Abstract
Interactions between maternal natural killer lymphocytes (NKs) and fetal tissues are important in mediating maternal–fetal tolerance. We therefore investigated the interactions of NKs to placenta-derived multipotent cells (PDMCs) isolated from the term human placenta. PDMCs have similar cell surface marker expression as bone marrow mesenchymal stem cells (BMMSCs) and additionally express human embryonic stem cell markers SSEA-4 and CD-9. Differentiation into the tri-mesodermal lineages of osteoblastic, adipocytic, and chondrogenic phenotypes can be readily achieved under the appropriate conditions. We found that PDMCs are more resistant to NK-mediated lysis than the major histocompatibility complex (MHC) class-I null target cell K562, and can suppress NK secretion of interferon-γ (IFN-γ). Moreover, as third-party cells, PDMCs suppressed the cytotoxic effects of cytokine-stimulated NKs on K562. Pretreatment of PDMCs with IFN-γ, a proinflammatory cytokine, surprisingly enhanced such immunosuppressive effects. Cell–cell contact between NKs and PDMCs is required for suppressive effects, which are partially mediated by slight upregulation of the NK inhibitory receptor killer inhibitory receptor and downregulation of the activating receptor NKp30. Moreover, enhancement of PDMC suppressive effects is also mediated by IFN-γ-induced surface expression of HLA-G—an immunomodulatory nonclassical MHC class I molecule—on PDMCs, as seen by partial reversibility with HLA-G neutralizing antibodies. With its broad immunosuppressive properties, PDMCs may represent a potential cell source for therapeutic use.
Introduction
The immunomodulatory effects found for multipotent mesenchymal stem cells (MSCs) from the adult bone marrow (BM) have enabled the use of these progenitor cells for clinical treatment of immune-related diseases (23,24). While in vivo mechanisms have yet to be clearly defined, much data have accumulated on the in vitro immune suppressive effects and mechanisms, especially against T lymphocytes (4,8,18,20,25,32). Interactions of MSCs with natural killer lymphocytes (NKs) have only more recently been studied. NKs are lymphocytes of the innate immune system, and, while arising from the same progenitor as T and B lymphocytes, NKs do not respond to antigen-specific stimulation but rather mount effector responses to the “missing self,” or the absence of self-major histocompatibility complex (MHC) class I molecules (19,39). While NKs were previously thought to contribute mainly to antiviral and antitumor responses, intense research in recent years has shown that these lymphocytes can be implicated in a wide range of interactions including transplantation (21). However, data on NK interactions with MSCs are still scarce, with reports focused almost exclusively on adult BMMSCs (33).
Because adult-source progenitors require invasive procedures for procurement and cell numbers decrease with age (31), fetal-associated tissues have become attractive sources of progentitor cells. Moreover, in clinical practice, the use of fetal umbilical cord blood hematopoeitic stem cells is associated with a lower incidence of immune-related complications compared to adult BM source (3,40). Multipotent cells have been derived from the placenta (13,17,43) and we have found that these placenta-derived multipotent cells (PDMCs) harbor immunosuppressive effects towards T lymphocytes (7). Throughout fetal development, the innate immune system is particularly important; much evidence has accumulated regarding the key roles of maternal NKs and fetal trophoblastic tissues in mediating maternal–fetal tolerance (28). We therefore investigated the interactions of NKs and PDMCs. We found that PDMCs can suppress a number of NK effector functions via mechanisms including the modulation of NK surface receptors and interferon-γ (IFN-γ)-mediated surface expression of human leukocyte antigen (HLA)-G—an immunomodulatory, nonclassical MHC I molecule—on PDMCs.
Materials and Methods
Cell Culture
Term (38–40 weeks gestation) placentas from healthy donor mothers were obtained with informed consent approved by the institutional review board. Isolation, expansion, and differentiation of PDMCs were performed as previously described (42–44). IFN-γ (200 ng/ml; R&D Systems, Minneapolis, MN) stimulation of PDMCs was performed for 2 days prior to coculture with leukocytes.
NKs were isolated from peripheral blood mononuclear cells with an NK isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany) and cultured as previously described (41). Purity of isolated NKs was assessed by flow cytometric analysis for CD56 positivity, which was greater than 90% (data not shown). For coculture of PDMCs and leukocytes, PDMCs were first seeded on to six-well culture plates (Corning, Lowell, MA) and allowed to attach for 24 h before addition of the suspension cells NKs and K562, a positive control cell line sensitive to NK cytotoxicity.
Immunophenotyping
Flow cytometric analyses of cell surface markers were performed as previously reported (7,43). All antibodies were purchased from Becton Dickinson (San Diego, CA, USA) except for HLA-G from Exbio Antibodies (Prague, CZ) and stage-specific embryonic antigen-4 (SSEA-4) from R&D Systems.
Cytotoxicity/Apoptosis Assay
NK cytotoxicity was evaluated with the annexin V/propidium iodide (AV/PI) staining kit for apoptosis (Roche Applied Science, Mannheim, Germany) as previously reported (41). NKs were stimulated with interleukin-2 (IL-2, 200 ng/ml or as indicated) or IL-15 (200 ng/ml; R&D Systems). Target cells included PDMCs and K562. To assay the effect of PDMCs as third-party cells on NK cytotoxicity, IL-2 and PDMCs were added simultaneously with 5 × 104 NKs to 1 × 104 PDMCs unless otherwise indicated. Transwell studies were performed using 24-well transwell inserts (0.4 μm pores; BD Falcon, San Jose, CA). HLA-G neutralizing antibodies (nAbs, 20 μg/ml) and control antibodies (20 μg/ml) were purchased from Exbio Antibodies.
Quantification of IFN-γ
IL-2-stimulated (200 ng/ml) NKs were cultured at various concentrations (2.5 × 104, 5 × 104, or 1 × 105 cells) without or with PDMCs (1 × 104 cells). Supernatants were collected from the cell cultures for detection of IFN-γ by using a commercially available ELISA kit (R&D Systems) according to the manufacturer's instructions.
Statistical Analysis
Statistical analysis was performed with the statistical SPSS 12.0 software (SPSS Inc., Chicago, IL, USA). Values were expressed as the mean ± SEM. The Student's t-test with Bonferroni correction and/or ANOVA were used to determine statistical significance for comparison of multiple groups. All experiments were performed at least in triplicate with PDMCs from three different donors unless otherwise indicated.
Results
PDMCs isolated from the human term placenta possess similar cell surface markers as BMMSCs, expressing CD105, CD73, and HLA-ABC, but not endothelial markers such as CD31 nor hematopoietic markers such as CD14, CD34, or CD45 (43) (Fig. 1). In addition, PDMCs are positive for the human embryonic stem cell (ESC) marker SSEA-4 (7,43), which has also been found on primitive populations of somatic progenitor cells (30), as well as CD9 (9). Differentiation into the mesodermal lineages of osteblastic, adipocytic, and chondrogenic phenotypes can be readily achieved when cultured in the appropriate conditions (Fig. 1B–D, respectively).
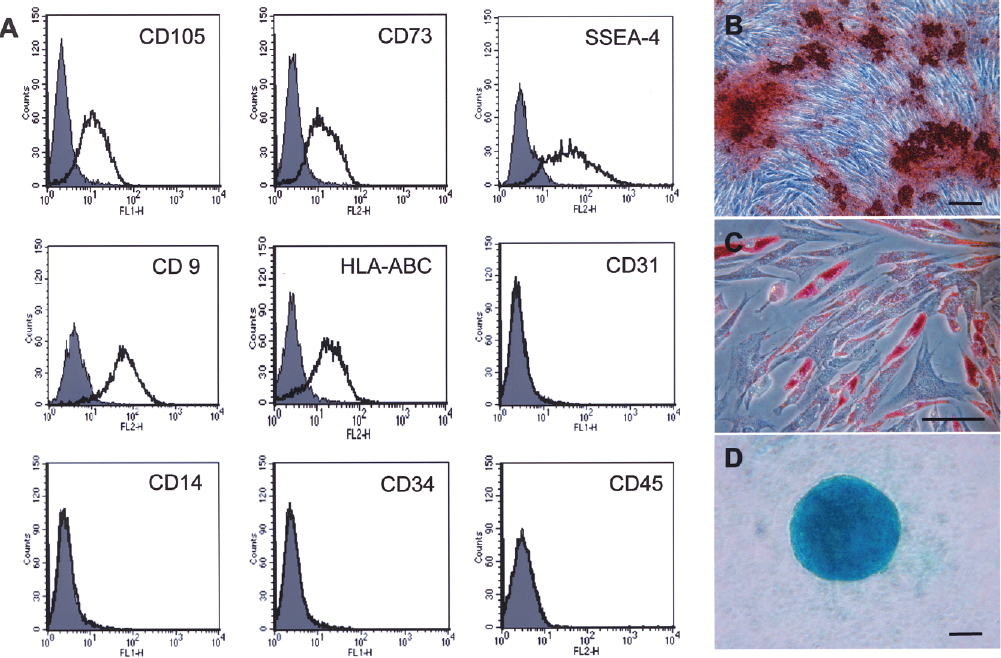
Characterization of placenta-derived multipotent cells (PDMCs). (A) Flow cytometric analysis of PDMCs for cell surface markers, and differentiation of PDMCs to (B) osteogenic (Alizarin Red staining), (C) adipogenic (Oil Red O staining), and (D) chondrogenic (alcian blue staining) phenotypes; phase-contrast microscopy. Scale bar: 200 μm. SSEA-4, stage-specific embryonic antigen-4; HLA, human leukocyte antigen.
We next evaluated the effects of PDMCs on two important NK effector functions. One of the most important functions of NKs is its cytotoxic capacity on cells with non-self MHC I molecules. We found that PDMCs from two different donors can resist lysis from freshly isolated or IL-2-stimulated NKs compared to K562 cells, a MHC I-null cell line (Fig. 2A). However, cytotoxicity is strongly reduced when the target cells are PDMCs, regardless of the dose of IL-2. In addition to cytotoxic capacity, NKs secrete a number of cytokines, the most important being IFN-γ. We found that coculture of NKs with PDMCs can decrease NK secretion of IFN-γ, and this effect remained even as the ratio of NKs to PDMCs is increased (Fig. 2B).

Immunosuppressive effects of PDMCs on natural killer lymphocyte (NK) effector functions of cytolysis and interferon-γ (IFN-γ) secretion. (A) NK [with or without interleukin-2 (IL-2) stimulation, 200 ng/ml] lysis of PDMCs with evaluation by flow cytometric analysis for apoptosis by annexin V/propodium iodine (AV/PI) staining. Three different donors of NKs against two different donors of PDMCs (donor #1 & #2) were used in the experiments. (B) Effects of coculture with PDMCs on NK (with and without IL-2 stimulation, 200 ng/ml) secretion of IFN-γ (x-axis, NK/PDMC ratio); two different donors of NKs against two different donors of PDMCs were used in the experiments. ∗p < 0.005.
In addition to resisting NK cytotoxicity, we investigated whether PDMCs as a third-party cell can suppress NK cytotoxicity towards target cells. We found that when PDMCs are added to the coculture of K562 and cytokine-stimulated NKs, apoptosis of K562 can be reduced, as seen by flow cytometric analysis for AV/PI double-positive apoptotic cells. Apoptosis of K562 cells was 56.47% and 56.06% for IL-2- or IL-15-stimulated NKs, respectively; after coculture with PDMCs, apoptosis of K562 was reduced to 23.67% and 29.17%, respectively (Fig. 3A). NK cytotoxicity towards K562 can be further reduced by PDMCs when PDMCs are pretreated with IFN-γ for 2 days prior to coculture, and this reduces K562 apoptosis to a low of 11.25% and 6.77% for IL-2- and IL-15-stimulated NKs, respectively. The suppressive effects of PDMCs on NK cytoxicity require cell–cell contact, as these effects are lost when the two type of cells are prohibited from direct contact by culturing in Transwell inserts (Fig. 3B).

Immunosuppressive effects of PDMCs as third-party cells on NK cytotoxicity against target cell K562 and role of cell contact. (A) Effect of PDMCs as third-party cells (104 cells), with or without IFN-γ pretreatment, on IL-2- or IL-15-stimulated NK (105 cells) cytotoxicity against target cell K562. (B) Coculture versus transwell culture of PDMCs and IL-2-stimulated NKs (2.5 × 104, 5 × 104, or 1 × 105 cells) and effects on NK cytolysis of K562. Cytotoxic effects were evaluated by flow cytometric analysis for apoptosis of K562 cells by AV/PI staining. Three different donors of NKs against two different donors of PDMCs were used in the experiments. ∗p < 0.05.
To elucidate the mechanisms involved in the suppressive effects of PDMCs on NKs, we analyzed for phenotypic changes on both cell populations because cell–cell contact appears to be required. After coculture with PDMCs, NK surface markers undergo slight modification, with the inhibitory receptor killer inhibitory receptor (KIR) slightly upregulated [Fig. 4, left column, upper and middle graph; mean fluorescence intensity (MFI) in upper right corner] and the activating receptor NKp30 slightly downregulated (Fig. 4, right column, upper and middle graph). IFN-γ stimulation of PDMCs did not further change the surface expression pattern of these two NK receptors (Fig. 4, bottom row). We also assessed for changes in the expression of CD94, an inhibitory receptor, and two other activating receptors, NKG2D and NKp46, but did not see any alterations (data not shown).

Changes in NK surface receptor expression after coculture with PDMCs as evaluated by flow cytometric analysis. KIR, killer inhibitory receptor; mean fluorescence intensity indicated in upper right corner.
Since no additional enhancement of NK surface receptor expression alteration could be seen with coculture of IFN-γ-treated PDMCs, we further investigated for the mechanism behind the enhanced suppressive effects of IFN-γ-treated PDMCs. We specifically assayed for changes in MHC class I molecule expression on PDMCs since NK activation can be triggered by these molecules. We found that after 2 days of IFN-γ stimulation, PDMCs do not express surface HLA-DR, a MHC II molecule, but strongly expressed surface HLA-G, a nonclassical MHC I molecule found on extravillous trophoblasts with immunomodulatory properties (6) (Fig. 5A; percentage of positive cells indicated in upper right corner). This profile is in contrast to that of BMMSCs, in which IFN-γ stimulation results in strong expression of surface HLA-DR but not HLA-G. To see whether IFN-γ-induced expression of surface HLA-G is involved in PDMC-mediated suppression of NK cytotoxicity, we performed cytotoxicity assays with HLA-G nAbs (Fig. 5B). We found that the suppressive effects of PDMCs as third-party cells against NK cytotoxicity are significantly stronger if PDMCs are pretreated with IFN-γ for 2 days than without (-25.1 ± 3.9% for PDMCs only versus −33.7 ± 3.2%, for IFN-γ-treated PDMCs). When nAbs to HLA-G are added, partial reversal of suppressive effects can be seen only with IFN-γ-pretreated PDMCs but not untreated PDMCs; addition of HLA-G nAbs to IL-2-stimulated NKs cocultured with unstimulated PDMCs results in nonsignificant changes compared to addition of control antibodies, whereas addition of HLA-G nAbs to IL-2-stimulated NKs cocultured with IFN-γ-stimulated PDMCs results in significant reduction of suppressive effects (approximately 30% reduction, from −30.8% with control antibodies to −20.3% with HLA-G nAbs). BMMSCs minimally suppressed NK cytotoxicity in our hands, and neither pretreatment with IFN-γ nor the addition of HLA-G nAbs altered effects significantly (data not shown).
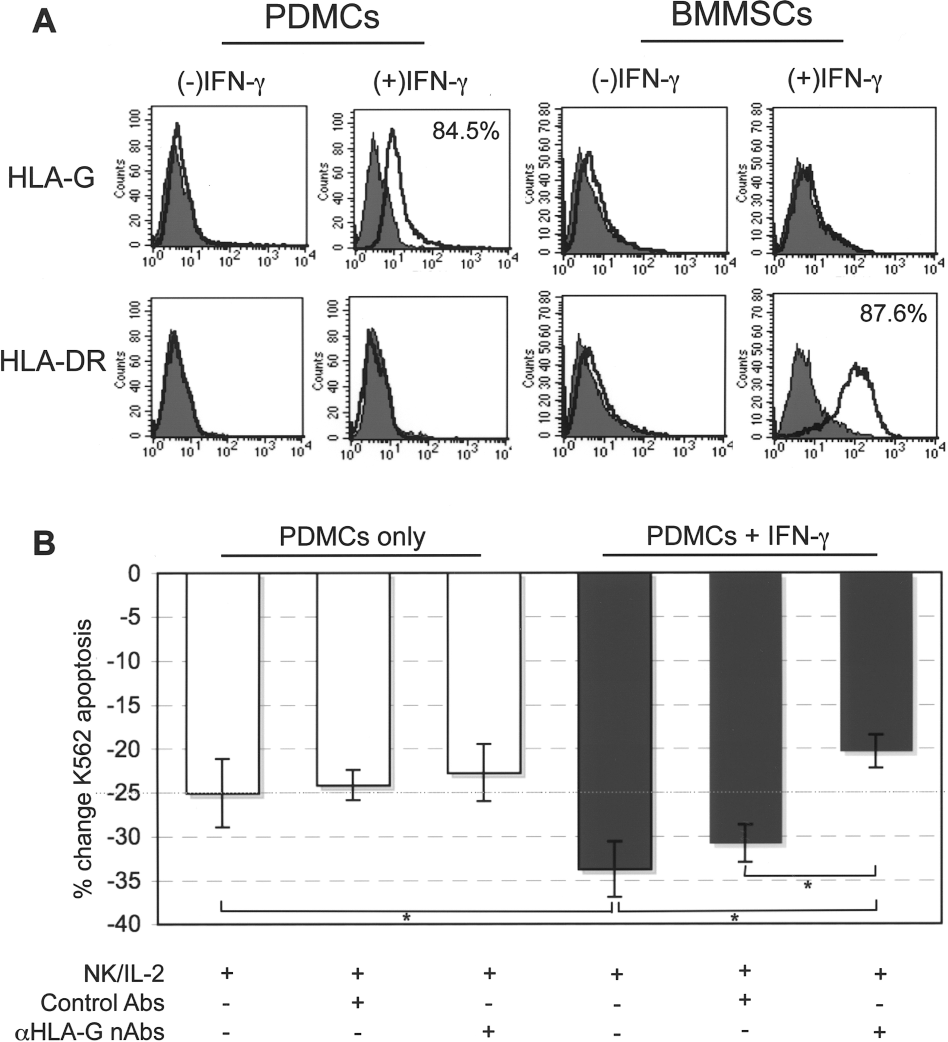
IFN-γ enhancement of immodulation by PDMCs on NKs and role of surface HLA-G. (A) Cell surface expression of HLA-G and HLA-DR on PDMCs and bone marrow mesenchymal stem cells (BMMSCs) with or without 2 days of IFN-γ pretreatment as evaluated by flow cytometric analysis (percentage of positive cells indicated in upper right corner). (B) Effect of neutralizing HLA-G antibodies (nAbs) on IL-2-stimulated NK cytotoxicity towards K562 (flow cytometric analysis of AV/PI staining) with coculture of PDMCs alone or PDMCs pretreated with 2 days of IFN-γ. y-axis, percent decrease in IL-2-stimulated NK cytotoxicity towards K562. Theee different donors of NKs against three different donors of PDMCs were used in the experiments. ∗p < 0.00625 after Bonferroni correction.
Discussion
We have shown that PDMCs possess immunosuppressive effects towards NKs. While PDMCs share many characteristics with adult BMMSCs, the developmental origins are vastly different, and embryonic/fetal stem cells, including fetal MSCs, are relatively immunologically naive (10,15). Moreover, in particular with NK immunomodulation, the placenta has unique interactions with maternal NKs that has important consequences for the successful “engraftment” of the fetal semiallograft (14,27). During pregnancy, decidualization of the uterus occurs and is associated with leukocyte infiltration, the majority of which are NKs (28). Under the influence of locally produced hormones and factors, it also appears that NKs of the uterus are uniquely altered to have phenotypic and functional properties different than peripheral populations of NK cells. The preponderance of NK cells in the uterus and decidua during pregnancy along with a number of other mechanisms point to the importance of the innate immune system in modulating fetomaternal tolerance.
Our data suggest that the placental origin of PDMCs may have a role in the strong immunomodulatory effects towards NKs. Under certain circumstances, trophoblasts have been found to confer immune protection for allogeneic stem cells (12). We found that the expression of surface HLA-G can be induced on PDMCs, but not BMMSCs, after IFN-γ stimulation, and this has a partial role in inhibiting NK cytoxicity. HLA-G is a nonclassical MHC class I molecule that has been found to have immunomodulatory effects. First identified in choriocarcinoma and migratory trophoblasts, HLA-G is thought to confer for the fetus a protective effect against the maternal immune system, including directly suppressing maternal NK cytotoxicity (34,35). HLA-G can exist in several forms, with the best characterized being the complete transmembrane form (HLA-G1)—the predominant in vivo form—and one of the three soluble, truncated forms (HLA-G5 or sHLA-G) (2). Unlike most MHC I molecules, HLA-G has very low polymorphism and its expression in the adult is highly restricted; however, in certain pathologic states, including cancer and inflammatory diseases, expression can be induced (6). While adult BMMSCs have been reported to secrete soluble HLA-G, significant surface expression of this restricted MHC I molecule on BMMSCs has not been shown (29). Moreover, there is active ongoing debate regarding the detection of sHLA-G (5,16,36). While surface HLA-G is expressed on PDMCs after IFN-γ stimulation, HLA-DR, a MHC class II molecule, is not. This is in contrast to BMMSCs, in which HLA-DR is strongly induced by IFN-γ. Our finding is in line with other reports on fetal- and embryonic-source stem cells in terms of lack of expression or much delayed HLA-DR expression after IFN-γ stimulation (7,15). The IFN-γ-stimulated expression of surface HLA-G on PDMCs is likely reflective of its organ of origin. We previously have reported that intracellular HLA-G can be found in unstimulated, undifferentiated PDMCs but not BMMSCs (7). HLA-G has been reported to have immunodulatory effects on a wide range of immune cells, including dendritic cells and monocytes (6). It would be interesting in future investigations and useful clinically to assess the possible immunomodulatory interactions of PDMCs towards these other populations of immune cells.
The focus in stem cell immunobiology has so far been mainly centered on interactions with T cells, since for successful transplantation, these lymphocytes are arguably the most important subpopulation in mediating rejection. In transplantation immunology, the role of NKs had previously been believed to be limited to BM transplantation. Recent data, however, show that these lymphocytes participate in cross-talk with the adaptive arm of the immune response and in rejection of solid organs (21,26). Nonetheless, research on the immune interactions of this important population of lymphocytes with MSCs has lagged behind that of T lymphocytes. While it appears that MSCs also suppress some effector responses of NKs, overall the immunosuppressive effects are not as clearly defined as for T cells. BMMSCs are resistant to cytolysis of freshly isolated NKs but not cytokine-stimulated NKs (33,37,38); and the production of IFN-γ by NKs is reduced in the presence of BMMSCs (1,22,33,37,38). However, reports on the ability of BMMSCs to inhibit NK cytolysis towards its target cells are not as clear cut (22,33). Our work shows that PDMCs can suppress IFN-γ secretion by NKs and resist NK cytotoxicity, similar to data on BMMSCs. In addition, PDMCs can suppress NK cytotoxicity towards known target cells as well. These broader immunomodulatory effects of PDMCs on NKs may partially be due to its earlier developmental stage, since both human ESCs and ESC-derived mesenchymal progenitors can also resist NK cytotoxicity to a great extent (11,41). More research is needed for definitive conclusion regarding NK interactions with MSCs of different developmental stages.
Conclusion
In summary, PDMCs are a multipotential population of progenitor cells with strong immunomodulatory effects on NKs, affecting NK cytotoxicity and cytokine secretion. Inhibition of cytotoxicity effects are cell-contact dependent, and partially mediated through modulation of NK surface activating and inhibitory receptors. Moreover, the immunomodulatory effects of PDMCs on NKs can be enhanced by IFN-γ stimulation of PDMCs, which induces surface HLA-G expression on these progenitor cells and increases suppressive effects. With its broad immunosuppressive properties, PDMCs may be an attractive, alternative source of multipotent progenitor for therapeutic use.
Footnotes
Acknowledgments
This work was supported by grants from the National Health Research Institutes of Taiwan (97A1-SCPP05-014 to B.L.Y.) and National Science Council of Taiwan (NSC98-3112-B-400-012 to K.J.L.; NSC100-2321-B-400-022 to B.L.Y.) The authors declare no conflicts of interest.
