Abstract
Injection of autologous bone marrow cells into infarcted myocardium has been proposed to limit the deterioration of cardiac function following myocardial infarction (MI); unfortunately, the beneficial effects observed have been modest. One of the limiting factors is believed to be poor local survival of the injected cells, but the potential impact of apoptosis among the injected cells has yet to be assessed. Therefore, this study aimed to quantify the apoptosis rate in bone marrow mononuclear cells (BMMCs) prepared for cardiac therapy, and to analyze their effects in vitro on cardiomyoblast apoptosis and in vivo on cardiac function recovery following MI. Using rabbit BMMCs prepared by Ficoll gradient, apoptotic cells were detected via Annexin V (AnV) staining. The effects of depleting the apoptotic cell population by means of AnV magnetic beads was tested in vitro after coculture with cardiomyoblasts (H9c2 cells) and in vivo after cell injection into the infarcted area. Left ventricular ejection fraction and scar extent were assessed by echography and histology 2 months later. After Ficoll gradient isolation, 37.3% (33.4–37.9%) of BMMCs were found to be apoptotic (ApoBase BMMCs). AnV depletion decreased the proportion of apoptotic cells to 20% (17.6–32%) (ApoLow BMMCs). Rabbits treated in vivo with ApoLow BMMCs after MI presented with significantly improved left ventricular ejection fraction [41.4% (41.0–43.6%) vs. 34.6% (34.6–35.9%),
Introduction
Heart failure following myocardial infarction (MI) remains a deadly and economically costly disease (2,22). Transplantation of autologous bone marrow mononuclear cells (BMMCs) has been proposed as a new method to limit the deterioration of cardiac function after MI. The first clinical trials involving cardiac cell therapy after MI have reported discordant and overall modest results (6,12,15,18). It is believed that poor local survival of injected cells is a limiting factor to the efficacy of this therapeutic approach (17,25). Indeed, the rate of early cell death among BMMCs is relatively high, probably due to the restricted supply of oxygen and nutrients, local inflammation, and loss of microenvironment survival signals (5,10). In order to inject a sufficient amount of viable cells into the patient, a battery of quality control tests must be performed on the BMMCs immediately prior to injection (6,12,18,24). Unfortunately, routine viability tests do not assess the presence of cells that are still alive but engaged in an apoptotic process; thus, this aspect remains poorly documented. As apoptotic cells could be exerting influence on numerous biological processes through release of microparticles to induce apoptosis in neighboring cells (9,21,23), we speculated that injection of apoptotic cells could impair the efficacy of cell therapy.
In this study, we first aimed to document the presence of apoptotic cells among BMMCs that had been routinely prepared for use in cardiac cell therapy. Then, in order to assess the role of apoptotic cells in this setting, we compared the effects of BMMCs, partially depleted from apoptotic cells (ApoLow) or not (ApoBase), on H9c2 survival in vitro, and in vivo using a rabbit model of MI.
Materials and Methods
Animals
All experiments were conducted in syngeneic New Zealand rabbits weighing 3.5–4.5 kg. Rabbits were housed in individual cages under standard conditions of temperature (14–20°C) and light (12 h per day) with food and water provided ad libitum. A delay of 1 week between each animal's arrival and the initiation of experiments was systematically respected. All experiments were conducted in accordance with the European Communities Council directive (86/609/EEC) after approval was obtained from the local Ethics Review Board of Lille University, and conformed to the US National Institutes of Health guidelines for the care and use of laboratory animals.
BMMC Isolation and Assessment of Cell Viability
Bone marrow (BM) was isolated from the rabbit's posterior leg bone. Bone extremities were surgically opened under sterile conditions and bones flushed with anticoagulant solution (ACD-A; Maco Pharma, Mouvaux, France) to isolate the crude BM, which was then filtered through a 100-μm filter and centrifuged to pellet (400 ×
Assessment of Cell Apoptosis
Simultaneous to the evaluation of cell viability, assessment of apoptosis was performed by incubation of BMMCs with FITC-labeled Annexin V (Miltenyi Biotec, Bergisch Gladbach, Germany) in order to detect the redistribution of phosphatidylserine on the external cell membrane of apoptotic cells. Samples were analyzed on a Beckman Coulter flow cytometer (Beckman Coulter; Villepinte, France), and results analyzed by accompanying Expo32 software. Apoptotic BMMCs were defined as Annexin V positive – PI negative, dead cells as Annexin V positive – PI positive, and viable nonapoptotic cells as Annexin V negative – PI negative.
The proportion of apoptotic BMMCs was also assessed by detecting the presence of DNA lesions by using cytoimmunochemistry (Apostain technique; Bender MedSystems, Vienna, Austria). The Apostain technique is based on the characteristic of increased sensitivity of apoptotic cells' DNA to thermic denaturation. Briefly, BMMCs were spun down onto histological slides, and permeabilized with saponine. The DNA denaturation capacity was determined by heating cells at 56°C in the presence of formamide. Slides were cooled down for 5 min in cold PBS. Nonspecific blocking was performed by incubation in 3% fat milk for 20 min at 37°C. Slides were incubated with monoclonal antibody (MAb) F7–26, specific to single-stranded DNA, for 30 min at room temperature. MAb F7–26 antigen detection was performed by using mouse anti-IgM FITC-labeled antibody for 30 min at room temperature. Finally, slides were counterstained with DAPI and the proportion of apoptotic cells was determined as the ratio of green FITC-positive cells to DAPI-positive cells.
Assessment of Apoptosis Pathway Activation
To evaluate the activation of apoptotic pathways in BMMCs, we analyzed the activation of mitochondrial and Fas ligand receptor pathways. We analyzed the mitochondrial pathway through evaluation of mitochondrial membrane potential by flow cytometry. In apoptotic cells, the decrease of mitochondrial membrane potential results in the loss of mitochondrial capacity to accumulate lipophilic and cationic probes like JC1 (5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolcarbocyanin iodide; Sigma-Aldrich, St. Louis, MO, USA). As a consequence, monomers of JC1 remain in the cytosol and emit green fluorescence (527 nm). In nonapoptotic cells aggregates of JC1 accumulate in the mitochondria, due to the high membrane potential, and emit red fluorescence (590 nm). The ratio of red to green fluorescence is dependent only on mitochondrial membrane potential. We used FCCP (mesoxalonitrile-4-trifluoromethoxyphenylhydrazone; Sigma-Aldrich) as a positive control. FCCP artificially depolarizes the mitochondrial membrane, which in turn simulates the decrease of the mitochondrial membrane potential as observed in apoptotic cells. Each analysis was carried out with samples of 0.5 × 106 BMMCs.
To analyze the Fas ligand receptor pathway, we used a reverse-transcriptase polymerase chain reaction (RT-PCR) technique. Briefly, samples of 5 × 106 cells were incubated with a primer designed from the messenger Ribosomal Nucleic Acid (mRNA) of
Preparation of ApoLow BMMCs and Assessment of Cell Function
Immediately after Ficoll gradient isolation, a portion of BMMCs was incubated with Annexin V-labeled microbeads (Annexin V Microbeads Kit; Miltenyi Biotec, Bergisch Gladbach, Germany). Cells were then sorted in MS type columns through a magnetic separator (MIDI-MACS; Miltenyi Biotec, Bergisch Gladbach, Germany) that retained Annexin V-positive cells. The residual cell suspension was depleted of apoptotic BMMCs and designated as ApoLow BMMCs. In contrast, BMMCs that did not undergo the depletion procedure were designated as ApoBase BMMCs. We compared the capacity of ApoLow and ApoBase BMMCs for colony-forming unit (CFU) and migration capacity. To assess CFU capacity, 104 cells were plated in METHOCULT media (GF H4435; Stem-Cell Technologies Inc., Vancouver, BC, Canada) for 14 days. Colonies were then counted by two independent investigators. The experiments were performed in triplicate and results expressed as number of colonies/cell culture plates (CFU-GM; colonies = 50 cells). In order to assess migration capacity of BMMCs, 250 μl of cell suspension (4 × 105 cells/ml) were plated in Boyden chambers with either 500 μl of standard medium, or standard medium + SDF-1 (100 ng/ml) or standard medium + VEGF (50 ng/ml) for 24 h at 37°C. The supernatant was then discarded, the chambers fixed with 4% PFA for 10 min, and then colored with Giemsa to obtain cell count. Transmigrated cells were counted by two independent investigators. Results are expressed as the ratio between the number of cells that migrated in the presence or in the absence of cytokine.
Model of MI and Intramyocardial Injection of BMMCs
Rabbits were anesthetized with ketamine (50 mg/kg) and xylazine (6 mg/kg) and then intubated for mechanical ventilation (SAR-830A Ventilator, Harvard Apparatus, MA, USA). Myocardial infarction was performed by transient ligation of the main left descendant coronary artery for 1 h, followed by reperfusion. Coronary ligation was confirmed by myocardium pallor and kinetic alteration. BMMCs injection was performed 1 week after MI. Animals were anesthetized using the same protocol and the chest was reopened. Scar tissue was identified as pale akinetic tissue. Using a 29-gauge needle, placebo (buffer PBS) or 2 × 106 freshly isolated syngeneic BMMCs (either ApoBase or ApoLow) were injected into two spots, at 1 × 106 BMMCs each, in the border zone of the MI scar. Success of the injection was controlled by the formation of a little epicardial wheal. The needle was cautiously removed in order to limit leakage of the cell suspension.
Assessment of BMMC Survival and of Cardiomyocyte Apoptosis Following Intramyocardial Injection
In order to determine the impact of apoptotic BMMCs on cell survival after intramyocardial injection, intramyocardial injection of 2 × 106 ApoBase BMMCs or ApoLow BMMCs was performed 1 week after MI. Immediately after Ficoll gradient isolation, cells were labeled with Hoechst 33342 (Molecular Probes, Invitrogen, Carlsbad, CA, USA) and then injected. Animals were sacrificed at 1, 2, and 24 h after cell injection to perform histological analysis (
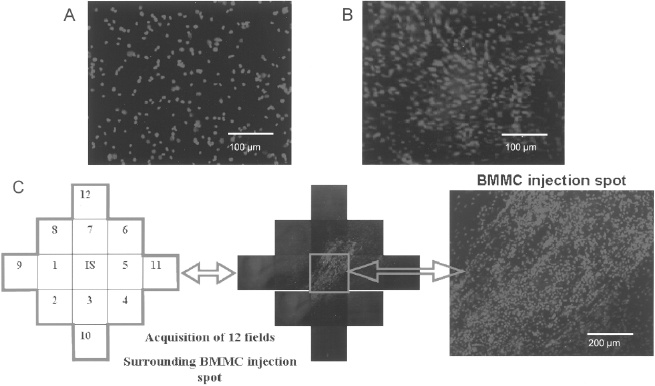
BMMC count after intramyocardial injection. (A) Representative aspect of Hoechst-labeled BMMCs before injection. (B) Representative aspect of Hoechst-labeled BMMCs after intramyocardial injection. (C) After heart excision (at 1, 2, and 24 h following BMMC injection,
Apoptosis in myocardial host tissue was assessed by Apostain on frozen cardiac tissue sections sampled 24 h and 2 months after injection of BMMCs.
Assessment of Left Ventricular Remodeling and Left Ventricular Ejection Fraction
Five rabbits were included in each group: placebo, ApoBase BMMCs, and ApoLow BMMCs. In vivo measurements of left ventricular (LV) dimensions and systolic function were performed under anesthesia by transthoracic echocardiography with a sequoia Acuson 512 and a 15 MHz linear pediatric probe. LV dimensions and systolic function were assessed before MI, 1 week after MI, just before the BMMC injection, and 2 months after cell therapy and just before sacrifice. In order to evaluate LV remodeling, we measured LV end diastolic and end systolic diameters (LVEDD and LVESD, respectively) and estimated the corresponding volumes (LVEDV and LVESV) with the following simplified Simpson's formula: (0.85 × short axis area × long axis area)/long axis diameter. Left ventricular ejection fraction (LVEF) was estimated by the Simplified Simpson method (LVEF = LVEDV – LVESV/LVESV).
Assessment of Cardiac Scar After MI
Two months after MI and cardiac cell therapy, rabbits were sacrificed; hearts were then excised, and placed in OCT solution before freezing in isopentane and liquid nitrogen. LV cross sections, 7 μm in thickness, were obtained as oriented along the LV short axis. A Masson's trichrome staining was performed to delineate the extent of the fibrotic infarct region. Using Sigma ScanPro software, the extent of both endocardial and epicardial scar formation was measured. For each tissue section, endocardial scar was calculated as extent of endocardial fibrosis over total endocardial circumference. Similar calculation was performed for the epicardial side. MI size was reported as the average of the extent of the endo- and epicardial scars over three section levels (apex, midventricular, and basal).
In Vitro Assays: Impact of Apoptotic BMMCs and of Apoptotic Microparticles on Cardiomyoblast Apoptosis
H9c2 cells (European Collection of Cell Cultures) were plated in modified DMEM (DMEM + 10% FCS + 1% penicillin-streptomycin) in hypoxic condition (1% oxygen for 24 h) in order to mimic the condition of the myocardial border zone after MI. To evaluate the level of oxidative stress, H9c2 cells were exposed for 30 min to H2DCF (2′,7′-dichlorodihydrofluorescein diacetate; Molecular Probes, Invitrogen), a nonfluorescent dye that is converted into fluorescent DCF in the presence of hydrogen peroxide and peroxynitrite. The level of oxidative stress was then evaluated by fluorescence microscopy. We assessed the impact of apoptotic BMMCs on surrounding H9c2 in different culture conditions: H9c2 + control media; H9c2 + 2 × 106 ApoBase BMMCs; H9c2 + 2 × 106 ApoLow BMMCs; H9c2 + supernatant from ApoBase BMMCs; and H9c2 + supernatant from ApoLow BMMCs. After 24 h, nonadherent cells (i.e., BMMCs) were removed and adherent cells were trypsinized and morphological analysis was performed on cytospins to confirm that apoptotic cells were H9c2. The rate of H9c2 apoptosis was assessed by Apostain technique.
Microparticles were isolated from ApoBase and ApoLow BMMC supernatants by double centrifugation, as previously described for platelets (7). Microparticles were analyzed and quantified by flow cytometry after identification of their size (0.5–0.8 μm) using fluorescent beads of different sizes for gating (MEGAMIX; Biocytex, Marseille, France) and Annexin V expression.
Statistical Analysis
Results were expressed as median (25th and 75th percentiles). Comparison of continuous variables between more than two groups was performed using the Kruskall-Wallis test. In cases of significant values of
Results
Evidence for the Presence of Apoptotic BMMCs After BM Isolation
After isolation of BMMCs by Ficoll gradient, cell viability was determined to be 91.7% (86.1–95.4%) as assessed by flow cytometry and IP staining. After gating the viable BMMCs, we observed an average of 37.3% (33.4–37.9%) apoptotic cells as assessed by Annexin V staining (Fig. 2A), and 33.85% (27.31–39.47%) as assessed by immunocytochemistry (Fig. 2B).
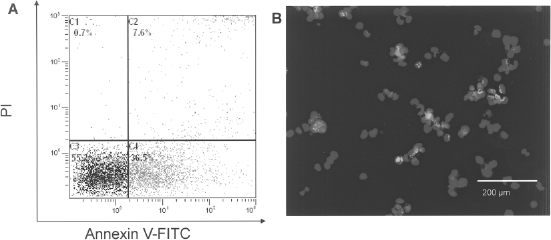
Identification and quantification of apoptotic BMMCs. Representative aspects of quantification of apoptotic BMMCs among viable BMMCs by flow cytometry and immunostaining. (A) Quantification of apoptotic BMMCs by flow cytometry after incubation with FITC-Annexin V. Apoptotic cells (Annexin V positive/PI negative) are in the lower right square whereas nonviable cells (Annexin V positive/PI positive) are in the upper right square. (B) DNA lesions of apoptotic BMMCs revealed by Apostain technique. Apoptotic BMMCs were identified by colocalization of blue nuclei and FITC fluorescence of F7–26 Ab that bind single DNA strain. BMMCs: bone marrow mononuclear sells; PI: propidium iodide.
Efficacy of Apoptotic BMMC Depletion and Assessment of Apoptosis Pathway Activation
After depletion of apoptotic cells with the use of Annexin V microbeads, the proportion of apoptotic BMMCs was reduced from 37.3% (33.4–37.9%) (ApoBase group) to 20.0% (17.6–32.0%) (ApoLow group) as assessed by flow cytometry (

Efficacy of apoptotic cells depletion. (A) After depletion with Annexin V microbeads, the number of apoptotic BMMCs was significantly reduced as assessed by Apostain technique. (B) Representative aspect of mitochondrial membrane potential assessment with the use of cationic probe JC1. In nonapoptotic cells, JC1 aggregates in the mitochondria emit predominantly red fluorescence (i.e., high red/green ratio). In apoptotic cells, the decrease of mitochondrial potential membrane results in the release of JC1 monomers in the cytoplasm that emit predominantly green fluorescence (i.e., low red/green ratio). In this example, the proportion of BMMCs with low mitochondrial potential membrane was 25% in ApoBase and 8% in ApoLow BMMCs. (C) Expression of FAS receptor pathway was assessed by polymerase chain reaction technique. Fas receptors were overexpressed in ApoBase BMMCs. BMMCs: bone marrow mononuclear cells; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Depletion of Apoptotic Cells From BMMCs Does Not Modify BMMC Functions
The number of CFU-GM colonies was not different between ApoLow BMMCs and ApoBase BMMCs [138 (49–312) and 129.5 (59–219), respectively,
Assessment of BMMC Survival and of Cardiomyocyte Apoptosis Following Intramyocardial Injection
Animals received unselected (ApoBase) BMMCs or selected (ApoLow) BMMCs 1 week after MI, and were sacrificed for histological analysis at 1, 2, and 24 h following cell injection. The number of residual BMMCs detected at 1 and 2 h after injection was similar in the two groups (Fig. 4A). Twenty-four hours after injection, the number of BMMCs detected in animals who had received ApoLow BMMCs was twice as high as that in animals who had received ApoBase BMMCs [8,383 (6,649–10,470) vs. 4,264 (4,053–5,555), respectively,
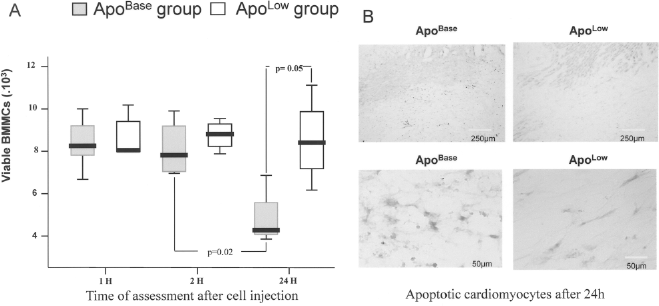
Depletion of apoptotic BMMCs improves BMMC survival after intramyocardial injection and decrease surrounding cardiomyocytes apoptosis. One week after MI, labeled BMMCs were injected in the MI border zone. Hearts were excised at 1, 2, and 24 h after injection for histological analysis. (A) No difference was observed between ApoBase and ApoLow group at 1 and 2 h after intramyocardial injection. After 24 h, the number of surviving cells was significantly higher in ApoLow group. (B) The apoptotis of cardiomyocytes in the border zone was assessed ex vivo by Apostain staining 24 h after BMMCs injection. Positive cells for Apostain (i.e., apoptotic cells) are brown. The pictures show a representative sample of the border zone after injection of ApoBase BMMCs (left column) and ApoLow BMMCs (right column). The upper line is a low magnification of the border zone. At a high magnification, there are more brown positive cells for Apostain (i.e., apoptotic cells) in the ApoBase section (lower left) than in the ApoLow group (lower right).
Depletion of Apoptotic BMMCs Limits LV Remodeling, Improves LVEF, and Reduces Scar Extent After MI
The effects of ApoBase, ApoLow BMMCs, and placebo (buffer PBS) injection on cardiac function were assessed in vivo using the same rabbit model of MI. At baseline before MI, and 1 week after MI, we observed no differences in echocardiographic parameters between the three groups. After 2 months, LVEF had significantly improved in animals treated with ApoLow BMMCs [41.4% (41.0–43.6%)] versus control [32.7% (32.7–33.1%),

Apoptotic BMMCs impair the efficacy of cell therapy after MI. (A) ApoBase BMMCs injection after MI slightly improved left ventricular ejection fraction. Significant improvement was observed in animals receiving ApoLow BMMCs. (B) Slight reduction of left ventricular diameters was obtained with ApoBase BMMCs therapy after MI compared to control group. Significant improvement was obtained in animals receiving ApoLow BMMCs. (C) Compared to control group, ApoLow BMMCs were more efficient than ApoBase BMMCs to reduce scar extension, although not significantly (
Apoptotic BMMCs Increase Apoptosis of Cardiomyoblasts
In a single culture model of H9c2, the level of oxidative stress after 24 h of hypoxia was significantly higher than after culture in normoxia conditions [885 AU (870–902) vs. 468 AU (452–482),
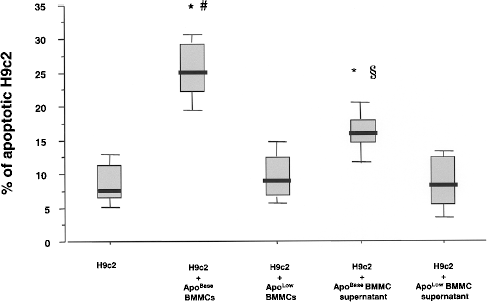
Apoptotic BMMCs increase apoptosis of surrounding H9c2 cardiomyoblasts. Coculture of H9c2 with ApoBase BMMCs increased the rate of apoptotic H9c2. This was not observed when H9c2 were cocultured with ApoLow BMMCs. Incubation of H9c2 with the supernatant of ApoBase BMMCs partially reproduced this effect. *
Discussion
In this study, we demonstrate that up to 30% of BMMCs, prepared for cardiac cell therapy, are engaged in the apoptotic process. Partial elimination of these apoptotic BMMCs from the cell therapy product prior to injection into infarcted myocardium 1) increased in vivo survival of injected BMMCs, 2) decreased cardiomyocyte apoptosis in the border zone, and 3) improved the efficacy of cell therapy on cardiac function. Moreover, coculture of BMMCs with cardiomyoblasts induced cardiomyoblast apoptosis when BMMCs or their supernatants were used. This effect was blunted after depletion of apoptotic BMMCs was carried out before coculture. Taken together, these results demonstrate that apoptotic BMMCs, which are commonly present among the cell therapy product, act to minimize the benefit of cardiac cell therapy. Our results suggest that it could be at least partially related to an increase in cardiomyocyte apoptosis, through both direct and paracrine mechanisms.
A Significant Proportion of Apoptotic BMMCs Is Observed After Density Gradient Isolation
The first observation in this study was a significant proportion of BMMCs being apoptotic before their injection into the myocardium. Since apoptosis is a dynamic process that is difficult to assess, we performed three complementary techniques to explore three different steps of the process. The decrease of mitochondrial potential membrane was used to represent an early step, whereas translocation of phosphatidylserine (Annexin V labeling) explored an intermediate step, and chromatin condensation (Apostain technique) explored the last step (20). We observed a similar proportion of apoptotic BMMCs with these three techniques. These results are in line with the notion that apoptosis is a natural regulatory process of hematopoiesis that might be further increased by sampling and isolation procedures (8). The proportion of apoptotic BMMCs before injection had not yet been explored in experimental or clinical studies (either cardiac or hematological). In our study, the proportion of apoptotic cells was assessed immediately after bone marrow isolation. The time between bone marrow harvest and BMMCs isolation will certainly be longer in clinical settings and the use of autologous serum might limit the number of apoptotic cells, although it remains to be demonstrated. Temperature variation should be avoided and cells kept at room temperature during all the process of BMMCs preparation.
In order to evaluate the role of apoptotic cells in the setting of cardiac cell therapy, we chose to deplete BMMCs in apoptotic cells by immunoadsorption on Annexin V-coated beads. This method is based on Annexin V binding to phosphatidylserine translocated to the external side of the cell membrane. This method of magnetic cell sorting led to a significant decrease of Annexin V-positive cells from the overall BMMC population, although the depletion was only partial. This method has the advantage of producing clinical grade cells while preserving proliferative and migration functions (3).
Depletion of Apoptotic Cells Increases BMMC Survival and Decreases Cardiomyocyte Apoptosis in Infarcted Myocardium
Twenty-four hours after injection, the number of injected BMMCs detected in the myocardium was higher in animals who had received depleted, rather than nondepleted, cells. Obviously, this effect may be the direct result of cell death occurring in injected apoptotic cells. However, this effect remained significant after normalization to the initial number of injected nonapoptotic cells. This suggests that the limitation of the number of apoptotic cells improved the overall survival of injected cells.
Simultaneously, Apostain analysis showed a lower rate of apoptotic cardiomyocytes in the border zone after injection of those BMMCs depleted in apoptotic cells, compared to nondepleted ones. Together, these results suggest that decreasing the number of injected apoptotic cells not only increased local survival of injected cells but also limited cardiomyocyte apoptosis. Other groups have previously reported that the transplantation of mesenchymal stem cells modified to resist apoptosis improved the efficacy of cell therapy. Hence, mesenchymal stem cells genetically modified to overexpress antiapoptotic protein such as akt (14) and bcl-2 (11) limited scar extension and improved cardiac function recovery. However, the use of genetically engineered cells is not ready for clinical application.
Impact of Apoptotic BMMCs on Surrounding Cardiomyoblasts
In order to investigate the possible link between apoptosis of BMMCs and that of cardiomyocytes, we analyzed the effect of ApoBase BMMCs and ApoLow BMMCs after in vitro coculture with H9c2. The rate of apoptotic H9c2 was significantly higher when H9c2 were cocultured with ApoBase BMMCs than with ApoLow BMMCs. Interestingly, the deleterious effect of ApoBase BMMCs was partially reproduced when H9c2 were cultured in the presence of supernatant from ApoBase BMMCs, but not in the presence of supernatant from ApoLow BMMCs. This finding demonstrates that H9c2 apoptosis is at least partially induced by paracrine mechanisms, which could be mediated by microparticles or by microparticle-derived factors.
Limitation of Apoptotic BMMCs Improves the Efficacy of Cell Therapy After MI
In the rabbit model, we assessed the effects of BMMCs on LV remodeling and systolic function recovery after MI. Cell therapy based on ApoBase BMMCs ended up in the improvement of LV remodeling and systolic function. Of note, the depletion of apoptotic BMMCs significantly enhanced the magnitude of LVEF recovery (absolute gain of +8.7% vs. control and +6.8% vs, ApoBase BMMCs).
Overall, both our in vitro and in vivo results suggested that apoptotic BMMCs are able to induce apoptosis of neighboring cardiomyoblasts and cardiomyocytes. As cardiac function is also improved after injection of BMMCs containing less apoptotic cells, we concluded that the presence of apoptotic cells among BMMCs is deleterious to local survival of injected cells and also to the surrounding myocardium. This may be surprising since apoptotic cell death induces less inflammatory response than necrosis and is known to have little deleterious effects on surrounding cells in other cellular environments. It has even been suggested that apoptotic transplanted cells may exert a beneficial effect in the infarcted myocardium by changing the immune status from inflammation to an anti-inflammatory state (21). However, it should be noted that this has been mainly demonstrated for macrophages, which express anti-inflammatory cytokines after ingestion of apoptotic cells. Direct effects on cardiomyocytes have not yet been explored. Apoptotic cells release microparticles that have various effects depending on cell type and apoptosis conditions (1,4,13,16,19,23). Thus, it remains possible that apoptotic BMMCs and/or apoptotic microparticles, growth factors, and paracrine factors released by apoptotic cells may induce apoptosis in cardiomyocytes, as suggested by our results.
Study Limitations
In this study, we deliberately chose to avoid the use of autologous serum in cell suspensions before cell injection. Serum is a potent antiapoptotic agent that is routinely added in cell suspensions in order to preserve cell viability and has been used in some (but not all) clinical trials, especially those in which cells had to be stored for several hours prior to injection. As serum harbors various growth and angiogenic factors that might interfere with the results of subsequent experiments, and independently of the apoptosis issue, we chose to modulate the rate of apoptotic cells through immunodepletion rather than through serum addition.
Conclusions
BMMCs prepared for cardiac cell therapy were found to contain a significant proportion of apoptotic cells, which impaired the efficacy of cell therapy through direct and indirect mechanisms. The depletion of apoptotic BMMCs prior to injection improved the efficacy of cell therapy after acute MI. Our results suggested that improvement of cell quality through limitation of apoptotic cells prior to injection has the potential to improve cardiac function recovery after cell therapy.
Footnotes
Acknowledgments
