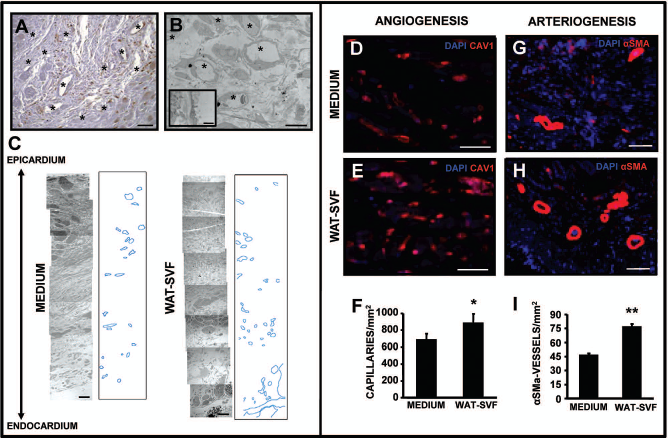
SVF injection induces increased vasculogenesis in vivo. Higher vessel density was detected inside the engrafted area and in the vicinity (A), which was confirmed by TEM, showing a multitude of vessels in the peri-infarct area with an extremely thin but active cytoplasm (B), characteristic of neovessels. TEM analysis of treated tissues also allowed reconstruction by serial imaging, showing a higher degree of vascularization within the area of injection (C). Capillary and arterioles/arteries densities (mean ± SD) were respectively determined by quantification of caveolin-1-positive capillaries/mm2 (D—F) and α-SMA-positive arterial vessels/ mm2 (G—I) in the perinfarcted area of animals treated with media (D, G) or SVF (E, H), 3 months after transplantation. *p < 0.05 and **p < 0.01. Scale bars: (A), 20 μm (B), 100 μm (C), 25 μm (D, E), 100 μm (G, H).
SVF Transplantation Strongly Influences Positive Tissue Remodeling
Despite this process of vascular differentiation, the great majority of transplanted cells (30.0 ± 5.4% of engrafted cells at 3 months) retained a fibroblastic phenotype (Fig. 7A—E). Some of the transplanted cells acquired characteristics of myofibroblasts (Fig. 7F) with expression of αSMA (Fig. 7G). They could be distinguished from vascular cells by absence of expression of other smooth muscle markers such as SM myosin (not shown) and also by TEM analysis (Fig. 7H, I), although their levels remained relatively low through the duration of the study (1.1 ± 0.4% at 3 months) (Fig. 7J). As the main mediators of remodeling processes, engrafted cells were screened for the expression of MMPs and their natural inhibitors TIMPs, demonstrating that SVF cells in vitro and in vivo expressed TIMP1 (Fig. 7K; Fig. 5A) and TIMP4 (Fig. 7L) but not other MMPs (2, 9, and 14) or TIMP2, as shown by double immunofluorescence at 3 months. Moreover, immunofluorescence for TIMP4 revealed that SVF-derived cells caused a marked increase in the expression of this inhibitor, not only in the area of injection, but also in the remote myocardium (Fig. 7M, N). Also, cell injection correlated with a reduction in the expression of MMP2 in the peri-infacted area (Fig. 7O, P). These results were further confirmed by zimographies performed on peripheral blood sera of SVF- and medium-treated rats, which showed a significant decrease in MMP2 activity in the SVF-treated animals (Preimplant: 0.81 ± 0.15%; 3 months: 0.62 ± 0.10%; p = 0.02) unlike control animals where no changes were found (Preimplant: 0.79 ± 0.05%; 3 months: 0.79 ± 0.07%; p = 0.98).

SVF cells positively affect tissue remodeling in vivo. A percentage of engrafted cells retained a fibrocytic phenotype (A), not staining with antibodies specific for smooth or cardiac muscle or endothelium (B). After colloidal-gold staining for GFP, this subset of cells were also found by TEM (C, D). Some fusiform cells (F) stained positive for α-SMA (and negative for SM myosin), identifying them as myofibroblast (G), which was confirmed by TEM (H, I). Quantification of transplanted cell differentiation towards fibroblasts and myofibroblasts was performed by counting the number of GFP-positive cells and double-positive for GFP and α-SMA. Levels of fibroblasts (E) and myofibroblasts (J) were expressed as a percentage of the total number of engrafted cells. Fibroblastic GFP cells expressed TIMP1 (K) and TIMP4 (L). Cell injection increased TIMP4 expression in the myocardium (N) in comparison with the nontreated ones (M) and also decreased MMP2 in the peri-infarcted area of the SVF hearts (P) versus the control hearts (O). Infarct size (percentage of infarcted area vs. total left ventricle area; %IA vs. LVA) (Q—S) and scar fibrosis (percentage of collagen volume fraction; %CVF) (T—V) were determined in Sirius Red serial stained sections while hypertrophy was determined by quantification of the cardiomyocyte size (μm2) in laminin immunostained serial sections (W—Y), 3 months posttransplantation. Results represent the mean ± SD. *p < 0.05; **p < 0.01. Scale bars: 15 μm (A, B, F, G), 2 μm (C, H), 0.25 μm (D, I), 25 μm (K, L), 2 μm (M, N), 50 μm (O, P, T—X), 250 μm (Q, R).
These changes were consistent with the morphometric analysis. Transplantation of SVF cells induced positive remodeling of the heart in comparison with control animals, suggesting a protective effect exerted by the cells. Thus, a significant smaller infarct size (medium: 19.7 ± 1.2%; WAT-SVF: 10.9 ± 3.4; p < 0.001), greater infarct wall thickness (medium: 432.6 ± 60.9 μ m; WAT-SVF: 739.6 ± 104.1 μm; p < 0.03) (Fig.7 Q—S) with lower scar collagen content (medium: 60.9 ± 1.7%; WAT-SVF: 46.8 ± 5.6%; p < 0.001) (Fig. 7T—V), and a lower degree of cardiac hypertrophy (medium: 293.3 ± 51.0 μm2; WAT-SVF: 205.9 ± 34.3 μm2; p < 0.02) (Fig. 7W—Y) were all detected 3 months after SVF treatment. On the other hand, no significant direct contribution of SVF to the cardiac tissue was detected. GFP-MLC2v double-positive cells were found but the percentage was indeed very low (0.017 ± 0.001% of engrafted cells at 2 weeks), indicating that the differentiation of SVF cells to cardiomyocytes is at most a rare event (Fig. 8).
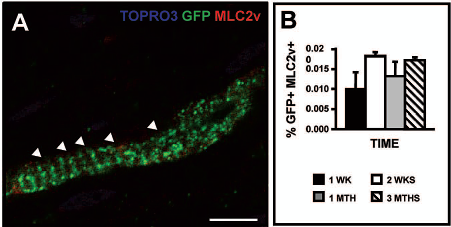
SVF-derived cells differentiate into cardiomyocyte-like cells in vivo. Confocal laser images demonstrated presence of GFP+ cells with the morphology of CM, positive for the cardiac marker MCL2v (A). Quantification of cardiac differentiated cells was assessed by counting the number of double-positive cells (GFP and MLC2v), and expressed as a percentage of total number of engrafted cells (B). Results represent the mean ± SD. Scale bars: 15 μm.
Finally, no ectopic tissue formation, such as bone, cartilage, or tumor, was detected in any of the transplanted animals (not shown) and, although a relatively high percentage of the engrafted cells differentiated towards adipocytes (23.6 ± 7.7% after 3 months) (Fig. 9), there was no significant increase in adipose cells in the SVF-injected hearts versus the media-injected ones (not shown), suggesting that there was no significant induction of adipogenesis in the transplanted hearts.

SVF-derived cells give rise to adipocytes. Transplanted cells also gave rise to adipocytes (A), which was further confirmed by TEM showing colloidal gold-stained cells with characteristics of adipose cells (B). Quantification between 1 week and 3 months was assessed by counting the number of GFP+ cells with morphology of adipocytes and expressed as a percentage of total number of engrafted cells (C). Results represent the mean μ SD. Scale bars: 15 μm (A), 2 μm (B), 1 μm (B, inset).
Discussion
Although the unexpected vasculogenic and cardiomyogenic potential of adipose tissue-derived stem cells has prompted their use in models of acute MI (43), significantly less information has been gathered regarding their use in chronic models. Thus, unlike recent studies in which subpopulations of adipose derived stem cells (22,44) or previously in vitro cultured SVF cells were used (18,43), our research tested freshly isolated SVF, since it may contain several stem/progenitor cell populations (17) and can also be readily obtained from patients without the need for extensive processing. Albeit stromal-cultured cells may benefit from their mesenchymal nature, they may also lose the potential to give rise to all the lineages needed for the healing of the disease. Moreover, fresh adipose cells could be implemented as an autologous therapy not only for the chronic but for the acutely infarcted patients as well. The results of our study demonstrate that transplantation of SVF cells in chronically infarcted tissue promotes a persistent benefit in cardiac function and metabolism by inducing tissue revascularization and protection against deleterious tissue remodeling. Furthermore, an ongoing clinical trial (PRECISE trial: NCT00426868), in which SVF cells have induced a beneficial effect in chronically infarcted patients, support the feasibility and safety of this therapy.
Furthermore, in contrast to previous studies in which culture adipose cells were used, our results indicate that SVF cells responded in vivo to tissue signals that induced their differentiation towards cardiovascular cells (15,18). In fact, the differentiation capacity of SVF into endothelial (30) and smooth muscle (11,41) cells was consistent with recent in vitro and in vivo studies, which demonstrated direct incorporation of the SVF cells into mature vessels, both capillaries and arterioles/arteries, in a hind-limb ischemia model (22,41). Similarly, cardiac-like progenitors have also been isolated in vitro from fresh SVF (26,29) but not from cultured adipose cells. In this sense, despite the low percentage, our study provides proof of the concept that SVF can also differentiate in vivo into cardiomyocytes. An important question that remains to be answered is which population of cells within the SVF is endowed with cardiac potential.
An overwhelming issue in cell therapy remains the low engraftment of transplanted cells, which diminishes the efficiency of cell therapy [reviewed in (34)]. Recent studies have demonstrated that a combination of prosurvival factors (13) or the use of a matrix support (12, 23,37) favors their survival. Remarkably, in our study, despite the fact that “bare” cells were directly injected into a chronically infarcted myocardium (a more severe situation than the acute model), they could be detected up to 3 months later in relatively high levels in comparison with reported findings with other (stem) cell types (34). On the other hand, the low degree of engraftment indicates that despite the direct contribution of the transplanted cells, they cannot be responsible for the beneficial effect, making more likely that the paracrine effect is the mechanism, which explains the functional results observed (32). SVF transplantation strongly induced revascularization of the heart tissue along with the secretion of the angiogenic factors VEGF and HGF, together with the inflammatory factor MCP-1. MCP-1 is known to be involved in the recruitment of a subpopulation of monocytes that positively affect the initiation of angiogenesis (2,5), which was clearly detected in the transplanted cells, adding support to the trophic hypothesis. The in vitro culture studies with SVF-conditioned media further confirm this mechanism.
In this regard, several reports have demonstrated the capacity of the SVF or SVF-derived cells to secrete cytokines with angiogenic properties like VEGF (15,25, 33) and HGF (10) and to directly enhance the chemotaxis of progenitors by producing SDF-1 (38), possibly contributing to the increased vasculogenesis of the heart (33). Additionally, HGF could also exert a wide array of protective effects beyond induction of angiogenesis. In cardiomyopathic hamster hearts, treatment with HGF not only decreased cardiac hypertrophy, tissue fibrosis, remodeling, and dysfunction, but also induced a significant benefit in cardiac function (24). Along similar lines, we have recently demonstrated in a chronic model of MI in rats that the functional benefit associated with skeletal myoblast transplant is at least partially related to the production of angiogenic cytokines by the transplanted cells (28).
On the other hand, as an evolving tissue, the remodeling heart is a complex system in which MMPs and TIMPs are tightly regulated (40). Even minor changes in their balance can end up in major histopathological and functional consequences, as reported in animal models (4) and patients (16). TIMP1 is downregulated in ischemic cardiomyopathy (16) and is able to inhibit CM apoptosis (39). TIMP4, on the other hand, is preferentially expressed in the heart (14) and has been reported to be downregulated in cardiac diseases (16,35). TIMP4 inhibits the activity of MMP2 (7) so it is plausible that upregulation of TIMP4 in transplanted SVF cells and injured tissues may be responsible for the downregulation of MMP2 in the peri-infarct region and the significant decrease in peripheral serum MMP2 activity. This could intrinsically have therapeutic implications, as MMP2 is upregulated in MI (40) and may also influence cardiac regeneration by inactivating SDF1 (20), thus decreasing progenitor homing to the damaged tissue. The paracrine potential of TIMP4 may be related to its antithrombotic features (31) and its ability to regulate proper cardiac fibroblast behavior (42). Globally, SVF-derived regulation of TIMP/MMP can induce a protective balance upon injured hearts which, together with the increased revascularization of the tissue that could rescue myogenic cells at the border zone of the infarct, would avoid the progressive deleterious remodeling of the heart (as shown by detection of a smaller infarct size, thicker infarct wall, and lower degree of scar fibrosis and cardiac hypertrophy in the SVF-treated hearts).
Finally, although no adverse effects were detected in the SVF-transplanted animals and no local induction of adipogenesis of host cells was induced in the hearts, it would be interesting to analyze the effects of SVF cells depleted for the preadipocytic population (sorted, for example, for their pref-1-specific marker) in order to elucidate the best treatment and potential clinical application.
In summary, this report demonstrates the long-term benefit of SVF cell injection in chronic myocardial infarction and provides evidence for the in vivo multilineage capacity and paracrine activity of this population. Furthermore, compared with ADSC (cultured adipose-derived stem cells), the significantly faster and inexpensive processing of the cells makes them much more affordable in the clinical setting. If proven safe and effective, SVF would offer an attractive population to consider for future clinical applications for cardiac repair.
Footnotes
Acknowledgments
This work was supported in part by ISCIII PI050168, PI070474, CP09/00333 and ISCIII-RETIC RD06/0014, MICCIN PLE2009-0116, and PSE SINBAD, Gobierno de Navarra (Departamento de Educación), Comunidad de Trabajo de los Pirineos (CTP), European Union Framework Project VII (INELPY), Caja de Ahorros de Navarra (Programa Tu Eliges: Tu Decides) and the “UTE project CIMA.”
