Abstract
Endothelial progenitor cells (EPCs) have shown tropism towards primary tumors or metastases and are thus potential vehicles for targeting tumor therapy. However, the source of adult EPCs is limited, which highlights the need for a consistent and renewable source of endothelial cells for clinical applications. Here, we investigated the potential of human embryonic stem cell-derived endothelial cells (hESC-ECs) as cellular delivery vehicles for therapy of metastatic breast cancer. In order to provide an initial assessment of the therapeutic potency of hESC-ECs, we treated human breast cancer MDA-MB-231 cells with hESC-EC conditioned medium (EC-CM) in vitro. The results showed that hESC-ECs could suppress the Wnt/β-catenin signaling pathway and thereby inhibit the proliferation and migration of MDA-MB-231 cells. To track and evaluate the possibility of hESC-EC-employed therapy, we employed the bioluminescence imaging (BLI) technology. To study the therapeutic potential of hESC-ECs, we established lung metastasis models by intravenous injection of MDA-MB-231 cells labeled with firefly luciferase (Fluc) and green fluorescent protein (GFP) to NOD/SCID mice. In mice with lung metastases, we injected hESC-ECs armed with herpes simplex virus truncated thymidine kinase (HSV-ttk) intravenously on days 11, 16, 21, and 26 after MDA-MB-231 cell injection. The NOD/SCID mice were subsequently treated with ganciclovir (GCV), and the growth status of tumor was monitored by Fluc imaging. We found that MDA-MB-231 tumors were significantly inhibited by intravenously injected hESC-ECs. The tumor-suppressive effects of the hESC-ECs, by inhibiting Wnt/β-catenin signaling pathway and inducing tumor cell death through bystander effect in human metastatic breast cancer model, provide previously unexplored therapeutic modalities for cancer treatment.
Keywords
Introduction
Tumor growth and metastasis depend on neovascularization, the growth of new blood vessels. Tumor vasculatures are mainly developed through angiogenesis by sprouting from preexisting vessels and vasculogenesis via recruitment of endothelial progenitor cells (EPCs) (16,19). Accumulating evidence suggests that circulating bone marrow-derived EPCs contribute to tumor neovascularization, and thus, tumor growth may be retarded by inhibiting their incorporation (14,25,26,29). In addition to autologous EPCs, exogenous EPCs via intravenous injection could also reside in sites of tumor development (3,18). Exploitation of this tumor tropism offers numerous potential therapeutic applications. The feasibility of using genetically modified EPCs as angiogenesis-selective gene-targeting vectors and the potential of this approach to mediate nontoxic and systemic antitumor responses have been demonstrated (5,15, 32), highlighting the need for a consistent source of endothelial cells to make clinical applications possible.
With their capacity for unlimited self-renewal and pluripotency, human embryonic stem cells (hESCs) may provide an alternate source of therapeutic cells by allowing the derivation of large numbers of endothelial cells. Due to the limited sources for adult EPCs, differentiation of hESCs into endothelial cells (hESC-ECs) may provide an unlimited source of cells for novel transplantation therapies of ischemic diseases by supporting angiogenesis and vasculogenesis (13,17). At present, hESCs have been approved by the US Food and Drug Administration (FDA) for use in clinical trials to treat acute spinal cord injury (www.geron.com) and Stargardt's Macular Dystrophy (www.advancedcell.com) (11). In addition, previous studies have demonstrated that hESC microenvironment suppresses the tumorigenic phenotype of cancer cells and that this effect is exclusive to hESCs and unavailable to other types of stem cells derived from amniotic fluid, cord blood, or adult bone marrow (4,20). Hence, we hypothesize that hESC-ECs may have the potential to serve as cellular vehicles for antitumor therapy in vivo. Given the possibility to form or promote tumors by transplanted cells, the development of technologies to noninvasively diagnose cellular misbehavior and monitor the response to therapy is a top priority of hESC-EC therapy (9,12,30).
In this study, we sought to develop a model procedure to analyze the behavior and efficiency of cellular vehicle combinations used to deliver localized bystander therapy to tumors and applied it to explore the therapeutic potential of hESC-ECs on metastatic breast cancer. We also introduced noninvasive bioluminescence imaging (BLI) to evaluate the efficiency of hESC-ECs armed with the herpes simplex virus truncated thymidine kinase (HSV-ttk) suicide gene for cancer therapy in metastatic models.
Materials and Methods
Cell Culture
The undifferentiated human female embryonic stem cell line (H9) was purchased from Wicell Research Institute (Madison, WI, USA), and the female breast adenocarcinoma- derived MDA-MB-231 cell line was purchased from ATCC (Manassas, VA, USA). Differentiation of hESCs to endothelial cells was performed using the two-step protocol we described previously (13). Briefly, we cultured hESCs in ultra-low attachment plates (Corning, Lowell, MA, USA) to form embryoid bodies (EBs), the three-dimensional aggregates of hESCs. Next, we harvested 12-day-old hEBs, seeded them in rat tail collagen type I (BD Biosciences, Bedford, MA, USA), and obtained hEB sproutings 3 days later. Cluster of differentiation 31-positive (CD31+)/CD144+ cells of hEB sprouting were isolated as hESC-ECs via flow cytometry as previously described (13) and cultured in endothelial cell growth medium (EGM-2; Lonza, Walkersville, MD, USA). Human breast cancer cell line MDA-MB-231 was cultured with Dulbecco's modified Eagle's medium (DMEM; ThermoFisher Scientific, Hudson, NH, USA) supplemented with 10% fetal bovine serum (FBS; Corning), 1% penicillin–streptomycin solution (Gibco, Rockville, MD, USA), and 1% minimum essential medium (MEM) non-essential amino acid solution (Gibco). To track transplanted cells in vivo, hESC-ECs and MDA-MB-231 cells were transduced with a self-inactivating lentiviral vector carrying a ubiquitin promoter driving firefly luciferase and green fluorescence protein (Fluc-GFP) double-fusion (DF) reporter gene. In addition, hESC-ECs were transduced with a self-inactivating lentiviral vector carrying an elongation factor-1α (EF-1α) promoter driving renilla luciferase, red fluorescence protein, and herpes simplex virus truncated thymidine kinase (Rluc-RFP-HSV-ttk) triple fusion (TF) reporter gene for gene therapy.
Plasmid Constructs
The pLV-EF1α-TF-blasticidin (Bsd) plasmid was generated by inserting the TF fragment between BamHI and XbaI sites of the multiple cloning site (MCS) of pLV-EF1α-MCS- internal ribosome entry site (IRES)-Bsd (Biosettia, San Diego, CA, USA). Double fusion reporter gene plasmid was described in a previous study (12).
Collection of Conditioned Medium
For treatment of MDA-MB-231 cells, hESC-ECs or MDA-MB-231 cells were cultured to 80% confluence, and the medium was changed with 5 ml endothelial basal medium (EBM-2) per 10-cm dish (Corning). For assessment of tumor tropism in vitro, MDA-MB-231 cells were cultured to 50% confluence, and the medium was changed with 5 ml DMEM per T75 flask (Corning). Twenty-four hours later, the supernatant was harvested and restored at −80°C until use.
Cell Proliferation Assay
MDA-MB-231 cells were seeded in six-well plates (Corning) at a density of 1 × 105 per well. After the cells attached, the medium was changed with MDA-MB-231 conditioned medium (231-CM) or hESC-EC conditioned medium (EC-CM) mixed with the same amount of DMEM (supplemented with 10% FBS). Five fields per well were chosen randomly and marked. Cells in the chosen fields were counted at 0, 24, 48, and 72 h, respectively.
Cell Cycle Analysis and Apoptosis Analysis
MDA-MB-231 cells were cultured with EC-CM mixed with the same amount of DMEM (supplemented with 10% FBS for cell cycle analysis or 2% FBS for apoptosis analysis). After 48 h, cells were harvested. For cell cycle analysis, cells were fixed and stained with 1 mg/ml propidium iodide (PI) solution (Keygene, Nanjing, Jiangsu, China), and stained cells were analyzed by fluorescenceactivated cell sorting (FACS). For apoptosis analysis, cells were prepared using Annexin V-fluorescein isothiocyanate (FITC) apoptosis detection kit (Keygene) according to the manufacturer's directions, and stained cells were analyzed by FACS.
Transwell Migration Assay
MDA-MB-231 cells (1 × 105) or hESC-ECs (1 × 104) in 200 μl basic medium were seeded upon the 24-well Millicell Hanging Cell Culture Inserts (Millipore, Billerica, MA, USA) with attractants in the lower chamber. After incubation for 24 h, the inserts were taken out, and the membranes were fixed and stained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen, Carlsbad, CA, USA). Cells of five randomly chosen fields per membrane were counted under the microscope at 200× and statistically analyzed.
Wound-Healing Assay
MDA-MB-231 cells were seeded in six-well plates at a density of 2.5 × 105 per well. After the cells attached, the medium was changed with 231-CM or EC-CM mixed with the same amount of DMEM (supplemented with 10% FBS). When the cells were cultured to 90% confluence, the medium was removed, and three separate wounds were scratched with sterile 20-μl tips (Corning). Then the cells were rinsed with PBS (Invitrogen), and 2 ml DMEM supplemented with 2% FBS per well was added. Photographs were taken at 0, 24, and 48 h, respectively.
Western Blot Analysis
Proteins were examined with specific antibodies against β-catenin (1:1,000 dilution, Cell Signaling Technology, Danvers, MA, USA), c-myc (1:500 dilution, Santa Cruz Biotechnology, Santa Cruz, CA, USA), survivin (1:1,000 dilution, Abcam, Cambridge, MA, USA), fibronectin (1:500 dilution, Santa Cruz Biotechnology), α-smooth muscle actin (α-SMA; 1:1,500 dilution, Sigma, St. Louis, MO, USA), and β-actin (1:5000 dilution, Santa Cruz Biotechnology), followed by peroxidase-conjugated secondary antibodies (Abcam). The reactions were detected using Immobilon Western Chemiluminescent HRP Substrate (Millipore).
WST-1 Assay
WST-1 assay kit was bought from Beyotime (Beijing, China). Cells for assay were cultured in 96-well plates (Corning), and the culture medium was changed with 100 μl fresh medium per well before each measurement. WST-1 working solution (10 μl) was added to each well, and the cells were incubated for another 2 h. Two hours later, the absorbance of the samples at 450 nm was measured using GloMax-Multi Detection System (Promega, Madison, WI, USA).
Assessment of Bystander Effects
hESC-ECs or EC-TF (Rluc-RFP-HSV-ttK) cells were mixed with MDA-MB-231-GFP (231-GFP) cells and seeded in 96-well plates. Ganciclovir (GCV; Keyi Pharmaceutic, Wuhan, Hubei, China) was added after the cells attached. After 5 days, the survival of 231-GFP cells was evaluated by GFP fluorescence intensity of cell lysates on GloMax-Multi Detection System.
Assessment of Tumor Tropism
Six-week-old female nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice were purchased from the Experimental Animal Institute, Chinese Academy of Medical Sciences (Beijing, China). All experimental procedures were conducted in conformity with institutional guidelines for the care and use of laboratory animals at Nankai University, Tianjin, China, and conformed to the National Institutes of Health (NIH) guidelines for the care and use of laboratory animals. MDA-MB-231 cells (1.5 × 106 per mouse) were intravenously injected into the mice. After 21 days, mice with pulmonary metastases (n = 6) and those without treatment (n = 6) were all given an intravenous injection of 1 × 106 hESC-EC-DF (Fluc-GFP) cells. BLI of Fluc was performed using the IVIS Lumina II system (Xenogen, Alameda, CA, USA). After intraperitoneal injection of d-luciferin (Invitrogen; 150 mg/kg), each mouse was imaged for 1–5 min. Bioluminescence signals were quantified in units of maximum photons per second per square centimeter per steridian (photons/s/cm2/sr).
Treatment of Metastatic Breast Cancer
MDA-MB-231-DF cells (1.5 × 106) were intravenously injected to each mouse (day 0). After 11 days, tumor-bearing mice began to receive (1) no treatment (control group), (2) GCV (GCV group), (3) EC-TF cells (EC-TF group), or (4) EC-TF cells combined with GCV (EC-TF + GCV group) (six mice for each group). Specifically, 1 × 106 EC-TF cells were intravenously injected to mice of groups 3 and 4 on day 11, 16, 21, 26. Twelve hours after every EC-TF cell injection, mice of groups 2 and 4 were given 25 mg/kg GCV every 12 h intraperitoneally on days 11–15, 16–20, 21–25, and 26–30. Four cycles of treatment were given. The development of pulmonary metastases was evaluated by BLI of Fluc and the tropism of hESC-ECs toward tumor was tracked by Rluc imaging. For tumor area calculation, we euthanized all mice at day 36 and performed hematoxylin and eosin (H&E; Zhongshanjinqiao, Beijing, China) staining of lung tissue. The sections were observed under microscope. For each mouse, five to seven representative 40× fields were chosen and photographs were taken. Using Image J software (NIH, Bethesda, MD, USA), we circled tumor foci and calculated the percentage of tumor area in the lung.
Immunohistochemical Staining
Specific antibody against GFP (1:50 dilution, Invitrogen) followed by peroxidase-conjugated goat anti-rabbit IgG (Vector Laboratories, Burlingame, CA, USA) were used. DAB peroxidase substrate kit (Vector Laboratories) was employed for detection. Then the sections were costained with hematoxylin, hydrated, mounted, and observed under the microscope.
Immunofluorescence Staining and TUNEL Assay
For immunofluorescence staining, specific antibodies against tight junction associated zona occludens protein 1 (ZO-1; 1:50 dilution, Santa Cruz Biotechnology), β-catenin (1:100 dilution, Cell Signaling Technology), GFP (1:50 dilution, Invitrogen), and mouse CD31 (1:100 dilution, BD Biosciences, Bedford, MA, USA), followed by Alexa Fluor 488/594 labeled-secondary antibodies (Invitrogen), were used for detection. Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay was performed using DeadEnd Fluorometric TUNEL System (Promega) according to the manufacturer's directions. Then the sections were counterstained with DAPI, mounted, and observed under the microscope.
Statistical Analysis
Statistics were calculated using SigmaStat for Windows Version 3.5 (Systat, San Jose, CA, USA). For comparison between two groups, two-tailed Student's t test was used. And for comparison between multiple groups, we used oneway ANOVA followed by Bonferroni test. Differences were considered as significant at values of p < 0.05.
Results
hESC-ECs Inhibit Proliferation and Induce Apoptosis of Breast Cancer Cells In Vitro
The direct influence of potential therapeutic vehicles on tumor cells is of great importance. In order to test whether cultured hESC-ECs can release any soluble factors that affect the growth of tumor cells, we collected conditioned medium from hESC-ECs (EC-CM). Treatment with EC-CM resulted in a remarkable inhibition of MDA-MB-231 cell proliferation, while conditioned medium from MDA-MB-231 cells (231-CM) could hardly affect cell proliferation rate (Fig. 1A). We also tested the effects of EC-CM on MDA-MB-231 cell cycle progression. After treatment with EC-CM, the percentage of cells in G0/G1 phase increased from 43.1% to 55.1%, while cells in G2/M phase declined from 9.5% to 0.6% (p < 0.01) (Fig. 1B). To determine if hESC-ECs can induce apoptosis of breast cancer cells, the apoptosis analysis was performed. Treatment with EC-CM led to a higher apoptosis rate of MDA-MB-231 cells, and both the rate of early apoptotic and late apoptotic cells increased (Fig. 1C). To uncover the underlying mechanism, we tested the expression of proliferation-associated molecule c-myc and inhibitor of apoptosis protein survivin in MDA-MB-231 cells, both of which were found to be downregulated by EC-CM (Fig. 1D).

hESC-ECs inhibit proliferation and induce apoptosis of breast cancer cells in vitro. (A) The female breast adenocarcinomaderived MDA-MB-231 cells cultured with the conditioned medium of female human H9 embryonic stem cell line-derived endothelial cells (hESC-EC conditioned medium; EC-CM) proliferated more slowly, and there was no significant difference between the MDA-MB-231 conditioned medium (231-CM) group and control group. (B) After treatment with EC-CM for 48 h, MDA-MB-231 cells in G0/G1 phase increased, and those in G2/M phase decreased significantly. (C) Treatment with EC-CM for 48 h increased the apoptosis rate of MDA-MB-231 cells. (D) c-Myc and survivin expression of MDA-MB-231 cells were downregulated by EC-CM. The results were representative of three independent experiments. #p < 0.01.
hESC-ECs Inhibit Breast Cancer Cell Migration In Vitro
We also detected a possible influence of hESC-ECs on breast cancer cell migration. Using a transwell migration assay, treatment with EC-CM resulted in a dramatic decrease of migrated MDA-MB-231 cell numbers compared to control, and only a slight decrease on migrated cell number was caused by 231-CM (Fig. 2A). A wound-healing assay was also performed, which showed that the speed of wound healing was slowed down by EC-CM at both time points we tested (Fig. 2B). Epithelial–mesenchymal transformation (EMT) is considered a significant mechanism of tumor cell migration; we therefore investigated the expression of EMT markers. We found that both of the mesenchymal markers, fibronectin and α-SMA, were downregulated by EC-CM (Fig. 2C). By contrast, the epithelial marker ZO-1, which was not expressed on the membrane of control MDA-MB-231 cells, was upregulated by EC-CM, and translocated to the cell membrane (Fig. 2D). ZO-1, fibronectin, c-myc, and survivin are all downstream molecules of β-catenin, and considering the significant roles of wingless-type mouse mammary tumor virus (MMTV) integration site family (Wnt)/β-catenin signaling pathway in tumor growth and metastasis, we proposed that soluble factors released by hESC-ECs may suppress Wnt/β-catenin signaling pathway in MDA-MB-231 cells. Corresponding to our hypothesis, immunofluorescence staining revealed that treatment with EC-CM downregulated β-catenin expression and also decreased its nuclear assembly (Fig. 2E, right), as confirmed by Western blot analysis (Fig. 2E, left).

hESC-ECs suppress migration of breast cancer cells in vitro. (A) Migrated MDA-MB-231 cells per field were decreased after being treated with EC-CM in transwell migration assay with 10% fetal bovine serum (FBS) in the lower chamber. (B) Representative photographs of wound-healing assay of MDA-MB-231 cells treated with 231-CM and EC-CM. (C) Expression of mesenchymal markers, fibronectin (FN), and α-smooth muscle actin (α-SMA) in MDA-MB-231 cells were downregulated by EC-CM. (D) Expression of tight-junction-associated protein zona occludens protein 1 (ZO-1) in MDA-MB-231 cells treated with EC-CM was upregulated and translocated to cell membrane. Scale bar: 100 μm. (E) Treatment with EC-CM downregulated β-catenin expression and reduced its nuclear assembly in MDA-MB-231 cells. Scale bar: 50 μm. The results were representative of three independent experiments. #p < 0.01. DAPI, 4′,6-diamidino-2-phenylindole.
hESC-ECs Exhibit Bystander Effects on MDA-MB-231 Cells via HSV-ttk/GCV Suicide System
To obtain cells that can be tracked and also express suicide gene for therapy as well, we transduced hESC-ECs with pLV-EF1α-TF-Bsd to establish an EC-TF cell line (Fig. 3A). More than 80% of EC-TF cells were killed in the presence of 100 μM to 1 mM GCV, while the viability of nontransduced hESC-ECs was not apparently affected (Fig. 3B). In the presence of 200 μM GCV, the toxicity effect reached its maximum degree in 3 days, demonstrating the utility of the HSV-ttk/GCV suicide system (Fig. 3C). To evaluate the bystander effect, we mixed EC-TF cells with 231-GFP cells in variable ratios, and 200 μM GCV was given. After 5 days, the viability of MDA-MB-231 cells as measured by the relative GFP intensity, declined by more than 50% in the 25%, 50%, 75%, and 90% mixture groups (Fig. 3D). More than 30% of MDA-MB-231 cells mixed with the same number of EC-TF cells could be killed in the presence of 10 μM GCV. By contrast, a similar effect was only achieved when 1 mM GCV was given to the mixture of MDA-MB-231 cells and hESC-ECs, which demonstrated that the bystander effect was mediated by HSV-ttk/GCV system, but not by hESC-ECs themselves (Fig. 3E).

HSV-ttk expression in hESC-ECs increases sensitivity to prodrug ganciclovir (GCV) and HSV-ttk-expressing hESC-ECs exhibit bystander effect on MDA-MB-231 cells in vitro. (A) Schema of triple fusion (TF) reporter gene containing fusion of renilla luciferase, red fluorescence protein, and herpes simplex virus truncated thymidine kinase (Rluc-RFP-HSV-ttk). IRES, internal ribosome entry site; Bsd, blasticidin. (B) hESC-ECs or EC-TF cells were treated with increasing concentrations of GCV, and viability of cells was measured by WST-1 assay 5 days later. (C) hESC-ECs or EC-TF cells were cultured with 200 μM GCV for 4 days, and WST-1 assay was performed every day. (D, E) hESC-ECs exert significant bystander effects on 231-green fluorescent protein-positive (GFP) cells via HSV-ttk/GCV suicide gene system. (D) Mixtures of EC-TF cells and 231-GFP cells in the ratios indicated were treated with medium alone or 200 μM GCV for 5 days, and the viability of 231-GFP cells was indicated by the relative GFP intensity compared to control. (E) Mixtures of EC-TF cells and the same number of 231-GFP cells were treated with 10 μM to 1 mM GCV for 5 days, and the viability of 231-GFP cells was measured as mentioned above. The results were representative of three independent experiments, #p < 0.01.
hESC-ECs Aggregate in Tumor Sites After Intravenous Injection
To effectively deliver localized therapy to tumors, potential vehicles must possess tumor tropism. Using a transwell assay, we observed that hESC-ECs could be attracted by MDA-MB-231 conditioned medium in the lower chamber (Fig. 4A). To track injected hESC-ECs in vivo, we transduced them with Fluc reporter gene. hESC-ECs (1 × 106) labeled with DF reporter gene were intravenously injected into mice with MDA-MB-231 pulmonary metastases, and BLI was performed to monitor the injected cells. For tumor-free mice, signals diffused in 18 h after injection, whereas for mice with pulmonary metastases, signals stayed in the lung without apparent diffusion until 72 h later (Fig. 4B, left). The pulmonary signal ratios between tumor-bearing and control mice increased gradually from 0 to 48 h after injection (Fig. 4B, right). To further investigate the distribution of hESC-ECs, we euthanized the mice 48 h after cell administration, and lungs were isolated and processed for immunohistochemical and immunofluorescence staining. hESC-ECs were detected mostly near the periphery of tumor foci or around tumor vessels, with a few diffused sparsely in the adjacent normal lung tissue (Fig. 4C, D). We also performed a TUNEL assay and found that injected hESC-ECs could induce the apoptosis of adjacent tumor cells in vivo without affecting normal tissue (Fig. 4E).

Systemically administered hESC-ECs can aggregate in tumor sites. (A) Transwell migration assay of hESC-ECs with Dulbecco's modified Eagle's medium (DMEM), 231-CM, and 15% fetal bovine serum (FBS) in the lower chamber. Data were representative of three independent experiments. (B) After MDA-MB-231 pulmonary metastases were formed, 1 × 106 hESC-ECs labeled with double fusion (DF) reporter gene were intravenously injected to tumor-bearing mice, and bioluminescence imaging (BLI) was performed. hESC-ECs were sequestered much longer in the lung of mice with metastases compared to control (n = 6). (C) Tail vein-injected hESC-ECs aggregate in pulmonary metastases. The injected cells were detected by immunofluorescence staining using specific anti-GFP antibody. hESC-ECs were found mostly around tumor foci instead of in normal lung tissue. Scale bar: 100 μm. (D) Intravenously administered hESC-ECs were also found around the tumor vessels (cluster of differentiation 31, CD31) by immuno-histochemical and immunofluorescence staining. Scale bar: 100 μm. (E) Tail vein-injected hESC-ECs induced apoptosis of adjacent tumor cells (terminal deoxynucleotidyl transferase dUTP nick end labeling, TUNEL). Scale bar: 200 μm. #p < 0.01.
In Vivo Inhibition of Tumor Growth by hESC-ECs Armed with HSV-ttk
To evaluate the efficacy of hESC-ECs as vehicles in tumor therapy, a breast cancer pulmonary metastasis model in NOD/SCID mice was used. Figure 5A showed the outline of treatment with EC-TF cells. MDA-MB-231-DF cells (1.5 × 106) were injected intravenously to each mouse to form tumor lesions in the lung. The mice bearing tumor were divided into four groups: (1) control group, (2) GCV group, (3) EC-TF group, and (4) EC-TF + GCV group, with each group receiving treatments as described in Materials and Methods. We monitored the development of tumor by BLI of Fluc from the 10th day. hESC-ECs were tracked by Rluc imaging (data not shown). The average Fluc signal increased dramatically in control and GCV groups, with no significant difference between them. By contrast, in the EC-TF group, signals increased much more slowly and showed significant difference compared to the first two groups. This inhibition effect was further amplified in EC-TF + GCV group (Fig. 5B). To elucidate the progression of metastasis, H&E staining was also performed, which confirmed the results of BLI (Fig. 5C).
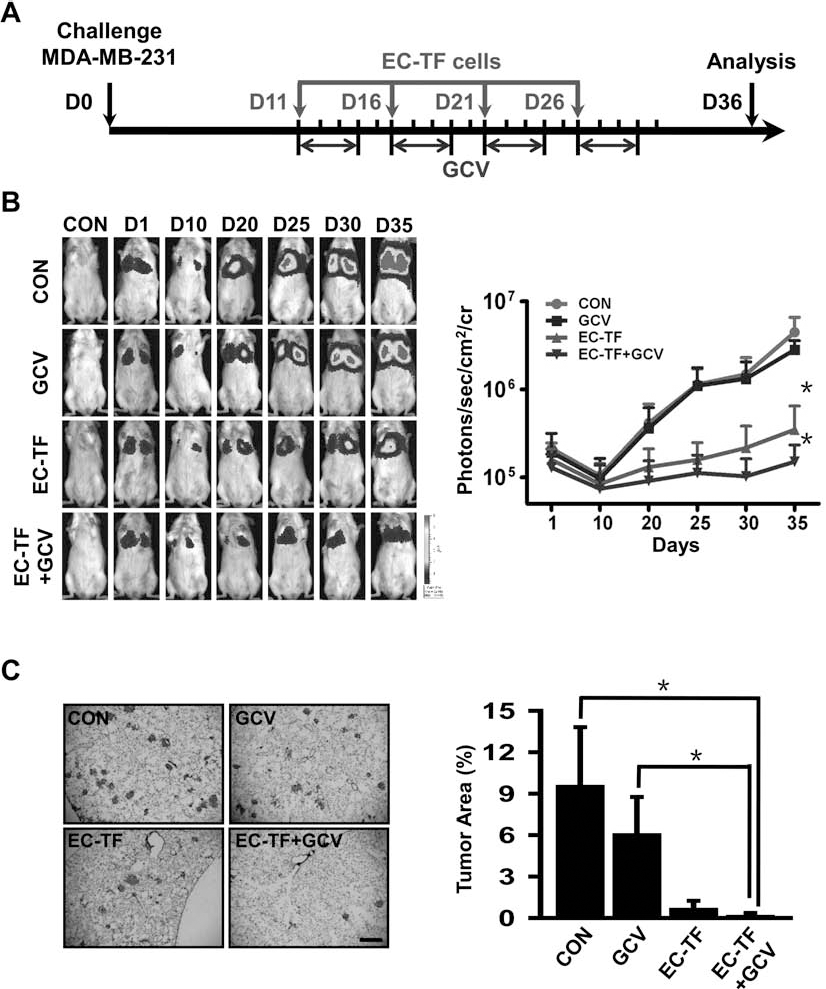
In vivo inhibition of tumor growth by hESC-ECs via HSV-ttk/GCV system. (A) Schema of treatment of metastatic breast cancer with EC-TF cells. (B) Firefly luciferase (Fluc) imaging of pulmonary metastases development in vivo. For each group, a representative mouse at each time point was shown. And quantitative analysis of BLI signals was shown as photons/s/cm2/sr on the right. (C, left) Representative hematoxylin and eosin (H&E) staining of lung sections from each group. Scale bar: 500 μm. (C, right) Percentage of tumor area in lung sections from each group. *p < 0.05.
Given the observation of significant inhibition of tumor by hESC-ECs, we hypothesized that hESC-ECs can inhibit the proliferation and migration of tumor cells via suppression of Wnt/β-catenin signaling pathway, and hESC-ECs can effectively kill nearby tumor cells via the HSV-ttk/GCV suicide system (Fig. 6).

Putative model outlining the tumor-suppressive effects of the hESC-ECs by inhibiting Wnt/β-catenin signaling pathway or inducing tumor cell death through bystander effect in human metastatic breast cancer.
Discussion
In order to improve the efficacy and minimize the toxicity of current gene therapy approaches, stem and progenitor cells are emerging as ideal delivery vehicles in tumor-selective therapy for primary and metastatic cancer (1,6,8,28). Mesenchymal stem cells (MSCs) have been modified into anticancer vectors carrying various cytokine, suicide genes, and other oncolytic genes, and this approach has showed significant antitumor effect in vivo (2,23,24). Due to their ability to circulate, home in on ischemic sites, and differentiate into mature endothelial cells, EPCs also hold enormous potential as delivery vehicles in antitumor therapies (33,34).
In preclinical experiments, therapeutic gene-modified mouse or human EPCs could successfully aggregate in tumor sites and offer a novel strategy for treating cancer using gene therapy (7,22,32). However, the exact mechanisms by which EPCs might contribute to tumor angiogenesis and the origin of neovascular endothelial cells of tumors are controversial (21). Previous reports revealed that hESCs have an inhibitory effect on tumor proliferation, but an analogous effect by hESC-ECs has not been investigated (4,20). In order to provide an initial assessment of the potential therapeutic index of hESC-ECs, our current study tested whether these cells could emit regulatory cues to cancer cells in vitro. Our findings provide evidence that soluble factors secreted by hESC-ECs can suppress the Wnt/β-catenin signaling pathway and thereby inhibit the proliferation and migration of MDA-MB-231 cells. The apparent paracrine findings we describe here may help introduce a new dimension on the use of hESC-ECs in regenerative medicine and cancer therapy. Detailed characterization of the properties of hESC-ECs after tumor engraftment should be addressed in future studies.
Further demonstration of tumor tracking properties of hESC-ECs in vivo could present attractive strategies for systemic transgene delivery into the sites of tumor formation. However, the successful clinical application of future hESC-ECs transplantation-based tumor therapies needs noninvasive imaging approaches to monitor the fate and tissue distribution of transplanted cells (31,35). In this study, we investigated the feasibility of using hESC-ECs as both gene carriers and imaging probes in a breast cancer animal model. We showed that the introduction of a suicide gene into hESC-ECs could produce a tumor-specific pro drug converting cellular vehicle. Moreover, the teratoma formation, a major hurdle to treating patients with hESCs, was not detected after hESC-ECs transplantation by bioluminescence imaging.
Breast cancer metastasis causes nearly 90% of the mortality among breast cancer patients, and metastatic breast cancer is the most difficult type of breast cancer to treat and control (10,27). In this study, we used hESC-ECs for systemic cancer therapy for the first time, with encouraging initial results. Here, we showed for the first time the capability of hESC-ECs to aggregate in tumor sites. This is consistent with other reports describing tumor-directed migratory abilities of EPCs derived from bone marrow or cord blood (16,22). The lung metastatic tumors were suppressed in mice that received hESC-ECs, and this effect was ameliorated by GCV administration. These results clarify the feasibility and effectiveness of using these “tumor tracking” cells as therapeutic delivery vehicles to treat tumors by utilizing their antiangiogenic and/or cytotoxic effects. Given their unique property to migrate to pathological lesions, MSC or EPC delivery systems carrying various cytokine, suicide genes, or other oncolytic genes have become the focus of many investigations (22,23). In the case of EPC-based therapeutic approaches, their particular advantages lie in the favorable (i.e., hypoxic) microenvironment can guide EPCs to the tumor site by homing signals.
In conclusion, hESC-ECs can suppress the Wnt/β-catenin signaling pathway by soluble factors and thereby suppress the proliferation and migration of breast cancer cells. After systemic administration, hESC-ECs can home in on established pulmonary metastases. Moreover, we demonstrated the bystander effect of hESC-ECs transduced with HSV-ttk on adjacent MDA-MB-231 cells, suggesting the possibility of using hESC-ECs in tumor therapy. These desirable properties make hESC-ECs ideal vehicles for cancer gene therapy. Although the mechanisms and the magnitude of hESC-ECs' involvement in tumor-associated angiogenic and vasculogenic processes are not certain, these cells have great potential to become valuable tools in designing future antitumor strategies.
Footnotes
Acknowledgments
This work was partially supported by grants from the National Basic Research Program of China (2011CB964903) to Z.L. and (2013CB967201) to R.X., Program for New Century Excellent Talents in University (NCET-12-0282) to Z.L., National Natural Science Foundation of China (31071308) to Z.L. and (30830096) to R.X., Tianjin Natural Science Foundation (12JCZDJC24900) to Z.L., and the Key Project of Tianjin Scientific and Technological Commission for China-Sweden Cooperation Research Program (09ZCZDSF04000) to R.X. The authors declare no conflicts of interest.
