Abstract
Our objective was to explore the mechanism of cell-assisted adipose transplantation by using freshly isolated human stromal vascular fraction (SVF) cells and to observe the dynamic changes of the graft after transplantation. The SVF was isolated from human liposuction aspirates, and 0.5 ml adipose tissue was mixed with 1 × 106 SVF cells or culture medium then injected to nude mice subcutaneously. At 1, 4, 7, 14, 30, 60, and 90 days after transplantation, samples were harvested for 1) general observation and retention rate; 2) whole-mount stain; 3) H&E stain; 4) immunohistochemical staining for S100, CD68, and CD34; 5) ELISA for VEGF and bFGF; 6) peroxisome proliferator-activated receptor-γ (PPARγ) fluorescence in situ hybridization. The retention rate in the experiment group was markedly higher than that in the control group. Whole-mount stain shows most of the cells in the center of the graft could not survive the ischemia until day 14. Histology showed new vessels on the surface of the graft at 3 days. However, in the control group, fewer newly formed vessels were detected until day 7. In the late stage of transplantation, gradual fibrosis was found in the graft, and the tissue was divided into a grid-like structure. A large number of round neonatal adipocytes with big nuclei in the center were found surrounding the new vessels, which were S100 and CD34 positive and CD68 negative. In the late stage of transplantation, most of the neonatal adipocytes were human PPARγ positive. Moreover, the SVF group showed a higher level of VEGF and bFGF. SVF assisting adipose transplantation could increase the retention rate of the graft through promoting adipose tissue regeneration via secretion of growth factors, promotion of angiogenesis, and increasing the density of mesenchymal stem cells.
Introduction
There have been great controversies as to what manner adipose cells survive after transplantation of autologous adipose tissue. In 2009, Yoshimura reported the clinical application of stromal vascular fraction (SVF) cell-assisted adipose transplantation, indicating that SVF cells could remarkably raise the retention rate for transplanted adipose tissue (35). One of our previous studies also proved that SVF cells from rabbits could markedly raise the retention rate of the autogenous adipose tissue transplantation (9). Numerous animal and clinic studies proved that SVF-assisted transplantation could effectively raise the retention rate of grafts (28). However, the survival mode of adipose tissue and whether tissue regeneration was involved during the transplantation was not well studied. Therefore, we hypothesize that SVF cells could promote the angiogenesis of the graft by secreting angiogenic factors and that some subpopulations of SVF may differentiate into adipocytes, both of which could promote adipose tissue regeneration after transplantation. The goal of the present study is to clarify the regulation mechanism of the SVF in adipose tissue regeneration after SVF-assisted fat transplantation. Therefore, we focused on the histological changing of the graft after grafting as well as the cell morphology and phenotype changes within the fat graft.
Materials and Methods
Human SVF Cell Harvest
To obtain SVF cells, human adipose tissue collection and cell harvests were approved by the Southern Medical University Institutional Review Board (IRB approval No. 15402). Abdominal human liposuction aspirates were obtained from 10 female patients aged from 32 to 35 (Plastic Surgery Department of Nanfang Hospital, Southern Medical University). Each patient provided written informed consent. The body mass indexes (BMIs) were within the normal range, and the following were exclusion criteria: history of serious illness in vital organs (as cardiovascular system, liver, lungs, nervous system, kidneys, and others) or history of autoimmune disease (immune abnormality or immune compromise), such as rheumatism, erythematous lupus, infectious disease, and others.
Briefly, the lipid part of fat aspirate was rinsed with sterile phosphate-buffered saline (PBS) (HyClone, Logan, UT, USA) to remove blood, saline, and local anesthetic. Samples were centrifuged (Heraeus, Hanau, Germany) within radius 13 cm, at 1,000 rpm for 3 min to remove lower lavage fluid and upper grease, taking the middle of the fat particles 10 ml or so, 6 ml of which was shaken on rocker with 0.1% type I collagenase (Sigma, St. Louis, MO, USA) at 37°C for 30 min. During that time, shaking and blending were done repeatedly two to three times. Neutralization was achieved by placing fractions in complete medium [high glucose Dulbecco's modified Eagle's medium (HG DMEM; HyClone) + 10% fetal calf serum (Hyclone) + 1% penicillin–streptomycin (Gibco, Gaithersburg, MD, USA)], filtration through a 200-mesh sieve (Millipore, Billerica, MA, USA), centrifugation within radius 13 cm, at 2,590 rpm, for 5 min. Lipid droplets and cell impurities were expelled from the supernatant. A complete culture mixed suspension of 1 ml containing the pellet was added to six times the volume of erythrocyte lysis buffer (Sigma), which was then mixed and blended, incubated at room temperature for 6 min, centrifuged within radius 13 cm at 2,590 rpm for 5 min, supernatant discarded, and again resuspended in DMEM for precipitation. SVF concentrations were saved for assisting fat transplantation.
Cell Yields
The cell solution was centrifuged, the supernatant was discarded, and the cells were resuspended in the complete culture medium. The total cell number was counted by a hemocytometer (Qiujing, Zhejiang, China). We counted the cells to ensure that 1 × 106/ml SVF cells could be separated and obtained from each gram of fat tissue. Based on the total cell number, the yield of cells per gram of fat tissue was calculated. Approximately 45 ml adipose tissue (with 0.5 ml being equivalent to 470 mg wet weight) was stored at 4°C for transplantation. For the control group, the fat sources of the same patients were used for pairwise comparison. We used flow cytometry (LSR II; BD Biosciences, San Jose, CA, USA) to analyze the specific surface antigens of cells including cluster of differentiation 34 (CD34), CD31, CD45, and CD14 (2 ml each; BD Biosciences) to determine if there were any marked differences in SVF cellular components between samples.
Description of Experimental Groups
Two different treatment groups were investigated for seven time points: S group: 1 × 106/ml SVF cells were blended with 0.5 ml of fat; F group: 0.3 ml complete DMEM was uniformly mixed with 0.5 ml of fat particles (negative control group). Six nude (BALB/c-nu/nu; Experimental Animal Center, Southern Medical University, Nanfang, China) mice were examined at each time point.
Surgical Procedure
Following an IACUC-approved protocol at the Southern Medical University (Animal Use Statement No. 2011–040), 42 BALB/c-nu/nu mice of health specific pathogen free (SPF) level were utilized (provided by Experimental Animal Center). Mice weighed 15–18 g at age 4–6 weeks, regardless of gender. At present, there is no evidence to show that different sexes result in different retention rates after adipose tissue transplantation in nude mice. Therefore, in this experiment, we did not take the sex of nude mice into consideration. According to the principle of randomization, we allocated the mice into groups. This method might have potential influence, which we will strive to exclude in future studies. In brief, the back areas were disinfected with three alternate scrubs of Betadine (Likang, Shanghai, China) and 70% ethanol and then draped. Only strict aseptic techniques and sterile instruments were used, and the surgeon wore sterile gowns, masks, and head covers. All surgical tools were sterilized prior to surgery. No. 16 syringe needle (Shifeng, Chengdu, China) was used for 0.5 ml of fat particles, which were injected into the two recipient sites of the back areas of 42 experimental nude mice subcutaneously. After operation, all of the mice were cared for at the animal center (SPF level) of Southern Medical University.
General Observation, Wet Weight, and Volume Measurement
The wet weight and general observation, including the volume size, texture, color, vascularity, and fibrous capsule formation of the graft, were analyzed (n = 6) at 1, 4, 7, 14, 30, 60, and 90 days. Wet weights were measured immediately with electronic balance, and their volumes were measured with a displacement method as soon as the grafts were harvested.
Histological Preparation and Staining
Harvested adipose grafts from all time points were immediately placed in 4% formalin (Yulu, Jiangxi, China). After fixation, the grafts were embedded in paraffin and sectioned at a thickness of 4 μm using a Cryostat (Leica CM1900, Wetzlar, Germany). Hematoxylin & eosin (H&E; Yulu) staining was performed for histological assessment. Immunohistochemical staining was used to detect different cell types in the tissue. Briefly, tissue samples were zinc fixed (Zinc Fixative; BD Biosciences) and paraffin embedded. We prepared 4-μm-thick sections and performed immunostaining using the following primary antibodies: mouse anti-human CD34 (1:400 dilution; Santa Cruz Co, Santa Cruz, CA, USA), mouse antihuman CD68, and mouse anti-human S100 (1:400 dilution; Santa Cruz Co). Endogenous peroxidase activity was inhibited using 1% hydrogen peroxide (Rongbai, Shanghai, China) and incubated with primary antibodies. Following the incubation, the sections were then incubated with an EnVision complex (Dako, Carpinteria, CA, USA) and secondary antibody (rabbit anti-mouse; Gibco). Color was developed by 3,3′-diaminobenzidine (DAB; Sigma).
Immunohistochemical Staining Software Was Used for Analysis of Survival Rate of Nascent Fat Cells, Stem Cells, and Expression Level of Macrophages
S100, CD34, and CD68-positive expression of nascent adipocytes was viewed with a biological microscope for photographing. The photos were analyzed by means of professional image analysis software IPP6.0 (Media Cybernetics Co, Rockville, MD, USA). The data from the items to be analyzed [density (mean)] were statistically analyzed.
Whole-Mount Staining
Visualization of grafted fat tissue was performed using the procedure of Eto et al. (8). Accordingly, fresh tissue samples from day 14 and day 90 were cut into 0.5- to 1-mm pieces and incubated with the following reagents for 60 min: boron-dipyrromethene (BODIPY) 558/568-conjugated phalloidin (1:200; Molecular Probes, Eugene, OR, USA) to stain adipocytes, Alexa Fluor 488-conjugated Griffonia simplicifolia isolectin B subunit 4 (GS-IB4; 1:200; Molecular Probes) to stain endothelial cells, Alexa Fluor 488-conjugated anti-human CD34 (1:100; Biolegend, San Diego, CA, USA), and Hoechst 33342 (20 μg/ml; Sigma) to stain nuclei. The samples were then washed and observed directly with a confocal microscope system (Leica TCS SP2). Eight to ten images were acquired at 3-μm intervals and used for reconstructing three-dimensional images.
Enzyme-Linked Immunosorbent Assay (ELISA)
Adipose tissue specimens were homogenized in lysis buffer (PanEra Bioscience, Guangzhou, China) according to the manufacturer's instructions and centrifuged at 1,000 × g for 15 min at 4°C. The aqueous layer was collected and assessed for vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) using an ELISA kit (BIOO Scientific, Austin, TX, USA) according to the manufacturer's instructions. After washing, the plates were incubated with biotin-conjugated anti-cytokine primary antibodies followed by washes and incubation with horseradish peroxidase-conjugated streptavidin. The color was developed using the enzymatic substrate o-phenylenediamine, and the optical density values of absorbance were read on a microplate reader (Molecular Devices, Sunnyvale, CA, USA) at 450 nm.
Peroxisome Proliferator-Activated Receptors-γ Fluorescence In Situ Hybridization
Fluorescence in situ hybridization (FISH) was performed according to the manufacturer's instructions (GP Medical, Beijing, China). In brief, paraffin sections were dewaxed. Sections were digested by proteinase K (Sigma) for 10 min, washed by Rinse A for 5 min twice, dehydrated by precooled 70%, 85%, and 100% alcohol, respectively, each for 5 min. Sections were soaked in preheated 73°C denaturing solution, denaturing at 73°C for 5–8 min. Sections were dehydrated by precooled 70%, 85%, and 100% alcohol. Probe Cot-1 DNA and hybridization solution (GP Medical) were mixed, denaturing at 73°C for 5–8 min, and then placed in a water bath at 37°C for prerenaturation for 30–60 min. Hybridization mixture was added dropwise onto the sections, for hybridization at 37°C overnight. Sections were then washed with preheated Rinse B for 5 min, three times, with Rinse C for 5 min, twice, and dehydrated by precooled 70%, 85%, and 100% alcohol. Then sections were restained by 4′,6-diamidino-2-phenylindole (DAPI) staining (Golden Bodhisattva Medical Corp.) solution and then sealed by antifluorescence decay sealed tablet (Golden Bodhisattva Medical Corp.) for observation under a fluorescent microscope (Olympus Bx51, Tokyo, Japan).
Statistical Analysis
All data were expressed by mean and standard deviations, and statistical software SPSS 13.0 (IBM, Armonk, NY, USA) was used for data processing. An independent samples t test was used between the two groups. Analysis of covariance was used for different time points in one group. Analysis of covariance is a general linear model, which blends analysis of variance and regression. Tukey's test was used as the post hoc test to detect the significant differences between the samples in the same group after the analysis of covariance, and p < 0.05 was considered to be statistically significant.
Results
Cell Components of SVF and Observation Findings on Fat Graft
Flow cytometry suggested that the SVF cells contained 30.2 ± 4.4% CD45+ cells (blood-derived cells), 27.4 ± 3.2% adipose stem cells (ASCs) (CD45–/CD31–/CD34+), and 6.7 ± 2.1% monocytes (CD45+/CD14+) (n = 10; data not shown).
General observation indicated there was no remarkable difference between the sizes of the two groups of grafts at 7 days in the early postoperative stage, yet at 14 days, the graft volume of the SVF group was larger than that of the control group, with obvious blood vessels, tuff texture, normal gloss, and light coated fibers. The texture in the control group was hard, grayish, and lackluster with obvious fibrous encapsulation (Fig. 1).
General observational findings at different time points after transplantation. Volume change is not remarkable at 1–7 days in the two groups of grafts; at 14–90 days, the graft volume in S group is obviously larger than that in control (F) group and soft in texture with luster and light coated fibers and more remarkable vascularization than that in the control group. In the control group, the texture of the graft is hard, gray with no luster, with remarkable fibrous encapsulation (S group, freshly isolated auto-SVF group; F group, complete culture control group).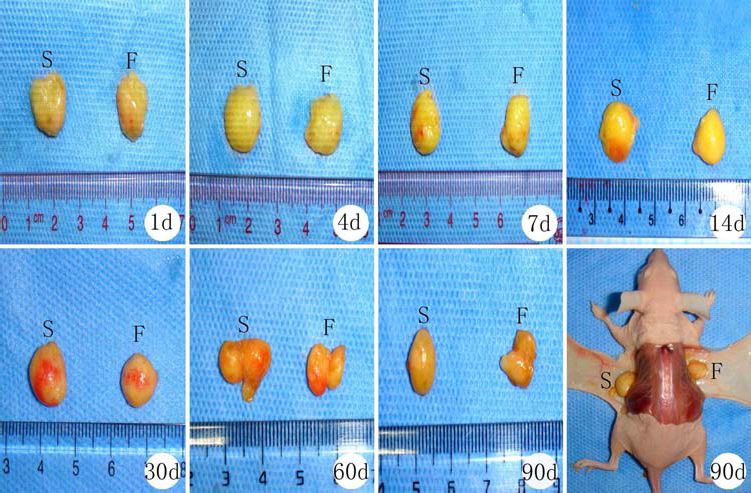
Graft Retention and Survival Determination
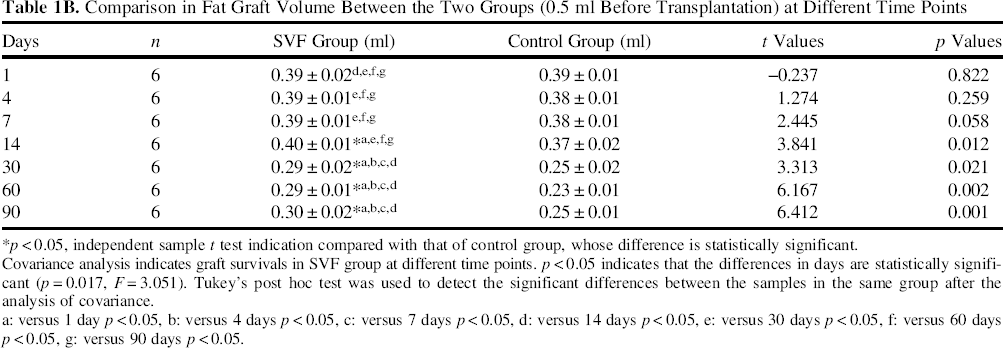
The retention rates of transplanted adipose tissue at different time points were calculated, respectively, in accordance with the wet weight of 470 mg before transplantation. The graft retention curves of the two groups at different time points indicated that the tendency of posttransplantation grafts assisted by SVF was basically the same as that of the simple fat group. However, the graft retention rate of the SVF group was slightly higher than that of control group at 14, 30, 60, and 90 days (p < 0.05), but the difference was not significant at the other time points (p > 0.05) (Fig. 2). Retention rates of each group at different time points were performed with covariance analysis, indicating that the differences were statistically significant. Then Tukey's test was used as the post hoc test to detect the significant difference between the different time points in the same group after the analysis of covariance. The results suggested that the retention rate of both groups remained stable from day 1 to day 7, then increased slightly from day 7 to day 14, and decreased sharply from day 14 to day 30, and finally remained stable again from day 30 to day 90 (Table 1A). For verification, we also calculated the retention rate of the graft by using the tissue volume. The volume change remained insignificant from day 1 to day 14 in both groups, then decreased sharply from day 14 to day 30 and became steady until day 90, which was quite similar to the retention rate of graft wet weight. Moreover, the graft volume of the SVF group was significantly higher than that of control group at day 14 and the subsequent time points (Table 1B).
Retention rate curve for the two groups of fat grafts at different time points postoperatively. The tendencies for the two groups are basically the same. The graft retention rate of SVF group was slightly higher than that of control group (FAT) at 14, 30, 60, and 90 days (p < 0.05) but not significantly different at the remaining time points. The retention rate of both groups remained stable from day 1 to day 7 then increased slightly from day 7 to day 14 and decreased sharply from day 14 to day 30, finally remained stable again from day 30 to day 90. Comparison in Fat Graft Retention Rate Between the Two Groups According to the Wet Weight (470 mg) Before Transplantation at Different Time Points x±S (%) Covariance analysis indicates graft survivals in the SVF group at different time points. p < 0.05 indicates that the differences in days are statistically significant (p = 0.017, F = 3.051). Tukey's post hoc test was used to detect the significant differences between the samples in the same group after the analysis of covariance. p < 0.05, independent sample t test indication compared with that of control group, whose difference is statistically significant. versus 1 day p < 0.05, versus 4 days p < 0.05, versus 7 days p < 0.05, versus 14 days p < 0.05, versus 30 days p < 0.05, versus 60 days p < 0.05, versus 90 days p < 0.05. Comparison in Fat Graft Volume Between the Two Groups (0.5 ml Before Transplantation) at Different Time Points Covariance analysis indicates graft survivals in SVF group at different time points. p < 0.05 indicates that the differences in days are statistically significant (p = 0.017, F = 3.051). Tukey's post hoc test was used to detect the significant differences between the samples in the same group after the analysis of covariance. p < 0.05, independent sample t test indication compared with that of control group, whose difference is statistically significant. versus 1 day p < 0.05 versus 4 days p < 0.05 versus 7 days p < 0.05 versus 14 days p < 0.05 versus 30 days p < 0.05 versus 60 days p < 0.05 versus 90 days p < 0.05.

To further investigate the survival of the grafts, we used whole-mount stain to identify different cell types that survived within the graft. Results showed that almost all of the mature adipocytes in the central area of the graft died at 14 days after transplantation in both groups (Fig. 3). The sharp decrease of the graft weight may have been due to necrosis. Only a few mature adipocytes near the edge survived. Endothelial cells (Fig. 4, red arrow) and CD34-positive cells (mainly contain precursor cells and pericytes) also survived in this area (Fig. 4, white arrow). At day 90, well-organized adipose tissue appeared in both groups, and the number of CD34-positive cell decreased slightly over time.
Whole-mount stain of BODIPY 558/568-conjugated phalloidin to stain adipocytes. Whole-mount stain showed that almost all the adipocytes (BODIPY 558/568-conjugated phalloidin; yellow) including mature adipocytes and SVF cells in the central area of the graft die at 14 days after transplantation in both groups (scale bar: 50 μm). Whole-mount stain identified that only a few mature adipocytes near the edge survived. Endothelial cells (red arrows; isolectin) and cluster of differentiation 34 (CD34)-positive cells (green; mainly contain precursor cells and pericytes) (white arrow) also survived in this area. At day 90, well-organized adipose tissue appeared in both groups, and the number of CD34-positive cells decreased slightly over time (scale bar: 50 μm). Hoechst 33342 nuclear staining is in blue.

H&E-Stained Section Analysis for Fat Grafts
Histological observation demonstrated coenocytes, blood cells, and lymphocytes infiltrated into the graft with small numbers of vacuoles appearing at day 1 after transplantation. On day 4, the growing of new vessels around the tissue capsule could be detected in the SVF group (control group on day 7), and the vessels grew into the graft from the edge to the center. The vacuoles and cystlike changes increased over time (Fig. 5, day 14). However, the number of vacuoles was smaller than that in control group. The gaps between adipocytes were broadened with interstitial tissue that grew in on day 7 in both groups, but new adipocytes with big round nuclei appeared around blood vessels within the interstitial tissue in the SVF group (Fig. 6, day 7). These new adipocytes grew bigger over time and presented with deviation of nuclei and then unilocular signet ring-like cells (Fig. 6, day 14). Though these new adipocytes also appeared in the control group, the number of cells was much smaller than that of the SVF group.
The overall view of H&E staining at multiple time points after operation. H&E staining shows that at 40 days after transplantation, in the SVF group, graft adipocyte gaps are broadened with remarkable ingrowth of interstitial tissue; there occur less vacuoles and cyst-like changes. At 30 days, in some parts of SVF group arise nascent adipose tissue areas with complete structure composed by differentiated and mature adipocytes (indicated by yellow arrows), and in another part, adipocytes of original adipose tissue areas are divided by interstitial tissue into adipocytes of various sizes, with obvious interstitial tissue reaction. In the control group, no obvious nascent adipose tissue area was seen, interstitial tissue gradually moving to fibrosis and vacuoles being remarkable. At 90 days, in the SVF group, adipose tissue structures characterized by close-to-normal adipose lobular tissue occur. In smaller numbers, the original adipocytes shrink and move toward fibrosis, which is lighter in extent compared with control group. Obvious nascent adipose tissue is not seen in the control group area; adipocytes are divided by abundant fibrous connective tissue into grid structure, which is mainly made up of unilocular adipocytes. There arise large numbers of vacuoles and cyst-like changes (S, experimental group; F, control group; d, days, scale bar: 200 μm). Dynamic changes of neonatal adipose tissue after transplantation. H&E staining shows that at 7 days after transplantation, in the SVF group, small round cells with nucleus in the middle could be detected around the neovascularized graft, which have abundant cytoplasm inside and are interspaced by microfilament. At 14 days, small cells are gradually increased and enlarged, with the nucleus moving from middle to deviation. There occur adipocytes as round and multivesicular ring-like cells with nucleus pushed to the periphery and others at various differentiation stages. At 30 days of late stage, small cells at various differentiation stages are continuously increasing and maturing. At 90 days, small cells are gradually decreasing, and the most common cell type was mature adipocytes. In the control group, the number of small cells is smaller, and they are maturing more slowly than the SVF group. At 90 days, part of adipocytes is fused to form fat bullous and separated by fibrous connective tissue (yellow arrow indicates nascent adipocytes) (d, days, scale bar: 50 μm).
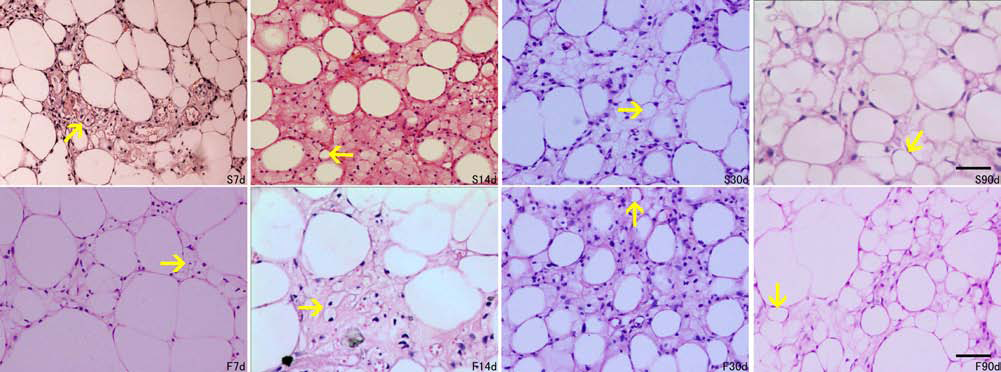
On days 14 to 30, fat lobule-like nascent adipose tissue parts composed of differentiated and mature nascent adipocytes could be found in the SVF group (Fig. 5, day 30). Meanwhile, adipocytes at different stages of differentiation with varying sizes arose. Interestingly, during this period, the histological and morphological changes in the control group were obviously lagging behind that in the SVF group (Fig. 6, day 14). On days 30 to 60, the number of nascent adipocytes at various stages of differentiation reached a peak. Most were unilocular signet ring-like cells that tended to be mature, while there were occasionally multilocular adipocytes (Fig. 6, day 30). The nascent adipose tissue area increased within the complete structure.
On days 60 to 90 after transplantation, most areas presented with a nascent adipose tissue makeup that was characterized by lobular structures. A small part of the adipocytes of the original fat area shrunk, undergoing fibrosis. Adipocytes in the control group were divided by abundant fibrous connective tissue into a grid structure, which were mainly unilocular adipocytes, without nascent adipose tissue. There arose large numbers of vacuoles and cyst-like changes (Figs. 5 and 6, day 90).
Immunohistochemical Staining Analysis
The cell membrane S100 protein expression of mature adipocytes was seen in the nuclei of nascent adipocytes at various differentiation states. The expression of S100 in mature adipocytes was high, but the expression of primitive mesenchymal cells and nascent adipocytes was low during the early stages of transplantation. The expression in nascent adipocytes increased through their maturation. In our study, after 3 months, the expression of S100 in nascent adipocytes increased, further suggesting that the new cells were adipocytes.
At the early stage of transplantation, mild infiltration of macrophages could be detected near the capsule of the graft. At the late stage, the expression of CD68 increased, and these positive cells were mainly located in the gaps between adipocytes; however, the expression in nascent adipocytes was negative, suggesting the small cells that appeared in the early stage were not macrophages but nascent adipocytes (Fig. 7).
S100 immunohistochemical staining. At 7 days of the early stage of transplantation, the nucleus and membrane of small cells and mature adipocytes are stained brown with S100 (A) (indicated by yellow arrow). H&E was used as a counterstain. Primitive mesenchymal cells are weakly positive. Nascent adipocytes at various differentiation stages occurring during the early and late stages of transplantation stain strongly. CD68 immunohistochemical staining (B): cytoplasm is brown stained, while the nucleus is unstained (indicated by red arrow). At 7 days of the early stages of transplantation, macrophage expression is mainly concentrated on the envelope and in small amounts. At the late stages, positive staining gradually increased, appearing in the adipocyte interstitial substance, while most areas of the adipocytes are negative (A: S100, B: CD68, d, days; scale bar: 50 μm).
CD34 is considered a mesenchymal stem cell and immature cell marker and is also expressed by cells with differentiation ability. Our results indicated that CD34-positive short-spindle cells could be detected in the grafts of the SVF group at the early stage of transplantation (Fig. 8). Part of the small round nascent adipocytes (nucleus in the middle) at day 14 were also CD34 positive, and then more nascent adipocytes in various differentiation states expressed CD34 at day 30. Finally, the expression of CD34 decreased at day 60. The dynamic changes of CD34 expression suggested that mesenchymal stem cells derived from grafts, and the SVF could promote regeneration of the transplanted adipose tissue.
Immunohistochemical staining of CD34. At 7 days after transplantation, CD34-positive spindle cells in adipose tissue are evident. At 14 days, a portion of the small round neonatal adipocytes with a central nucleus is CD34 positive. At 30 days, neonatal adipocytes at various differentiation states are CD34 positive (yellow arrow) (d, days; scale bar: 50 μm).
Immunohistochemical Staining Software Was Used for Analyzing Nascent Fat Cells, Stem Cells, and Macrophages
We analyzed S100-positive nascent adipocytes by means of immunohistochemical software. By comparing the percentage of nascent adipocytes within the graft between the two groups at 7, 14, 30, 60, and 90 days, it was found that the percentage of nascent adipocytes increased gradually during the early stages then reached a peak at 60 days and decreased gradually until day 90. Moreover, the percentage of the SVF group was higher than that in the control group, and the differences between the two groups at 14, 30, 60, and 90 days were statistically significant (p < 0.05) (Fig. 9A). Covariance analysis for nascent adipocyte percentages in the SVF group at different time points indicated that there were significant differences among the seven time points (p < 0.05) (Table 2). In addition, the analysis of CD34 and CD68-positive cells suggested that the percentage of CD34-positive cells was higher at the early stages and lower at the late stages. It was much higher in the SVF group than in the control group (p < 0.05) (Fig. 9B). However, the percentage of CD68-positive cells was lower at the early stages and higher at the late stages and was higher in the control group (p < 0.05) (Fig. 9C).
Cell number, CD34, and CD68 changes over time in the grafts. (A) Early stage nascent adipocytes are increased and reach a peak at 90 days, which is higher in the SVF group than in the control group. The differences between the groups are statistically significant at 14, 30, 60, and 90 days (p < 0.05). (B) The expression of CD34 decreases at 60 days. It is higher in the SVF group than in the control group, and the difference between the two is statistically significant (p < 0.05). (C) The expression of CD68 increases at 60 days. It is lower in the SVF group than in the control group, and the difference between the two is statistically significant (p < 0.05). Neonatal Fat Cell Survival in the Two Groups at Different Time Points After Transplantation (%) Covariance analysis indicates graft survivals in SVF group at different time points. p < 0.05 indicates that the differences in days are statistically significant (p = 0.000, F = 10.912). Tukey's post hoc test was used to detect the significant differences between the samples in the same group after the analysis of covariance. p < 0.05, independent sample t test indication compared with that of control group, whose difference is statistically significant. versus 7 days p < 0.05 versus 14 days p < 0.05 versus 30 days p < 0.05 versus 60 days p < 0.05 versus 90 days p < 0.05.

Expression of VEGF and HGF
The expression of VEGF in the SVF group was markedly increased compared with that in the control group, which is higher at the early stages and achieves its peak value at 7 days after transplantation and decreases at the late stages. The expression level was significantly higher in the SVF group than the control group at days 4–30; however, at the rest of the time points, there was not a statistical difference (Fig. 10A). Interestingly, the trend of bFGF was very similar to that of VEGF in both the SVF group and control group (Fig. 10B).
Expression level of VEGF and bFGF by ELISA. The expression of both VEGF and bFGF in the SVF group is greatly increased compared with the control group, which is higher at the early stages and achieves its peak value at 7 days after transplantation and decreases at the later stages. There is a significant difference between the two groups in the expression levels at days 4, 7, 14, and 30 (p < 0.05).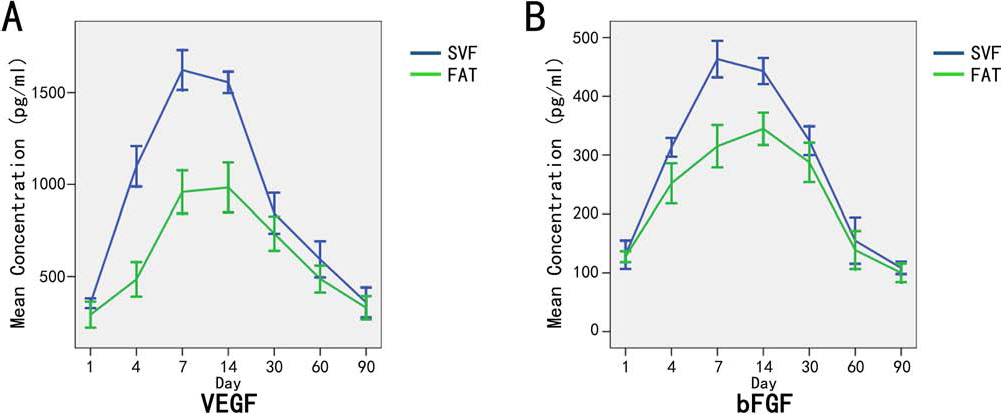
PPARγ Fluorescence In Situ Hybridization
We chose human PPARγ genes for FISH to confirm the source of nascent adipocytes. Bright green fluorescent nuclei and cytoplasm showing significant color were indicative of PPARγ-positive expression. In the interstitial adipose tissue, scattered spot-film-shaped bright green fluorescent expression was observed. The nuclei and cytoplasm of nascent adipocytes were PPARγ positive (Fig. 11), which confirmed that the nascent adipocytes were derived from human tissue. These cells were at a differentiation state. Besides, there were still a few nascent adipocytes that were PPARγ negative, indicating that these nascent adipocytes might derive from hematogenous interstitial cells of host mice.
Human PPARγ fluorescence staining. (A) Intranucleus and cytoplasm show bright green spot-shaped PPARγ-positive fluorescence (red arrow). (B) DAPI restained nuclei. (C) H&E-stained section from the same area. Yellow arrow indicates nascent adipocytes. (D) Merged image of (A) and (B). Red arrows show PPARγ-positive nascent adipocytes, confirming that these were derived from the transplanted human cells. The yellow arrow showed a PPARγ-negative nascent adipocyte, indicating that a small proportion of the nascent adipocytes might differentiate from hematogenous interstitial cells of mice [scale bar: 25 μm (A, B, D), 20 μm (C)].
Discussion
Currently, there have been two theories on the survival mechanism for free transplanted adipose tissue. The host replacement theory states that host macrophages were once proposed to acquire lipid material and eventually replace all adipose tissue of the graft that had disappeared. The cell survival theory states that transplanted living cells partly survive and remain alive for a long time after grafting (4,32).
Although many researchers have suggested that increases in the ratio of SVF/ASCs within the graft will lead to a higher retention rate in adipose transplantation, the survival mechanism of free fat transplantation remains to be identified (3,10,12,15). As each adipocyte needs to connect with a capillary (8), there is a close link between the adipose tissue formation and angiogenesis. The major mechanism of SVF that contributes to angiogenesis is the secretion of angiogenic factors like VEGF and hepatocyte growth factor (HGF) (17). It has been reported that ASCs could proliferate, migrate, and then differentiate into blood endothelial cells (BECs) due to the tissue loss caused by necrosis and apoptosis during transplantation (35), and the increasing SVF cell ratio of the graft would lead to a significant protective effect in reducing necrosis and increasing vasculogenesis (22). In addition, BECs within the SVF also play a key role in angiogenesis and become incorporated into the new vessels through dynamic reassemblies (14).
However, the crux of the matter is whether adipose tissue can survive the ischemia before restoring blood supply and what kind of cells can survive the ischemia. Besides the angiogenic mechanism of SVFs, there might be other roles of SVFs that promote the retention of transplanted adipose tissue. In this study, whole-mount staining showed that the most mature adipocytes in the center of the graft died during the early stages of transplantation; however, the ones located on the edge survived. Moreover, another of our studies showed, at day 7 after transplantation, most SVF cells located at the edge of the graft still survived, though all the SVF and mature adipocytes in the central area of the graft died (unpublished data). The research by Eto et al. also suggested that the outer layer of the graft contained mature adipocytes and SVF cells that survived, and this layer may play an important role in the remodeling process (7). However, neither the host replacement theory nor the cell survival theory could perfectly explain these findings.
In our research, the histological examination found that some stromal cells change their morphology from spindle cells to round, multilocular adipocytes, unilocular adipocytes, signet ring-like cells, and finally mature adipocytes. Zeve et al. presumed that adipose tissue derived from ASCs that shared morphology similar to fibroblasts also had multidifferentiation potential (36). Moreover, the immunohistochemical staining of CD34 in our study showed a proportion of the small round nascent adipocytes with nuclei in the middle were CD34 positive during the early stages of transplantation. CD34 is an adhesion protein and is considered a marker of multipotent stem cells, such as stromal stem cells and progenitor cells. The marker is thought to play an important role in the migration and homing process of stem cells (18). The CD34-positive cells in SVF are localized in the blood vessels in adipose tissue, and their staining is different from that of CD31 in that CD34-positive cells are visible in the adventitia of all blood vessels (16). Some data indicate that CD34-positive SVF cells have a higher proliferation rate than the CD34-negative cells (27). Another study showed that the CD34-positive cell population from cultured SVF (but not CD34-negative cells) was able to produce lipid-droplet formation activity under adipogenic culture conditions (18). These findings show that CD34-positive stromal cells have the potential for differentiating into mature adipocytes and may be the key cells in adipose tissue regeneration after transplant.
In addition to the morphological changes of nascent adipocytes in both the SVF group and the control group after transplantation, we detected nascent adipocytes at various differentiation states as early as 14 days after transplantation in the SVF-assisted group. We hold that nascent adipocytes may derive from stromal stem cells. The sources of stromal stem cells may be as follows: 1) Inherent stromal stem cells in the grafts. It was reported that adipose stromal cells could maintain cell activity even under ischemic and hypoxic conditions for 3 days after transplantation and could further promote adipogenesis and angiogenesis (25,26,35). 2) Exogenous stromal stem cells could also be the cell source (8). In a recent report, Hollenback and colleagues demonstrated a novel finding of adipose stromal cells migrating from the engraftment point in the flap across a wound interface into the surrounding native tissue when injected into rat ischemic flaps (13). This finding suggests that regenerative cells may also come from adjacent tissues. 3) Some cells may be derived from the dedifferentiation of mature adipocytes in the body. In a hypoxic environment, mature adipocytes may expel lipid droplets and change their morphology from a round cell into fibroblast-like cells. This is called dedifferentiation of adipocytes (31). In a study by Zhu et al., dedifferentiated adipocytes could better survive the ischemia and hypoxia (37). We hypothesized that at the early stages of transplantation, a portion of the mature adipocytes might survive the ischemia and hypoxia by dedifferentiation, then differentiate to adipocytes again when the local tissue's blood supply is restored. Hausberger suggested that only a small number of the adipocytes in the graft survived after transplantation and then dedifferentiated into a special precursor, which was called preadipocyte (11). Interestingly, this cell could differentiate into mature adipocytes again with proper stimulation.
These three sources of stem cells are all from donor cells. We found that most of the new cells were derived from humans in our study. Besides, a small number of nascent adipocytes were PPARγ negative, indicating that some cells were hematogenous cells from mice; therefore, hematogenous stromal stem cells were also one of the sources of nascent adipocytes.
Macrophages could form foam cells after the phagocytizing of lipid droplets from necrotic adipocytes. However, these foam cells may be mistaken for nascent adipocytes. In order to exclude this possibility, we used immunohistochemical staining of CD68 (a macrophage marker) to identify foam cells from nascent adipocytes. At the early stages of transplantation, the macrophages were mainly located in the gaps between adipocytes, and the number of macrophages was small at first, but then increased over time until the late stages of transplantation. Interestingly, most S100-positive cells were CD68 negative, indicating that most small cells were nascent adipocytes. The regeneration of the graft began from the edge along with the ingrowth of capillaries. Lipid droplets are phagocytized by macrophages, which infiltrated from blood vessels to the necrotic area. That means there should be a peak of macrophage infiltration during the angiogenic process of the graft. However, it is a very slow process and will continue into the stabilization stage for as long as 3–12 months (34). Therefore, we could find a higher ratio of CD68 expression in both groups on day 60 than on day 14. Many studies demonstrated that adipose mesenchymal stem cells hold an immunoregulatory capacity. These cells could inhibit cell proliferation of immune cells. Moreover, they also have an antiinflammatory function, which could reduce the secretion of inflammatory factors, increasing the expression of antiinflammatory factors (5,6,23,33). This may explain why the SVF group revealed a mildly lower percentage expression compared to that in the control group. Besides, in the early stages of transplantation, inflammatory cells such as macrophages could secret matrix metalloproteinases, which could degrade the extracellular matrix and release the stromal stem cells and endothelial cells. This process may also promote adipose tissue regeneration (1,21).
PPARs are a group of nuclear receptor proteins that function as transcription factors regulating the cellular differentiation, adipose tissue development, and lipid metabolism (30). Remarkably, PPARγ is the only transcription factor that is highly expressed in adipose tissue, and it is the key regulator of adipogenic differentiation (29). Therefore, PPARγ is a specific marker of adipogenic differentiation of mesenchymal stem cells (20). The expression of PPAR begins at the early stages of adipogenic differentiation, and the expression level increases with the maturation of adipocytes over time. In our study, we found that most of the nascent adipocytes were human PPARγ positive, which suggests that the nascent adipocytes were differentiated from human stem cells within both the SVF and transplanted adipose tissue. However, some of the nascent adipocytes were PPAR negative, which means they were derived from hematogenous cells.
In summary, we hold that the SVF cells may promote tissue regeneration in SVF-assisted adipose tissue transplantation, and the mechanisms are described below. The stromal stem cells in the SVF may act as preadipocytes and may have been involved in the following adipogenic differentiation. Under the stimulation of the growth factors (VEGF, FGF, etc.) secreted by SVFs (2,24), the adipose stromal stem cells and endothelial cells from the graft migrated to the neovascularized area and formed new adipose tissue and vessels through reassembly (19,34). SVF-assisted fat transplantation has the prospect for extensive application. Some prospective clinical trials achieved inspiring outcomes, but cell therapy based on SVF cells is still at the early stage of preclinical studies. Studies on the microenvironment for SVF transplantation, differentiation mechanisms, regulation mechanisms, and so on will provide new theories and evidence for SVF to be used for clinical application.
Footnotes
Acknowledgments
This work was financially supported by the National Nature Science Foundation of China (81171834, 81071589), Medical Scientific Research Foundation of Guangdong Province (A2013449, A2011453). The authors declare no conflicts of interest.
