Abstract
The therapeutic benefits of bone marrow-derived mesenchymal stem cell (BM-MSC) transplantation for ischemic stroke have been extensively demonstrated. However, studies on the optimal cell dose for intravenous administration are still limited. This study aimed to determine an appropriate cell dose for BM-MSC intravenous transplantation and to investigate the effect of cell dose on vascular remodeling in a rat model of ischemic stroke. BM-MSCs at doses of 5 × 10 4 (low-dose group), 5 × 10 5 (medium-dose group), and 2 × 10 6 (high-dose group) were intravenously injected into rats at 72 h after ischemia. The therapeutic efficacy of BM-MSCs was evaluated by measuring infarct volume, vascular diameters, capillary area in the peri-infarct zone, level of basic fibroblast growth factor (bFGF) in the peri-infarct zone, and serum vascular endothelial growth factor (VEGF) level at 7 days after ischemia. Compared with the low-dose and control groups, medium-dose and high-dose BM-MSC transplantation significantly reduced the volume of the infarct area, enlarged the diameters of pial vessels and the basilar artery, and increased the capillary area in the peri-infarct zone of the cerebral cortex. Furthermore, transplanted BM-MSCs elevated the expressions of bFGF in the peri-infarct zone and the serum VEGF level. Administration of 5 × 10 5 BM-MSCs is an appropriate cell dose for ischemic stroke therapy in rats. These findings may be helpful for designing future clinical trials.
Keywords
Introduction
Ischemic strokes are most commonly caused by occlusion of an intracranial artery 1 , which results in the sudden interruption of local cerebral blood flow and irreversible neuron death in the ischemic core within minutes due to complete ischemia. Neurons in the ischemic penumbra, however, may still recover due to reperfusion and collateral circulation. Although significant efforts had been made toward developing therapies for ischemic stroke, it is still the second leading cause of death and disability in adults worldwide 2 . In China, the annual stroke mortality rate is 157 per 100,000 persons and is the first leading cause of adult death and disability 3 . Currently, the only treatment that has been proven to improve the outcome of stroke patients is recanalization of occluded vessels via thrombolysis 4 . However, given a narrow therapeutic window of thrombolysis therapy, only a minority of stroke patients actually benefit from this therapy 4 . Therefore, developing a more effective therapy for ischemic stroke is urgently required.
Bone marrow-derived mesenchymal stem cells (BM-MSCs) are adult nonhematopoietic cells that are commonly used to perform various cellular therapies because of their pluripotency and trophic abilities 5 . BM-MSCs have been shown to pass through the blood–brain barrier (BBB) 6 and localize to the injured tissue in an ischemic stroke animal model 7 . Accumulating evidence suggests that transplanted BM-MSCs significantly reduce ischemia-induced histological and motor impairment in animal models of ischemic stroke8–11. In addition, BM-MSC transplantation has been demonstrated to reduce cell apoptosis12–14 and enhance vascular remodeling15–17, neural remodeling18–20, and endogenous cell proliferation 21 around the injured tissue in animal models of ischemic stroke. Furthermore, BM-MSC transplantation has been recently demonstrated to provide clinical benefits in patients who had had an ischemic stroke22–24. While the aforementioned findings support transplantation of BM-MSCs as a promising therapy for ischemic stroke, the mechanism underlying the therapeutic benefit of BM-MSC transplantation is still largely unexplored. To advance the use of BM-MSC transplantation in clinical therapy, it is necessary to comprehensively gain more data from preclinical studies. We have previously demonstrated that transplantation of allogeneic 5 × 10 6 BM-MSCs into middle cerebral artery occlusion (MCAO) rats at 3 days after ischemia significantly improved motor impairment, reduced infarct volume, and promoted neurogenesis and angiogenesis in the infarction area at 1 and 2 weeks after ischemia 25 . The results also showed that the injected BM-MSCs localized, exclusively, to the periinfarct area and significantly enhanced cell proliferation not only at the site of their location (peri-infarct area) but also in a remote area.
There are currently three different administration routes for BM-MSC transplantation, namely, intracranial, intra-arterial, and intravenous 26 . Of these routes, intracranial injection results in the greatest number of transplanted cells in the infarcted area, but it is also the most invasive. In addition, surgery is necessary for animals to receive an intracranial injection 27 , which may raise the mortality rate of the animals. Furthermore, the safety of intracranial administration of stem cells for stroke has yet to be adequately proven in clinical trials 26 . Intra-arterial administration implants stem cells via the ipsilateral internal carotid artery 27 , and the transplanted cells bypass the systemic circulation and directly migrate along the blood vessel to the injured brain tissue. However, this administration route still requires surgery and is more invasive compared with intravenous injection. Currently, intravenous administration is the most common method used for stem cell transplantation in both preclinical and clinical settings because of its simplicity and noninvasive properties 28 . The intravenous transplanted cells need to migrate along blood vessels and pass through the BBB to reach the infarct zone, and thus the number of effective therapeutic cells in the target ischemic lesion is far less than that of intracranial injection.
Although the therapeutic benefits of BM-MSC transplantation have been extensively demonstrated in animal models of ischemic stroke, studies focusing on the optimal cell dose of BM-MSCs administered intravenously for ischemic stroke are still limited. The appropriate cell dose of BM-MSCs is especially crucial for clinical practice in terms of biosafety. Previous studies have adopted different numbers of BM-MSCs for transplantation into animal models 29 of ischemic stroke, and the results of these studies are difficult to compare because of differences in the animal models, cell types, timing of transplantation, and timing of therapeutic efficacy evaluation. Therefore, the differences in the therapeutic efficacy between different cell doses of BM-MSCs in the same animal model and with the same experimental conditions should be evaluated. Recently, Wang et al. 29 investigated the optimal cell dose for intravenous BM-MSC transplantation in MCAO rats. Five different doses ranging from 1 × 10 4 to 1 × 10 7 BM-MSCs were used, and the results showed that intravenous transplantation of 1 × 10 6 , 2 × 10 6 , and 1 × 10 7 BM-MSCs at 24 h after MCAO significantly reduced the infarct volume at 14 days after ischemia. In addition, transplantation of 2 × 10 6 and 1 × 10 7 BM-MSCs significantly improved rat motor performance. However, the effect of transplanted cell dose on vascular remodeling in the infarct area remains to be investigated.
In our previous study, we used a cell dose of 5 × 10 6 BM-MSCs for transplantation 25 . In that study, a preliminary experiment for determining the appropriate cell dose (5 × 10 5 , 5 × 10 6 , and 5 × 10 7 BM-MSCs) was conducted and showed that intravenous administration of a high cell dose (5 × 10 7 BM-MSCs) resulted in a high mortality rate because of cell aggregation and pulmonary embolism. Therefore, the purposes of the current study are to evaluate the feasibility and therapeutic efficacies of low-cell dose BM-MSC transplantation and to investigate the effect of cell dose on vascular remodeling in a rat model of ischemic stroke. Using a permanent MCAO rat model, 5 × 10 4 (low-dose group), 5 × 10 5 (medium-dose group), and 2 × 10 6 (high-dose group) BM-MSCs were intravenously injected into rats at 72 h after ischemia. The therapeutic efficacy of BM-MSCs at the three different doses was evaluated by determining the infarct volume, the vascular diameters, the capillary area in the peri-infarct zone, the level of basic fibroblast growth factor (bFGF) in the peri-infarct zone, and the serum vascular endothelial growth factor (VEGF) level at 7 days after ischemia.
Materials and Methods
Establishment of MCAO Cerebral Infarction Rat Model
Male adult Sprague–Dawley rats weighing 250–300 g were purchased from Guangdong Medical Laboratory Animal Center. All the protocols in this study were approved by the Institutional Animal Care and Use Committee (IACUC) of Guangzhou Medical University. The MCAO procedure was performed as described in a previous study 25 . Briefly, rats were anesthetized, and the right common carotid artery, external carotid artery, internal carotid artery, and vagus nerve were separated. MCAO was achieved by insertion of a nylon monofilament in the middle cerebral artery to block blood flow.
Isolation and Culture of Rat BM-MSCs
The rat BM-MSCs were isolated from the tibia and fibula of Sprague–Dawley rats (weighing 50–100 g) as previously described 25 . In brief, the rats were sacrificed, and the tibia and fibula were obtained. The marrow cavities were repeatedly flushed with 10 ml of L-Dulbecco's modified Eagle's medium (DMEM)/F12 (Invitrogen, Carlsbad, CA, USA) to obtain the cell suspension. After washing twice with phosphate-buffered saline (PBS), the cells were suspended with 6–8 ml of L-DMEM/F12 medium containing 10% fetal bovine serum (FBS) (Invitrogen) and seeded in 75-cm 2 flasks. The cells were passaged when 90% confluence was reached. The cells at passage 5 (P5) were characterized by immunostaining with antibodies specific for surface markers CD29+, CD44+, CD31-, and CD45- (Fig. S1A, supplementary material available at https://drive.google.com/open?id=0B6vjgOZzIwv_SFpfUlVNQXlrU1E) using a Beckman Coulter EPICS ALTRA Flow Cytometer (Beckman Coulter, Brea, CA, USA).
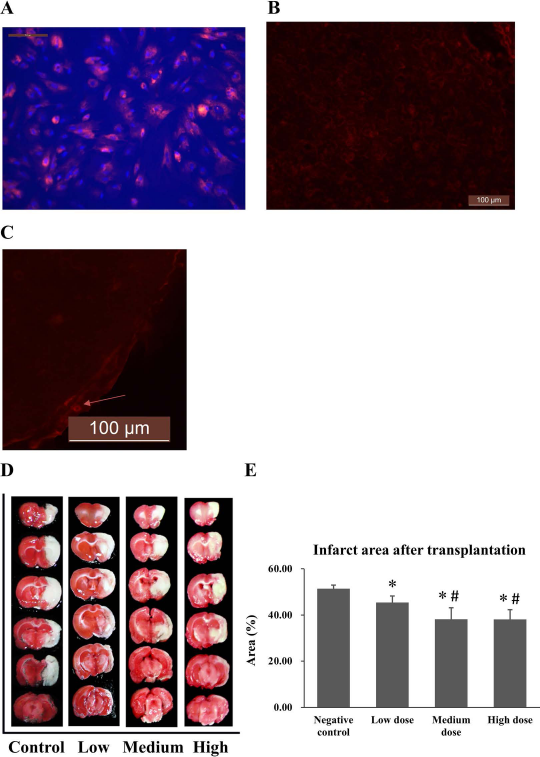
Transplanted BM-MSCs migrated to the infarct zone and reduced the volume of infarct area in MCAO rats. Bone marrow-derived mesenchymal stem cells (BM-MSCs) were prelabeled with the cell tracker marker CM-DiI fluorescent dye (4 μg/ml) for 5 days, followed by treatment with 4′,6-diamidino-2-phenylindole (DAPI) nuclear staining dye (1 μg/ml) for 30 min. Scale bars: 100 μm. (A) CM-DiI-labeled BM-MSCs were intravenously injected into middle cerebral artery occlusion (MCAO) rats 72 h after ischemia. Seven days after transplantation, brain slices were prepared and examined by a fluorescence microscopy. Images showed CM-DiI-labeled BM-MSCs in the peri-infarct area of rats in the high-dose group (B, C). (D) 2,3,5-Triphenyltetrazolium chloride (TTC)-stained serial brain slices of rats. The red and white areas represent healthy brain tissue and the infarct zone, respectively. The percentages of infarct zone were quantitatively determined as described in the Materials and Methods section (E). ∗p < 0.05 compared to the negative control group, #p < 0.05 compared to the low-dose group.
Transplantation of BM-MSCs
Three days after MCAO, the rats were randomly divided into control, low-dose, medium-dose, and high-dose groups. Before transplantation, BM-MSCs were prelabeled with CM-DiI fluorescent dye (4 μg/ml; Molecular Probes, Eugene, OR, USA), a cell tracker marker. BM-MSCs at P5 were used for transplantation. The growth curve of P5 BM-MSCs and CM-DiI-labeled P5 BM-MSCs was determine by the 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) assay and shown in Figure S1B (supplementary material available at https://drive.google.com/open?id=0B6vjgOZzIwv_SFpfUlVNQXlrU1E). The rats were injected with 1 ml of DMEM/F12 medium (control group) 29 and with 5 × 10 4 (low-dose group), 5 × 10 5 (medium-dose group), or 2 × 10 6 (high-dose group) P5 BM-MSCs through the tail vein. A total of three batches of BM-MSCs were used in this study, and the transplantation experiments were mainly repeated with different batches of cells. Seven days after transplantation, the rats were sacrificed and the brain tissue and serum samples were collected for analyses.
Brain Slices and Fluorescence Microscopy
P5 BM-MSCs were labeled with the cell tracker marker CM-DiI fluorescent dye (1 μg/ml) by incubation at 37°C for 5 min and then at 4°C for 15 min. The CM-DiI-labeled BM-MSCs were observed by fluorescence microscope (Olympus Corporation, Tokyo, Japan). A dose of 2 × 10 6 CM-DiI-labeled BM-MSCs was injected into MCAO rats at 3 days after ischemia. Seven days after transplantation, the brain of the rat was removed and submerged in 4% paraformaldehyde (PFA; pH 7.4) at 4°C for 4 h. The brain was then dehydrated in ethanol solutions, cleaned with xylene, and embedded in paraffin. Four-micrometer-thick serial sections were sliced from the brain at a distance of 1.6 and −0.2 mm from the anterior fontanelle. The slices were examined by fluorescence microscopy.
2,3,5-Triphenyltetrazolium Hydrochloride (TTC) Staining
Seven days after transplantation, rats were sacrificed, and the brains were immediately obtained and placed into a brain slice mold, followed by freezing at −20°C for 20 min. The brain was then cut into 2-mm blocks and immersed in PBS at room temperature. The slice was then incubated in 0.05% TTC solution (Sigma-Aldrich, St. Louis, MO, USA) at 37°C in the dark for 30 min, followed by fixation with 4% PFA for 10–30 min. The stained brain was photographed by a Leica S6D camera (Leica Microsystems Ltd., Wetzlar, Germany). The red and white areas represented, respectively, healthy brain tissue and infarct zone. The volume of the infarct area was calculated with Image-Pro Plus 6.0 software (Media Cybernetics Inc., Bethesda, MD, USA). The infarct zone percentage was determined by the equation: total area of infract zone/total area of cerebral hemisphere in affected side × 100%.
Latex Perfusion
The latex perfusion solution was prepared by mixing carbon ink (Shanghai Ink Factory, Shanghai, P.R. China) and latex (60% natural latex concentrate; Tavorn Para Rubber Industry, Sadao, Thailand) at a ratio of 1:20. The latex perfusion solution was connected to a perfusion device and intravenous pump Terfusion Syringe Pump TE-331 (Terumo Corporation, Tokyo, Japan). Seven days after transplantation, rats were anesthetized, and 10 ml of latex perfusion solution was injected into the aortic arch at a rate of 12.8 ml/min. The rat was then sacrificed, and the brain was removed and fixed in 4% PFA for 24 h, followed by photographing with a Leica S6D stereomicroscope. The observation zone for the vascular anastomosis between the anterior cerebral artery and the middle cerebral artery cerebral in the peri-infarct area was at a distance of 2 and −1.1 mm from the anterior fontanelle, and 1–3 mm from the median line. The observation zone for the basilar artery was at a distance of 5 mm from the bifurcation of the posterior cerebral artery. The mean vascular diameter was measured and calculated with Image-Pro Plus 6.0 software [National Institutes of Health (NIH), Bethesda, MD, USA].
Quantitative Determination of Vessel Area by Uptake of FITC
Seven days after transplantation, 1 ml of fluorescein isothiocyanate (FITC)–dextran (50 mg/ml) solution was injected into the tail vein of the anesthetized rats. After 4 min, the rats were sacrificed, and the brain was removed and fixed for 24 h in 4% PFA. The brains were dehydrated and fixed in a solution of 30% sucrose and 4% PFA for 48 h. The resultant tissue was then sliced into 50-μm sections. The slices within 1.6-0.2 mm from the anterior fontanelle were visualized by an EVOS fluorescence microscope (Life Technologies, Carlsbad, CA, USA). Image-Pro Plus 6.0 software (Media Cybernetics Inc.) was used for area calculations.
Western Blot and Enzyme-Linked Immunosorbent Assay (ELISA) Analysis
Seven days after transplantation, brain tissue including the hippocampus, striatum, and cerebral cortex from the affected side was lysed and homogenized in 200 ml of radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime, Beijing, P.R. China). The lysate was centrifuged at 14,000 × g for 5 min, and the supernatant was collected. The protein extract was electrophoresed by 12% sodium dodecyl sulfate (SDS)/polyacylaminde gel electrophoresis (PAGE) and transferred onto polyvinyl difluoride (PVDF) membranes (Millipore, Billerica, MA, USA). Antibodies specific for bFGF (Abcam, Cambridge, UK) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Cell Signaling Technology, Danvers, MA, USA) were used for Western blot analysis. For ELISA analysis, rat serum was collected through the eye 7 days after transplantation. The serum VEGF level was determined using a rat VEGF ELISA Kit (Invitrogen) according to the manufacturer's protocol.
Statistical Analysis
All quantitative results were presented as mean ± standard deviation (SD). Shapiro–Wilk test and Levene's test were used for testing data normality and homogeneity of variance, respectively. One-way analysis of variance (ANOVA) with Fisher's least significant difference (LSD) test for post hoc testing was utilized for the comparisons among all groups. The statistical significance level for all comparisons was set at a two-tailed value of p < 0.05. All analyses were performed using IBM SPSS Version 20 (SPSS Statistics V20; IBM Corporation, Armonk, NY, USA).
Results
Transplanted BM-MSCs Can Migrate to the Infarct Zone and Efficiently Reduce the Volume of Infarct Area in MCAO Rats
We established a MCAO rat model to evaluate the therapeutic efficacy of BM-MSCs on brain ischemic stroke. To determine an appropriate dose of transplanted cells, P5 BM-MSCs at 5 × 10 4 (low-dose group), 5 × 10 5 (medium-dose group), and 2 × 10 6 (high-dose group) were intravenously injected into MCAO rats at 72 h after ischemia. To address if the injected BM-MSCs could migrate to the infarct area, the cells were prelabeled with the cell tracker marker CM-DiI fluorescent dye (Fig. 1A). The results showed that the transplanted BM-MSCs could be clearly observed in the peri-infarct area of MCAO rats in the high-dose group at 7 days after transplantation (Fig. 1B and C).
The therapeutic efficacy of transplanted BM-MSCs was evaluated by determining the volume of infarct area using the TTC staining method. The TTC-stained brain slices of the rats are shown in Figure 1D, and the percentage of infarct zone was quantitatively compared among the four groups (Fig. 1E and Table 1). The control group had a significantly higher infarct zone percentage than the other three transplanted groups (all p < 0.05), while the low-dose group also had a significantly higher percentage than the medium- and high-dose groups (both p < 0.05). However, there was no significant difference between medium- and high-dose groups (p > 0.05). These results suggest that transplanted BM-MSCs can migrate into the infarct zone and significantly reduce the volume of the infarct zone.
Comparisons of Cerebral Results Among Different Doses of BM-MSCs

The sample sizes of each variable were as follows: infarct area, n = 5; diameter of pial anastomosis vessel, n = 6; diameter of basilar artery, n = 3; capillary counts (MVD), n = 3; bFGF-hippocampus, n = 3; bFGF-striatum, n = 3; bFGF-cerebral cortex, n = 3; VEGF expression, n = 5. Abbreviation: BM-MSCs, bone marrow-derived mesenchymal stem cells; PBS, phosphate-buffered saline; MVD, microvessel density; bFGF, basic fibroblast growth factor; VEGF, vascular endothelial growth factor.
p < 0.05 compared to control group
p < 0.05 compared to low-dose group
p < 0.05 compared to medium-dose group.
Transplanted BM-MSCs Induce Vasodilation in Pial Vessel and the Basilar Artery
Since transplanted BM-MSCs can reduce the infarct zone, we further investigated if vascular remodeling contributed to this effect. The diameters of pial vessels and the basilar artery after transplantation of BM-MSCs were measured by the latex perfusion method. The mean diameters of pial vessels (Fig. 2A) and the basilar artery (Fig. 2B) were both significantly different among the four groups (both p < 0.05, one-way ANOVA). The mean pial vessel diameters of the medium- and high-dose groups were significantly larger than that of the control or low-dose groups (all p < 0.05), while there was no significant difference between medium- and high-dose groups (both p > 0.05) (Fig. 2C). As for the basilar artery, the high-dose group had a significantly larger mean diameter of the basilar artery than the other groups (all p < 0.05) (Fig. 2D). These results suggest that transplanted BM-MSCs can significantly induce vasodilation in both pial vessels and the basilar artery.

Transplanted BM-MSCs enlarged the diameters of pial vessels and the basilar artery. The diameters of pial vessels and basilar artery after transplantation of BM-MSCs were measured by the latex perfusion method. Pial vessels (A) and basilar artery (B) in the affected side of MCAO rats 7 days posttransplantation. The solid line rectangle on the left indicates the area in the infarct zone magnified (right) and used for analysis. The mean vascular diameter of pial vessels (C) and the basilar artery (D) was quantitatively determined. ∗ p < 0.05 compared to the negative control group, #p < 0.05 compared to the low-dose group, Δp < 0.05 compared to the medium-dose group.
Transplanted BM-MSCs Increased the Capillary Area in the Cerebral Cortex Peri-Infarct Zone
Next, we determined if the transplanted BM-MSCs had an effect on the capillary area in the peri-infarct zone of the cerebral cortex by the FITC–dextran method. The location where the capillary area was measured in the brain is shown in Figure 3A. The capillaries were filled with FITC–dextran (Fig. 3B–E), and the total capillary area was quantitatively determined (Fig. 3F). The capillary areas were significantly higher in the medium- and high-dose groups than in the other two groups (all p < 0.05), while there was no significant difference between the medium- and high-dose groups (both p > 0.05) (Fig. 3F and Table 1). These results indicate that transplanted BM-MSCs significantly increased the capillary area in the peri-infarct zone of the cerebral cortex.

Transplanted BM-MSCs increased the capillary area in the peri-infarct zone of the cerebral cortex. (A) The solid line square indicates the location where the capillary area was measured in the brain. Fluorescence images showed that the capillaries were filled with intravenously injected FITC–dextran in the selected area of rats in the control, low-dose, medium-dose, and high-dose groups (B–E, respectively). The total capillary area was quantitatively determined using Image-Pro Plus software (F). ∗p < 0.05 compared to the negative control group, #p < 0.05 compared to the low-dose group.
Transplanted BM-MSCs Elevated the Expression of bFGF in the Peri-Infarct Zone and Serum VEGF Level
To elucidate the molecular mechanism of BM-MSC-mediated vascular alterations, the expression of bFGF in the infarct zone and serum VEGF level were examined. The protein expression of bFGF in the hippocampus, striatum, and cerebral cortex was examined by Western blotting (Fig. 4A). All three transplanted groups had a significantly higher expression of bFGF in the hippocampus and striatum than the control group, while there was no significant difference among the three transplanted groups (all p < 0.05) (Fig. 4B and C and Table 1). In the cerebral cortex, bFGF expression was significantly higher in the high-dose group than in the other three groups (all p < 0.05) (Fig. 4D and Table 1).

Transplanted BM-MSCs elevated the protein expressions of bFGF in the infarct zone. Seven days after transplantation, the protein expression of basic fibroblast growth factor (bFGF) in different areas of the affected side was examined by Western blotting (A). The expression level of bFGF in the hippocampus (B), striatum (C), and cerebral cortex (D) was quantified by densitometric analysis. ∗p < 0.05 compared to the negative control group, #p < 0.05 compared to the low-dose group, Δp < 0.05 compared to the medium-dose group.
The serum VEGF level was significantly higher in the medium- and high-dose groups than in the other two groups (all p < 0.05), while there was no significant difference between the medium- and high-dose groups (Fig. 5 and Table 1). These results demonstrate that transplanted BM-MSCs can significantly elevate the expressions of bFGF in the peri-infarct zone and the serum VEGF level.

Transplanted BM-MSCs elevated the serum VEGF level. Seven days after transplantation, the serum VEGF level was measured by ELISA. ∗p < 0.05 compared to the negative control group, #p < 0.05 compared to the low-dose group.
Discussion
In this study, we used three different doses to perform BM-MSC transplantation in MCAO rats. The results showed that BM-MSCs can migrate to the infarct zone. Compared with the low-dose and control groups, medium-dose and high-dose BM-MSC transplantation significantly reduced the volume of the infarct area. In addition, the transplanted BM-MSCs enlarged the diameters of the pial vessel and the basilar artery and increased the capillary area in the peri-infarct zone of the cerebral cortex. Furthermore, the transplanted BM-MSCs elevated the expression of bFGF in the peri-infarct zone and serum VEGF level.
In our experiments, P5 BM-MSCs were used for transplantation because P5 BM-MSCs were of high purity and exhibit a homogeneous population. Although P2 or P3 BM-MSCs had a higher proliferation rate than P5 cells (data not shown), our data demonstrated that P5 BM-MSCs were still in the logarithmic phase of growth (Fig. S1B, supplementary material available at https://drive.google.com/open?id=0B6vjgOZzIwv_SFpfUlVNQXlrU1E). In this study, cell tracker marker CM-DiI was used to label BM-MSCs before transplantation. CM-DiI labeling has been shown to have no negative effects on cell vitality, proliferation, and functionality 30 , which has been widely applied to track cells in the cell transplantation studies31,32. Our data also showed that there was no significant difference in the growth kinetics between BM-MSCs and CM-DiI-labeled BM-MSCs (all p > 0.05) (Fig. S1B, supplementary material available at https://drive.google.com/open?id=0B6vjgOZzIwv_SFpfUlVNQXlrU1E), indicating that CM-DiI labeling has no negative effects on cell proliferation.
CM-DiI-labeled BM-MSCs could be observed in the peri-infarct zone at 7 days after intravenous transplantation, suggesting that the transplanted BM-MSCs possess the ability to pass through BBB and localize to the injured area in the brain. The mechanism underlying BM-MSC transmigration across the BBB is not fully understood. BM-MSCs have been shown to express lymphocyte homing receptors, such as chemokine receptors CXCR4 and CCR2, and cell adhesion molecules CD44, CD99, integrin α4, and integrin β1, which mediate cell adhesion on the vessel wall 33 . Some evidence suggests that transplanted and circulating BM-MSCs may cross the BBB as a leukocyte via rolling, adhesion, and transmigration34–36.
We utilized TTC staining to quantify the infarct size in this study. TTC staining is an efficient and economic method, which is widely used for determining infarct size. In addition to TTC staining, several currently available methods, such as MRI-based methods T2-weighted imaging (T2WI) and diffusion-weighted imaging (DWI) 37 , as well as neuronal nuclear antigen (NeuN) staining 38 , are of high sensitivity and could be adopted for quantification of infarct size. Our results showed that intravenous transplantation of BM-MSCs significantly reduced the infarct volume of MCAO rats and increased the capillary area in the peri-infarct zone of the cerebral cortex, which is in agreement with the findings of our previous study. Results from animal studies have shown that ischemia triggers angiogenesis in the brain and heart and is correlated with reduced injury39,40. In humans, ischemic stroke-induced angiogenesis seems more developed in the ischemic penumbra 41 . It has been shown that intravenous transplantation of BM-MSCs 24 h after ischemia increased the number of enlarged and thin-walled blood vessels and the number of newly formed capillaries at the peri-infarct zone of rats 42 . In an ischemic situation, BM-MSCs may enhance angiogenesis through upregulation of the VEGF–vascular endothelial growth factor receptor 2 (VEGFR2) signaling pathway and expression of phosphorylated v-raf-leukemia viral oncogene 1 and extracellular signal-regulated kinase 1/2 (ERK1/2), which eventually reduces the damaged zone 43 .
After occlusion of the cerebral artery, redistribution of cerebral blood flow increases shear stress in collateral small vessels, which in turn induces cell proliferation and structural remodeling of existing vessels, and eventually formation of permanent vasodilation in the compensatory collateral circulation 44 . We observed vasodilation of pial vessels in the peri-infarct area 7 days after BM-MSC transplantation. Our result also demonstrated that BM-MSC transplantation induced vasodilation in the basilar artery, which would further improve the blood supply in the ischemic region. The BM-MSC-induced permanent vasodilation may be attributed to the fact that ischemia stimulates the homing BM-MSCs to secrete bFGF, VEGF, and other angiogenic growth factors 45 . However, the underlying mechanism remains to be further investigated.
BM-MSC transplantations have been demonstrated to be capable of stimulating the expression of various growth factors, such as brain-derived neurotrophic factor (BDNF), nerve growth factor, bFGF, and VEGF in the ischemic zone. These factors can facilitate functional recovery, such as vascular remodeling 46 . Consistent with these observations, our data showed that BM-MSC transplantation upregulated the protein expression of bFGF in several tissues in the injured side of the cerebrum. bFGF mediates migration and proliferation of endothelial cells, and smooth muscle cell migration, proliferation, and differentiation into tubelike structures, as well as formation of compensatory collateral circulation 47 . Exogenous bFGF has been proven to promote angiogenesis in an animal model of cardiac infarction 48 and in a clinical trial of patients with critical limb ischemia 49 . Intravenous transplantation of BM-MSCs increased bFGF expression in the ischemic boundary area and improved functional recovery in a rat stroke model 13 . In our study, migration of transplanted BM-MSCs to the injured area may also enhance the expression of bFGF in the injured cerebral tissue, which in turn promotes formation of compensatory collateral circulation.
Our result demonstrated that BM-MSC transplantation elevated the serum VEGF level in MCAO rats. VEGF is mitogenic for endothelial cells and has the potential to increase vascular permeability. Intranasal administration of VEGF has been shown to promote angiogenesis and functional recovery in a rat stroke model 50 . In humans, serum VEGF levels are correlated with long-term prognosis in acute ischemic stroke patients, and an increase in serum VEGF level in the acute stage was proportional to an improved National Institutes of Health Stroke Scale (NIHSS) score after 3 months 51 . Therefore, in our study, the BM-MSC-induced elevated serum VEGF level may mediate vascular remodeling, including vasodilation of pial vessels and the basilar artery, and the increased capillary area in the peri-infarct zone.
Although growing evidence in preclinical and pilot studies has suggested benefits of administration of BM-MSCs in stroke 52 , a randomized controlled trial by Prasad et al. has shown that intravenous administration of autologous BM-MSCs has no beneficial effect on stroke outcome 53 . Possible reasons accounting for these discrepancies may be stroke severity of patients and timing and dose of cell administration. In the study by Prasad et al., only moderate stroke patients were included.
Timing of cell administration also affects the therapeutic efficacy. An animal study suggested that cell transplantation should be performed as soon as possible after ischemia to obtain better therapeutic outcomes 29 . However, in clinical studies, only patients after 1 week postischemia could receive cell transplantation because the patient's condition is not completely stabilized within a week postischemia. In Prasad et al.'s study, patients received transplantation at 2–4 weeks after ischemia, which may reduce the therapeutic efficacy. In our animal study, we conducted a preliminary experiment to investigate the optimal time point for BM-MSC transplantation. Our preliminary results showed that at 72 h postischemia, the infarct size was more homogeneous (data not shown). In addition, at 72 h postischemia, the effects of anesthesia (used in model generation) were largely eliminated. Therefore 72 h postischemia was selected as time window for BM-MSC transplantation. An appropriate timing of cell injection is crucial for optimal efficacy in both preclinical and clinical studies.
Studies focusing on the optimal cell dose for intravenous transplantation of BM-MSCs are limited. In the present study, we tested three different cell doses to determine an appropriate one for BM-MSC transplantation. The results showed that low-dose (5 × 10 4 ) BM-MSC transplantation had almost no therapeutic efficacy compared with the control group, whereas medium-dose (5 × 10 5 ) and high-dose (2 × 10 6 ) BM-MSC transplantation significantly improved cerebral ischemia-induced impairment. Our data also showed that there were no significant differences between the medium-dose and high-dose groups in all the evaluations, except for the diameter of the basilar artery and bFGF expression in the cerebral cortex, indicating that their therapeutic efficacies were comparable. This observation is consistent with previous studies29,54, suggesting that there is an optimal threshold of cell number for transplantation, and transplantation of a cell number exceeding the optimal threshold does not further improve the therapeutic efficacy. In addition, our previous study showed that intravenous administration of 5 × 10 7 BM-MSCs resulted in a high mortality rate in transplanted rats due to cell aggregation and pulmonary embolism. In the current study, rats in the three transplanted groups all survived for 1 week after transplantation. It is noteworthy that rats in the high-dose group had a significantly larger basilar artery diameter and significantly higher bFGF expression than rats in the medium-dose group, indicating that high-dose transplantation may be more beneficial than medium dose for vascular remodeling after ischemic stroke. Wang et al. 29 demonstrated that intravenous transplantation of 1 × 10 6 , 2 × 10 6 , and 1 × 10 7 BM-MSCs significantly reduced infarct volume, and transplantation of 2 × 10 6 and 1 × 10 7 BM-MSCs significantly improved the motor performances in rats after ischemia. There was no significant difference in histological and behavioral results between a cell dose of 2 × 10 6 and 1 × 10 7 BM-MSCs. Taken together with our results, the data suggest that intravenous BM-MSC transplantation at cell number greater than 5 × 10 5 has an effective therapeutic effect on vascular remodeling and reducing the infarct volume, and the optimal threshold of cell number for BM-MSC transplantation is between 2 × 10 6 and 1 × 10 7 .
In summary, our study demonstrated that BM-MSC transplantation promoted expression of bFGF in the periinfarct zone, elevated serum VEGF level, induced permanent vasodilation in pial vessels and the basilar artery, and increased the capillary area in the peri-infarct zone. Administration of 5 × 10 5 BM-MSCs is an appropriate cell dose for ischemic stroke therapy in rats. Our findings may be helpful for designing future clinical trials of BM-MSC transplantation for ischemic stroke therapy.
Footnotes
Acknowledgments
This work was supported by the Guangdong Technology Department (2013B022000098 and 2015A030313454), the National Natural Science Foundation of China (81471292, U1503222, and 81430021), the keypoint Science Foundation of Guangdong of China (2015A030311021), a grant supported by the technology project of Guangzhou (20151260), and a grant supported by the assisting research project of science and technology for Xinjiang (201591160). The authors declare no conflicts of interest.
