Abstract
Autologous platelet-rich plasma (PRP) has been extensively investigated for large bone defect treatment, but its clinical application is harassed by controversial outcome, due to highly variable PRP quality among patients. Alternatively, allogeneic PRP from well-characterized donors cannot only generate more consistent and reliable therapeutic effect but also avoid harvesting large quantities of blood, an additional health burdens to patients. However, the use of allogeneic PRP for bone defect treatment is generally less investigated, especially for its immunogenicity in such application. Here, we meticulously investigated the immunogenicity of allogeneic PRP and evaluated its healing efficacy for critical-sized defect treatment. Allogeneic PRP contained 4.1-fold and 2.7- to 4.9-fold higher amount of platelets and growth factors than whole blood, respectively. The intramuscular injection of allogeneic PRP to rabbits did not trigger severe and chronic immunoresponse, evidenced by little change in muscular tissue microstructure and CD4+/CD8+ T lymphocyte subpopulation in peripheral blood. The implantation of allogeneic PRP/deproteinized bone matrix (DPB) constructs (PRP + DPB) successfully bridged 1.5-cm segmental radial defects in rabbits, achieving similar healing capacity as autologous MSC/DPB constructs (MSC + DPB), with greater bone formation (1.1–1.5x, p < 0.05) and vascularization (1.3–1.6x, p < 0.05) than DPB alone, shown by histomorphometric analysis, bone mineral density measurement, and radionuclide bone imaging. Furthermore, the implantation of both allogeneic PRP- and autologous MSC-mediated DPB constructs (PRP + MSC + DPB) resulted in the most robust bone regeneration (1.2–2.1x, p < 0.05) and vascularization (1.3–2.0x, p < 0.05) than others (PRP + DPB, MSC + DPB, or DPB alone). This study has demonstrated the promising use of allogeneic PRP for bone defect treatment with negligible immunogenicity, great healing efficacy, potentially more consistent quality, and no additional health burden to patients; additionally, the synergetic enhancing effect found between allogeneic PRP and autologous MSCs may shed a light on developing new therapeutic strategies for large bone defect treatment.
Keywords
Introduction
Large bone defect treatment still remains as a major clinical challenge, and implantation of autologous bone grafts are currently considered as the gold standard. However, the use of autologous bone grafts is beset by several problems including limited availability and donor site morbidity (2, 13). Recently, bone tissue engineering (BTE) approaches based on the use of platelet-rich plasma (PRP) have been proposed and achieve certain success (1, 31).
PRP is a concentrate of platelets in a small volume of plasma, containing many types of highly concentrated growth factors with potentially beneficial effect for bone regeneration and vascularization (9, 16). In 1998, Marx et al. first showed the healing efficacy of PRP for bone defect treatment (29); in the past decade, PRP, especially the autologous PRP, has been extensively investigated for BTE application and demonstrated defect healing efficacy in animal models and clinical trials, especially in dental application (17, 25, 32, 43, 46, 47, 49, 50). However, the therapeutic outcome of autologous PRP remains highly controversial, as a number of studies have claimed no positive or even negative effect in defect treatment (18, 21, 23). This controversy could be explained by PRP's highly variable quality in terms of platelet number and growth factor concentration and activity (16, 35). Preparation procedures could easily contribute to this variation, including centrifuge conditions, type of reagents for blood collection, and platelet activation (16, 31, 35). Moreover, the physiological conditions of human body may have a profound influence on the quality of PRP as well; for instance, the trauma condition in patients may activate the platelets in the blood and subsequently reduce activities and amounts of growth factors in PRP prepared, leading to the compromised therapeutic outcome (27, 34). While many investigations has been conducted to optimize the PRP preparation technique to minimize the variations (16), the inherent physiological restriction could be hardly overcome, especially for traumatic patients, where most of the bone fractures are accompanied with bleeding, platelet activation, and blood coagulation.
Alternatively, PRP from allogeneic source could be explored to overcome the inherent deficiencies of autologous PRP. Blood isolated from healthy adults in consistent physiological conditions can be used to prepare allogeneic PRP with high-quality and reliable therapeutic efficacy. Furthermore, unlike autologous PRP, the use of allogeneic PRP avoids the additional procedure to harvest large quantity of blood, thus eliminating the extra health burdens on the patients, especially in the severe traumatic conditions (16, 31). However, so far the use of allogeneic PRP for BTE application is rarely investigated, in particular its immunogenicity in such application remaining unknown. Hence, in this study, we first investigated its immunogenicity without major histocompatibility complex (MHC) matching by analyzing the changes of T lymphocyte population and tissue morphology at the injection site following the intramuscular injection of allogeneic PRP gel into rabbits. Then, we evaluated the defect healing potential of allogeneic PRP in a rabbit segmental radial defect model by implanting allogeneic PRP-mediated tissue engineered (TE) constructs with or without autologous mesenchymal stem cells (MSCs).
Materials and Methods
Ethics and Experimental Design
All animal experiments were performed in strict accordance with protocols approved by the Institutional Animal Care Committee of Xijing Hospital. Male New Zealand White rabbits (6 months old, 1.8–2.2 kg) were obtained from the Experimental Animal Center of the Fourth Military Medical University (Xi'an, China). All materials used were purchased from Sigma-Aldrich (China) unless otherwise stated. This study is consisted of two experiments. In first experiment, we evaluated the allo-immunogenicity by injecting the allogeneic PRP into the gluteus maximus muscle of rabbits and analyzing the changes of T lymphocyte population in the peripheral blood and tissue morphology at the injection site. In the second experiment, we investigated the defect healing efficacy of allogeneic PRP by implanting allogeneic PRP-mediated deproteinized bone graft (DPB) constructs with or without autologous MSCs into the segmental radial defects in rabbits (Fig. 1A).

(A) Experimental design. (B) PRP preparation procedure. (C) Experimental procedure of Experiment 2. PRP, platelet-rich plasma; DPB, deproteinized bone matrix; MSC, mesenchymal stem cell.
Allogeneic PRP Preparation and Activation
Sixty milliliter of blood was drawn from each rabbit donor through the cardiac puncture, and PRP was prepared using a typical two-step centrifugation procedure (Fig. 1B) (19, 20). Briefly, first centrifugation at 2,400 rpm for 10 min was employed to separate the layers of PRP and platelet-poor plasma (PPP) from red blood cells. Then PRP and PPP portions were spun again at 3,600 rpm for 15 min to concentrate PRP. Upon activation, PRP mixture was added with activating solution (10% CaCl2 and 400 IU/ml thrombin) in the ratio of 9:1 to form PRP gel. The platelet number and growth factor concentrations [transforming growth factor-β1 (TGF-β1), platelet-derived growth factor (PDGF), epidermal growth factor (EGF), and vascular endothelial growth factor (VEGF)] of PRP and whole blood were measured respectively by hemocytometer and commercial enzyme-linked immunosorbent assay (ELISA) kits (R&D System, USA) according to the manufacturer's instructions.
Immunogenicity of Allogeneic PRP
Under the general anesthesia and sterile condition, immediately after the activation, allogeneic PRP gel was injected into the animal gluteus maximus muscles bilaterally (0.5 ml of PRP gel in each injection site), and its immunogenicity was investigated as follows.
CD4+ and CD8+ T Lymphocyte Subpopulation Analysis
One milliliter of blood was drawn from the ear vein of rabbits before and 1, 4, and 12 weeks after the surgery. Ninety microliters of blood was incubated first with 10 μl of CD4-fluorescein isothiocyanate (FITC) or CD8-FITC monoclonal antibodies (Antigenix, USA) for 15 min, then with 2 ml of red blood cell lysate solution (Biomiga, USA) for 20 min, followed by centrifugation of 1,000 rpm for 5 min. Supernatant was discarded, and cell pellet was resuspended at 0.2 ml of fixation buffer (0.2% formalin in PBS) and analyzed by flow cytometry (Coulter, USA).
Histology and Immunohistochemistry (IHC) Analysis
Tissue samples at the injection site were harvested at 1, 4, and 12 weeks postsurgery after euthanizing the animals. Tissue samples were paraffin-embedded, sectioned at the thickness of 5 μm, and hematoxylin and eosin (H&E)-stained for the tissue morphology. In addition, IHC staining with interleukin-2 (IL-2) rabbit-specific antibody (Boster, China) was conducted to investigate the expression of IL-2 at the injection site, according to manufacturer's manual.
Defect Healing Efficacy of Allogeneic PRP
DPB was prepared from the euthanized rabbits, loaded with allogeneic PRP and/or autologous MSCs, and implanted into a 15-mm segmental defect in the radius (Fig. 1C).
Preparation of Deproteinized Bone Graft (DPB)
DPB was prepared as previously reported (53). Briefly, ilium bone tissue of freshly euthanized rabbits was harvested and sawn into suitable bone grafts (3 mm x 3 mm x 15 mm). Bone grafts were immersed in 30% H2O2 solution for 48 h, washed thoroughly with water, incubated in chloroform and methanol solutions (1:1) for 4 h, and followed by ether extraction treatment for 24 h. After complete removal of ether by 100% ethanol, DPB was freeze-dried and sterilized by ethylene oxide gas before use (Fig. 1C).
Autologous MSC Isolation and Cultivation
One month before the surgery, 3 ml of bone marrow was aspirated from rabbits (Fig. 1C), mixed with Dulbecco's modified Eagle's medium (DMEM), centrifuged, and plated in petri dishes in DMEM supplemented with 15% fetal bovine serum (Hyclone, China), 50 U/ml penicillin, and 50 mg/ml streptomycin. Media were changed every 2–3 days, and cells were subpassaged at 80% confluence. MSCs at passage 4 were used for implantation.
Surgical Procedure
Immediately before the surgery, 1 ml of allogeneic PRP or MSC suspended PRP solution (5 × 10 × 106 cells/ml) was loaded onto each DPB under the negative pressure and activated with activating solution. A longitudinal incision over the front limb was performed to expose the radius, and a 1.5 cm in length segmental defect was created. Forty-eight defects in 24 rabbits were divided into four groups and implanted with Group I: allogeneic PRP and autologous MSC-mediated DPB constructs (PRP + MSC + DPB), Group II: autologous MSC-mediated DPB constructs (MSC + DPB), Group III: allogeneic PRP-mediated DPB constructs (PRP + DPB), and Group IV: DPB alone (Fig. 1C). After the implantation, wound was closed in layers with antibiotics and painkiller administrated for 3 days postsurgery.
Radiographic Imaging and Bone Mineral Density (BMD) Analysis
At 4, 8, and 12 weeks postsurgery, under the anesthesia, the defect region of rabbits was subject to the radiographic imaging (distance: 1 mm; X-ray source: 46 Kv, 50 mA; exposure time: 0.14 s), and the BMD of the regenerated new bone (n = 4 for each group) was analyzed by the dual-energy X-ray bone densitometer (Norland XR 36, USA).
Radionuclide Bone Imaging
At 4, 8, and 12 weeks postsurgery, rabbits were subject to radionuclide bone imaging as previously described (41). Briefly, radionuclide reagent, 99mTechnetium-methyl diphosphonate (99mTc-MDP) was injected into rabbits through the ear vein at the dosage of 0.8 mCi/kg body weight. Four hours later, rabbits were anesthetized and analyzed by the single-photon emission computed tomography (SPECT, GE HawK Eye VH, USA) at the acquisition rate of 1 frame/5 min, matrix size of 256 × 256 pixel, and energy peak at 140 kev. Region of interest (ROI) with the size of 0.3 cm x 1.5 cm was selected at the defect region to calculate the accumulation of 99mTc-MDP (n = 4).
Histology and Histomorphometric Analysis
At 4, 8, and 12 weeks postsurgery, radius bones were harvested for analysis. After fixation and decalcification, samples were embedded in paraffin, sectioned at the thickness of 5 μm, and stained with H&E. Histomorphometric analysis was performed to study the new bone regeneration. For each sample, six sections spaced 50 μm apart were selected; in each section, six equidistantly spaced microscopic fields within the defect region were analyzed by Image J software. New bone regeneration was evaluated as bone volume/tissue volume (BV/TV, %), as suggested by the “ASBMR Histomorphometry Nomenclature Committee” (33).
Statistical Analyses
Parametric data were represented as mean ± SD. Student's t test was used to analyze the platelet count and growth factor concentration; one-way ANOVA with Bonferroni posttest was used to analyze the subpopulation change of CD4+ and CD8+ T-cell in peripheral blood; and two-way ANOVA with Bonferroni posttest was applied for the analysis of histomorphometry, bone mineral density, and 99mTc-MDP uptake. A value of p < 0.05 was taken as significant.
Result
Platelet Counts and Growth Factor Concentrations
The platelet concentrations in PRP (12.87 ± 0.848 × 106/μl) have been enriched for more than fourfold, as compared to whole blood (3.172 ± 0.4864 × 1 0 6/μl, p < 0.001, n = 5) (Fig. 2A). Moreover, ELISA assay demonstrated a significant higher level of growth factors existing in PRP than whole blood, specifically, 3.3-fold higher in TGF-β1, 4.9-fold higher in PDGF, 2.7-fold higher in EGF, and 3.8fold higher in VEGF (p < 0.01, n = 5) (Fig. 2B).
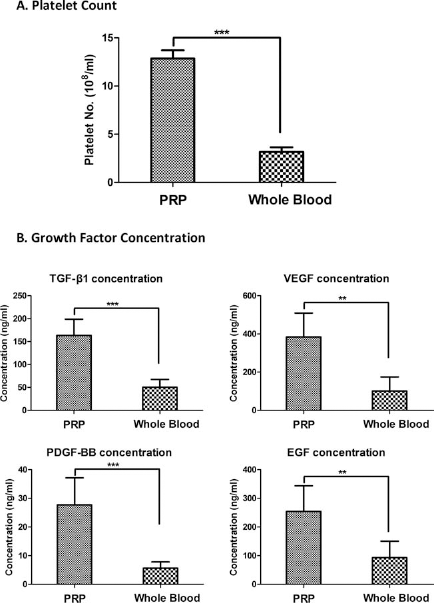
Platelet counts and growth factor levels. (A) The hemocytometer counting showed that the platelet number in PRP is more than fourfold higher than whole blood. (B) ELISA assay demonstrated a 2.7- to 4.9-fold increase of growth factor concentration in PRP compared to whole blood (**p < 0.01, ***p < 0.001, n = 5). TGF-β1, transforming growth factor-β1; VEGF, vascular endothelial growth factor; PDGF-BB, platelet-derived growth factor; EGF, epidermal growth factor.
Immunogenicity of Allogeneic PRP
After the injection of allogeneic PRP into the gluteus maximus muscle, rabbits exhibit normal appetite, elimination, behavior, and weight gain with no sign of redness and swelling at the injection site, indicating minimal immunoreactions. The normal physiological level of CD4+ and CD8+ cell subpopulation was measured by flow cytometry before the PRP injection as the baseline value, which is 32.40 ± 2.67% for CD4+ cells, 14.99 ± 4.53% for CD8+ cells, and a ratio of 2.35 ± 0.84 for CD4+/CD8+ cells (Fig. 3A). Allogeneic PRP injection resulted in a slight increase of CD4+ lymphocytes subpopulation at week 1 (36.09 ± 2.93%), which then decreased to 34.69 ± 1.84% at week 4 and restored to a normal level at week 8 (32.22 ± 1.77%), although the differences of change were statistically nonsignificant (p > 0.05, n = 4) (Fig. 3A). Nevertheless, the CD8+ cell subpopulation and CD4+/CD8+ cell ratio remained relatively steady throughout the whole study with no significant changes after PRP injection (Fig. 3A). In addition, the intramuscular injection of allogeneic PRP did not cause any visible tissue bruise or fibrous tissue encapsulation around the injection site by gross examination, and H&E histological staining revealed no obvious alteration in the microscopic structure of surrounding skeletal muscle tissue (Fig. 3B). Furthermore, subtle inflammation was found at the injection site at early time point (week 1), but the inflammation did not persist till week 4, which is evidenced by the infiltration of monocytes spotted at week 1, but not at weeks 4 and 12 (black arrow, Fig. 3B). This result is further corroborated by the IHC staining with IL-2-specific antibody, showing the short-term expression of IL-2 only at week 1 (gray arrow, Fig. 3B).
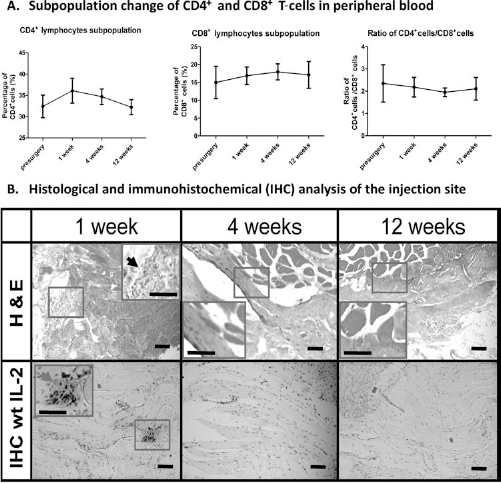
Immunogenicity of allogeneic PRP. (A) The flow cytometry analysis demonstrated no significant difference in CD4+, CD8+ lymphocyte subpopulations and the ratio of CD4+ cells/CD8+ cells before and 1, 4, and 12, weeks after the injection of allogeneic PRP into the gluteus maximus muscle, despite of the slight increase of CD4+ cells at week 4. (B) H&E histological staining showed that the injection of allogeneic PRP did not change the normal morphology of muscular tissue around the injection site throughout the period of study, while monocyte infiltration was only observed at week 1 (as indicated by the black arrow); immunohistochemical (IHC) staining with interleukin (IL)-2-specific antibody showed a weak positive staining around the injection site at week 1 (as indicated by the gray arrow), whereas there is negative staining of IL-2 at weeks 4 and 8 (n = 4). Scale bars: 100 μm.
New Bone Formation
Next, allogeneic PRP-mediated TE constructs with or without autologous MSC were implanted into the 1.5-cm segmental radial defects. All the rabbits survived throughout the whole study with no evidence of infection and fully recovered the wound and movement of operated limbs. Rabbits from Group I (allogeneic PRP + autologous MSC + DPB), Group II (autologous MSC + DPB), and Group III (allogeneic PRP + DPB) experienced better bone regeneration and faster resorption of DPB compared to Group IV (DPB alone). In spite of the clear observation of DPB in all the four groups at week 4 (white arrow, Fig. 4A), in the later time points of Groups I–III (weeks 8–12), DPB was resorbed rapidly and replaced with newly formed bone tissue, which successfully bridged and integrated well with the two defect ends (green arrow, Fig. 4A). By contrast, Group IV showed a poor osteointegration between DPB and host bone tissue at week 4 with clear borderlines observed between DPB and the two defect ends (pointed by the blue arrow, Fig. 4B). At weeks 8 and 12, the subsequent absorption of DPB in Group IV led to the nonunion defect healing, as shown by both macroscopic observation and radiographic images (yellow arrow, Fig. 4A, B). Callus formations were found in Groups I–III, while the callus formed in Group I was bigger than those in Groups II and III at weeks 4 and 8, as shown by radiographic images (red arrow, Fig. 4B). Furthermore, bone remodeling occurred between weeks 8 and 12, leading to the absorption of callus and maturation of the newly formed bone tissue, indicated by the reduced callus size and the increased X-ray density of bone tissue in defect area (Fig. 4B).

Macroscopical and radiographic images of the bone defects. Both macroscopical (A) and radiographic images (B) showed a better bone healing process in Groups I–III at weeks 4 and 8, which successfully bridged the segmental defect, while in Group IV nonunion healing was observed at week 12 with the gradual resorption of DPB. Bone defects from Group I experienced slightly bigger callus formation at weeks 4 and 8 compared to defects from Groups II and III (as indicated by red arrow, B) and the bone remodeling process occurred in Groups I–III from weeks 4 to 12, which reduced the size of callus tissue with increased radiographic density. Scale bars: 1 cm. White arrow, implanted DPB; green arrow, newly formed bone tissue; blue arrow, borderlines between DPB and the two defect ends; yellow arrow, nonunion healing of defect.
Histology and Histomorphometric Analysis
At weeks 4, 8, and 12, samples from all groups were retrieved for histological evaluation. H&E staining showed partial degradation of DPB in Groups I–III accompanied with trabecular bone formation at week 4, while Group IV experienced the least amount of DPB degradation and bone formation. With the progress of time, the trabecular bone structure in all groups continued to grow and fuse together to form larger neobone tissue with the gradual degradation of DPB (black arrow, Fig. 5A) and infiltration of blood vessel (gray arrow, Fig. 5A). Highest amounts of neobone tissue formation were found in Group I, followed by Groups II and III, and least bone formation was observed in Group IV, as suggested by the area of bone tissue (BT, Fig. 5A). Quantitative histomorphometric analysis showed that Group I generated a significantly higher amount of neobone tissue formation compared to Group IV throughout the entire study (1.5–2.1x, p < 0.001, n = 4) (Fig. 5B), while the neobone volume in Group I was 1.16- to 1.4fold more than Groups II and III with significant difference in week 12 (p < 0.05, n = 4) (Fig. 5B). Furthermore, Groups II and III have achieved more bone formation than Group IV (1.24–1.74x, p < 0.05, n = 4) (Fig. 5B), although no significant difference were found between Groups II and III within 12 weeks (p > 0.05, n = 4) (Fig. 5B).

Histological and histomorphometric analysis of new bone regeneration. (A) H&E histological staining showed better bone regeneration with more neobone tissue formation in Group I compared to other groups and in Groups II and III compared to Group IV, while Group IV showed the least bone formation, while obvious absorption of DPB can be found at week 8 (as indicated by the black arrow). (B) Histomorphometric analysis demonstrated the highest amount of new bone regeneration in Group I, followed by Groups II and III, with least bone amount in Group I V. Scale bars: 100 μm. Gray arrow, infiltrated blood vessel; BT, bone tissue. *p < 0.05, **p < 0.01, ***p < 0.001, n = 4.
BMD Analysis and Radionuclide Bone Imaging
BMD examination and radionuclide bone imaging were utilized to evaluate the neobone tissue quality and the activities of vascularization and metabolism during bone regeneration. No significant differences were found among the four groups at week 4 in terms of BMD, whereas Groups I–III demonstrated better quality of newly formed bone in defect region with higher mineral density compared to Group IV at week 8 (1.26–1.50x, p < 0.05, n = 4) and week 12 (1.10–1.24x, p < 0.05, n = 4) (Fig. 6A). Generally the BMD examination result correlated well with the radiographic findings in Figure 4B. Radionuclide bone imaging demonstrated that intravascularly injected 99mTc-MDP mainly accumulated in the skeletal system, especially in regions of shoulder, elbow, skull, spine, and segmental defects, which were lighted up during the imaging (Fig. 6B). In the bone defect region, at weeks 4 and 8, Group I showed a highest amount of 99mTc-MDP accumulation, followed by Groups II and III, with least amount in Group IV, although all groups shared a similar degree of 99mTc-MDP accumulation at week 12 (regions in red circles, Fig. 6B). Quantitatively, 99mTc-MDP uptake in Group I is 1.28–2.00 and 1.14–1.56 fold significantly higher than Groups II–IV (n = 4, p < 0.05) at weeks 4 and 8, respectively; Groups II and III are 1.47–1.57 and 1.22–1.37 fold significantly more than Group IV (n = 4, p < 0.05) at weeks 4 and 8, respectively. Moreover, in all groups, the 99mTc-MDP accumulation at week 8 is generally higher than weeks 4 and 12, suggesting active metabolism and vascularization occurring at week 8.

Bone mineral density (BMD) analysis and radionuclide bone imaging [single-photon emission computed tomography (SPECT) analysis]. (A) BMD analysis showed a significantly higher degree of BMD at the defect regions in Groups I–III compared to Group IV at weeks 8 and 12. (B) Highest metabolic activities and vascularization at the defect region were observed in Group I, followed by Groups II and III, while Group IV experienced the least metabolic rate and vascularization (as indicated by the color density in red circle). (C) This result was confirmed by the quantitative analysis of 99mTechnetium-methyl diphosphonate (99mTc-MDP) accumulation at the defect region. *p < 0.05, **p < 0.01, ***p < 0.001, n = 4.
Discussion
PRP has demonstrated favorable applications for large bone defect treatment; however, its therapeutic efficacy is widely debated, especially for the use of autologous PRP. This can be explained by its highly variable quality due to preparation procedures and patients' physiological conditions. One promising solution to obtain high-quality PRP with consistent therapeutic efficacy is to prepare PRP from allogeneic sources. However, the use of allogeneic PRP for large bone defect treatment is rarely investigated, especially for its immunogenicity. In this study, we first investigated its immunogenicity by intramuscular injection of allogeneic PRP gel into rabbits, which showed little immunogenicity of allogeneic PRP with no significant change of CD4+ and CD8+ lymphocyte subpopulation in blood and tissue structure at the injection site. Furthermore, we evaluated the defect healing efficacy of allogeneic PRP in a rabbit segmental defect model and demonstrated that allogeneic PRP can significantly promote new bone regeneration with comparative healing potential as autologous MSCs, while the synergetic use of allogeneic PRP and autologous MSCs achieved the best healing result compared to the single use of PRP or MSCs.
Platelets are a small anucleate cell fragment, whose main physiological role is to prevent the blood loss by sustaining haemostasis (35). Platelets possess complex surface antigens: on one hand, their membrane bears the typical immuno molecules, human leukocyte antigen (HLA), and the blood group antigen ABO; on the other hand, a specific type of glycoprotein complex with polymorphisms constitutes the human platelet alloantigen (HPA). During the activation of immunization, HLA-I may play a major role in triggering the alloimmune response via a CD8+ T-cell-mediated approach; additionally, HPA can contribute substantially to the formation of alloantibodies by direct or indirect pathways mediated mainly by CD4+ T-cell (15, 38, 39, 42). Therefore, in our study, in order to understand immunoresponse of allogeneic PRP, we investigated the subpopulation change of both CD4+ and CD8+ T-cells. Similar approaches have been widely adopted to analyze the immune response after organ or tissue transplantation (3, 14, 22). In our study, a mild immunoresponse against the intramuscular injection of allogeneic PRP was observed at week 4 postoperatively, evidenced by the subtle increase in CD4+ cell subpopulation, infiltration of monocytes, and low amount of IL-2 expression. This temporal immunoresponse did not last till week 8, as shown by the restoration of CD4+ and CD8+ subpopulation to normal physiological level and negative IL-2 expression, implying a limited influence of early alloimmunization. These findings kept in consistent with the gross examination showing the uneventful healing of the wound with no swelling and exudates as well as the observation of rapid new bone regeneration and remodeling, as it is known that chronic alloimmune reaction is one of the major reasons leading to the failure of defect treatment using allogeneic bone grafts (44). The low immunogenicity of allogeneic PRP without proper HLA and HPA matching could be possibly explained by the fact that allogeneic PRP was delivered in the form of gel, which did not enter the circulation system and encapsulated most of the alloantigens (HLA and HPA) inside the gel with limited exposure to host antibodies and lymphocytes. Moreover, the activation of platelets may change surface immune antigens in terms of their structure and expression levels (40), reducing their immunogenicity. Last but not least, unlike the solid organ or tissue transplantation, PRP can be degraded and absorbed completely within weeks, thus eliminating the possibility for chronic immune reaction. Hence, the immunogenicity of allogeneic PRP may not become a major hurdle for its clinical application.
When loaded into DPB to generate TE constructs (Groups I and III) and implanted to the segmental defects, allogeneic PRP can promote faster and more robust new bone formation, as shown by the histology and radiographic imaging (Figs. 4 and 5), and achieved more mature bone structure, as shown by the BMD analysis (Fig. 6). This beneficial effect of PRP for new bone regeneration was first reported by Marx et al. in 1998, showing the addition of PRP to bone grafts can achieve 1.62–2.16 times better bone formation than grafts without PRP (29) and subsequently has been confirmed by many other researchers in several animal models and clinical settings (17, 25, 32, 43, 47, 49, 50). PRPs are known to contain a large variety of concentrated growth factors: besides the enriched levels of TGF-β1, PDGF, EGF, and VEGF as shown in our study (2.7–4.9x) (Fig. 2B), angiopoietin-2 (Ang-2), basic fibroblast growth factor (bFGF), insulin-like growth factor-I (IGF-I), and so on are also found in PRP (16, 31). Many of these growth factors have been demonstrated favorable effects for vascularization and/or the proliferation and maturation of osteoblast or osteoprogenitor cells, which could be the leading reason to explain for the capability of PRP-based BTE approaches to foster bone defect healing.
The Ang-2, bFGF, PDGF, and VEGF found in PRP are known to be able to promote neovascularization and angiogenesis (16, 31, 52), which could likely explain for the enhanced vascularization in defect regions when implanted with PRP-mediated TE constructs (Group I and III), evidenced by histology and radionuclide bone imaging. Radionuclide bone imaging is a well-established clinical examination technique to assess the abnormalities of bone tissue; after the intravascular injection, radio nuclide reagents, such as 99mTc-MDP, will be accumulated mainly into the bone tissue, especially in the highly vascularized region (e.g. bone tumor), and the accumulation dosage is closely correlated to the degree of vascularization (26, 51, 54). In current study, we observed the implantation of PRP-mediated DPB TE constructs (Groups I and III) led to a 1.2- to 2-fold increase of 99mTc-MDP accumulation in the defect region than DPB alone (Group IV) at weeks 4 and 8, implicating an enhanced vascularization activity. This finding is mirrored by the reports of Li et al., showing that the addition of PRP led to increase vascularization in the ectopic tissue-engineered bones (24) and Rai et al.'s study showing that PRP-treated femoral defect displayed 70.3% higher vascularization than control femurs without PRP treatment in a rat model (37). Nevertheless, at week 12, the differences of 99mTc-MDP accumulation among groups disappeared, indicating a similar vascularization level. This phenomenon could be possibly caused by bone remodeling process occurring at the last stage of defect healing, which can remove the excess vascular structure for the deposition of mature bone tissue (6). Moreover, bone remodeling process also contribute to the removal and replacement of excess immature bone tissue (the major component of bone callus) with much denser mature bone tissue, as reflected in this study by the reduce in callus size (Fig. 4B) but the increase in mineral density (Fig. 6A) (6).
Apart from the angiogenic factors, another group of growth factors found in PRP including bFGF, IGF-1, PDGF, and TGF-β1 demonstrates favorable capabilities to promote the proliferation, differentiation, and mineralization of osteoblast or osteoprogenitor cells such as MSCs (16, 31, 35). This could possibly be responsible for the increased new bone formation (Fig. 5B) and higher value of BMD (Fig. 6A) in the PRP-mediated DPB TE constructs (Groups I and III) as compared to DPB alone (Group IV). In addition, a synergetic enhancing effect was observed between allogeneic PRP and autologous MSCs (Group I), which led to the most robust new bone regeneration with highest amount of vascularization and new bone formation (Figs. 4–6), as compared to MSC- or PRP-mediated TE constructs (Groups II and III). MSCs, firstly identified by Friedenstein in 1960s (11, 12), have demonstrated great potential for bone defect treatment in various animal models and a few clinical reports (4, 5, 10, 28, 30, 36). It is known that they cannot only directly contribute to the new bone formation by osteogenic differentiation but also generated a local microenvironment favorable for bone formation by secreting a large spectrum of growth factors (6–8). This unique cellular or paracrinal behavior of MSCs could probably account for the capability of MSC-mediated TE constructs (Group II) to promote neovascularization and new bone formation. When PPR and MSCs were delivered simultaneously, on one hand, the growth factors released from PRP can induce neovascularization and stimulate the proliferation and mineralization of local osteoblast and osteoprogenitor (such as MSCs), as well as the externally delivered MSCs, on the other hand, the addition of externally expanded MSCs enlarge the stem cell pool responsible for bone regeneration. In such a synergetic manner, the codelivery of allogeneic PRP and autologous MSC-mediated DPB (Group I) achieved more bone regeneration than the single use of PRP or MSC-mediated DPB (Groups II and III). This is corroborated by the study of Yamada et al. showing a better defect treatment with the integrated use of autologous PRP and MSCs as compared to PRP alone (48). Furthermore, both Yamada et al. and Wojtowicz et al.'s studies demonstrated that this synergetic use of PRP and MSCs can even achieve a similar healing efficacy as the autologous bone graft, the gold standard of defect treatment (45, 48), thus alluding to a new therapeutic paradigm for large bone defect treatment.
Conclusion
In this study, we demonstrated that allogeneic PRP possesses negligible immunogenicity and great efficacy for large bone defect treatment by virtue of inducing neovascularization and enhancing new bone formation and maturation. Therefore, allogeneic PRP presents a promising potential to replace autologous PRP for the defect treatment, allowing potentially more consistent therapeutic efficacy and no additional health burden to patients. In addition, we found that synergetic enhancing effect between allogeneic PRP and autologous MSCs can further promote bone regeneration, which may represent a prologue for the development of a new BTE therapeutic strategy for large bone defect treatment.
Footnotes
Acknowledgments
This work was supported by Chinese Ministry of Science and Technology (973 Program No. 2009CB930003), NSF of China (Grant No. u0732003 and No. 30901532) to Pei Guoxian, National Natural Science Foundation of China (No. 81101353), and Xijing Zhutui Project (SJZT10Z09) to Zhang Zhiyong. The authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. The authors declare no conflicts of interest.
