Abstract
Primary human fetal and adult hepatocytes have been considered feasible donor cell sources for cell transplantation. We compared the engraftment efficiencies between adult human, fetal human, and adult porcine hepatocytes after transplantation into MUP–uPAtg(+/+)Rag2-/-γC-/-mice. Transplantation of adult human hepatocytes yielded a 1,000-fold higher serum albumin level compared to transplantation of fetal human hepatocytes, while transplantation of adult porcine hepatocytes resulted in a 100-fold higher serum albumin level than adult human hepatocytes. These results suggest that adult liver cells are superior to fetal liver cells for transplantation, and caution should be applied if porcine hepatocytes are used for preclinical studies as a proof of concept for human hepatocytes.
Keywords
Introduction
During development, fetal cells have abundant proliferative capacity and differentiation potential, which makes them an attractive cell source for regenerative medicine. However, transplantation of fetal liver cells is still controversial, due to the conflicting reports about their engraftment capability (2,7,12,16). In this report, we investigated and compared the engraftment efficiencies of three different donor hepatocyte sources: murine, porcine, and human hepatocytes under similar conditions of transplantation into a new stable immunodeficient animal model of liver failure.
Chimeric human–mouse livers (“humanized livers”) have been generated previously in immunodeficient mice that lack the fumarylacetoacetate hydrolase gene (Fah-/-) or that overexpress the urokinase-type plasminogen activator (uPA) (1,24). The Fah-/- mouse is invaluable for liver cell transplantation with one caveat; no studies can exceed 9 months due to the spontaneous generation of hepatocarcinoma in older animals (6). In albumin–uPA mice, uPA expression is controlled by the albumin enhancer/promoter, which results in diffuse vacuolization within hepatocyte rough endoplasmic reticulum that increases in severity with age and is accompanied by sporadic hepatocyte death (20, 27). Transplantation of healthy donor hepatocytes in this environment led to high repopulation of human donor cells (24). However, these mice have increased neonatal death due to frequent hemorrhaging and require hepatocyte transplant at an early age because the albumin promoter is activated during embryonic development (8,20,24,27). On the other hand, the major urinary protein (MUP) promoter is activated approximately 2–3 weeks after birth in the liver, which eliminates neonatal hemorrhaging and allows for postnatal transplantation that is not possible in albumin–uPA mice (27). Therefore, we crossed MUP–uPA mice with the immunodeficient recombination-activating gene 2 interleukin (IL-2) receptor common γ chain double knockout (Rag2-/-γC-/-) mice, which lacks B, T, and natural killer (NK) cells, to allow cell repopulation from xenogeneic donors, to alleviate neonatal lethality, and to expand the window of opportunity for hepatocyte transplant (25). Our aim was to establish and validate this new animal model for human hepatocyte transplantation and compare side by side the degree of liver repopulation of various hepatocyte populations.
Materials and Methods
Animals
Green fluorescent protein (GFP) transgenic mice [C57BL/6-Tg(UBC-GFP)30Scha/J] were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). To generate MUP–uPAtg(+/+)Rag2-/-γC-/- mice, MUP–uPA mice (a kind gift from Dr. Eric Sandgren, University of Wisconsin-Madison, WI, USA) were crossed with Rag2-/-γC-/- mice (Taconic, Germantown, NY, USA). PCR-based genotyping was carried out on 200 ng genomic DNA isolated from tail tissue as previously described (24). Sow 6 weeks of age (around 70 kg) were purchased from Animal Biotech Industries (Danboro, PA, USA). Animals were bred and housed in the Division of Laboratory Animal Resources facility at the University of Pittsburgh Center for Biotechnology and Bioengineering. Experimental protocols followed the National Institutes of Health guidelines for animal care and were approved by the Institutional Animal Care and Use Committee at the University of Pittsburgh.
Preparation of Primary Hepatocytes From Mouse, Human, and Swine
All research protocols were reviewed and approved at the University of Pittsburgh by the Institutional Review Board for Human Research Studies and the Institutional Animal Care and Use Committee for murine and porcine studies. The tissue dissociation and subsequent hepatocyte isolation procedures used were developed by Seglen (21) and modified as described previously (4). For adult human liver (gender not identified), a total of 12 liver tissues were used in this study. Most of the patients (age 20 to 80) were undergoing scheduled liver resection, and residual liver tissue not needed for diagnostic purposes was transported to the laboratory from the operating rooms in cold Eagle's minimum essential medium (EMEM; Lonza, Atlanta, GA, USA) within 30 min of removal. Most of these liver resections were the result of metastatic colon cancer. Several livers were obtained from non-heart-beating donors and were reported to have 15 and 40 min of warm ischemia. Fetal human liver tissues were obtained from the tissue bank at the Magee Women's Hospital of the University of Pittsburgh Medical Center (UPMC). Samples were between 19 and 23 weeks of gestation (gender not identified). Fetal livers were placed in Hank's balanced salt solution (HBSS; Thermo Scientific, Waltham, MA, USA) and minced into small pieces. Liver samples were incubated with Earle's balanced salt solution (EBSS; Thermo Scientific), 10 mM ethylene glycol tetraacetic acid (EGTA; Sigma, St. Louis, MO, USA), and 1% 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; Cellgro, Manassas, VA, USA) for 15 min at 37°C and treated with 1 mg/ml collagenase II (Life Technologies, Carlsbad, CA, USA) + 1 mg/ml hyaluronidase (Sigma) + 100 μg/ml of DNaseI (Roche, Basel, Switzerland) for 1–1.5 h to obtain a cell suspension. Adult mouse hepatocytes were isolated using the classic two-step collagenase perfusion technique described by Seglen (21) from 6- to 12-week-old mice. Adult porcine hepatocytes were isolated using the same method and enzymes as for human adult hepatocytes. Briefly, the Landrace pigs (females, 6 weeks of age and 70 kg) underwent a partial hepatectomy (left lateral resection), and the graft was immediately flushed with cold solution (4°C). A resected liver segment was transported (ice chest) to the lab in a sterile plastic bag while fully immersed at 4°C. The cell isolation was performed within 1 to 3 h from the initial hepatic resection as described previously (4). Cell viability for each fetal or adult hepatocyte preparation was assessed by mixing an aliquot of the final cell suspension with an equal volume of 0.4% (w/v) trypan blue (Cellgro) in phosphate-buffered saline (PBS; Mediatech, Manassas, VA, USA) and counting only the number of viable (unstained) and dead (blue) hepatocytes with the aid of a hemocytometer (Hausser Scientific, Horsham, PA, USA). Viabilities are expressed as a percentage of the total hepatocyte number.
Splenic Injection of Murine, Human, and Porcine Hepatocytes
One million viable hepatocytes isolated freshly in 50 μl of HBSS were injected into the spleen via a 28-gauge needle of an insulin syringe.
Human and Porcine Albumin Measurement
Blood collection (100 μl) was performed using the submandibular bleeding technique every 2–4 weeks. Human and porcine albumin concentration was measured with the Human and Porcine Albumin Enzyme-Linked Immunosorbent Assay (ELISA) Quantitation Kit, respectively (Bethyl, Montgomery, TX, USA) according to the manufacturer's protocol.
Immunohistochemistry
Frozen sections (5 μm) were fixed in cold acetone (Thermo Scientific) for 5 min. For immunohistochemistry staining, sections were washed with PBS and blocked with 5% skim milk. Sections were then incubated with primary antibody, goat anti-human, or porcine albumin (1:200; Bethyl Laboratories, Inc.) for 1 h; secondary antibody, Alexa Fluor 594 anti-goat IgG (1:200; Invitrogen, Carlsbad, CA, USA) 1 h; and a third antibody, fluorescein isothiocyanate (FITC) conjugated anti-mouse albumin (1:200; Bethyl Laboratories, Inc.) for 1 h. Sections were mounted with Hoechst mounting media (Life Technologies). Images were captured with an Olympus IX71 inverted microscope (Olympus, Center Valley, PA, USA).
Statistical Analyses
Statistical significance was determined with an unpaired two-tailed Student's t test and Bonferroni correction (Prism; Graphpad, San Diego, CA, USA).
Results
Generation of MUP–uPAtg(+/+)Rag2-/-γC-/- (uRG) Mice
In MUP–uPA mice, hepatocyte damage begins to increase at 4 weeks of age, peaks at 5 weeks, and returns to normal levels (no damage) by 13 weeks of age (27). Two-to 4-week-old recipients demonstrated high repopulation of healthy donor hepatocytes 4 weeks after transplantation (27). First, we transplanted, via splenic injection, C57BL/6 GFP+ mouse hepatocytes into immunocompetent MUP–uPA mice (C57BL/6 background). Repopulation 4 weeks after transplant in the livers of homozygous MUP–uPA transgenic [MUP–uPAtg(+/+)] mice was distinctly higher than that in heterozygous MUP–uPA transgenic [MUP–uPAtg(+/-)] mice, due to the increased uPA expression in homozygous mice (Fig. 1). This result was similar to what was observed with the albumin–uPA mice (19,20). As a result, MUP–uPAtg(+/+)Rag2-/-γC-/- (uRG) mice were used for all syngeneic and xenogeneic transplantations to maximize engraftment efficiencies.
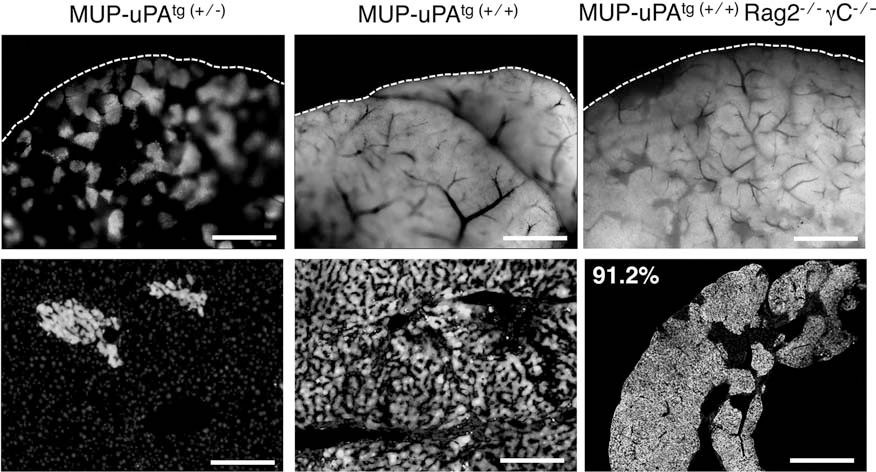
Repopulation in MUP–uPA mice and MUP–uPAtg(+/+)Rag2-/-γC-/- (uRG) mice following splenic injection of GFP-expressing mouse hepatocytes. Top panels: whole-mount imaging of the transplanted liver 4 weeks after splenic injection of donor green fluorescent protein-positive (GFP+) hepatocytes (bright gray). Dotted lines indicate the liver boundary. Bottom panels: frozen section with donor GFP+ hepatocytes (bright gray) counterstained with Hoechst 33342 (light gray, primarily seen in the bottom left panel). Scale bar: 1 mm, except bottom left and bottom middle panels (200 μm). MUP–uPAtg(+/+)Rag2-/-γC-/- (uRG): major urinary protein urokinase-type plasminogen activator transgenic immunodeficient recombination-activating gene 2 interleukin (IL-2) receptor common g chain double knockout mouse.
Syngeneic Transplantation of Murine Hepatocytes Into uRG Mice
To define a successful repopulation of hepatocytes in uRG mice, we transplanted 1 × 106 C57BL/6 GFP+ primary adult mouse hepatocytes by splenic injection. The syngeneic repopulation ranged from 91.2% to 99.9% GFP+ hepatocytes engrafted in the liver 4 weeks after transplantation, a level of repopulation similar to what we observed with the immunocompetent MUP–uPa (Fig. 1) or previously with Fah-/- mice (17,18). This result indicates that the cross of MUP–uPA mice with immunodeficient Rag2-/-γC-/- mice does not affect the evolution of liver disease or the engraftment of wild-type hepatocytes in this animal model.
Xenogeneic Transplantation of Porcine Hepatocytes Into uRG Mice
Next we transplanted 1 × 106 primary adult porcine hepatocytes from six donors into C57BL/6 mice (n = 4), MUP–uPA mice (n = 4), and uRG mice (n = 17). One uRG mouse died before first bleeding at 2 weeks after transplantation for unknown reasons. Serum porcine albumin level in uRG mice reached over 5 mg/ml at 2 weeks after transplant, while, as expected, the levels in immunocompetent C57BL/6 and MUP–uPA control mice were undetectable 2 weeks after transplant (Fig. 2A). Serum porcine albumin levels in uRG plateaued 4 weeks after transplantation and remained relatively stable for an additional 10 weeks (Fig. 2B). Splenic injection of 1 × 106 xenogeneic porcine hepatocytes repopulated over 90% of the uRG host liver (Fig. 2C–E), as expected by the high porcine albumin level detected in the mouse serum (over 10 mg/ml). It is interesting to note that, although the porcine albumin concentration is high, it is still not at normal mouse serum levels. Presumably mouse albumin is still expressed from the remaining mouse hepatocytes present in the liver, as demonstrated by immunostaining in Figure 2E. The relationship of host and donor hepatocytes in the context of serum albumin concentration after xenogeneic transplantation in mice has been discussed previously (24).

Donor porcine hepatocyte repopulation in uRG mice. (A) Porcine albumin concentration in mouse blood serum was assayed by enzyme-linked immunosorbent assay (ELISA) 2 weeks after transplant of immunocompetent C57BL/6 (n = 4) and MUP–uPatg(+/+) (n = 4), and immunodeficient MUP–uPatg(+/+)Rag2/γc-/- mice (n = 7). ND stands for not detected. (B) Porcine albumin concentration in mouse blood serum of 16 immunodeficient MUP–uPatg(+/+)Rag2/γc-/- mice 2 to 20 weeks after transplantation. (C) Macroscopic appearance of the mosaic liver after splenic injection of adult porcine hepatocytes in uRG mice. Dark red spots show host remaining murine liver cells. (D) Control immunofluorescence of a normal porcine and mouse liver frozen sections. Porcine albumin is shown in red, and mouse albumin is shown in green. (E) Immunofluorescence of two representative chimeric livers 4 weeks after adult porcine hepatocyte transplantation. Staining of frozen sections is the same as that of the controls above. Of the uRG mouse liver, 94.7% and 94.2% were repopulated with porcine cells with 14.8 mg/ml and 10.8 mg/ml of porcine albumin concentration detected in the mouse serum, respectively. Scale bar: 5 mm (C), 1 mm (D, E).
Strikingly, the high engraftment of xenogeneic porcine hepatocytes (Fig. 2) is at a similar level as syngeneic murine hepatocytes (Fig. 1). Our results suggest that there is no detectable xenogeneic barrier to adult porcine hepatocyte transplantation in uRG mice, although a xenogeneic barrier was present in immunocompetent animals because no transplanted adult porcine hepatocytes engrafted.
Xenogeneic Transplantation of Human Adult and Fetal Hepatocytes Into uRG Mice
Finally, we transplanted 1× 106 primary adult human hepatocytes from 12 different donors (n = 75 mice) and 1 × 106 primary fetal human hepatocytes from five donors (n = 36 mice). Two mice transplanted with adult human hepatocytes and one mouse transplanted with fetal human hepatocytes died before first bleeding 2 weeks after transplantation for unknown reasons. Therefore, we analyzed 108 of 111 transplanted mice (97.3%). The serum albumin level of uRG mice transplanted with adult human hepatocytes was significantly higher (p < 0.05) than in mice transplanted with fetal human hepatocytes 4–5 weeks after transplant (Fig. 3A). Experimental animals were necropsied at the termination of the experiment and tested for species-specific donor blood serum albumin concentration, which correlates with donor repopulation in the host liver (24). Thus, fetal human hepatocytes engraft significantly lower than adult human hepatocytes. It is interesting to note that despite the variability in age or other factors concerning the source of human hepatocytes isolated (see Materials and Methods), serum albumin difference is highly significant between fetal and adult human hepatocyte transplants (p < 0.05). This result might be interpreted that proliferative immature fetal liver cells could produce less “mature functional marker” during liver regeneration. However, mRNA and protein expression levels of human albumin in fetal hepatocytes are at least equal to that of adult hepatocytes (1,15,28).

Donor human hepatocyte repopulation in uRG mice. (A) Human and porcine albumin concentration in uRG mouse blood serum was assayed by enzyme-linked immunosorbent assay (ELISA) 4–5 weeks after transplant. *p < 0.05, ***p < 0.0001 (unpaired two-tailed Student's t test and Bonferroni correction). (B) Comparison of human albumin concentration in uRG mouse blood serum at 4–5 weeks and 8–10 weeks after transplant. Human fetal hepatic engraftment decreases significantly (**p < 0.005, unpaired two-tailed Student's t test), while human adult hepatic engraftment remains constant. (C) Immunofluorescence of a representative chimeric liver 4 weeks after adult human hepatocyte transplantation. Staining of frozen liver sections with anti-human albumin. A small area of the uRG mouse liver was repopulated with human hepatocytes with 0.015 mg/ml of human albumin concentration detected in the mouse serum. Scale bar: 1 mm (left panel) and 200 μm (right panel).
It has been reported that ED14 and ED16 fetal rat liver cells exhibited high repopulation potential under highly selective conditions (retrorsin/partial hepatectomy) at late phase (8 weeks after transplantation) (22). There is a possibility that uRG mice might not be suitable for growth of human fetal liver cells because of the lack of sufficient liver damage to induce proliferation at late phase. To confirm whether the phenomenon in rat can be reproduced after human cell transplantation in uRG mice, we measured albumin levels at 8–10 weeks. Mice transplanted with adult human and porcine hepatocytes maintain their respective albumin levels 8–10 weeks after transplant (Figs. 2B and 3B), while all of the mice transplanted with fetal human hepatocytes had significantly lower albumin levels (p < 0.005) compared to 4 weeks after transplant (Fig. 3B).
Moreover, we compared human engraftment levels in uRG mice with hepatocytes isolated from swine. Serum albumin levels and hepatic cell engraftment after porcine hepatocyte transplant was significantly higher (p < 0.0001) than that of both fetal and adult human hepatocyte transplant (Figs. 2B and 3A). For all transplantation experiments, there was no noticeable difference in donor cell viability by trypan blue exclusion after hepatocyte isolation from the liver. Viability of freshly isolated cells before transplantation was 70.5% for human adult hepatocytes (n = 9), 88% for human fetal hepatocytes (n = 5), and 74.3% for adult porcine hepatocytes (n = 6). In addition, all transplanted cells were isolated from donor livers within a couple of hours of liver resection (see Materials and Methods). Taken together, our results suggest a potential barrier for human hepatocyte transplantation in uRG mice.
Discussion
We have generated a new model suitable for xenogeneic hepatocyte transplantation by crossing MUP–uPA mice with Rag2-/-γC-/- mice. In this study, homozygous MUP–uPAtg(+/+)Rag2-/-γC-/- mice were used for comparison of various donor cells to exclude the potential variability of repopulation due to zygosity of the MUP–uPA transgene. One of the features of this model is that the donor-derived albumin was detectable in serum in 124 of 128 uRG mice (97%) that were transplanted with three different types of liver cells from various aged donors. One potential disadvantage is that long-term liver failure in this model is uncontrollable because native hepatocytes can spontaneously recover from uPA-induced liver failure after 13 weeks of age (20).
The enormous demand to treat liver-diseased patients has prompted increased interest in the clinical application of hepatocyte transplantation. Recently, several groups have tested fetal liver cells as donor cells for hepatocyte transplant in early phase clinical trials (5,13). Fetal hepatocytes are an attractive cell source due to their high proliferative and immature nature. Furthermore, developing fetal liver cells should have the capacity to differentiate into functional adult hepatocytes. However, it is still debatable whether human fetal liver cells have the potential to mature efficiently in a diseased adult liver environment. Our data show in a comparative analysis that fetal human hepatocytes have a significantly reduced engraftment potential compared to adult human hepatocytes in the uRG mice. Our results suggest that the stage of differentiation of human hepatocyte is important for having the most efficient human hepatocyte engraftment in an adult liver environment, and in consequence, fetal liver cells might require additional extrinsic factors to promote efficient engraftment (2,11). It is interesting to note that transplantation of fetal human cells from other tissues, like brain cells, has been promising in rodent models (9,26). Clinical trials of both fetal and adult human hepatocytes have been reported previously in patients with no clear results (3,5,10,13,23), and few studies have compared side by side the degree of liver repopulation of various human cell types in the same recipient. In consequence, very little is known about the potential barriers to human hepatocyte transplantation.
So far, only our study has compared two xenogeneic sources of hepatocytes, human versus porcine, in a mouse model of liver injury. Furthermore, we used larger numbers of samples and animals than previously reported studies. In agreement with others (7,12), we found that fetal human hepatocytes have a limited engraftment when compared to adult human hepatocytes, but unexpectedly, adult porcine hepatocytes have an extraordinary engraftment capability in the uRG mouse model, largely superior to human hepatocytes, and comparable to syngeneic mouse hepatocytes. Why porcine hepatocytes have a unique advantage for cell engraftment, while human hepatocytes are limited in their engraftment potential, is unclear at the moment. Such an advantage does not appear to be due to a reduced xenogeneic immune response, as porcine hepatocytes are promptly rejected in immunocompetent mice. This engraftment potential was not due to viability either, as all three sources of xenogeneic hepatocytes (fetal and adult human, and adult porcine and murine hepatocytes) were freshly isolated, with comparable viability. It seems that intrinsic and/or extrinsic factors that promote engraftment, survival and growth, or unknown alternative mechanism(s) might be responsible for this extraordinary potential. Regardless, it will be interesting to determine why porcine hepatocytes are able to engraft so efficiently, as it could be translated to improve human hepatocyte engraftment. We speculate that the barriers limiting efficient human hepatocyte engraftment in mice may also be present in patients.
Our results also lead to another important point to consider for hepatocyte transplantation; the results generated from adult porcine hepatocyte transplantation for preclinical studies may not necessarily correlate with transplantation of human hepatocytes, despite the xenogeneic similarity (14,29). Therefore, a cautious approach must be taken when trying to determine the best source of donor hepatocytes for clinical use using porcine hepatocytes.
Footnotes
Acknowledgments
We would like to thank Eric Sandgren for providing MUP–uPA mice. This work was supported by the NIH grant R01 DK085711 (J.K., A.D., and E.L.). The authors declare no conflicts of interest.
