Abstract
Bone marrow cells are used for cell therapy after myocardial infarction (MI) with promising results. However, cardiac persistence of transplanted cells is rather low. Here, we investigated strategies to increase the survival and cardiac persistence of mononuclear (MNC) and mesenchymal (MSC) bone marrow cells transplanted into infarcted rat hearts. MNC and MSC (male Fischer 344 rats) were treated with different doses of PDGF-BB prior to intramyocardial injection into border zone of MI (syngeneic females, permanent LAD ligation) and hearts were harvested after 5 days and 3 weeks. In additional experiments, untreated MNC and MSC were injected immediately after permanent or temporary LAD ligation and hearts were harvested after 48 h, 5 days, 3 weeks, and 6 weeks. DNA of the hearts was isolated and the number of donor cells was determined by quantitative real-time PCR with Y chromosome-specific primers. There was a remarkable though not statistically significant (
Introduction
Cardiac cell therapy with bone marrow cells after myocardial infarction was demonstrated to improve cardiac function in several large randomized clinical trials (12,17), but, on the other hand, there was no benefit observed in other studies (4,5,10). Although it is still unknown how bone marrow cells take action on cardiac regeneration, the number of transplanted cells within the injured myocardium is likely to be an important factor. In previous studies, we showed that after a good engraftment efficacy the number of mononuclear and mesenchymal bone marrow cells within the infarcted rat heart decreases rapidly to only a few percent of the originally transplanted cell number at 6 weeks after intramyocaridal cell injection (7). The aim of this study was to increase the midterm survival by either cell treatment with the growth factor and inhibitor of apoptosis PDGF-BB prior to transplantation or reperfusion of the infarcted vessel (improved nutrient and oxygen supply of the cells).
Materials and Methods
Protocols were approved by the regional government's Animal Care and Use Committee (Bezirksregierung Köln, No. 50.203.2-K47, 34/03, and 50.203.2-K47, 2/03), and conformed with the Guide for the Care and Use of Laboratory Animals of the NIH.
Bone Marrow Cell Isolation From Male Rats
After dissecting femurs, humeral and tibial bones (male Fischer 344 rats, 2 months old, Charles River, Sulzfeld, Germany) from surrounding muscle and connective tissue, the bone marrow was eluted with a 19-gauge needle and DMEM, high glucose, 10% FCS, 1% penicillin/streptomycin. The cell suspension was filtered through 70-μm filters, centrifuged, and resuspended in PBS. For recovery of mesenchymal bone marrow cells (MSC), 50 × 106 bone marrow cells were plated on 10-cm dishes and kept in culture for 10 days. At day 10 MSC were detached with Trypsin/EDTA, washed, and resuspended (DMEM, no FCS) at a concentration of 100,000 cells/50 μl (for experiments on PDGF treatment) or 1,000,000 cells/50 μl (for experiments comparing permanent and reperfused myocardial infarction). For isolation of mononuclear cells, the crude bone marrow cell suspension was subjected to a Ficoll gradient centrifugation (NycoPrep, Progen GmbH, Heidelberg, Germany). The interphase cells were collected, washed, and kept in suspension (DMEM, no FCS) until use (12 h at 4°C).
Treatment with PDGF-AA and PDGF-BB In Vitro
For in vitro studies, MSC of five animals were isolated as described above and the cells of each animal were portioned out and kept in culture under six different conditions. A Cell Death Detection ELISA (Roche Diagnostics, Basel, Switzerland) was used to measure cell death by the detection of cytoplasmic histone-associated DNA fragments (mono- and oligonucleosomes).
Cells (5,000 cells/well) were plated into 96-well plates in DMEM containing 10% FCS followed by serum deprivation (conditions 1–6). Stimulation with PDGF-AA (50 ng/ml, condition 2; +AA) or PDGF-BB (50 ng/ml, condition 3; +BB) was compared to unstimulated starving for 24 h (condition 1; control). Apoptosis was intensified by adding H2O2 (100 μM) after 24 h of unstimulated starving and incubation for another 15 h (conditions 4–6; +H2O2) in the absence (condition 4; +H2O2) or presence of PDGF-AA (50 ng/ml, condition 5; +H2O2+AA) or PDGF-BB (50 ng/ml, condition 6; +H2O2+BB). The cell lysates were collected, and apoptosis was measured using the Cell Death Detection ELISA with each condition's optical density data being normalized to its respective unstimulated starving control experiment.
Myocardial Infarction in Female Rats and Cell Injection
Myocardial infarction was induced in female syngeneic adult rats (2–3 months old) by ligation of the left coronary artery under ketamine and xylazine anesthesia as described previously (9). LAD ligation was either permanent or temporary (i.e., for 30 min). Animals of the reperfusion group received an IP injection of 10 IU heparin prior to surgery in order to prevent thrombosis during LAD occlusion. Infarction was evidenced by paling of left ventricular wall, which disappeared with reperfusion. Immediately after infarction or infarction/reperfusion 50 μl of cell suspension containing either 100,000 (for experiments on PDGF treatment) or 1,000,000 cells (for experiments comparing permanent and reperfused myocardial infarction) were injected into the border zone of the myocardial infarct with three separate injections using a Hamilton syringe (H. Faust GmbH, Rheinbach, Germany) attached to a 29-gauge needle. The higher cell number in the reperfusion experiments was chosen because of the unexpectedly high cell loss we observed already in preliminary experiments. After cell therapy, rats were allowed to survive for 48 h, 5 days, 3 or 6 weeks.
Treatment with PDGF-BB In Vivo
Prior to surgery PDGF-BB was added to the suspension of MNC or MSC resulting in a concentration of 10 or 50 ng/ml. After 10 min of incubation at room temperature the whole suspension was injected as described above.
DNA Preparation and Quantitative Analysis of Cell Survival by Real-Time PCR
Genomic DNA preparation and quantitative real-time PCR analysis of cell survival in the heart was performed as described previously with primers against the Sry genes (male genomes) and osteopontin as internal standard (to normalize for the amount of DNA) (9). Samples with known dilutions of male with female DNA were included to verify consistency and to derive a calibration curve for the calculation of male/total DNA ratio. During this technique's establishment, Y chromosome signal from dead male cells was investigated as a time course and showed a loss of signal within several hours (9). Additionally, to ensure that Y chromosome signal does arise from intact cells, we used gold and dye labeling techniques to track the injected cells in a related study without reperfusion and we found good correlation between the time course of cell loss resulting from our PCR analysis and that resulting from semiquantitative histological analysis (data not shown). For the calculation of absolute cell number in the heart, the male DNA/total DNA ratio (as obtained from PCR) was multiplied by the total number of nuclei in the hearts analyzed. The latter number was calculated from the heart wet weight and the number of 217,400 cells in rat heart per milligram wet weight as observed by van der Laarse et al. (14). This mode of analysis was previously validated in several models (7–9).
Statistical Analysis
All data are presented as mean ± SEM. Statistical analysis was performed with the help of computerized calculations with InStat® (GraphPad Software Inc., USA) and SPSS® (SPSS Inc., USA) using Student's
Results
Treatment with PDGF-AA and PDGF-BB In Vitro
With H2O2 added to standard cell culture conditions apoptosis of cultured MSC was 2.6 ± 0.2-fold higher compared to control (
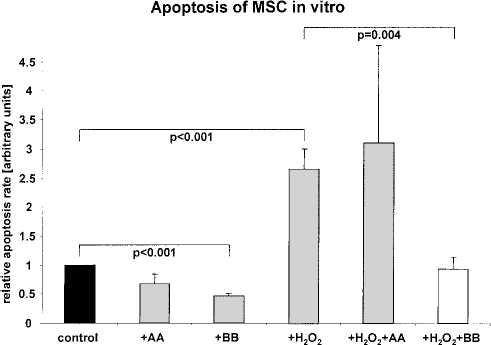
Apoptosis of MSC in vitro (each
Treatment with PDGF-BB In Vivo
As the changes in cell survival after treatment with PDGF-BB prior to cell injection were similar in MNC and MSC, the data of both groups were pooled for further analysis. Treatment with PDGF-BB did not change the number of detectable male cells 5 days after LAD ligation and cell transplantation, neither with the lower dose of 10 ng/ml (121 ± 44% with PDGF-BB vs. 100 ± 16% without,
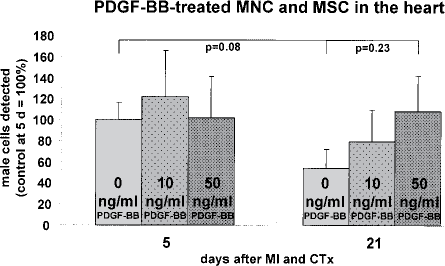
PDGF-BB-treated MNC and MSC in the heart (each
Reperfusion
For both MNC and MSC, there were lower numbers of transplanted cells detected in reperfused than in permanent myocardial infarction at all time points studied. For MNC, after reperfusion only 1.1 ± 0.5% of injected cells were detected in the heart after 48 h and 1.1 ± 0.2% after 5 days, which was significantly lower than in permanent MI with 5.6 ± 0.6% after 48 h (
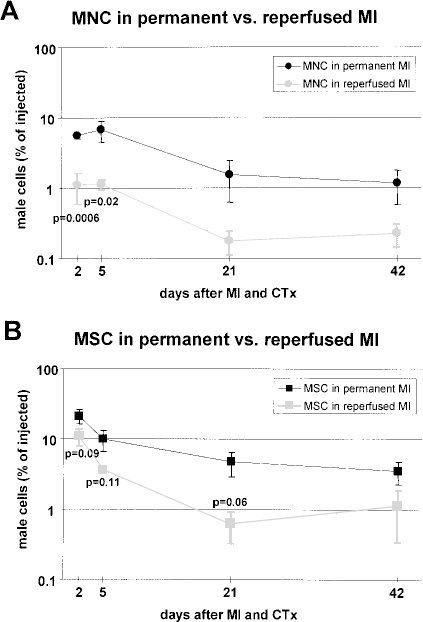
Mononuclear (MNC) (A) and mesenchymal (MSC) (B) in permanent versus reperfused MI (each
Mononuclear (MNC) and Mesenchymal (MSC) in Permanent Versus Reperfused MI (Each
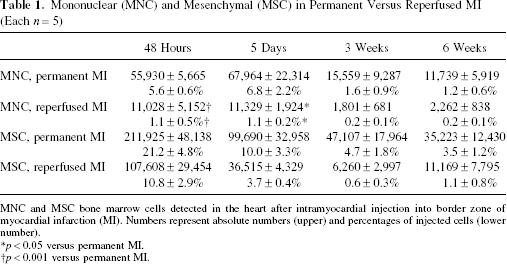
MNC and MSC bone marrow cells detected in the heart after intramyocardial injection into border zone of myocardial infarction (MI). Numbers represent absolute numbers (upper) and percentages of injected cells (lower number).
Discussion
The number of cells successfully implanted into the injured heart and their persistence within the heart might be major limiting factors for cardiac cell therapy. For example a dose dependency was shown for the decrease of infarct size in a rat model after transplantation of human umbilical cord blood mononuclear cells, which contain hematopoietic and mesenchymal stem cells (2). In our previous studies, we demonstrated that midterm survival of MNC and MSC bone marrow cells injected into the periinfarct region in rats is very low (7). Here, we evaluated two strategies to increase cardiac cell persistence (and/or survival) of MNC and MSC: 1) cell treatment with antiapoptotic PDGF-BB prior to transplantation and 2) reperfusion of the infarcted artery.
PDGF-BB was used as an antiapoptotic agent. In multiple previous studies it has been proven to protect several cell types from death (1,11,15,16). In our in vitro experiments, PDGF-AA and PDGF-BB were investigated because both are known to play different roles in healing myocardial infarction (19) and might therefore show differences in antiapoptotic effects as well. Our results showed that PDGF-BB, but not PDGF-AA, was able to prevent apoptosis in vitro, suggesting that the selective activation of PDGF-receptor-α by PDGF-AA does not, but unselective activation of both PDGF-receptor-á and PDGF-receptor-β by PDGF-BB does lead to antiapoptotic effects. Therefore, these effects might be mediated by the PDGF-receptor-β alone.
In vivo, no statistically significant change in the number of surviving MNC and MSC was observed, even though a remarkable cell loss of ~46% in the untreated control group between day 5 and day 21 was completely inhibited by treatment with high-dose PDGF-BB. Thus, cell loss at later times, but not early cell loss, may be prevented by the antiapoptotic effect of high-dose PDGF-BB on transplanted cells. Therefore, cell death rather than cell wash-out might be the predominant mechanism of cell loss at this time. Improved survival might result in improved functional benefit after treatment, as suggested by previous data from Mangi et al. (6). Overexpression of antiapoptotic Akt was also effective to reduce death of neonatal cardiomyocytes injected into infarcted hearts (18), while pretreatment with the caspase inhibitor AcYVADcmk was not (9). Hsieh et al. established a model of PDGF-BB-releasing nanofibers whose injection lead to decreased cardiomyocyte death, improved cardiac function, and decreased infarct size after myocardial infarction in rats (3). Maybe combining this constant release of PDGF-BB with cell application might multiply beneficial effects of both therapies. Nevertheless, it has to be noted that, although cell persistence was almost doubled by treatment with PDGF-BB in the present study, cell survival at 21 days is still low (5–7% of the initially injected cells).
Our second approach for improvement of cell persistence was to establish reperfusion of the infarcted artery, assuming that improved oxygen and nutrient supply to the cells would improve cell survival. However, somewhat surprisingly, reperfusion of the infarct resulted in decreased survival or persistence of transplanted MNC and MSC within the injured heart at all time points studied. A significantly lower number of MNC in reperfused hearts was detected at the earliest time points studied, indicating that cell loss due to reperfusion is an immediate phenomenon. Obviously, this finding suggests that wash-out of the transplanted cells plays an important role at early times. In contrast, after day 2 the cell survival curves in reperfused hearts run parallel to those in nonreperfused hearts, indicating that those cells that initially persist in the reperfused heart share the same fate with those that initially persist in the nonreperfused hearts. In previous studies using neonatal rat cardiomyocytes, we found that cell persistence was much lower when cells were injected into healthy myocardium (9) than when cells were injected into chronic infarcts (8), and we could show that wash-out is crucial even after intracoronary infusion in stop–flow technique as used in clinical trials (13). Therefore, healthy and/or reperfused tissue with reduced tissue damage and intracoronary application may be disadvantegous for the persistence of grafted cells.
From our data on the influence of pretreatment with PDGF-BB and reperfusion on cardiac persistence of injected bone marrow cells, it can be concluded that early after cell injection (up to 5 days) wash-out is the predominant mechanism of cell loss (promoted by reperfusion) while later after cell injection cell death is more important (prevented by treatment with PDGF-BB). This conclusion, albeit not very surprising, is supported by quantitative analyses of cell survival for the first time.
In summary, this study demonstrates that effective strategies to improve cardiac persistence of MNC and MSC are difficult to identify. The only successful approaches thus far were the increase in injected cell number (which increased absolute, but not relative, cell survival) (7) and, in trend, the pretreatment of cells with PDGF-BB. The other strategies, which we have studied so far, have failed to enhance cardiac cell persistence, which are injection of cells late (7 days) after infarction (7) and injection of cells in infarcted/reperfused myocardium. In conclusion, further investigations are needed to substantially improve the persistence and survival of grafted bone marrow cells in infarcted rat hearts, in order to fully explore the therapeutic potential of this novel treatment modality for myocardial repair.
Footnotes
Acknowledgments
