Abstract
The methodology of cryopreservation of human hepatocytes remains unsatisfactory. Even when the viability of thawed cells is tolerable, the cells often lose the attaching capability to a culture dish, resulting in the cells' inability to survive. Previously, we described the effectiveness of maltose on the attachment of hepatocytes. This article demonstrates that a silk-derived high molecular protein, sericin, improves the cell-attaching capability in the serum-free freezing medium. When human hepatocytes [initial viability: 60.9 ± 3.1% (mean ± SD, n = 3)] were frozen with serum-free Dulbecco's modified Eagle medium (DMEM) containing 10% dimethyl sulfoxide (DMSO), the viability was 29.4 ± 3.2% and the cell-attaching capability 20.4 ± 4.1%. On the other hand, DMEM containing 10% DMSO and 1% sericin increased the values to 45.0 ± 0.8% and 26.2 ± 3.2%. Moreover, the addition of 0.1 mol/L maltose to the sericin-containing medium improved to 42.2 ± 3.2% and 51.1 ± 1.0%, as we demonstrated in a previous report. The present results indicated that sericin combined with maltose is a novel additive in the serum-free freezing medium for human hepatocytes.
Introduction
An improvement in the cryopreservation of hepatocytes is expected to greatly benefit not only the research field but also the clinical cell transplantation field. The present major requirement is that pharmaceutical companies use human hepatocytes for predicting the metabolism of drug candidates (2). The potential demand will be elevated in clinical cell transplantation and the bioartificial liver. The number of cases of hepatocyte transplantation is gradually increasing, and in some cases the cells were transplanted repeatedly from the same donor to confirm effectiveness (5). Consequently, a reliable cryopreservation protocol needs to be established. However, hepatocytes easily lose their cell viability after cryopreservation.
We noticed that the cell viability immediately after thawing does not reflect the cells' survival in the culture condition. Therefore, we have been trying to improve cell survival by a modification of the freezing medium. Previously, we reported the different efficacies of oligo-saccharides on the attachment capability of cryopreserved hepatocytes and arrived at the conclusion that maltose is the most effective additive (6), while trehalose, which belongs to the same dissacharide as maltose, is thought to be effective elsewhere (3, 4). Therefore, we embarked on the development of a serum-free freezing solution to ensure the safety of cells for clinical transplantation, especially to avoid xenozoonoses.
Sericin is a silk-derived high molecular protein and was reported recently to be useful for cell cryopreservation (7), but the effectiveness for cryopreservation of primary hepatocytes has not been tested. Here we demonstrate that sericin can replace serum in the freezing solution, and that the addition of maltose resulted in a marked improvement in the cell-attaching capability after thawing.
Materials and Methods
Materials
The materials used in the present study were obtained as follows. Dulbecco's modified Eagle medium (DMEM), Williams medium E, and antibiotics (penicillin, streptomycin, and kanamycin) were from GIBCO BRL, Life Technologies (Grand Island, NY). Fetal bovine serum (FBS) was produced from Gemini Bio-Products (West Sacramento, CA). Insulin, dexamethasone, and dimethyl sulfoxide (DMSO, D-2650) were purchased from Sigma-Aldrich (St. Louis, MO). Maltose was obtained from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). A Live/Dead Viability Cytotoxicity Kit (L3224) was obtained from Molecular Probes (Eugene, OR). All other materials and chemicals not specified above were of the highest grade. Sericin hydrolysate with an average molecular mass of 30 kDa was generously supplied from Seiren Co. Ltd. (Fukui, Japan).
Procurement of Human Primary Hepatocytes
Human hepatocytes were isolated from the nontransplantable livers of three different donors (age/race/sex/cold ischemia time: 34/African-American/female/38 h; 36/Caucasian/male/30 h; 57/Asian/male/27 h) originally procured by the National Disease Research Interchange (NDRI) in Philadelphia (USA), and imported by the Human and Animal Bridging Research (HAB) Organization (Chiba, Japan). The cell isolation was performed by a collagenase perfusion method as described previously (6).
Hepatocyte Freezing and Thawing Procedures
The basic composition of the cryopreservation medium was DMEM containing 100 U/ml penicillin, 100 U/ml streptomycin, and 10% DMSO. Maltose was added to this medium at concentrations of 0.1 mol/L and sericin at 1%. One milliliter of cell suspension containing 5 × 106 cells was quickly transferred to a 2.0-ml freezing tube and placed in a controlled rate freezer (Kryo10, Planer, Middlesex, UK). The freezing protocol was described previously (6). Immediately after freezing, the tubes were transferred to liquid nitrogen and stored for 1–3 months. In order to thaw cells, the tubes were placed in a 37°C water bath for 90 s. The cell suspension was then diluted one- to ninefold with ice-cold DMEM. The suspension was centrifuged for 1 min at 50 × g. After the supernatant was removed, the cells were resuspended in fresh medium and the cell viability was assessed by a trypan blue exclusion test. The final concentration of trypan blue (GIBCO BRL) was 0.2%.
Hepatocyte Culture and Determination of Cell Survival
Freshly isolated or cryopreserved/thawed hepatocytes (4 × 105 live cells) were seeded into 35-mm collagen-coated dishes (Biocoat Cellware, Bedford, MA) in 2 ml of Williams medium E containing 10% FBS, 1 μmol/L insulin, 1 μmol/l dexamethasone, 100 μg/ml kanamycin, 100 U/ml penicillin, and 100 U/ml streptomycin. The cells were incubated at 37°C under a humidified 5% CO2 atmosphere. The culture medium was changed at 6 and 24 h after seeding. The cell survival rates were estimated by microscopic observation of the attached cells 24 h after seeding, using the NIH IMAGE program (National Institutes of Health, http://rsb.info.nih.gov/nih-image/). Two fields in each well of three wells (total six fields) were examined and the average was calculated. In order to confirm the viability of cultured cells, hepatocytes were stained with calcein and ethidium bromide (Live/Dead Viability/Cytotoxicity Kit).
Osmolality Measurements and Differential Scanning Calorimetry (DSC) Analysis
The osmolality of freezing medium was measured using a Vapor Pressure Osmometer (VAPRO 5520, Wescor, USA). Differential scanning calorimetry (DSC) was performed by a Perkin Elmer differential scanning calorimeter, Diamond DSC, under a helium atmosphere using a cooling attachment with liquid nitrogen. For the measurement, 5 μl of the sample was taken into a small aluminum pan. The temperature of the sample was changed according to the same programmed schedule as was used for the hepatocyte cryopreservation.
Ethical Consideration
All experimental procedures using human hepatocytes were performed after permission from the institutional review board of National Center for Child Health and Development.
Results
Survival of Human Hepatocytes Cryopreserved Under Different Conditions
The representative microscopic appearances of the cultured hepatocytes from a 34-year-old African-American female are shown in Figure 1. Almost all attached cells had transparent cytoplasm with a firm smooth surface. Because the culture medium was aspirated and plated, washed extensively, and supplied with fresh medium at 6 h after inoculation, there were few dead cells attached to the plate except for small-sized round nuclei. In fact, when cells were stained with calcein and ethidium bromide, all cells are calsein-positive live cells and only the small, probably naked nuclei were stained red by ethidium bromide (Fig. 1K). It was apparent that the cells cryopreserved in DMEM-containing DMSO and maltose (Fig. 1D), and containing DMSO, maltose, and sericin (Fig. 1G) attached well onto the culture plate and that these cells had a better appearance than that of the freshly isolated hepatocytes (Fig. 1A). The attachment of cells stored in DMEM-containing DMSO and sericin (Fig. 1E) was slightly better than the attachment of the cells stored in DMEM containing DMSO only (Fig. 1B), containing DMSO and FBS (Fig. 1C), and containing DMSO, FBS, and maltose (Fig. 1H). In contrast, the combination of FBS and sericin did not appear favorable for the cryopreservation (Fig. 1F, I). The cell attachment after storage by a commercially available cryopreservant, Cell Banker 1 (Fig. 1J) was the same as that for DMEM-containing DMSO and FBS (Fig. 1B).

The phase-contrast photomicrographs of human hepatocytes before (A) and after the cryopreservation (B–G). The hepatocytes were cultured on type I collagen cell culture dish for 24 h after the inoculation. Scale bar: 200 μm. The hepatocytes were cryopreserved with DMEM containing 10% DMSO (base medium) (B), base medium + 10% FBS (C), base medium + 0.1 mol/L maltose (D), base medium + 1% sericin (E), base medium + 10% FBS, 1% sericin (F), base medium + 0.1 mol/L maltose, 1% sericin (G), base medium + 10% FBS, 0.1 mol/L maltose (H), base medium + 10%FBS, 0.1mol/L maltose, 1% sericin (I), Cell Banker 1 (J). (K) Fluorescent photomicrograph of (G) after staining with calcein and ethidium.
Quantitative Evaluation of Cell Viability and Cell-Attaching Capability
In order to quantify the morphological differences, we determined the cell viability immediately after thawing and the cell-attaching capability (Fig. 2A and B, respectively). The values of the two indices are of independent nature, one from numbers of viable and total cells and the other from an occupied area in the microscopic field. The initial viability of freshly isolated hepatocytes was 61 ± 3.1% (condition 1, average ± SD, n = 3). The viability of cells cryopreserved in DMEM-containing DMSO only was 29 ± 3.2% and cell attachment was 20.4 ± 4.1%. When we used the other control, Cell Banker 1, the cell viability was reasonably good (46.0 ± 1.1%), but the cell attachment capability was quite low (13.4 ± 1.5%). This type of discrepancy between the initial cell viability and cell survival was often observed with the cryopreserved hepatocytes. The addition of FBS did not improve the cell viability (condition 3, 30.2 ± 4.7%), but maltose (condition 4, viability: 54.0 ± 2.1%, attaching capability: 40.2 ± 2.2%) and sericin (condition 5, viability: 45.0 ± 0.8%, attaching capability: 26.2 ± 3.2%) increased both the cell viability and attaching capability. The most marked effect was observed with DMEM-containing DMSO, maltose, and sericin (condition 7, viability: 42.2 ± 3.2%, attaching capability: 51.0 ± 1.0%). As expected from the morphological observation (Fig. 1), the combination of sericin and FBS decreased both the cell viability and attaching capability (condition 6), while the addition of maltose resulted in more favorable values for the corresponding controls (conditions 8 and 9).
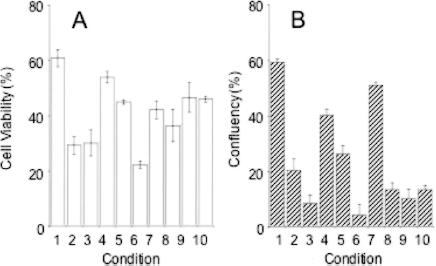
Cell viability (A) and attaching capability (B) of cryopreserved human hepatocytes with a different composition of the preservation solution. 1: Freshly isolated hepatocytes; 2: DMEM containing 10% DMSO (base medium); 3: base medium + 10% FBS; 4: base medium + 0.1 mol/L maltose; 5: base medium + 1% sericin; 6: base medium + 10% FBS, 1% sericin; 7: base medium + 0.1 mol/L maltose, 1% sericin; 8: base medium + 10% FBS, 0.1 mol/L maltose; 9: base medium + 10% FBS, 0.1 mol/L maltose, 1% sericin; 10: Cell Banker 1. The data are the means and SD of three independent experiments.
Comparison of Physical Characteristics of Freezing Solutions
The osmolality of the freezing solution is listed in Table 1. The value of DMEM culture medium was 0.312 mol/kg, almost equal to that of typical isotonic solutions (0.28–0.29 mol/kg, data not shown). When DMSO was added, the value increased to 1.922 but no marked elevation occurred by the addition of FBS, maltose, or sericin. Figure 3 shows the DSC analysis of the effect of maltose and sericin in the freezing solution. The spike indicates the starting point of crystallization. Each additive affected the crystallization temperature. Interestingly, the solution containing both sericin and maltose showed crystallization at the earlier phases containing sericin or maltose only. However, no obvious relationships were observed between the cell biological protecting activity and the physical parameters this time.

The effect of the additives in the freezing media by differential scanning calorimetry (DSC) analysis. s(spike)1: DMEM containing 10% DMSO (base medium); s2: base medium + 0.1 mol/L maltose, 1% sericin; s3: base medium + 1% sericin; s4: base medium + 0.1 mol/L maltose. Tmp indicates the temperature of the test medium. The solution temperature of each spike: s1: −28.0°C, s2: −29.1°C, s3: −31.8°C, s4: −32.5°C.
Osmolality of Various Freezing Media Used for Human Hepatocytes

Discussion
A lot of factors have been proposed to cause cell damage by cryopreservation, such as ice crystal formation, condensation of salts the free water, dehydration, etc. (8). After laborious efforts by numerous researchers, DMSO has emerged as one of the major cryoprotective agent for cells. However, DMSO itself does not sufficiently protect the cell from damage during the cryopreservation process. We previously reported that the addition of maltose to DMSO-based serum containing cryopreservation medium improved the cell-attaching capability of hepatocytes (6). In this report, we demonstrated that silk origin protein, sericin, can replace serum in the medium.
The advantage of serum-free medium is not merely the avoidance of xenozoonoses, but the emergence of quality differences of the serum lots. Sericin is a protein hydrolysate from raw silk and it is rich in serine (an average molecular weight of 30 kDa). Recently, sericin was reported as a novel cryopreservation agent of mammalian and insect cell lines (7). We expanded the utility of sericin to primary hepatocytes, cells that are perhaps the most difficult to storevia freezing. Because of the advantages described above, sericin-containing medium may become one option for the clinical transplantation of cryopreserved hepatocytes.
Ice crystal formation is likely to be a major hurdle to succeed cryopreservation. DSC measurement is often used to study ice formation in the multicomponent liquid phase (1). Unfortunately, no meaningful relationship between cell biological assessment and DSC as well as osmolality was gleaned from the present results, but the diligence of these physical approaches should be conducted in conjunction with biological evaluations.
Footnotes
Acknowledgments
Firstly, we would like to express deep gratitude to the persons who donated the livers for research and to their family members. We also thank Dr. Satoshi Suzuki of HAB Research Organization, Japan for supplying the human hepatocytes and Seiren Co. Ltd., Fukui, Japan for the generous gift of sericin. This research was supported by Grant-in-aid (KH71066 and KHD1027) from the Japan Health Sciences Foundation, Tokyo, Japan and by the Sasakawa Scientific Research Grant from The Japan Science Society.
