Abstract
The in vivo repopulation of hepatocytes depends on donor cell growth potential and recipient conditioning. We herein demonstrate the successful cell transplantation of a human hepatocyte cell line, THLE-5b, into the SCID mouse liver by means of a rather mild conditioning using a 55% hepatectomy and p21 transfection. Adult human liver-derived cells, THLE-5b, are SV40 T antigen-immortalized epithelial cells. A phenotypic examination of THLE-5b showed they expressed hepatic stem cell markers such as EpCAM, OCT3/4, and Thy-1, thus indicating the immature nature of the cells. A three-dimensional aggregate culture of THLE-5b showed a higher expression level of liver-specific genes such as albumin, α1-antitrypsin, and CYP3A4, thus suggesting that THLE-5b possess the capability to differentiate into hepatocytes. In a cell transplantation experiment, the cell cycle regulator p21 was transfected with adenoviral vector into the SCID mouse liver. On the next day, 8 × 105 cells of GFP-transfected THLE-5b were injected intrasplenically, together with the intraperitoneal administration of anti-asialo GM1 antibodies. The following day, a partial hepatectomy was performed. The GFP-THLE-5b cells were observed to have migrated and become integrated into the liver parenchyma 14 days after transplantation. The present protocol is thus considered to be a novel experimental model to elucidate the mechanism of hepatocyte repopulation and to develop efficient stem cell therapy in the liver.
Introduction
Although some cases of clinical hepatocyte transplantation have been reported (2,4,11), the therapeutic performance is not yet considered to be sufficient for organ transplantation and this treatment is recognized as being a bridge to liver transplantation. In experimental hepatocyte transplantation, preconditioning of the host liver damage caused by hepatotoxins is indispensable for successfully inducing the repopulation of transplanted cells (1,5,6,10,12,15,17). Otherwise, transplanted cells are normally quickly eliminated from the host liver, probably due to inflammatory and immune responses. In addition, the damage of transplanted cells by the isolation and culture process will be another factor that makes it difficult to overcome the host response.
Clinically, there are two target disorders. One is the occurrence of congenital metabolic disorders and the other hepatic failure (2,8,9,11). The former diseases, especially, single gene mutations such as ornithine transcarbamylase deficiency, Crigler-Najjar syndrome, glycogen storage disease, etc., will be a good indication of hepatocyte transplantation, because the host liver cells are almost completely intact except for the responsible gene and the transplantation of cells that compensate for any defect is desired due to medical reasons rather than a whole liver replacement from an allogeneic donor. However, the therapeutic outcome of hepatocyte transplantation is not equivalent (and not even semiequivalent) to liver transplantation. On the other hand, surgeons do not seem to be encouraged to try hepatocyte transplantation for hepatic failure such as fulminant hepatic failure and liver cirrhosis, because there are fewer cases reported than for metabolic disorders (2,11). Under those conditions, the host cells are damaged in a manner similar to that of experimental cell transplantation but the circumstances will be so severe, not only for the host cells but also for any transplanted cells to survive. Therefore, a more feasible target of hepatocyte transplantation will be congenital metabolic disorders, although the preconditioning of host liver damage is clinically unacceptable.
The present report demonstrates a successful protocol for hepatocyte transplantation using a hepatocyte progenitor-like cell line and cell cycle regulator. The protocol is not applicable for clinical cell transplantation, but it is considered to help elucidate the mechanism of repopulation and efficient stem cell therapy.
Materials and Methods
Cell Line and Primary Human Hepatocytes
The human hepatocyte cell line, THLE-5b, was originally established from human primary hepatocytes by the transfection of SV40 T antigen and it was kindly provided by Dr. Harris (National Cancer Institute, Bethesda, MD, USA) (7). The cells were maintained by culturing in RPMI-1640 containing 10% fetal bovine serum and the cell split ratio at passage was 1:5. A stable transfectant of THLE-5b with green fluorescent protein (GFP) was established by the lipofectamine transfection of the GFP gene and subsequent single cell cloning. Human parenchymal hepatocytes as a source of mRNA were obtained from the HAB Research Organization (Tokyo, Japan).
Primers
The primers used for reverse transcription and polymerase chain reaction (RT-PCR) were as follows. Octamer binding transcription factor 4 (Oct-4): sense 5′-gacaacaatgaaaatcttcaggaga-3′ and antisense 5′-ttctggcgc cggttacagaacca-3′; epithelial cell adhesion molecule (EpCAM): sense 5′-ctggccgtaaactgctttgt-3′ and antisense 5′-agcccatcattgttctggag-3′; Thy-1: sense 5′-ctagtggacca gagccttcg-3′ and antisense 5′-tggagtgcacacgtgtaggt-3′; c-kit: sense 5′gtgaatctacttggagcctg-3′ and antisense 5′-tcattcttgatgtctctggc-3′; glucose-3-phosphate dehydrogenase (G3PDH): sense 5′-accacagtccatgccatcac-3′ and antisense 5′-tccaccaccctgttgctgta-3′; α1-antitrypsin (AAT): sense 5′-tcttctccacaccctcaacc-3′ and antisense 5′-cggtgt ctttgtcaagctca-3′; albumin: sense 5′-tgtgttgctgatgagtca gc-3′ and antisense 5′-tcagcagcaatgagacagag-3′; cytochrome P450 1A1 (CYP1A1): sense 5′-tcccctcttgaat gaaggtg-3′ and antisense 5′-atccgacagctggatattgg-3′; cytochrome p450 3A4 (CYP3A4): sense 5′-tcctcagcca gagagagagc-3′ and antisense 5′-ggctaccgtaagggtgttga-3′; p21Cip1: sense 5′-ccaggaggcccgtgag-3′ and antisense 5′-ggtaccacccagcggacaag-3′.
p21Cip1 Adenoviral Vector
A replication incompetent adenovirus, AxCAp21, which contains a chicken β-actin promoter with cytomegalovirus enhancer and the human p21Cip1 gene was constructed as described by Terada et al. (13). A replication incompetent adenovirus containing the lacZ gene, AxCALacZ, generously provided by Dr. Saito (3) was used in the nontreatment control group. The viral preparations were purified using a cesium chloride density gradient and then were titrated with a plaque-forming assay on HEK293 cells.
Reverse Transcription and Polymerase Chain Reaction (RT-PCR)
RT-PCR was performed according to the method described in previous reports (14,16). Briefly, total RNA was isolated by ISOGEN (Nippon Gene, Toyama, Japan) and converted into double-stranded cDNA, using ReverTraAce-alpha Kit (Toyobo, Osaka, Japan). The PCR program consisted of denaturation at 94°C for 1 min, annealing at 55°C for 1 min, and extension at 72°C for 2 min, and 25–40 cycles were applied.
Cell Aggregate Formation and Immunohistochemistry
THLE-5b cells (5 × 105) in 3 ml of the medium were cultured in a 25-ml Meyer's flask that was precoated with 2.5% poly(2-hydroxyethyl methacrylate) (P-3932, Sigma, St. Louis, MO, USA) in ethanol to avoid cell adhesion to the wall. After incubating at 37°C, 5% CO2 with continuous rotation at 70 rpm on an orbital shaker for 72 h, the formed aggregates were harvested for RT-PCR or an immunohistochemical study. For immunohistochemistry, the samples were fixed first with acetone/methanol (1:1), followed by 10% neutralized formalin. The paraffin sections were stained with rabbit anti-human AAT antibodies (Ylem SRL, Roma, Italy) and visualized with alkaline phosphatase-conjugated secondary antibodies.
Assessment of Liver Regeneration in SCID Mouse
A male severe combined immunodeficient (SCID) mouse (CB17, 6–14 weeks old, specific pathogen-free grade, CLEA Japan, Tokyo, Japan) was injected with adenoviral vector AxCAp21 (1 × 108 pfu in 1 ml saline) via tail vein and nontreated controls received the same dose of AxCALacZ. On the next day, the left lateral lobe of the liver was excised (55% partial hepatectomy). One hour before harvesting the liver, 100 mg/kg of bromodeoxyuridine (BrdU, B-5002, Sigma) was injected intraperitoneally to label DNA synthesizing cells. The liver sample was weighed and a portion of liver tissue was embedded in OTC compound (Sakura Seiki, Tokyo, Japan) and a section 4 μm thick was stained for BrdU immunohistochemically (BrdU Labeling and Detection Kit I, Roche Diagnostics, Mannheim, Germany). The ratio of stained cells to the total number of cells was determined by image analysis. The p21 expression in the liver was estimated by RT-PCR on days 0, 2, and 8.
THLE-5b Cell Transplantation Into SCID Mouse
The mouse was administered p21 adenoviral vector as described above 1 day before hepatocyte transplantation. GFP-transfected THLE-5b cells (8 × 105 in 0.1 ml saline) were injected intrasplenically together with the intraperitoneal administration of 100 μg of anti-asialo GM1 rabbit antibodies (Wako Pure Chemicals, Osaka, Japan) to inactivate natural killer cells. On the next day the left lateral lobe of the liver of removed (55% partial hepatectomy). The liver was excised 14 days after cell transplantation and embedded in OTC compound. Liver cryosections (4 μm thickness) were investigated using fluorescent microscopy.
Quantification of Cell Proliferation and Statistical Analysis
Image analysis of the proliferating cell ratio was performed using the NIH Image software program from the National Institutes of Health. The value for each group was obtained from three independent experiments as a mean ± SD, and the value in each sample was the mean of three separate areas. Statistical significance was determined by one-way ANOVA and Fisher's PLSD (Protected Least Significant Difference) test using the StatView software program, version 5.0 for Macintosh (SUS Institute, Cary, NC, USA).
Ethical and Regulatory Considerations
Experimental procedures using human primary hepatocytes, experimental animals, and recombinant viral vectors were performed under permission of the institutional review board, animal ethics committee, and experimental recombinant DNA committee of National Center for Child Health and Development (permission numbers: 98, 00-001, 05-18).
Results
Phenotypic Properties of THLE-5b Cells Under Monolayer and Aggregate Conditions
The expression of the undifferentiated cell markers, EpCAM, OCT4, c-Kit, and Thy1, was more obviously prominent in the THLE-5b cells than in the hepatic parenchymal cells (Fig. 1). In contrast, the expression of AAT and CYP3A4, the markers for well-differentiated parenchymal hepatocytes, was not very strong in these cells.

Reverse transcription and polymerase chain reaction (RT-PCR) profile of THLE-5b (T) and human hepatocytes (H). Hepatocytes were isolated from a 56-year-old Caucasian. EpCAM, epithelial cell adhesion molecule; OCT4, octamer binding transcription factor 4; ES, embryonic stem; HSC, hepatic stem cell; AAT, α1-antitrypsin; G3PDH, glyceraldehyde-3-phosphate dehydrogenase. General information regarding each molecule is indicated in the right column.
The expression of albumin, CYP3A4, CYP1A1, and AAT mRNA, which are considered to be parenchymal hepatocyte markers, was upregulated under the aggregation culture of THLE-5b (Fig. 2). The THLE-5b cells showed a fibroblast-like morphology under the monolayer culture (Fig. 2A). An immunocytochemical study of THLE-5b showed AAT and albumin (albumin data not shown) to be stained only in the aggregates (Fig. 2A′, B), thus suggesting that the cell character changed to mature hepatocytes along with the aggregate formation.
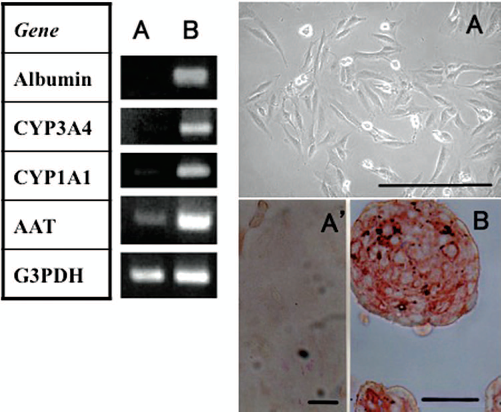
RT-PCR profile and microscopic morphology of THLE-5b cells under monolayer culture (lane A) or aggregate culture (lane B). (A) Phase contrast microscopy, (A′) immunostaining of THLE-5b under monolayer culture stained for α1-antitrypsin (AAT) (no positive staining was detectable), (B) immunostaining of THLE-5b under aggregate culture stained for AAT. AAT was stained red by alkaline phosphatase-conjugated secondary antibodies (B). Scale bars: 25 μm.
The Retardation of Liver Regeneration After p21 Transfection and THLE-5b Cell Transplantation
The transfection of p21 obviously delayed liver regeneration after a 55% partial hepatectomy (Fig. 3A). The weight of the remnant liver of the p21 transfected mouse decreased significantly on day 6 after a hepatectomy but survived thereafter. Since the hepatic parenchyma is a preferential site of adenoviral vector, the expression of human p21 was clearly confirmed on days 2 and 8 by RT-PCR (Fig. 3A, inset). The transfection also depressed cell proliferation after a hepatectomy (Fig. 3B). In a mouse hepatectomy, the peak of the proliferating activity is around day 3, as shown in Figure 3B, whereas the peak in the rat is day 1 (preliminary data). When the mice were transfected with p21, cell proliferation was clearly suppressed. In addition, the p21-transfected mice that received a 70% hepatectomy (median and lateral lobes) died within 3 days (data not shown). Therefore, the cell transplantation experiment was performed by removing only the left lateral lobe (55% hepatectomy).

Liver weight (A) and cell proliferation rate (B) after a left lateral robe resection (55% hepatectomy) with p21 (open circles, mean ± SD, n = 3) or lacZ (control) (filled circles, mean ± SD, n = 3) transfection. Transfection was done 1 day before. The inset in (A) shows the p21 expression by RT-PCR in the liver on the day indicated. *p < 0.05.
When the GFP stable transfectant of THLE-5b was transplanted, only the p21-transfected hepatectomized mice accepted the cells. When transplanted into either the p21-transfected or hepatectomized mice, no cells survived (data not shown). The GFP-THLE-5b cells were dispersed in hepatic parenchyma (Fig. 4A). At higher magnification, some cells were observed to form a small cluster (Fig. 4B), thus suggesting the occurrence of cell division after integration in the tissue.
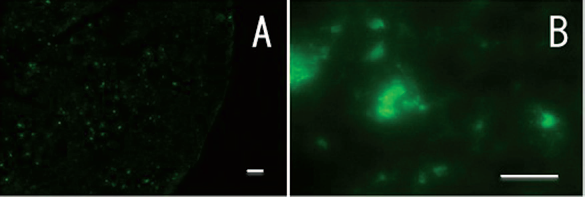
GFP-transfected THLE-5b survived in the sever combined immunodeficient (SCID) mouse liver 14 days after implantation. The cells were transplanted intrasplenically 1 day after p21 transfection and a partial hepatectomy was done 1 day after cell transplantation. A cryosection was observed using fluorescence microscopy. Original magnification: 40x (A), 400x (B). Scale bars: 50 μm.
Discussion
THLE-5b cells are a relatively new cell line established from adult human hepatocytes, using SV40 T antigen. In spite of immortalization, THLE-5b cells are not tumorgenic (7). Interestingly, these cells exhibit hepatic progenitor-like characteristics. Under monolayer culture conditions THLE-5b expressed undifferentiated markers, such as EpCAM, OCT4, c-kit, and Thy-1 but not AAT and CYP3A4, which are the typical differentiation markers of parenchymal hepatocytes (Fig. 1). The cells looked slightly like fibroblasts without expressing AAT protein, (Fig. 2A, A′). However, when grown as cell aggregates, THLE-5b expressed AAT (Fig. 2B). We previously reported that the aggregated form of THLE-5b showed a hepatocyte-like ultrastructure, ammonia metabolic activity, and several other indicators that suggest hepatocytic maturation, including the upregulation or induction of liver-specific genes such as albumin and tryptophan 2,3-dioxygenase, and the downregulation of biliary cell markers such as γ-glutamyl transpeptidase (14). Transcription factors such as hepatocyte nuclear factor (HNF)-1 and HNF-4α were also upregulated or induced. These results strongly indicated that THLE-5b should be considered a progenitor cell line. Recently, we detected side population cells in THLE-5b by a flow cytometry analysis after Hoechst 33342 staining (16).
Cell cycle regulator, p21Clip1 clearly suppresses the cell cycle and its artificial expression sometimes makes it possible to overcome proliferative inflammation (13). Its physiological function is effective across the species. Actually, we confirmed that the transfection of human p21 caused a decrease in the thymidine uptake of both rat and mouse primary hepatocytes with no sign of cell death (preliminary data). The use of p21 in combination with a 55% hepatectomy enabled the survival of transplanted cells under relatively milder conditions than the intoxication of carbon tetrachloride (1), retrorsine (6,17), heterotopic expression of tissue plasminogen activator (12), and anti-Fas antibody (15). Indeed, the present protocol itself is not yet recommended for the clinic, but this will be a useful experimental model for the development of both cell transplantation and stem cell therapy. These results therefore imply that bipotent progenitor cell populations similar to THLE-5b cells may exist in the adult human liver.
Footnotes
Acknowledgments
We thank Dr. Harris (Laboratory of Human Carcinogenesis, National Cancer Institute, Bethesda, MD, USA) for providing THLE-5b cells and Dr. Saito (University of Tokyo, Japan) for providing adenoviral AxCALacZ. We also thank Mr. Kazuhito Sato for his excellent technical assistance. This research was supported by Grant-in-aid (KHD1023 and 1027) from the Japan Health Sciences Foundation, Tokyo, Japan. The authors declare no conflict of interest.
