Abstract
Stem cells contained in the amniotic membrane may be useful for cellular repair of the damaged heart. Previously, we showed that amnion-derived cells (ADCs) express embryonic stem cell surface markers and pluripotent stem cell-specific transcription factor genes. These ADCs also possess the potential for mesoderm (cardiac) lineage differentiation. In the present study we investigated whether untreated naive ADC transplantation into the injured left ventricular (LV) myocardium is beneficial as a cell-based cardiac repair strategy in a rat model. ADCs were isolated from Lewis rat embryonic day 14 amniotic membranes. FACS analysis revealed that freshly isolated ADCs contained stage-specific embryonic antigen-1 (SSEA-1), Oct-4-positive cells, and mesenchymal stromal cells, while hematopoietic stem cell marker positive cells were absent. Reverse transcription-PCR revealed that naive ADCs expressed cardiac and vascular specific genes. We injected freshly isolated ADCs (2 × 106 cells suspended in PBS, ADC group) into acutely infarcted LV myocardium produced by proximal left coronary ligation. PBS was injected in postinfarction controls (PBS group). Cardiac function was assessed at 2 and 6 weeks after injection. ADC treatment attenuated LV dilatation and sustained LV contractile function at 2 and 6 weeks in comparison to PBS controls (p < 0.05, ANOVA). LV peak systolic pressure and maximum dP/dt of ADC-treated heart were higher and LV end-diastolic pressure and negative dP/dt were lower than in PBS controls (p < 0.05). Histological assessment revealed that infarcted myocardium of the ADC-treated group had less fibrosis, thicker ventricular walls, and increased capillary density (p < 0.05). The fate of injected ADCs was confirmed using ADCs derived from EGFP(+) transgenic rats. Immunohistochemistry at 6 weeks revealed that EGFP(+) cells colocalized with von Willebrand factor, α-smooth muscle actin, or cardiac troponin-I. Our results suggest that naive ADCs are a potential cell source for cellular cardiomyoplasty.
Introduction
Amnion-derived cells (ADCs) have notable advantages as a stem cell source for cellular therapeutic applications. Human amnion is readily available from normally discarded placentas and is easily procured without invasive procedures. Therefore, ADC use does not elicit an ethical debate related to the use of fetal-derived tissues. One of the clinical relevant advantages of ADCs is the potential of a wide representation of human leukocyte antigen (HLA) types (7). Abundantly available cell sources enable tissue banking of various HLA types to support immunocompatible allogeneic cell transplantations.
Recently, the differentiation capabilities of rodent and human ADCs have been described (12,14,19). Sakuragawa et al. reported human amniotic epithelial cells have characteristics of neural progenitor cells and subsequent studies have succeeded to induce functional neural differentiation, in vitro (21) and in vivo (6). Knezevic et al. transplanted rat amnion into rat kidney capsules and showed that donor cells differentiated to form epithelial structures including hair follicles (9). In 2004, Tamagawa et al. established a cell line from human amnion and generated chimeric mouse–human embryoid bodies to prove that human ADCs contribute to the development of all three germ layers (23).
Previously, we reported that the human amnion contains pluripotent stem cells based on cell surface markers and the expression of pluripotent stem cell-specific transcription factors such as Oct-4, Sox2, and Nanog (12). While some components of placenta derive from trophectoderm (extraembryonic), amniotic epithelial cells differentiate from pluripotent epiblasts at day 8 following fertilization and at least some cells retain pluripotent stem cell characteristics (13). Term amniotic epithelium may also contain various lineage committed progenitor cells, which have spontaneously differentiated during fetal development. In the previous study, we also showed the potential of human amniotic epithelial progenitor cells to differentiate along a cardiomyocyte lineage under specific culture conditions (12).
Given these observations, we hypothesized that untreated naive ADCs could contribute to the repair of damaged myocardium, prevent adverse remodeling, and improve cardiac function following myocardial infarction. We investigated the potential of syngeneic ADCs to facilitate cardiac remodeling and functional recovery through the injection of ADCs into a region of acute infarction produced by coronary ligation in adult rats.
Materials and Methods
Animals
All protocols followed National Institutes of Health (NIH) guidelines for animal care and were approved by the University of Pittsburgh's Institutional Animal Care and Use Committee and Children's Hospital of Pittsburgh Animal Research Care Committee. Gestational day 14 time-pregnant and 12-week-old adult female Lewis rats weighing 200–250 g were used (Harlan Sprague Dawley Inc., Indianapolis, IN). In order to track the fate of implanted ADCs, we used gestation day 14 positive EGFP-transgenic rat placentae and adult negative EGFP-transgenic rats as recipients (200–250 g body weight). The EGFP-transgenic rats were originally generated by Dr. Masaru Okabe (University of Osaka, Osaka, Japan) (25) and EGFP transgenic rat colonies were maintained within the animal facility of the Children's Hospital of Pittsburgh of UPMC.
Cell Isolation
Due to the logistical challenge of harvesting rat ADCs following delivery, we isolated ADCs from pregnant rats at gestational day 14. The uteri were excised from pregnant rats, transferred to cold phosphate-buffered saline (PBS), and then placentae were collected and pooled in a new petri dish. Harvested placentae were washed thoroughly with PBS. The almost transparent rat amnion was then dissected in sterile manner. The membrane was enzymatically digested using 0.05% trypsin/EDTA solution (Invitrogen) for 30 min at 37°C. Dissociated cells were collected through 100-μm cell strainer and washed three times with cold PBS. Viability of the rat ADCs was determined by exclusion of trypan blue dye and cell number was determined using a hemocytometer.
Flow Cytometric Analysis
Mixed rat ADCs isolated from 45–50 fetuses were used for flow cytometric analysis (FACS SCAN flow cytometer, Becton Dickinson). Cells were incubated for 30 min at 4°C with the FITC-conjugated mouse monoclonal antibodies against rat CD34 (clone ICO-115, Santa Cruz Biotechnology), CD45, and CD90 (clones OX-1 and OX-7, respectively, Becton Dickinson), and hamster anti-rat CD29 monoclonal antibody (clone Ha2/5, Becton Dickinson). Embryonic stem cell surface markers such as PE-conjugated anti-Stage Specific Embryonic Antigen-1 (SSEA-1) monoclonal antibody (clone MC-480, R&D systems) and PE-conjugated POU transcription factor, Oct-4, antibody were used. Isotype-identical antibodies served as controls for each analysis. FACS analysis was repeated at least three times. Following FACS analysis, the Oct-4/SSEA-1 double stained cells were embedded in 10% acrylamide and observed under the confocal microscope.
Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
Total RNA was prepared using Trizol solution (Invitrogen) and treated with TURBO DNA-free kit (Ambion, Austin, TX). Primers for target genes, Nkx2.5, GATA4, α- and β-cardiac myosin heavy chains (MHC), connexin 43 (Cx-43), α-smooth muscle actin (SMA), calponin-1 (Clp1), and PECAM were obtained from Qiagen Quanti-Tect Primer Assay with the target fragment sizes approximately 100 base pairs. Specific primers corresponding to cardiac α-sarcomeric actin (Card-α-Act) gene was designed. Rat Card-α-Act: forward 5′-GCC CTG GAT TTT GAG AAT GA-3′, reverse primer 5′-CCT TTT GCA TAC GAT CAG CA-3′. One-step RT was performed with a total volume of 1 μg RNA in a total volume of 25 μl that used MuLV (Roches, Pleasanton, CA) with program: 42°C 15 min, 99°C 5 min, 5°C 5 min, one cycle. cDNA (1 μl) was used for PCR, which used the program: 94°C 2 min, 95°C 50 s, 58°C 30 s, 72°C 1 min, 35 cycles with 72°C, 7 min extension. As an internal control a primer set for β-actin was used. All PCR products were confirmed by University of Pittsburgh DNA Sequence Core Facilities, performed by Eppendorf Mastercycles. All RT-PCR assays were completed in triplicate.
Myocardial Infarction and Cell Transplantation
Animal anesthesia was induced using 3.0% isoflurane inhalation followed by endotracheal intubation. The endotracheal tube was connected to a rodent volume-controlled mechanical ventilator. Hemodynamic monitoring was performed using limb lead electrocardiograms and tail cuff blood pressure recording. The heart was surgically exposed through a left thoracotomy and the proximal left anterior descending coronary artery was ligated with 7-0 polypropylene (3). Myocardial ischemia was confirmed by regional cyanosis and ST segment elevation. Following coronary ligation, 24 Lewis rats with infarcts were randomly divided into two groups: ADCs injection (ADC group n = 10), and PBS injection (PBS group; n = 14). In the ADC treatment group, after coronary artery ligation, 500 μl solution containing 2 × 106 ADCs in PBS were injected into the apical, proximal, lateral, and septal wall bordering the infarct and into the center of the infarct (5 portions, 100 μl per region). For the PBS injection group, 500 μl PBS was injected into the same location (100 μl per region). The incision was closed in layers with 4–0 silk continuous sutures in both groups. To investigate the fate of injected ADCs, naive 2 × 106 ADCs isolated from positive EGFP-transgenic amnion were injected into six negative EGFP-transgenic rats.
Assessment of Cardiac Function
Echocardiography was performed before coronary artery ligation, at 2 weeks, and at 6 weeks following ADC injection. Rats were sedated for echocardiography using 1.5% isoflurane inhalation delivered via nosecone. Standard transthoracic echocardiography was performed using the Acuson Sequoia C256 system with a 13-MHz linear ultrasonic transducer (15L8; Acuson Corporation, Mountain View, CA) in a phased array format. B-mode (2D) images were acquired in the LV short axis view at the level of the papillary muscles. End-diastolic (EDA) and end-systolic (ESA) LV internal cavity areas were measured by tracing the endocardial border. LV fractional area change (%FAC) was calculated as %FAC = [(LVEDA — LVESA)/LVEDA] × 100%. All measurements were performed using Scion Image software (Scion Image, Fredrick, MD) (3,18). LV pressure measurement (ADCs group, n = 7; PBS group, n = 8) was performed using a micropressure manometer (SRF-1000, Miller Instruments) introduced retrograde into the LV from the right carotid artery at 6 weeks. After standard echocardiography, the right carotid artery was exposed, secured with proximal and distal sutures, and a 1.0 French diameter pressure catheter was inserted to the LV cavity. The LV cavity pressure was recorded digitally using a custom program (LabVIEW, National Instruments, Austin, TX).
Histological Assessment
Six weeks after ADC injection, rats were sacrificed and their hearts were harvested, frozen in 2-methylbutane precooled in liquid nitrogen, and cryosectioned (from the apex to the base of each heart) in 8-μm thickness serial sections in the LV transverse direction. For cell tracking, EGFP-transgenic ADC-injected rats were sacrificed at 1, 2, and 6 weeks following cell injection (n = 1, 3, and 2, respectively). Hematoxylin and eosin (H&E) and Masson's trichrome staining were performed as previously described (2). Tissue sections for immunohistochemistry were fixed with 2% paraformaldehyde for 5 min and reacted with primary antibodies against basic fibroblast growth factor (bFGF, Serotec, Raleigh, NC), vascular endothelial growth factor (VEGF, Santa Cruz Biotechnology, Santa Cruz, CA), α-smooth muscle actin (α-SMA, Sigma, St Louis, MO), von Willebrand Factor (vWF, Abcam, Cambridge, MA), or cardiac Troponin-I (cTnI, Abcam). Subsequently the relevant antigen was visualized fluorescently (Alexa 594, Molecular Probes, Eugene, OR). Nuclei were stained with 4′,6-diamidino-2-phenyindole (DAPI, Sigma). We reconstructed 3D composite images from stacks of ζ-axis optical scans of each stained sample using a standard laser confocal microscope system (FV1000, Olympus, Tokyo, Japan) and Scion Image software (Scion Corp, MD) (24). The images were visualized at different rotation angles to identify colocalization of EGFP and individual fluorescent staining. For each LV sample five different microscopic fields at 100× for the wall thickness measurement and 10 different fields at 200× for capillary density were digitally photographed for each heart and analyzed using NIH Image software (NIH image and Adobe Photoshop, San Jose, CA), as previously described (3).
Statistical Analysis
All data were expressed as mean ± SD. All calculations were performed using SigmaStat (Systat Software Inc, Point Richmond, CA). Student' t-test was performed to compare wall thickness, capillary density, and LV cavity pressure between experimental groups. Two-way repeated ANOVA with Tukey test was performed to compare LV cavity areas and %FAC. Statistical significance was defined at p < 0.05.
Results
Rat Amnion Contains Embryonic Stem Cell Marker-Positive Cells
A total of 328 fetal amniotic membranes were collected from 34 pregnant rats. Most ADCs expressed the mesenchymal stem cell marker CD29 (94.3 ± 2.22%), while only 4.73 ± 0.94% cells were positive for CD90. Most of rat ADCs were negative for hematopoietic stem cell markers, CD34 and CD45. A small subpopulation of ADCs expressed the embryonic stem cell marker, stage-specific embryonic antigen-1 (SSEA-1, 0.10 ± 0.01%), and pluripotent stem cell-specific transcription factor, Oct-4 (0.62 ± 0.68%) and 0.10 ± 0.01% of the cells were SSEA-1/Oct-4 double positive cells (Fig. 1A). In one of the typical experiments, double positive cells were selected in the gated subset panel (right panel of Fig. 1B) and retrospectively investigated the cell size (forward scatter/FSC) and cell shape/cytoplasmic complexity (side scatter/SSC). The double positive cells were widely dispersed in the ungated panel. The result indicates that the signal was not an artifact signal and the double positive cells were low in number but contained in the naive rat ADCs. Following fluorescent microscopic observation, the presence of double positive cells was confirmed (Fig. 1C). Similar to the human amniotic epithelial cells, most of the Oct-4 expression was observed in the cytoplasmic area.

Rat amnion-derived cell (rADC) population contains embryonic stem cell markers, Oct-4 and SSEA-1, positive cells. (A) Freshly isolated rADCs labeled with FITC-conjugated anti-Oct-4 antibody and PE conjugated anti-SSEA-1 antibody. Corresponding fluorescein-conjugated isotype antibodies were used as control. (B) Oct-4/SSEA-1 double positive cells were scattered in the ungated cell profile plot panel. (C) The Oct-4/SSEA-1 double positive cells were embedded in 10% acrylamide and observed under the confocal microscope. Yellowish fluorescence represents Oct-4 (FITC/green) and SSEA-1 (PE/red) double positive cells. Scale bar: 10 μm.
Rat Amnion-Derived Cells Express Cardiac and Vascular Genes
To determine whether rat ADCs contain lineage committed cardiac progenitor cells, RT-PCR was performed using cardiomyocyte and vascular gene primers. Freshly isolated ADCs expressed both cardiac (Nkx2.5, GATA-4, cardiac α-sarcomeric actin, β-cardiac myosin heavy chains, and connexin 43) and vascular (α-smooth muscle actin, calponin-1, and PECAM) lineage genes (Fig. 2).

Expression of cardiomyocyte-specific genes and vascular cell relate genes in the naive rat ADCs. RT-PCR was performed with PCR primers that specific for rat genes encoding cardiac proteins and vascular cells related proteins. Most of cardiac specific genes were expressed in freshly isolated rat ADC except α-MHC. β-Actin was used as internal control.
Mortality and External Morphology
There were four deaths within 24 h after coronary artery ligation surgery (one from the ADCs group and three from the PBS group, 10% and 21.4% mortality rate, respectively). There were no late postoperative deaths in either surgical group (ADCs group, n = 9; PBS group, n = 11). ADC-injected hearts did not show tumor formation at 6 weeks after ADC transplantation. The pericardium overlying the region of myocardial infarction became opaque and in the PBS group, this opaque epicardium represented a much larger LV anterolateral surface area versus the ADC group (Fig. 3A, B).

Representative images, 6 weeks after surgery, of the anterior view of PBS control (A), and PEUU patched (B) hearts. Blue arrows point to the ligation suture of proximal left anterior descending coronary artery. Scale bar: 5 mm.
ADC Transplantation Reduced Postinfarction LV Cavity Dilatation and Improved LV Systolic Function
Longitudinal echocardiography showed that LV EDA increased and %FAC decreased at 2 weeks after coronary artery ligation in both ADC and PBS groups in comparison to precoronary ligation. However, at 6 weeks the PBS group displayed further increased LV EDA and decreased %FAC (p < 0.05 for each vs. 2 weeks), while the ADC group sustained both LV EDA and %FAC (p < 0.05 vs. PBS) (Fig. 4A, B). Of note, heart rate was similar between the groups. LV peak systolic pressure and maximum dP/dt (parameter of systolic function) were higher in the ADC group than in the PBS group at 6 weeks (p < 0.05); the LV end-diastolic pressure and minimum dP/dt (parameter of early diastolic function) was lower in the ADC group than in the PBS group (p < 0.05) (Table 1).

Echocardiographic assessment of PBS control, and ADCs injection during the study period. EDA (end-diastolic area) is shown in (A), and %FAC (fractional area change) in (B). ‡p < 0.05 between groups at 6 weeks; ∗p < 0.05 versus within group; †p < 0.05 versus 2 and 6 weeks within group.
Left Ventricular Pressure at 6 Weeks After ADC Injection
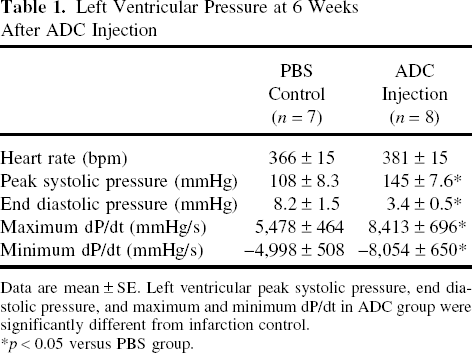
Data are mean ± SE. Left ventricular peak systolic pressure, end diastolic pressure, and maximum and minimum dP/dt in ADC group were significantly different from infarction control.
p < 0.05 versus PBS group.
Reduced Myocardial Infarct Area 6 Weeks After Rat ADC Implantation
Histological sections stained with H&E and Masson's trichrome revealed extensive anterior-lateral wall injury extending towards the posterior ventricular wall with fibrous tissue replacement following coronary ligation in the PBS-injected animals. In the ADC-treated group, fibrous tissue was limited to the LV anterior wall (–D). Anterior LV wall thickness was greater in ADC group than the PBS group (1013 ± 179 vs. 616 ± 82 μm, p < 0.05) (Fig. 6) and capillary density in the ADC group was significantly higher (120 ±13 vs. 76 ± 12/mm2, p < 0.01) (Fig. 6B).

Representative histological sections of PBS control (A, B) and ADCs injection (C, D) myocardial wall 6 weeks after surgery stained with H&E and Masson Trichrome, indicating collagen (blue), fibrous cells (red), and nuclei (black). Scale bar: 1000 μm. LV, left ventricular cavity; RV, right ventricular cavity.

The left ventricular myocardial wall thickness (A) and capillaries density (B) of ADCs injection group compared to that of PBS control group at 6 weeks following surgery.
Tracking and Immunohistochemistry of the Injected ADCs
Injected EGFP-positive ADCs were identified in the sections at all studied time points (1, 2, and 6 weeks following ADC injection). The injected ADCs distributed widely in the infarcted anterior wall and the border zone between infarcted and healthy myocardium. At 6 weeks immunohistochemistry revealed the EGFP colocalized with vWF, α-SMA, or cTnI (Fig. 7). Increased expression of two potent growth factors, bFGF and VEGF, was documented via immunohistochemistry of the infarcted ventricular wall transplanted with ADCs by 2 weeks after injection (Fig. 5A Fig. 8) versus PBS controls.

At 6 weeks immunohistochemical staining with vWF (upper), α-SMA (middle), and cTnI (lower). Green: EGFP, red: each immunohistochemical staining, blue: nuclei staining. Scale bar: 20 μm.

At 2 weeks immunohistochemical staining with bFGF (upper) and VEGF (lower). Green: EGFP, red: growth factor staining, blue: nuclei staining. Scale bar: 50 μm.
Discussion
The human amnion contains epiblast-like pluripotent cells, mesenchymal stromal cells, and lineage-committed progenitor cells (19). In the present study we demonstrated that rat naive ADCs represent a heterogeneous cell population including mesenchymal stromal cells and embryonic stem cell marker-positive cells. Flow cytometric analysis and immunofluorescent staining data also identified SSEA-1 and Oct-4 double positive cells, indicating that rat amnion contains pluripotent cells similar to the human amnion (12). Studies have shown that the rat amniotic epithelial cells differentiate into endoderm lineage (hepatocyte) (15,22) and ectoderm lineage (neuronal) cells (10,17). Unlike embryonic stem cells, amniotic epithelial cells (12) and mesenchymal stromal cells (26) do not produce teratomas when transplanted to immunodeficient animals. In the current study the observation period was only 6 weeks; however, we noted no tumor formation. In the present study we found that rat naive ADCs expressed some cardiac lineage specific genes including two key transcription factor genes required for cardiac differentiation, Nkx2.5 and GATA-4. Some cells also expressed mesenchymal fibroblast and endothelial cell marker genes. Like human ADCs, rat ADCs show potential of multipotent differentiation. Previous studies have shown that ADCs differentiate into neuronal (10,17), hair and skin (9), and hepatic cells (15,22). Also, the same as human amnion, rat amnion contains mesenchymal stromal cells (11). Thus, despite anatomical differences between rodent and human placental and amniotic architecture, rat ADCs contain analogous cell populations to the human amnion-derived epithelial (12) and mesenchymal stem cells (5,20).
In this study, naive ADC transplantation into the infarcted myocardium remarkably reduced myocardial scar and prevented myocardial wall thinning. Echocardiographic evaluation revealed that the significant salvage effects from a nonreperfusion ischemia appeared at 2 weeks after ADC injection and the beneficial effect of ADCs on postinfarcted myocardium was maintained at 6 weeks following ADC injection. Several possible mechanisms might explain the functional benefits associated with ADC injection. First, we found that some of the injected EGFP-positive cells colocalized with cardiac troponin-I at 6 weeks, indicating that injected ADCs may differentiate along a cardiomyocyte lineage. Zhao et al. showed freshly isolated human ADCs are positive for some cardiac genes, such as MLC-2a, MLC-2v, cTnT, and cTnI. They also showed a possibility to induce cardiomyocyte differentiation in vitro (28). Okamoto et al. reported that human chorionic plate-derived cells differentiate into cardiomyocytes under co-culture with mouse neonate cardiomyocytes (16). Our in vitro data of Nkx-2.5 and GATA-4 gene expression supports the potential of cardiac differentiation from ADCs. Second, we also observed that some of EGFP-positive cells colocalized with vWF or α-SMA, indicating that injected ADCs can differentiate into vascular endothelial and smooth muscle cells. Alviano et al. showed that human amnion-derived mesenchymal stem cells have greater in vitro expansion potential than bone marrow-derived stromal cells and also have angiogenic potential (1). Finally, the improvement of cardiac function and reduction in scar formation might be mediated by paracrine effects of ADCs. Ventura et al. reported that implantation of hyaluronan mixed esters of butyric and retinoic acid-treated human amnion-derived mesenchymal stem cells into rat postinfarcted myocardium differentiated into cardiomyocyte phenotype and increased capillary density of the infarcted myocardium (27). We found that two major growth factors, bFGF and VEGF (8), were expressed in ADC-injected myocardium. In addition, the capillaries density in ADC group was greater than PBS group at 6 weeks following ADC injection. We speculate that naive ADCs would differentiate into cardiomyocytes, vascular lineage cells (4), stimulate angiogenesis, and prevent residential cardiomyocyte death by paracrine effects (27).
We have demonstrated the beneficial effect of naive myocardial ADC injection following acute infarction without the requirement of cell preparation, including sorting and modification during protracted in vitro culture. Should these cells be applied to clinical setting, expeditious procedure would be expected avoiding time-consuming complicated pretreatment. It is important to note that we have documented beneficial effects in terms of cardiac function and LV remodeling only up to a 6-week period following cell transplantation and additional longer term studies are needed. Although no tumor-like tissues were observed in any of the ADC-treated animals and placenta-derived stem cells have been shown to be more genetically stable than embryonic stem cells (12), the fate of the injected ADCs and functional changes remains to be examined through longer term protocols.
In conclusion, this report demonstrates that unmodified fresh ADC transplantation into acute infarcted myocardium preserved cardiac function and reduced myocardial scar formation in a syngeneic rat model. Our results suggest that the naive ADCs are a potential cell source for cardiac repair and regeneration.
Footnotes
Acknowledgments
Recognition of financial support: Commonwealth of Pennsylvania Department of Health (FY-08), Children's Hospital of Pittsburgh Foundation.
