Abstract
Transplantation of human hepatocytes (HTx) has gained recognition as a bridge to, or an alternative to, orthotopic liver transplantation for patients with acute liver failure or genetic defects in liver function. Although the quality of the hepatocytes used for cell transplantation is critical, no consensus exists on protocols to assess the function of hepatocytes prior to HTx. Application of this cell therapy in clinical practice could be aided by fast and reliable assays to evaluate the functional competence of isolated hepatocytes prior to clinical transplantation. Traditional assays for measuring metabolic functions in primary hepatocytes frequently involve highly technical equipment, time-consuming methods, and large numbers of cells. We describe a novel approach for the rapid assessment of the metabolic capabilities of human hepatocytes. This report details simple procedures to evaluate 11 endpoints from cells isolated from human liver that can be performed by a single operator within approximately 2 h of isolation. Longer term cultured hepatocytes were also analyzed to determine if the results from the 2-h tests were predictive of long-term hepatic function. The assays simultaneously measure five cytochrome P450 activities, one phase II activity, plating efficiency, and ammonia metabolism in addition to viability and cell yield. The assays require fewer than 20 million cells and can be completed using commonly available and inexpensive laboratory equipment. The protocol details methods that can be used in a time frame that would allow analysis of hepatic functions in freshly isolated hepatocytes prior to their use for clinical transplantation.
Introduction
Orthotopic liver transplantation (OLT) is not able to meet the demand for therapy of liver disease, largely because of an inadequate supply of donor organs. Transplantation of human hepatocytes (HTx) has been examined as an alternative to OLT for acute liver failure and metabolic liver disease. During the last 20 years, HTx has been successfully used to treat a variety of conditions, from chronic/acute liver failure to inborn error of metabolism (2,8,21,22,24). Individuals with inborn errors of metabolism who have undergone HTx show clinical improvement and a partial correction of the underlying metabolic defect (7,23). Nevertheless, in many cases, sustained benefits have not been observed. This may be partly related to the transplantation of an inadequate cell dose and/or to variations in the quality of the hepatocyte preparations. Although a few transplants may be conducted with lobes or segments from reduced cadaveric grafts, the main source of tissue for cell isolation for HTx are livers deemed unsuitable for transplantation (7,18, 19,23). Steatosis is the most common reason for rejection of a donor tissue for transplantation. Notably, lower cell viability and yield and low levels of drug-metabolizing cytochrome P450 (CYP) enzymes have been reported in hepatocytes obtained from fatty livers compared to normal tissue (5,15). Damage produced by warm or cold ischemia is another factor that determines, in part, if useful hepatocytes can be isolated from the tissue (1). Evaluation of each hepatocyte preparation prior to transplantation is required by regulatory authorities; however, there is no consensus on methods to assess hepatocyte viability and function, and it is not known which tests, if any, will be useful to predict long-term engraftment and function following HTx.
Previous reports describe methods to evaluate hepatic functions, both before and after cryopreservation with the aim of identifying cells suitable for transplantation (1,4, 6,9,16). Metabolic activities in hepatocytes are usually assessed by incubation of drug or chemical substrates with hepatocytes in culture, and analysis frequently requires sophisticated and expensive high-pressure liquid chromatography (HPLC) or mass spectrometry equipment for metabolite detection (1,6). Recently, chemical substrates have become commercially available that, when metabolized by specific cellular enzymatic processes, form luminescent or fluorescent metabolites that can be directly analyzed with simple spectrophotometers without prior chromatographic separation. These fluorescent and luminescent methods have high signal-to-noise ratios, can be adapted and performed in standard culture conditions with intact cells, and provide rapid and sensitive methods to assess hepatic enzymatic activities.
This report describes simple procedures to analyze metabolic functions in human hepatocytes, where six hepatic phase I/II activities, cell attachment, ammonia metabolism, viability, and cell yield can be analyzed with common laboratory equipment in approximately 2 h of cell isolation by a single operator. Long-term culture of hepatocytes and CYP metabolism and induction studies and resorufin and ammonia metabolism were also analyzed to validate results observed in the short-term tests. Thus, large amounts of data on the viability and function of isolated hepatocytes could be collected and analyzed prior to the use of freshly isolated cells for a clinical transplant. Data from over 100 human hepatocyte isolations are presented. These data provide a range of values that one could expect using these assays so that activities measured on a new hepatocyte case could be immediately compared to a large group of existing data.
Materials and Methods
Hepatocyte Isolation
All human tissues were collected with informed consent following ethical and institutional guidelines. Human hepatocytes were isolated from 34 samples obtained from organ donors (OD) rejected for OLT for different reasons (prolonged warm/cold ischemia time, drug overdose, anoxia, steatosis, and fibrosis); 35 explanted tissues from patients undergoing OLT for genetic inborn errors or other liver diseases (LD); 44 specimens derived from patients undergoing scheduled liver resection usually for primary or metastatic cancer (LR). Whole livers not used for orthotopic liver transplantation were obtained 6–36 h after cross-clamp and in situ perfusion. The transportation was performed using specific cold preservation solutions [usually Belzer's, University of Wisconsin (UW) solution], and liver tissues were maintained on wet ice. Residual tissue not needed for diagnostic purposes after hepatic resection were transported to the laboratory from the operating rooms in cold Eagle's minimum essential medium (EMEM; Lonza, Walkersville, MD, USA) within 90 min of removal.
The tissue dissociation and subsequent hepatocyte isolation procedures used in this study were performed as previously described (11,12). In OD, the left lateral segment was processed after anatomical dissection from the right lobe, 2–3 cm from the falciform ligament. All LD tissues were from right lobe sterile dissection in the operating room, after whole organ removal during the OLT procedure. LRs originated from left or right lobe resections. The procedure for hepatocyte isolation was the same in all the tissues and performed as previously described (11). Briefly, the major hepatic vessels were cannulated and sutured on the cut surface of the parenchyma. A three-step perfusion by peristaltic pump was conducted, followed by mechanical tissue disruption with a sterile scissors and filtration through stainless steel mesh and/or gauze to produce a cellular suspension. Hepatocytes were enriched, and digestive enzymes were removed by three sequential low speed centrifugations (85–90 ×
Viability Determination
Cell viability was assessed immediately after isolation as previously described using trypan blue (Sigma-Aldrich) exclusion (TBE) (11). Time to complete evaluation: 5 min.
Plating Efficiency Evaluation
Cell adhesion was performed as previously described (11). Two hours after plating, a visual inspection by microscopy was conducted to evaluate the attachment of the cells, and after 9–17 h, cells from all the wells were scraped and collected separately. Plating efficiency (PE) was determined by measuring the protein content. Time to complete visual preliminary inspection: 2 h; to quantify protein content: 11–19 h. Number of cells required: 4.5 × 106.
Caspase-Glo™ Assays
Caspase 3 and 7 activity was measured with a luminescent assay, Caspase-Glo® 3/7 (Promega Corporation, Madison, WI, USA) according to the manufacturer's instructions with some minor changes, previously described (10). Immediately after isolation, 30,000 cells (40 μl of the cell suspension of 7.5 × 105 viable cells/ml) were seeded in each well of a 96-well white plate (Corning, Inc., Corning, NY, USA) and incubated at 37°C for 30 min, and then incubated with an equal volume of Caspase-Glo™ reagent. Running time: 60 min. Number of cells required: triplicate analysis, 9 × 104.
Drug Metabolism Studies
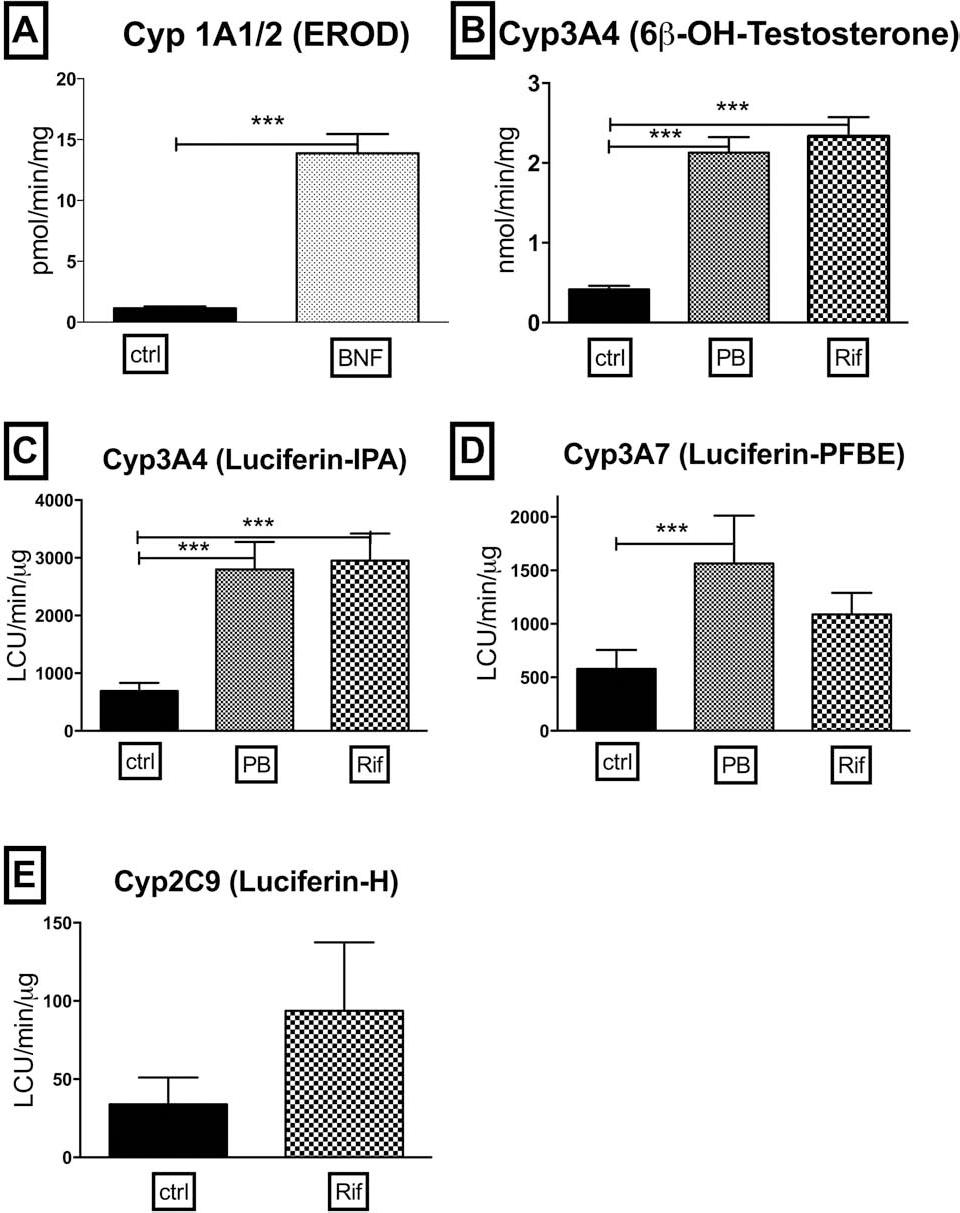
Each metabolite was measured in fresh, 1-ml cell suspensions (1 million viable hepatocytes/ml) or 40 μl (7.5 × 105 viable cells/ml) immediately after digestion and in adherent cultures on day 5 of culture and, where indicated, after 3 days of exposure to specific prototypical inducers. To evaluate the long-term activity, cells were plated in collagen-treated [made in-house from rat tail (25)] 12- and 96-well plates (1.5 × 105/cm2; Corning) with supplemented hepatocyte maintenance medium (HMM; Lonza) (11), and the medium was changed daily. The first 2 days, cells were cultured with HMM to remove dead cells and to allow the CYP450 gene expression to decrease to basal levels. On day 3, cells were incubated for 72 h in the presence of specific inducers [25 μM β-naphthoflavone (BNF) for cytochrome P450 family 1 subfamily A polypeptide 1 and 2 (CYP1A1 and 1A2); 10 μM rifampicin (Rif) or 1 μM phenobarbital (PB) for CYP2C9, 3A7, and 3A4; 0.1% dimethyl sulfoxide (DMSO) as vehicle control; all from Sigma-Aldrich, St. Louis, MO, USA] with replenishment every 24 h as previously described (11). Different CYP450 subunit activities were specifically measured using fluorescent and luminescent in vitro assays.
Results are expressed as luminescent counting units (LCU)/min, pmol/min, or nmol/min and normalized to 1 million viable cells (suspension cultures) or to the dsDNA or protein content (adherent cultures).
CYP450-Glo™ Assays
Different CYP450 activities were measured using commercial cell-based assays (P450-Glo™ Assays; Promega Corporation, Madison, WI, USA), as previously described (10). Briefly, a 96-well white plate containing five different luminescent probes [Luciferin-CEE (6′-chloroethyl ether), Luciferin-ME (6′-methyl ether), Luciferin-IPA (isopropyl acetal), Luciferin-PFBE (6′-pentafluoro-benzyl ether), and Luciferin-H (6′-deoxyluciferin)] specifically designed for different CYPs (CYP1A1, CYP1A2, CYP3A4, CYP3A7, and CYP2C9, respectively) was previously prepared and kept frozen until the hepatocyte suspension was ready. Every probe (10 μl) was seeded in triplicate and 40 μl of the cell suspension (7.5 × 105/ml) was added to every well of the 96-well white plate. Six additional wells were used to evaluate background signal, as described in the manufacturer's procedure. Total running time: 50 min. Number of cells required: 9 × 104 per CYP; total amount 6.3 × 105.
Cells in culture were read with the same procedure, treating the cells in a 96-well clear culture plate (Corning, Inc.) with different Glo™ substrates and by transferring the lysate suspension to a 96-well white plate, which was analyzed as described above.
7-Ethoxyresorufin-O-Deethylase (EROD) Assay
Cytochrome P450–1A1/2 activity was assessed by the conversion of 7-ethoxyresorufin to resorufin as previously described (11,26). Total running time: 65 min. Number of cells required: 3 × 106. Following sampling, cells were harvested in phosphate buffer (0.1 M, pH 7.4; made in house from Sigma-Aldrich components) and stored at −20°C until protein content was determined.
Testosterone Hydroxylation Assay
Testosterone 6b-hydroxylase activity, catalyzed by CYP3A4, was determined directly both in cell suspensions and in intact cultured hepatocytes, as described previously (11,17). After cell exposure to testosterone (250 μM; Sigma-Aldrich), media was collected, centrifuged to remove cells and fragments, and stored at −20°C until analyzed by high-pressure liquid chromatography (HPLC; Waters 2695 model; Waters Corporation, Milford, MA, USA). Running time: 35 min; evaluation time: 1–3 days. Number of cells required: 3 × 106.
Phase II Assay
Phase II activity was determined by the metabolism of the fluorescent compound resorufin as previously described (11). Total running time: 35 min. Number of cells required: 3 × 106. Resorufin conjugation was quantified by measurement of the decrease in fluorescent signal.
Ammonia Metabolism
Human hepatocytes were evaluated for ammonia metabolism by direct, in vitro, colorimetric determination of ammonia (20). Ammonia removal was evaluated as previously described (11), and results were compared to a linear calibration curve. Total running time: 2 h. Number of cells required: 3 × 106.
dsDNA Amount Quantification
Double-stranded DNA (dsDNA) quantification was performed after the Glo™ assays were complete with a Quant-iT™ PicoGreen® dsDNA ultrasensitive fluorescent nucleic acid staining kit according manufacturer's instructions (Molecular Probes, Invitrogen Corporation, Camarillo, CA, USA), with some minor modifications as previously described (10). Total running time: 5 min.
Protein Amount Quantification
Total protein content in cell cultures was determined after fresh fluorescent/colorimetric assays were performed using a commercial kit (Bio-Rad Protein Assay Kit; Bio-Rad, Richmond, CA, USA) based on the method of Bradford. It involves the addition of an acidic dye to protein solution and subsequent measurement at 595 nm with a spectrophotometer (Synergy HT; BioTek Instruments, Winooski, VT, USA). Comparison to a bovine serum albumin (Sigma-Aldrich) standard solution provides a measurement of protein concentration in cell suspension after isolation (1 million viable cells) and in different time points in cultured cells (5 days with different treatments).
Statistical Analysis
Analysis of variance (ANOVA) was used to determine statistical differences, and Tukey's multiple comparison test was performed on data from the three tissue cell sources. A value of
Results
For this study period, 113 human liver tissues were collected, and successful hepatocyte isolations were performed on all specimens. Not all the reagents were commercially available at the beginning of the study, so some of the assays were not conducted on all of the cell samples. The liver tissue was obtained from patients undergoing liver resection (LR;
There was a significant difference in size of tissue perfused between the three groups (
Most isolations produced hepatocytes with high viability as determined by TBE (78.8 ± 1.4%) with a significant difference between the OD and the other two groups (73.2 ± 3.1%, 81.8 ± 2.9%, and 80.8 ± 1.4%, respectively, in the OD, LD, and LR groups) (Fig. 1A). Although it can differ between transplant centers, an initial viability as low as 60% might still be considered for clinical transplant (14,24). Not all the cases reported here provided cells with >60% viability. Seven of 113 total isolations provided cells with viability between 7% and 57%. Cases with low viability resulted from three OD, three LD, and one LR and were characterized by prolonged cold ischemic times (more than 24 h) or a high degree of steatosis or cirrhosis. All of these parameters are known to influence the cell isolation. The low viability preparations were not excluded from the study. In Figure 1B and C, we present the data by dividing it into those that might be recommended for transplant (viability >60%) and those from preparations with viability below 60%.
Caspase 3/7 activity, a measure of apoptosis, was measured in cells from 52 cases. No difference in caspase activity between the tissue sample groups was observed, and an average activity of 32,621 ± 4,094 LCU/min per million viable cells was measured. Caspase activity was measured in only two of the seven cases with <60% viability, and those cases did not have higher than normal caspase activities (Fig. 1B).
Plating efficiency (PE), or the ability of isolated cells to attach to a culture substrate, is considered a more stringent assay for cell viability/vitality, since cell attachment requires continued energy expenditure by the cell. Final measurements of PE reported here were determined by protein content remaining on plates after an extensive washing procedure to remove dead or unattached cells. An early estimate of PE was obtained by visual inspection 2 h after plating cells. This estimated PE was found to correlate well with the final evaluation of PE by protein content (

Viability, caspase activity, and plating efficiency in freshly isolated hepatocytes. (A) Histograms of viability in human hepatocyte isolated from organ donors rejected for orthotopic liver transplantation (OLT) (OD,
Hepatic Phase I Activities Assessed Immediately After Isolation
Isolated hepatocytes must be capable of performing a number of metabolic functions if they are to be useful for clinical transplantation. The activity of several CYP enzymes that are responsible for the oxidative metabolism of drugs, xenobiotics, and endogenous compounds such as hormones can be measured with specific profluorogenic and luminescent substrates. Data are presented in Figure 2 on the activity measurement for several important cytochrome P450 family members including CYPs 1A1/2, 2C9, 3A4, and 3A7 immediately after isolation. Data are also presented for resorufin conjugation (Fig. 2H) and ammonia metabolism (Fig. 2I).
The conversion of the nonfluorescent substrate, 7-ethoxyresorufin, to the fluorescent product, resorufin, is mediated by a CYP1A activity, EROD, and is used to assess CYP1A1 and 1A2 activities in cells. Average EROD activity from the transplantable cases was 1.4 ± 0.2 pmol/min/106 (
When CYP 1A1 and 1A2 activities were individually analyzed with luminescent assays using Luciferin-CEE and Luciferin-ME, respectively, the results were generally similar to the EROD assay. As expected, since CYP1A1 is not liver specific, few preparations had any measurable CYP1A1 activity (166 ± 32 LCU/min/106;
Immediately postisolation, CYP1A2 activity was, on average, 30,664 ± 2,569 LCU/min/106 (
Traditionally, CYP3A4 activity has been measured with high-pressure liquid chromatographic techniques to separate and quantify testosterone metabolism to 6β-hydroxytestosterone. In addition to the traditional HPLC technique (Fig. 2D), CYP3A4 activity was measured with a rapid luminescent assay using Luciferin-IPA as the substrate (Fig. 2E). There was good agreement between the activity measured by HPLC and the luminescent assay. However, as shown in Figure 2D and 2E, the luminescent assay is more sensitive, providing values greater than two times the background in all samples tested, while 10 of the same samples showed background activity when measured by the HPLC technique. This greater range of response and increased sensitivity revealed a significant difference between cells with low viability and the rest of cell preparations (

Metabolic functions in freshly isolated hepatocytes. The indicated metabolic activity from freshly isolated hepatocytes divided in two groups, those with viability greater or lower than 60%. (A) Cytochrome P450 family 1 subfamily A polypeptide 1/2 (CYP1A1/2) assessed by the conversion of 7-ethoxyresorufin to resorufin [7-ethoxyresorufin-
Although CYP3A4 is frequently the most highly expressed CYP in adult liver, another CYP3A family member, CYP3A7, is expressed at the highest levels in fetal liver, and its expression gradually declines over the late fetal, neonatal, and pediatric periods (13). Since some donor tissues for this study were obtained from pediatric cases, CYP3A7 activity was also analyzed with a luminescent substrate (Luciferin-PFBE) in all cases analyzed for CYP3A4. Statistically, higher 3A7 expression was observed in the LD group (3,118 ± 674 LCU/min/106). A large number of the samples in the LD group were less than 2 years old. Significantly more CYP3A7 expression could be expected in the LD group because of the relative age of this group in comparison to the LR (268 ± 62 LCU/min/106,
Measurements of CYP2C9 with luciferin-H showed a range of activities that were similar in all the hepatocyte preparations, 200–1,200 LCU/min/106 with an average of 677 ± 58 LCU/min/106. No significant difference in CYP2C9 activity was observed between cases with high and low viability (Fig. 2G).
In addition to CYP-mediated metabolism, conjugation activity and ammonia metabolism were measured in isolated cells. All samples tested conjugated resorufin (4.23 ± 0.25 pmol/min/106,
Comparison of Results of the 2-h Assays with Longer Term Follow-Up
Cytochromes in the 1A, 2C, and 3A families are inducible genes. That is, exposure to specific chemical compounds leads to an induction in the expression of the target gene followed by increased protein synthesis and an increase in the corresponding metabolic activity. The induction of CYP enzyme activity with inducing agents is a robust test of longer term survival and function of the cells. Longer term culture and measurements of the induction of CYP450 genes was analyzed to determine if the results from the short-term (2 h) tests were validated in longer term experiments. All preparations with low viability after isolation (<60%) where long-term culture was attempted (
Long-term metabolic activity and CYP450 inductions in human hepatocytes after 5 days in vitro. Values represent the measured metabolic activities of hepatocytes isolated and cultured for 5 days in media, three of which contained specific prototypical inducers. Values are expressed as pmol or nmol or LCU/min and normalized to total protein content (mg) or total dsDNA content (ng). (A) CYP1A1/2 assessed by the conversion of 7-ethoxyresorufin to resorufin (EROD assay) in control cells [exposure to vehicle, dimethyl sulfoxide (DMSO)] and in cells after 3 days of exposure to inducer [β-naphthoflavone (BNF) 25 μl] (
The CYP3A family genes are inducible in cultured hepatocytes. As shown in Figure 3, the activity measured immediately after isolation was maintained after 5 days in culture, and it was significantly increased in a range of 2–28 times after exposure to PB or Rif (
Since CYP3A7 is not normally expressed in older donors, data presented in Figure 3D were taken from donors less than 3 years of age (
As was observed in the fresh assays, there was considerable variability in 2C9 expression in different cases (Fig. 3E), to the point where the exposures to Rif did not lead to a statistically significant induction in activity. The lack of induction of CYP2C9 is gene specific, since the same hepatocyte cases showed robust induction of CYP3A4 activity following exposure to Rif.
Long-Term Phase II and Ammonia Metabolism
As we previously reported (12), conjugation activity and ammonia metabolism increases over time in cultured cells. As shown in Figure 4A and B, a significant increase in both of these activities in plated cells was measured (
Discussion
Hepatocyte transplants have been proposed as an alternative to OLT in the management of some liver-based diseases. There is no consensus within the groups performing these transplants on assays that could, or even should, be conducted prior to using the cells for a transplant. Experiments reported here were designed to examine a variety of hepatic functions from adhesion to extracellular matrix to metabolic activities for specific chemical substrates. The criteria for inclusion in this battery of tests were that each assay measures a distinct hepatic function and that the test could be completed within 2 h after cell isolation by a single operator, a time that would not likely delay the use of the cells for a clinical transplant. In addition, the assays must be performed using common and inexpensive laboratory equipment such as a spectrophotometer or luminometer and a small number of cells (16 million). A large number of human hepatocyte isolations were analyzed with each assay to determine a normal range of activities to expect from isolated cells. Data from each assay were correlated with the viability of the cells measured immediately after isolation by TBE. The experiments reported here were conducted to examine the hypothesis that specific tests could be conducted on hepatocytes immediately postisolation and that the results of these tests would aid in the identification of cells most suitable for clinical transplant.
The data indicate that the results of no single test correlate completely with the viability of the isolated cells as determined by TBE. In every assay, cells with viability below 60% provided values that were also found in cells with viabilities above 60%. Assays such as EROD (Fig. 2A) and CYP3A activity (Fig. 2D–F) correlated best with viability of the cells. Using the EROD assay, from the analysis of 52 hepatocyte isolations, if a cutoff line was drawn at the highest value obtained from cells with viability below 60%, only five cases with viability above 60% would fall within that range and would have been considered not useful for transplant based solely on that assay. This would suggest that the EROD assay has a >90% chance of correctly predicting cells that would be useful for transplantation if 60% viability were the only other criteria. Activities mediated by CYP3A were also found to correlate with cell viability, in particular, when the evaluation was conducted using the more sensitive luminescence-based assay compared to the traditional HPLC method. The major CYP in the adult human liver, is CYP3A4, which is responsible for the metabolism of >50% of administered drugs. Here, CYP3A4 was analyzed with two different methods, the traditional 6β-OH-testosterone HPLC method and a new assay using the proluminescent substrate Luciferin-IPA (Fig. 2D, E). Isolated cells with viability below 60% displayed low CYP3A4 activity, particularly in the IPA-luminescent assay. The HPLC technique, as conducted here, was not sensitive enough to provide values significantly above background on many cases, while the dynamic range of response in the luminescent assay was considerably greater and covered several logs of activity. There was good correlation between CYP3A4 activity as measured with Luciferin-IPA and cell viability. If one applied the same exclusion criteria as described above for EROD activity, that of drawing a line at the highest activity obtained in the samples with lower than 60% viability, only 11 of the 67 samples with viability higher than 60% expressed might have been excluded as suitable for transplant (84% accuracy) based solely on CYP3A4 activity. While CYP3A4 is the major 3A family member in adult liver, fetal liver and donors below 5 years of age would be expected to express the fetal form of 3A, CYP3A7. Luciferin-PFBE is five times more specific for 3A7 compared to CYP3A4. Like CYP3A4 activity in adults, CYP3A7 activity, measured in the younger donors shows good correlation with the viability of the cells (80% accuracy). Thus, both the EROD assay and activity measurement of CYP3A family members, CYP3A4 in adult tissues or CYP3A7 in younger donors, were found to correlate well with the viability of the cells. Importantly, these assay activity measurements provide data on metabolic function that is not provided by a simple estimate of TBE viability and should be considered informative and useful in the decision to use or not to use specific cells for transplantation.

Long-term phase II activity and ammonia metabolism in human hepatocytes. Histograms represent phase II activity and ammonia metabolism in freshly isolated cells and in cells from the same cell preparation after 5 days in vitro. Value are expressed as pmol or nmol/min and normalized to total protein content (mg). (A) Phase II activity determined by the metabolism of the fluorescent compound resorufin (
When possible, additional cells were cultured for 5 days to analyze CYP450 inductions, ammonia metabolism, and resorufin conjugation. These longer term studies with isolated cells were conducted to determine if the results from the 2-h assays with the fresh cells were validated by additional long-term studies. In hepatocyte cases with greater than 60% viability, the long-term studies showed robust induction of CYP activity when cells were exposed to prototypical inducing agents and the typical increase in ammonia metabolism and conjugation capacity normally observed in cultured hepatocytes. The plating efficiency is also a useful indicator of cell viability and function. In cases with less than 60% viability, long-term follow-up studies could not be conducted because plating efficiency was too low, and too few cells remained attached to culture plates to conduct long-term studies.
In contrast, data from several other assays on the freshly isolated cells did not appear to correlate well with viability. The measurement of CYP1A1 (Luciferin-CEE, Fig. 2B) or CYP1A2 activity (Luciferin-ME, Fig. 2C) CYP2C9 activity (Luciferin-H, Fig. 2G), resorufin conjugation (Fig. 2H) caspase activity, and ammonia metabolism (Fig. 2I) were easily measured in isolated cells, but there was considerable overlap in the values obtained with cells below 60% viability with those from cells with greater than 60% viability. While these assays do not seem useful to discriminate between cells with high and low viability, the inclusion of some assays as part of a screening process for cells prior to a clinical transplant could still be considered in specific cases. For example, ammonia metabolism would still be considered an important assay to perform if the isolated cells were to be transplanted into a patient with a urea cycle defect. A quick estimate of phase II conjugation activity with a general substrate such as resorufin could still be employed on cases where the cells were to be transplanted into patients where immediate conjugation capacity would be needed. Urea cycle defects and conjugation defects are the most frequent metabolic diseases treated with HTx (3,7,22,23). As previously suggested by Bonora-Centelles et al., the analysis of specific functional activities required in the recipient provides useful information in addition to TBE viability estimates with which to choose the most appropriate cells for a clinical transplant (1).
In most assays, cells with the low viability displayed activity measurement below the average of the cells with greater than 60% viability, but in most cases, some activity was still measurable in cases with low viability. These data indicate that hepatic functions were maintained in the remaining viable cells from low viability isolations. One could speculate that a simple density separation technique to remove dead cells from the preparations might be sufficient to recover highly viable and functional cells from cases with low viability. We tested this hypothesis on two cases with 29% and 57% viability. In preliminary experiments, it was observed that following Percoll™ separation, there were considerable improvements in cell viability (65% and 90%, respectively), plating efficiency (10–40% higher), ammonia metabolism, and other metabolic activities. Additional studies with cell separation techniques with cells with low viability appear to be warranted. Removing dead cells by density separation techniques might be especially useful if the isolated hepatocytes were maintained in hypothermic storage for a prolonged time awaiting transplantation (10).
Methods described here permit a rapid assessment of up to 11 parameters and metabolic functions in freshly isolated human hepatocytes. All of the assays described here can provide useful data concerning hepatic function and metabolic activity to support the estimate of cell viability as determined by TBE. These assays are highly sensitive and allow the simultaneous measurement of a large number of endpoints using multiwell plate readers, thus enhancing sample throughput. Assays conducted with fresh cells were validated by measurements of the long-term function of cells from the same isolation. The assays use few cells and require simple and inexpensive laboratory equipment. All assays, including an estimate of plating efficiency, can be completed within 2 h of cell isolation and can be completed by a single operator. It is clear that the decision to use or not use cells for a clinical transplant will not be based on the results of a single assay. Data on several hepatic functions will help in the decision process. Data from over 100 cell isolations are included, and the range of values obtained with the different assays is provided. Measurements on newly isolated cells could easily be compared to this data bank. Cell preparations that fall into the lower range of activity values in several assays could be given lower priority for subsequent clinical use. In particular, measurement of activities of the CYP1A family with an EROD assay and the measurement of CYP3A activity with luminescent assays were found to correlate well with cell viability.
