Abstract
Myoblast sheet transplantation is a promising novel treatment modality for heart failure after an ischemic insult. However, low supply of blood and nutrients may compromise sheet survival. The aim of this study was to investigate the effect of mitochondria-protective Bcl-2-modified myoblasts in cell sheet transplantation therapy. In the Bcl-2-expressing rat L6 myoblast sheets (L6-Bcl2), increased expression of myocyte markers and angiogenic mediators was evident compared to wild-type (L6-WT) sheets. The L6-Bcl2 sheets demonstrated significant resistance to apoptotic stimuli, and their differentiation capacity in vitro was increased. We evaluated the therapeutic effect of Bcl-2-modified myoblast sheets in a rat model of acute myocardial infarction (AMI). Sixty-four Wistar rats were divided into four groups. One group underwent AMI (
Introduction
Although the first myoblast transplantation therapies were administered more than a decade ago for the treatment of heart failure (29, 34), problems associated with cell injections, such as the massive loss of donor cells, inadequate cell proliferation in the host myocardium, and arrhythmogenicity, remain to be solved. To overcome some of these problems, epicardial transplantation of cell sheets provides a conceptual alternative and a minimally invasive method for cell delivery. In this system, a tissue-engineered multicellular patch or cell sheet is made with a temperature-responsive cell culture dish (22, 31). With this technique freely transplantable cell sheets consisting of 3–6 million cells can easily be manufactured with no added scaffold material. Such sheets can then be implanted on top of the injured myocardium to which they adhere immediately. Moreover, therapy can be enhanced by piling two or more layers of sheets on top of each other. The superiority of sheet transplantation over intramyocardial injections for cell therapy of heart failure has been demonstrated in both small and large animal studies (12, 15, 20). The therapeutic effect of cell sheets is considered to be mediated via finite production of paracrine effectors that locally stimulate the underlying injured myocardium. In models of cardiac ischemia and infarction, myoblast sheet therapy has been shown to inhibit fibrosis and to stimulate angiogenesis (20).
These previous studies have shown that multiple myoblast sheets are required for ischemic heart failure therapy. The sheets on the injured myocardium are exposed to apoptotic stress and nutrient deprivation, and thus an increased number of cells are required for therapeutic benefit. In order to reduce the amount of transplanted sheets and to increase their tolerance of the death-promoting host environment, we investigated the effect of
The family of Bcl-2 proteins comprises several members with anti- or proapoptotic functions. Bcl-2 itself is antiapoptotic, functioning in the mitochondrial pathway by counteracting functions of the proapoptotic Bax and Bak, and inhibiting cytochrome c release (26). Bcl-2 over-expression has been shown to promote cell survival and to inhibit cell death induced by such apoptosis-inducers as staurosporine, or by nutrient deprivation (6). Bcl-2 gene therapy has been used in cardiac cell therapy to prevent apoptosis upon cell injection into the myocardium (17, 19).
In this study, we evaluated the Bcl-2 expressionmediated effects on L6 myoblast sheets both in vitro and in vivo. We first compared the apoptosis resistance and gene expression profiles of wild-type and Bcl-2-modified L6 myoblast monolayers and sheets. We then expanded these results to an in vivo setting, and evaluated the effects of wild-type and Bcl-2-overexpressing myoblast sheet transplantation in a rat model of AMI.
Materials and Methods
Cell Culture and Sheets
The L6 rat skeletal myoblast cell line was obtained from the American Type Culture Collection (CRL-1458, Manassas, VA). Cells were cultured at 37°C with 5% CO2 in growth medium (DMEM supplemented with 10% FCS and antibiotics), passaged three times weekly, and kept at 60% confluency to retain their differentiation potential. Passages 5 to 15 were used for the experiments. Cell sheets were formed by plating 6 × 106 myoblasts on thermoreactive culture dishes (CellSeed, Tokyo, Japan) to achieve sheet thickness of approximately five cell layers. Cells were incubated for 16 h at 37°C to induce sheet formation. The sheets detached spontaneously at room temperature within 45 min, and were then washed once with growth medium.
Measurement of Proliferation, Apoptosis, and Differentiation
Myoblast viability after 48-h serum deprivation or staurosporine treatment was assessed by the mitochondria-dependent reduction of 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) to formazan (Roche, Mannheim, Germany) as described previously (33). Briefly, NADPH produced by the cells was visualized by adding MTT (10 μl, 5 mg/ml in PBS) to 100 μl of cell suspension. Following a 4-h incubation period at 37°C, the samples were dissolved in DMSO, and the amount of formazan dye generated was quantified (A540 nm/A650 nm). Cell adherence was determined 48 h after treatment. Nuclei were stained with DAPI, and cells were washed thoroughly with PBS. Cell nuclei were counted with CellC software version 1.11 (Seli-nummi and Seppälä, Tampere University of Technology, www.cs.tut.fi/sgn/csb/cellc). The early phase of apoptosis, characterized by phosphatidyl serine translocation, was determined 24 h after serum deprivation or staurosporine treatment by cell surface annexin V binding (Invitrogen, Carlsbad, CA). Fluorescence (340 nm/460 nm) was measured with Wallac Victor2 (Perkin-Elmer, Wellesley, MA). Myoblast sheet apoptosis was determined by measuring caspase-3 activity after the induction of apoptosis by either serum deprivation or staurosporine for 24 h. Caspase-3 activity was measured using EnzChek® caspase-3 assay kit (Invitrogen). For differentiation, sheets were deprived of serum to induce myoblast differentiation into myotubes. Western blotting samples were then collected for evaluation of troponin T and myogenin expression.
VEGF-A Measurements
VEGF-A protein secretion from myoblast sheets was determined from growth medium conditioned for 48 h using a rat VEGF DuoSet™ ELISA kit (R&D Systems, Minneapolis, MN) according to the manufacturer's instructions. VEGF-A expression was determined from staurosporine-treated or nutrient-deprived sheets.
Bcl-2 Overexpression and Transfection
pBabepuro-
Western Blotting
Western blotting samples were prepared in Laemmli sample buffer (Bio-Rad Laboratories, Redmond, WA). Detection was carried out as previously described (3) using appropriate alkaline phosphatase-conjugated secondary antibodies. The primary antibodies used were mouse monoclonal anti-Bcl-2 (610539, clone 7, BD Biosciences, San Jose, CA), mouse monoclonal anti-myo-genin (sc-12732, clone F5D, Santa Cruz Biotechnology Inc., Santa Cruz, CA), and mouse monoclonal anti-troponin T (T6277, clone JLT-12, Sigma-Aldrich).
Immunofluorescence Imaging
Cells were grown on coverslips for 48 h prior to fixation with 4% paraformaldehyde and permeabilization with 0.2% Triton X-100. Coverslips were incubated with anti-Bcl-2 antibody, washed with PBS, and incubated with an appropriate Alexa-Fluor fluorophore-conjugated (Invitrogen) secondary antibody. Nuclei were stained with DAPI, and coverslips were mounted on microscopy slides with Vectashield mounting medium (Vector Laboratories, Burlingame, CA). Samples were visualized with an Olympus IX70 microscope (Olympus Finland, Vantaa, Finland).
RNA Extraction and Microarray
Myoblast sheets were prepared as described. After a 40-h incubation period to enable three-dimensional culture-induced gene expression in vitro, total RNA was isolated using the Trizol reagent (Invitrogen) according to the manufacturer's instructions. Further purification was carried out with an RNeasy Mini Kit (Qiagen, Hilden, Germany). RNA quality was verified with an Agilent 2100 bioanalyzer (Agilent Technologies, Palo Alto, CA). Purified RNA was synthesized into double-stranded cDNA with a Superscript Double-stranded cDNA Synthesis Kit (Invitrogen). A Bioarray High Yield RNA Transcript Labeling Kit (Enzo LifeSciences, Faiming-dale, NY) was then used to synthesize cRNA. cDNA and cRNA products were purified with a GeneChip Sample Cleanup Module (Affymetrix, Santa Clara, CA). cRNA products were then used for fragmentation reaction and hybridized into the Affymetrix Rat Genome 230 2.0 GeneChip Array. After 18 h, chips were washed and stained with a GeneChip Fluidics Station 400 (Affymetrix), followed by chip scanning with a GeneChip Scanner 3000 7G (Affymetrix). Data analysis was performed with GeneSpring GX software, version 7.3.1 (Agilent Technologies), normalized to the median 50 percentile, and presented as gene expression fold changes.
Animals
This study used 92 male Wistar rats (250–400 g) of which 64 rats survived surgery and follow-up. The rats were divided into four groups: Group 1—control, left anterior descending coronary artery (LAD) ligation (
LAD Ligation and Cell Sheet Transplantation
AMI was induced by ligation of LAD as previously described (23). Briefly, animals were anesthetized subcutaneously using 0.05 mg/kg of medetomidine (Orion Pharma Inc., Turku, Finland) and 5 mg/kg IP ketamine (Parke-Davis, Barcelona, Spain). They were then intubated, and a respirator served to maintain ventilation during surgery. The heart was exteriorized through a left thoracotomy and pericardiotomy. LAD was ligated 3 mm from its origin. Immediately after LAD ligation, two circular myoblast sheets (total 1.2 × 107 cells), approximately 25 mm in diameter, were placed on the left ventricular anterior wall for each rat in groups 2 and 3. Group 2 received L6-WT sheets and group 3 received L6-Bcl2 sheets. After sheet transplantation, the heart was returned to its normal position and covered with pericardium to avoid adhesion to the lung and to the chest wall, and to prevent movement of the sheets. Throughout surgery, normal body temperature was maintained with a thermal plate. After surgery, anesthesia was antagonized with atipamezole hydrochloride (1.0 mg/kg, SC, Orion Pharma Inc.). Buprenorphine hydrocholoride (0.05 mg/kg, SC, Reckitt and Colman Ltd, Hull, UK) was administered for postoperative analgesia.
Echocardiography
All rats underwent echocardiography under anesthesia 3 (baseline), 10, and 28 days after surgery. The animals were sedated with 0.5 mg/kg medetomidine and placed on a thermal plate. Measurements were performed with a 7.5 MHz transducer (MyLab®25, Esaote SpA, Genoa, Italy). Anterior wall thickness, posterior wall thickness at diastolic (AWTd, PWTd) and systolic (AWTs, PWTs) phases, and left ventricular diameter at diastolic (Dd) and systolic (Ds) phases were measured in the short-axis right parasternal projection just below the mitral valves. Units are presented in millimeters. Dd and Ds were used to calculate fraction shortening (LVFS) and ejection fraction (LVEF) percentage: LVFS (%) = (Dd - Ds)/Dd, andLVEF (%) = (Dd3 - Ds3)/Dd3.
Histology and Immunostaining
At 28 days after surgery, the rats were euthanized after echocardiography. The heart was then excised and cut into four equal transverse parts. Two middle parts (next apex and next basal) were fixed in 4% neutral-buffered formalin for 48 h. The samples were then embedded in paraffin, and were cut into 4-μm-thick sections for histology and immunostaining. Sirius Red stain served to analyze fibrosis. Immunostaining was performed using a Ventana Discovery Automate (Ventana Medical Systems Inc, Tuczon, AZ). To demonstrate vascular density, endothelial cells were stained with antibody against von Willebrand Factor (vWF, RB-281, Labvision Inc., Fremont, CA). Cell proliferation was evaluated using anti-Ki67 antibody (RM-9106, Labvision Inc.), and apoptosis was detected with antibody specifically recognizing active cleaved caspase-3 (CST #9664, Cell Signaling Technology Inc., Danvers, MA). The stem cell antigen, c-kit, was detected with an antic-kit antibody (RA14132, Neuromics, Northfield, MN). Sections stained for Ki67 proliferation-associated antigen were double-stained for myocytes using anti-tropo-myosin antibody (MS-1256, Labvision Inc.).
Analysis of Fibrosis
Fibrosis was evaluated from scanned images of Sirius Red-stained sections. Percentage of fibrosis was calculated as Sirius Red-stained area divided by whole section area as evaluated using Photoshop 7.0 (Adobe Inc, San Jose, CA).
Analysis of Vascular Density, Cell Proliferation, Apoptosis, and c-kit-Positive Cells
The vWF- and Ki67-immunostained sections were photographed with a microscope (100x magnification) from six fields (two from infarcted area, two from border area, two from remote area). Vascular density and proliferating cell number were calculated with ImageJ (National Institutes of Health, Bethesda, MD, http://rsb.info.nih.gov/ij). Briefly, RGB images were background subtracted and divided into Hue-Saturation-Brightness channels from which the Brightness channel was further analyzed by thresholding for the staining intensities of vWF or Ki67. All images were processed with the same parameters using a preprogrammed macro. Data were collected separately for infarcted, border and remote areas. Apoptotic cells were manually counted as the number of positive cells for cleaved caspase-3 from six fields per slide. From c-kit-stained sections, the c-kit-positive cells were counted manually from the entire section.
Analysis of Sheet Survival In Vivo
A substudy was designed to specifically evaluate sheet survival on top of the infracted area. Six rats were used. L6-WT and L6-Bcl2 myoblasts were labeled with green fluorescent protein (GFP) by incubation with a lentiviral vector carrying the
Statistical Analysis
Data are presented as mean ± SEM. Differences between groups were compared using the Student
Results
Bcl-2 Overexpression and Function in Myoblasts and Myoblast Sheets
Bcl-2 expression was introduced to the L6 rat myoblast cell line with pBabepuro retroviral vector carrying the

Expressional and functional characterization of Bcl-2 in L6 myoblasts. (A) Expression of Bcl-2 protein in wild-type (L6-WT) and Bcl-2-overexpressing (L6-Bcl2) myoblasts. (B) Immunofluorescence (IF) detection of Bcl-2 in L6 myoblasts, suggesting cytoplasmic and perinuclear localization. (C) Mitochondrial activity as measured with MTT assay in L6-WT and L6-Bcl2 in myoblast cultures treated for 48 h. (D) Number of adherent L6-WT and L6-Bcl2 myoblasts in cultures treated for 48 h. (E) Quantification of early apoptosis as measured by FITC-labeled annexin V binding to cell surface-translocated phosphatidyl serine. (C–E) Cultures deprived of serum (left panels), and cultures with apoptosis induced by staurosporine (80 ng/ml) (right panels).
After 48-h serum deprivation or staurosporine treatment the number of adherent L6-Bcl2 cells was 2-fold and 1.6-fold higher than that of L6-WT cells, respectively (
Both cell types efficiently formed cell sheets, and the introduced overexpression of Bcl-2 was retained. Both types of sheets showed similar proliferative activity as evaluated by Ki67 expression. L6-Bcl2 sheets showed fewer cells positive for active cleaved caspase-3 (Fig. 2A).
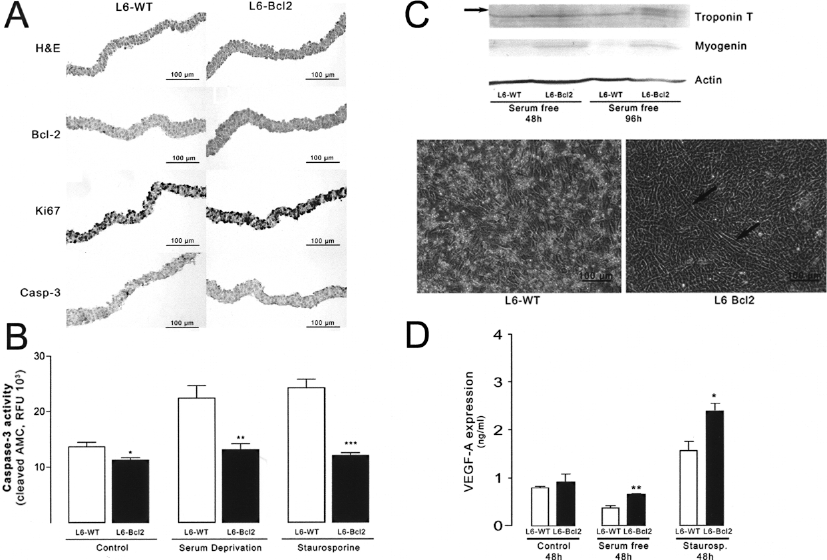
Immunohistochemistry, differentiation, and VEGF expression of wild type (L6-WT) and Bcl-2-overexpressing (L6-Bcl2) myoblast sheets. (A) Hematoxylin and eosin (H&E) stain, expression of Bcl-2 protein, cell proliferation as detected by the expression of proliferation-associated Ki67 antigen and the evaluation of apoptotic cells by immunodetection for active cleaved caspase-3. (B) Activation of caspase-3 in L6-WT and L6-Bcl2 myoblast sheets after 24 h in control, serum-deprived, and staurosporine-treated (10 ng/ml) sheets. (C) Immunoblots of myogenic differentiation markers troponin T and myogenin from L6-WT and L6-Bcl2 sheets after differentiation in serum-free medium for 96 h. Lower panels show phase contrast images demonstrating the formation of myotubes in L6-Bcl2 sheets. Cell detachment and death prevail in L6-WT sheets. (D) Amount of VEGF-A in culture medium of untreated, serum-deprived, and staurosporine-treated L6-WT and L6-Bcl2 sheets as determined with ELISA.
To study the functionality of Bcl-2, we measured the activity of caspase-3 in unstimulated, serum-deprived, or staurosporine-treated myoblast sheets after 24 h. We found that the introduction of Bcl-2 to myoblast sheets prevented the activation of effector caspase-3, whereas prolonged culture (
Because culture in reduced serum medium is standard practice for inducing differentiation of myoblasts (21), we used this method to investigate the differentiation of myoblast sheets. L6-Bcl2 myoblasts differentiated into myotubes as evaluated by the expression of differentiation markers troponin T and myogenin, whereas L6-WT myoblasts underwent cell death instead of differentiation under serum starvation. After prolonged culture, L6-WT sheets showed signs of disruption whereas cell survival in L6-Bcl2 sheets was predominant, and myotubes were visible (Fig. 2C).
Bcl-2 Induces Expression of Proangiogenic Mediators in Myoblast Sheets
Because Bcl-2 expression has been shown to affect several cellular processes (5), we used whole-genome microarrays to investigate the effect of
Genes Upregulated in Bcl-2-Overexpressing L6 Myoblast Sheets Compared to Wild-Type Cell Sheets

Bcl-2-Expressing Myoblast Sheets Enhance Ventricular Function After AMI
To study the increased therapeutic efficacy with L6-Bcl2 versus L6-WT sheet transplantation, we used the rat LAD ligation model. Table 2 shows cardiac performance data assessed with echocardiography at 3, 10, and 28 days after ligation and sheet transplantation.
Echocardiography Data at 3, 10, and 28 Days After LAD Ligation and Transplantation

Values represent mean ± SEM of anterior and posterior wall thickness in diastolic (AWTd, PWTd) and systolic phases (AWTs, PWTs) and left ventricular diameter in diastolic (Dd) and systolic (Ds) phases. Units are presented in mm. Dd and Ds were used to calculate fraction shortening (LVFS) and ejection fraction (LVEF) percentage.
The function of the left ventricle, as measured by LVEF and LVFS, improved only in the L6-Bcl2 sheet-treated group. These parameters were significantly enhanced at day 10, and remained elevated until the end of the study at day 28. At day 10, the LVEF of both the L6-WT and L6-Bcl2 groups recovered from baseline, whereas the LVEF of the control group decreased (L6-WT: 30.3 ± 2.3% to 32.2 ± 2.1%, L6-Bcl2 31.4 ± 2.3% to 37.80 ± 1.9%, control: 32.6 ± 1.7% to 29.1 ± 2.0%). Even at day 28, LVEF remained significantly higher in the L6-Bcl2 group than in the L6-WT and control groups (L6 Bcl2: 33.2 ± 2.0%, vs. control: 27.3 ± 2.2%,
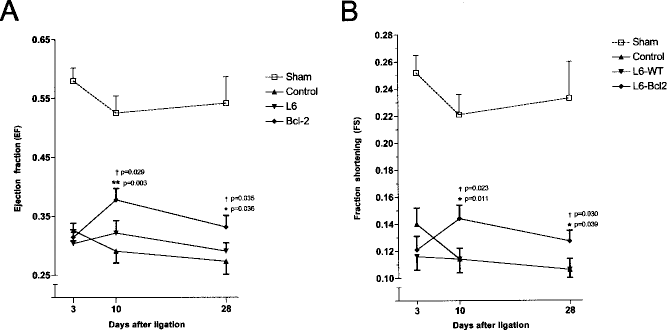
Echocardiography for (A) left ventricle ejection fraction (EF) and (B) left ventricle fraction shortening at the indicated time points after wild-type (
Myoblast Sheets Expressing Bcl-2 Exert Therapeutic Effects on Injured Myocardium
Because Bcl-2 expression improved the myoblast sheet production of angiogenic growth factors, such as VEGF and PlGF, we evaluated the end-point vessel density after sheet transplantation. Higher vessel density was evident with vWF immunostaining at the infarct area on which the sheets were placed (Fig. 4). This area thus received maximal concentrations of growth factors and paracrine stimulation deriving from the sheets. Paracrine effects of myoblast sheet transplantation were also observed in the border area, and to a lesser extent, in the remote area, suggesting an enhanced effect of L6-Bcl2 sheets. The higher vascular density was associated with decreased fibrosis. The L6-Bcl2 group showed a significantly lower percentage of fibrosis than did the control and L6-WT groups (L6-Bcl2: 21.2 ± 0.8%, vs. control: 25.6 ± 1.8%,

(A) Quantitative evaluation of vascular density using immunohistochemical staining for von Willebrand factor (vWF) expression in myocardial paraffin-embedded sections from sham-operated, wild-type (L6-WT), Bcl-2 (L6-Bcl2), and control animals. *

(A) Quantitative evaluation of fibrosis by Sirius Red staining of myocardial paraffin-embedded sections from sham-operated, wild-type (L6-WT), Bcl-2 (L6-Bcl2), and control animals. *
Cell death in AMI has been shown to occupy various forms ranging from overt necrosis to programmed apoptosis (27). To understand how myoblast sheet transplantation influences apoptosis and proliferation, we evaluated the expression of active caspase-3 as well as that of the proliferation-associated Ki67 antigen. No differences between groups in caspase-3-positive cells were found at the end of the study period (data not shown). In contrast, the number of proliferating cells within the myocardium was increased in groups receiving myoblast sheet transplantation (Fig. 6). In the L6-Bcl2 group, a significant number of proliferating cells was also found in the remote area (L6-Bcl2: 47.5 ± 3.7 vs. control: 30.9 ± 3.5,

(A) Quantitative evaluation of proliferative cells as assessed with the expression of proliferation associated Ki67 nuclear antigen (dark brown). Figure shows mean ± SEM densitometry values from the immunohistochemistry of myocardial paraffin-embedded sections of sham-operated, wild-type (L6-WT), Bcl-2 (L6-Bcl2), and control animals. *

(A) Quantitative evaluation of the number of c-kit-expressing cells in the myocardium. Paraffin-embedded sections were stained from sham-operated, wild-type (L6-WT), Bcl-2 (L6-Bcl2), and control animals. *
Bcl-2 Enhances Myoblast Sheets Survival In Vivo
To demonstrate the enhanced survival of sheets by Bcl-2 expression in vivo, we generated GFP-expressing L6-WT or L6-Bcl2 myoblast sheets. These sheets were then transplanted onto the infarcted hearts as in previous experiments. We found that after 3 weeks, animals receiving L6-Bcl2 (
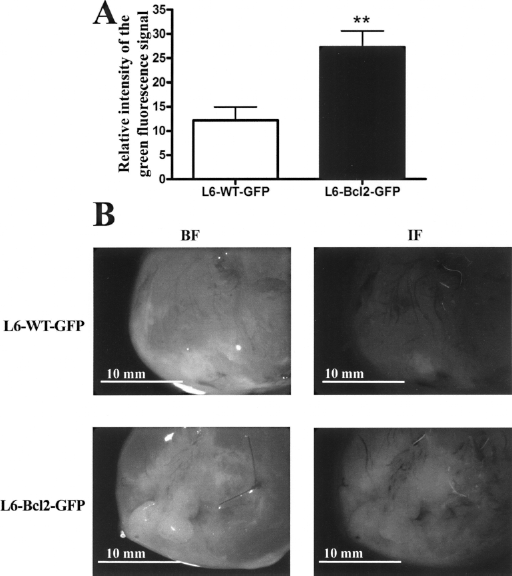
Quantitative evaluation of cell survival after myoblast cell sheet transplantation. (A) Analysis of green fluorescence intensity from the apical surface of hearts 3 weeks after transplantation of GFP-expressing wild-type (L6-WT-GFP,
Discussion
In AMI, the occlusion of a coronary artery blocks the oxygen and nutrient supply to the myocardium it supplied, and causes severe stress as well as cell death (16). Under these conditions, cell death occurs via several mechanisms ranging from programmed apoptosis to overt necrosis (27). The dying cells release mediators that not only alert the immune system, but also promote cell death and exacerbate the damage (32). Moreover, reperfusion may further aggravate the rate of cell death (28). Prior studies have shown that Bcl-2 expression can protect cells transplanted for therapy by injection under these conditions (19). In this study, we investigated the ability of antiapoptotic Bcl-2 in myoblasts to functionally enhance cell sheet transplantation in a rat model of AMI. We show here that Bcl-2 expression can enhance the novel technique of myoblast sheet transplantation. Sheets expressing Bcl-2 had increased tolerance for apoptotic stimuli, and survived longer when transplanted on top of infarcted myocardium. Moreover, we show that introducing Bcl-2 expression in myoblast sheets lead to their enhanced production of proangiogenic mediators, specifically VEGF and PlGF. Therapy with Bcl-2-expressing myoblast sheets significantly improved cardiac function, reduced fibrosis, enhanced myocardial neoangiogenesis and cell proliferation, and increased the amount of the stem cell antigen, c-kit, positive cells in the myocardium.
Bcl-2 localizes intracellularly to mitochondrial, nuclear, and endoplasmic reticulum membranes (1). It protects cells from death stimuli, such as nutrient deprivation (36), by stabilizing the mitochondrial membrane potential (30), reducing caspase activation, or inhibiting cytochrome c release (9). While tissue regeneration therapies and cell transplantation therapy aim to replace dead cells with progenitors as well as introducing growth-promoting signals to the tissue environment (19), it is frequently forgotten that transplanted cells face the same severe surroundings. Although studies have demonstrated the superiority of cell sheet therapy to intramyocardial injections in heart failure (20), transplantation of several sheets is required to counteract the struggle for survival, nutrients, and oxygen.
Mechanisms of cell death operating under ischemia and infarction depend on the location of cells in relation to the area supplied by the stenosed or occluded artery. Overt necrosis is predominant in the core areas, with programmed death by apoptosis gaining more ground towards the border areas with some collateral blood flow. The restricted oxygen and nutrient supply, along with cell debris released by necrotic cells, elicits cell death responses that operate through common mechanisms, such as the generation of reactive oxygen species (16) and target mitochondrial function (26). In this study, we mimicked these in vivo conditions in cell culture by removing serum from the culture medium, thus depriving cells of nutrients and growth factors. Moreover, to reproduce the proapoptotic environment of the infarcted area, we treated cells with staurosporine, a widely used inducer of apoptosis that elicits similar intracellular apoptotic cascades that spur cell death in infarcted myocardium (10). Bcl-2 overexpression in cells and cell sheets provided protection and resistance against both nutrient deprivation and staurosporine-induced apoptosis. Importantly, when stressed with nutrient deprivation myoblast sheets expressing Bcl-2 differentiated into myotubes. This suggests that expression of antiapoptotic Bcl-2 does not prevent the myoblasts to exit the cell cycle, fuse, and differentiate.
These results show the benefit of
The finding that Bcl-2 expression only influences gene expression in cell sheets suggests that Bcl-2 does not alter cell behavior under normal growth conditions, Bcl-2 activity specifically enhances apoptosis tolerance, and that formation of cell sheets may induce apoptotic stress to the cells. These results show that when applying cell sheet therapy, prevention of apoptotic death already at the initial steps of sheet formation is necessary for maximizing therapeutic efficacy. In the Bcl-2-expressing myoblast sheets we observed enhanced production of the VEGF family paracrine effectors, VEGF-A and PlGF. These growth factors are known to synergize in proangiogenic signaling (24), and further add to the beneficial profile induced by introduction of Bcl-2. In a previous study, Memon and colleagues reported that the myocardium under the myoblast sheets produces growth factors, such as VEGF (20). In this study, we show that wild-type myoblast sheets themselves exhibit enhanced production of proangiogenic factors compared to standard cultures of myoblasts, and that expression of Bcl-2 in sheets greatly enhances the production of these paracrine factors.
To further test the Bcl-2-mediated functional therapeutic benefit in vivo in AMI, wild-type, and Bcl-2-expressing sheets were transplanted onto the ischemic myocardium after LAD ligation. In the group undergoing Bcl-2 sheet transplantation, clearly enhanced left ventricular function was already evident at 10 days and was sustained until the end of the study period, 28 days after transplantation. LVEF paralleled LVFS as an alternate measure for left ventricular performance. Although some studies have reported enhanced cardiac function by unmodified myoblast sheets in experimental models of MI (11, 20), treatment with two layers of wild-type sheets in this study did not significantly increase LVEF. Here, however, transplantation of wild-type sheets also counteracted the LVEF decline after MI. This may be due to differences in rat strain (20), host species of donor myoblasts (11), the low number of cell sheets used, or the acute infarction model employed in the current study. Furthermore, due to generation and passage of the L6 cell line, differences to primary cells, as used in some studies, may emerge (35). Sheet transplantation increased vascular density in infarcted and border areas as evaluated by vWF immunostaining. Only the Bcl-2-modified sheet therapy increased both the number of proliferating cells in the remote area and the number of cells positive for stem cell antigen c-kit in the myocardium. In view of a recent report by Cimini et al. (8), these results suggest that more stem cells are stimulated to infiltrate the myocardium by Bcl-2-modified myoblast sheet therapy. Induction of stem cell infiltration can subsequently activate the endogenous repair processes of the myocardium (8). The observed effects on angiogenesis, proliferation, and c-kit expression were associated with an antifibrotic effect of the sheets at 28 days. Furthermore, overexpression of Bcl-2 resulted in prolonged survival of myoblast sheets in vivo, providing sustained secretion of angiogenic factors. The importance of such paracrine activators from myoblasts was demonstrated by Perez-Ilzarbe and colleagues on endothelial, smooth muscle, and cardiomyocyte cells (25). Activation of all these cell types is required for efficient repair of the damaged myocardium.
Taken together these results suggest that Bcl-2 over-expression in the myoblast sheets enhances their therapeutic potential by improving and sustaining the paracrine effects of the sheets in AMI. To our knowledge, this study presents the first combination therapy approach to use gene therapy-engineered cell sheets that can withstand apoptosis induction by means of Bcl-2 expression. These results provide the first insight on how antiapoptotic and mitochondrioprotective strategies not only functionally but also by specific modification of gene expression profile can enhance the novel approach of cell sheet transplantation therapy in treatment of heart failure. Because myoblast sheet transplantation has shown no adverse effects in either preclinical or clinical settings (Y. Sawa, personal communication), it seems feasible to adopt this cell transplantation methodology for gene modification and as vehicle in gene therapy as well. The transplantation of sheets can easily be carried out in conjunction with coronary artery bypass surgery, and it produces minimal damage to the heart muscle compared to cell injections. The cells can be transduced with an optimized minimal amount of viral vector, and no viral vectors are injected to the patient. However, clinical trials are warranted to unambiguously demonstrate the efficacy of cell sheet transplantation.
Footnotes
Acknowledgments
