Abstract
We previously reported that intracoronary implantation of bone marrow mononuclear cells (BMMC) into ischemic hearts improved cardiac function after myocardial infarction. However, the mechanisms have not been elucidated. The present study investigates whether apelin, a newly described inotropic peptide with important cardiovascular regulatory properties, contributes to the functional improvement in patients with severe heart failure after cell transplantation. Forty consecutive patients with severe heart failure secondary to myocardial infarction were assigned to the BMMC therapy group or the standard medication group according to each patient's decision on a signed consent document. In 20 patients intracoronary cell infusion was performed, and another 20 patients were matched to receive standard medication as therapeutic controls. An additional 20 healthy subjects were designated as normal controls. Clinical manifestations, echocardiograms, and biochemical assays were recorded. Plasma apelin and brain natriuretic protein (BNP) levels were determined by enzyme immunoassay. Baseline levels of plasma apelin were significantly lower in all heart failure patients compared to normal subjects. In patients who underwent cell transplantation, apelin increased significantly from 3 to 21 days after operation, followed by significant improvement in cardiac function. In parallel, BNP varied inversely with the increase of apelin. In patients receiving standard medical treatment, apelin remained at a lower level. Our findings indicated that increased apelin levels following cell therapy may act as a paracrine mediator produced from BMMCs and play an important role in the treatment of heart failure through autocrine and paracrine mechanisms.
Introduction
Despite major advances that have been achieved, ischemic heart failure is still a leading cause of death throughout the world. Progress of cardiomyocyte death and loss of myocardial cell mass inevitably results to advanced ischemic heart failure (24). This deficiency is compounded by the heart's own limited capacity for self-regeneration. Consequently, the development of new therapeutic approaches on the basis of cardiac cell regeneration initiates great excitement in the fields of cardiovascular investigation (19).
Transplantation of uncommitted adult stem cells has shown a new perspective for tissue repair (10,15,18). Recently, several randomized control trials of bone marrow mononuclear cell (BMMC) therapy for myocardial infarction showed the feasibility, safety, and potential efficacy in human studies (2,11,16). However, the mechanisms are controversial (9,17,29). Some authors suggested the mechanisms may relate to the differentiation of infused stem cells directly into functional reparative cells, which restores the tissue damage (10,15,18). However, other studies contradicted that differentiation of BMSC into reparative cell types is generally too inefficient to effectively replace damaged tissue and restore tissue function (9,17,29). We have previously reported that intracoronary infusion of bone marrow-derived mononuclear cells in patients with refractory chronic heart failure secondary to ischemic cardiomyopathy significantly increased urine output and improved heart function rapidly after BMMCs transplantation. This early remarkable effect cannot be readily attributed to myocardial regeneration from donor cells; instead, we hypothesize that paracrine actions exerted by the cells through the release of soluble factors is the direct mechanism involved in tissue repair and functional improvement (8). Other studies have supported hypothesis that BMSCs secrete a wide array of cytokines that are cardioprotective and promote vasculogenesis (4,23,27). However, it is still not clear which molecules BMSC release and by what mechanism they exert their beneficial effects. Therefore, understanding the mechanisms by which adult stem cells are capable of improving cardiac function in ischemic heart disease is of fundamental biological and clinical importance.
Apelin, a ligand for apelin-angiotension receptor-like (APJ), has recently been shown to be a potent positive inotropic agent in normal and failing heart (26). The apelin pathway has only recently emerged as an important regulator of myocardial cell specification and heart development (28). In the pathophysiology of heart failure, it has been reported that plasma apelin concentrations decrease as severe heart function impairment progresses in patients with ischemic heart failure (IHF) (7). Therefore, we hypothesized that BMMC transplantation may play a critical role in modulating cardiac function by releasing biologically active inotropic factor apelin. In this study, our objective was to assess how apelin plasma levels were changed and the relationship between the apelin plasma concentration and heart function after BMMC transplantation in patients with severe IHF.
Materials and Methods
Patients
Forty consecutive patients with heart failure secondary to myocardial infarction were selected to participate in the study based on the following criteria: a) a history of myocardial infarction; b) severe clinical symptoms of heart failure and/ or recurrent hospitalization; c) no option for standard revascularization as determined by a surgical committee and an interventional committee at the time of enrollment; d) left ventricular ejection fraction <40% as determined by two-dimensional echocardiography; e) NYHA class >3.0. Exclusion criteria included: a) hemorrhagic symptoms; b) severe renal and liver dysfunction (serum creatinine or alanine amino-transferase increased to two times above the normal range); and c) neoplasia or other comorbidity. The protocols were approved by the ethics committee of the Navy General Hospital (Beijing, China). The investigation conforms with the principles outlined in the Declaration of Helsinki. Patients were informed, in writing, regarding the advantages and disadvantages of the BMMC transplantation procedure. Forty patients with ischemia heart failure were selected to participate in the study based on the above criteria. Twenty patients were assigned to the BMMC therapy group, and the other 20 patients were assigned to the standard medication group according to each patient's decision on a signed consent document. In addition, 20 healthy age- and sex-matched subjects were also selected to serve as controls. These patients are a different cohort from those previously reported (8).
BMMC Preparation
One day before transplantation, 40 ml of bone marrow was aspirated from each patient's ilium at four different points as previously described (9). BMMCs were isolated by consecutive Ficoll density gradient separation in lymphocyte separation medium (Biowhittaker) and then placed into Teflon bags (Vuelife Cell Genix, Maryland) for overnight cultivation. On the next day, cells were harvested, washed with heparinized saline three times, and adjusted to a final concentration of 4 × 106 cells/2 ml, in which viable cells averaged 94 ± 2% and CD34-positive cells averaged 1.3 ± 0.1%.
Methods of Intracoronary Cell Transplantation
In the BMMC therapy group, patients (15 men and 5 women, with mean age of 63.6 ± 2 years) underwent BMMC transplantation by intracoronary infusion. A balloon catheter was inserted routinely into a main branch of the coronary artery supplying the infarcted area or a contralateral donor vessel supplying the target area. Balloon inflation was not performed if there was severe dyspnea. Two milliliters of BMMC suspension (4 × 106 cells) was infused each time, and repeated six to eight times. The total amount of infused BMMCs averaged 2.8–3.2 × 107 cells. In each case, the entire operative procedure was completed between 10 and 30 minutes. In the standard therapy group, patients (12 men and 8 women, with mean age of 62.6 ± 1.6 years) were administered the same medication as the BMMC group, except they did not undergo cell transplantation.
Two-Dimensional Echocardiograms
All patients underwent echocardiography consecutively before, and at 7 days and 21 days after cell infusion or standard therapy, respectively. Left ventricular (LV) end-systolic volume, end-diastolic volume, and LV ejection fraction (EF) were measured using standard methods. Data were analyzed independently by two experienced observers who were unaware of patient treatment assignments.
Blood Sample Collection
Blood samples were collected before and at 3, 7, and 21 days after therapy. Blood samples were obtained in tubes containing EDTA and centrifuged. Plasma was extracted, aliquoted, and stored at −80°C until analysis.
Enzyme Immunoassay Measurement of Plasma Apelin and Brain Natriuretic Peptide Levels
Total plasma myocardial apelin concentration in patients with ischemic heart failure was quantified in duplicate utilizing the commercially available Apelin-36 EIA Kit (Phoenix Pharmaceuticals, Belmont, CA, USA). This kit detects apelin-36 as well as smaller biologically active isoforms (i.e. apelin-19, -16, -15, -13, and -12). Both the standard peptide solutions and myocardial protein samples were loaded onto antibody-treated 96-well plates containing primary antiserum and biotinylated peptide, and allowed to incubate for 2 h at room temperature. Following the initial reaction, streptavadin-horseradish peroxidase solution was added to the samples and allowed to react for 1 h at room temperature. The samples were thoroughly rinsed, and substrate was added to each sample well, yielding a color reaction. Absorbance was recorded at 450 nm. A standard logarithmic curve was plotted and used to calculate protein concentration in the myocardial samples using apelin-36 as the standard. Plasma brain natriuretic peptide (BNP) levels were assayed with enzyme-linked immunosorbent assays (East-Asia, Inc., Beijing, the People's Republic of China).
Statistical Analysis
Data are presented as mean ± SEM. Comparison of data was performed using one-way ANOVA for repeated measures with Bonferroni's correction or chisquare test for categorical variables, as appropriate. For all tests a value of p < 0.05 was considered significant.
Results
Patient Clinical Baseline Characteristics
Baseline Characteristics of Patients Receiving BMMCs Infusion and Standard Treatment

MI, myocardial infarction; PCI, percutaneous coronary intervention; CBG, coronary bypass grafting; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; LVEF, left ventricular ejection fraction; LVESV, left ventricular end systolic volume; LVEDF, left ventricular end diastolic volume; SV, stroke volume; NYHA, New York Heart Association; BNP, brain natriuretic protein.
BMMC Transplantation Relieved Clinical Manifestations of Heart Failure
The Characteristics of Patients After BMMCs Transplantation

p < 0.05 compared to baseline with values measured at 7 days following cell grafting.
p < 0.01 compared to baseline with values measured at 21 days following cell grafting.
The Characteristics of Patients After Medication Therapy
No significant differences were shown in comparison of baseline with values measured after 7 and 21 days of medication therapy.
Plasma Apelin Levels Were Decreased in Both Patient Groups at Baseline
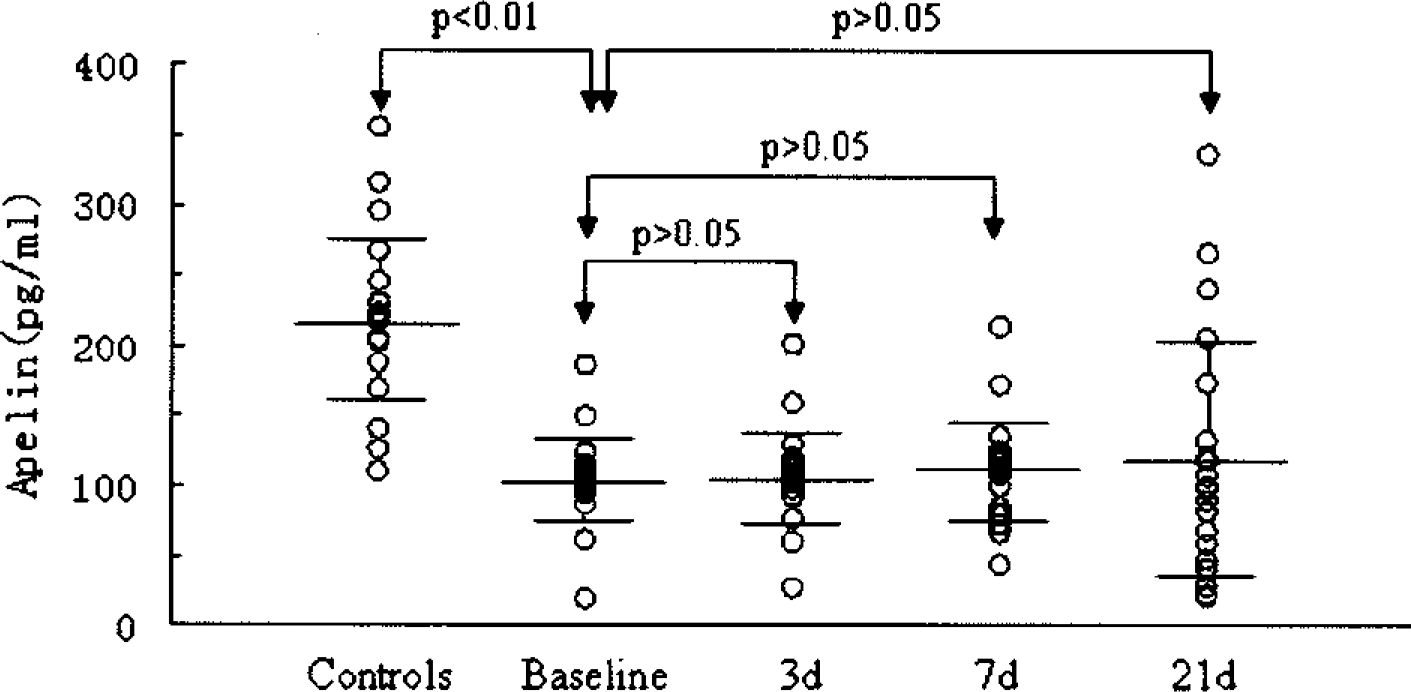
The baseline circulating apelin levels in patients with IHF were significantly reduced (mean apelin: 106.5 ± 32.2 vs. 219.4 ± 59.8 pg/ml, p < 0.01) and the plasma levels of BNP were markedly increased (mean BNP: 662.8 ± 83.0 vs. 167.7 ± 23.4, pg/ml, p < 0.001) compared to the healthy control subjects. However, there were no significant differences in apelin and BNP levels between the BMMC group and the standard medication therapy group at baseline, as shown in Tables 1–3 and Figure 1 and Figure 2.
Plasma apelin levels in control subjects and in patients with BMMCs grafted at baseline, and 3, 7, and 21 days after cell infusion, respectively. Apelin levels are reported as individual values, mean and 95% CI (confidence interval). Plasma apelin levels in control subjects and in patients receiving standard medication therapy at baseline, and 3, 7, and 21 days, respectively. Apelin levels are reported as individual values, mean and 95% CI.
Apelin Levels Increased Markedly After Transplantation of BMMCs
At 3 days after BMMC transplantation, plasma levels of apelin were slightly but not significantly increased compared to baseline (107.2 ± 32.2 vs. 112.2 ± 42.9 pg/ml, p > 0.05). At 7 days after the procedure, there was a significant increase of plasma apelin level (107.2 ± 32.2 vs. 168.4 ± 41.0 pg/ml, p < 0.05) compared to baseline. At 21 days after the procedure, plasma apelin levels increased threefold above baseline (p < 0.01) and were 49.4% higher than at 7 days (p < 0.05), as shown Figure 1. These changes in apelin were mirrored by significant reductions in BNP levels (p < 0.05–0.01) compared to baseline. However, in the standard medication therapy group, there were no significant changes of plasma apelin level and BNP level after medication treatment. The relation of apelin and BNP to LVEF at baseline, and 7 and 21 days after BMMC infusion or standard medication therapy are shown in Figure 3.
Relation of apelin and brain natriuretic peptide (BNP) to LVEF at baseline, and 7 and 21 days after BMM cells transplantation or standard medication therapy. (A) Relation of BNP to LVEF in BMMC group; (B) relation of apelin to LVEF in BMMC group; (C) relation of BNP to LVEF in control group (standard medication therapy); (D) relation of apelin to LVEF in control group.
Discussion
This study reports the novel finding that plasma apelin level is significantly increased after transplantation of BMMC in patients with severe ischemic heart failure. The increase of plasma apelin was accompanied by improvement of heart function, such as relief of dyspnea, increase in urine output, and increase of LVEF within 7 days after BMMC transplantation. These observations suggest that apelin-APJ signaling may play a very important role in the improvement of heart function with BMMC transplantation.
At present, despite the evolution of a broad array of treatment options, the conventional management for heart failure generally does little to replace lost cardiomyocyte mass or myocardial scar with new contractile cells (1). So cell therapy is a novel and promising option to improve vascularization and cardiac function. BMMCs have been used in preclinical and clinical experiments to treat ischemic heart disease (2,10,11,15,16,18,19,24). Several group randomized control trials have reported that intracoronary administration of BMMCs reduces postinfarction ventricular remodeling and improves left ventricular function (2,11,16). However, the efficiency of transplanted BMMCs to differentiate into functional reparative cells in damaged myocardium and in therapeutically relevant numbers has never been adequately documented or demonstrate (8,28). The mechanisms underlying these therapeutic effects have not been clearly defined (9,17). Recent reports have suggested that some of these reparative effects are not mediated by differentiation of BMMCs but rather by paracrine factors secreted by BMMCs (4,23,27). The conditioned medium from mesenchymal stem cells contains proangiogenic factors, such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), stromal cell derived factor-1 (SDF-1), and insulin growth factor-1 (IGF-1) (27). These factors have been postulated to promote arteriogenesis, to support stem cell survival in damaged myocardium, to protect against ischemia, and to support and maintain hematopoiesis through a paracrine mechanism (4,23,27). However, it is still not clear whether BMMCs can release small active peptides that exert direct positive inotropic contractility. In this study, we first reported that the relationship of changes of apelin plasma levels and the improvement of heart function after BMM cells transplantation in patients with severe ischemia heart failure, which shown that apelin-APJ signaling may exert a direct positive inotropic action on the failing heart through a paracrine and autocrine mechanism.
Apelin was found to be the endogenous ligand for the orphan receptor APJ in 1998 (26). The apelin-APJ system has been postulated to play an important role in cardiovascular homeostasis (25). Apelin and its G protein-coupled receptor regulate cardiac development as well as cardiac function (20,22). Apelin is one of the most potent endogenous stimulators of cardiac contractility with characteristics of circulating hormones in general (14). Studies in animal models of heart failure have shown that administration of apelin increased cardiac output and the contractility in failing heart (3,6). The apelin-APJ signaling pathway has also been identified as a potentially important mediator in the pathophysiology of heart ischemic injury (12,21). Although measurement of apelin plasma levels in chronic heart failure has yielded conflicting results (13), most studies showed that plasma apelin levels were decreased in patients with heart failure (5). Some showed that plasma apelin levels increased in early heart failure before a decrease in late disease, suggesting that the apelin-APJ system is recruited to support the failing heart in mild to moderate left ventricular dysfunction (5).
In our study, all patients with IHF were confirmed to be in severe heart failure resulting from ischemic cardiomyopathy (LVEF <40%, NYHA mean classification >III). The levels of plasma apelin-36 were significantly decreased compared to the control subjects. Although there are discrepancies in the plasma apelin levels reported by others (13), they may be due to the difference in experimental techniques used to determine plasma apelin concentrations. We extracted and prepurified plasma samples prior to apelin analysis. The changes in apelin after BMMC transplantation were also mirrored by significant reductions in BNP levels. The low baseline apelin levels in severely sick patients may reflect the inability of the failure heart patients to compensate for the decline of left ventricular performance.
After BMMC transplantation we were surprised to find that the plasma levels of apelin significantly increased. This increase occurred gradually from day 3 to day 21. The plasma concentrations of apelin were as high as threefold above baseline at 21 days after cell grafting. This was accompanied by improvement of heart function reflected by relief of dyspnea, increase in urine output, and an increase in LVEF at 7 days, which was maintained at 21 days. To eliminate the effects of percutaneous coronary intervention (PCI) and medication on concentration changes of apelin, patients had received maximal tolerated medical therapy at the time of enrollment. In the standard medication therapy group, there were no significant changes in the plasma apelin concentrations, BNP, or LVEF from day 3 to day 21. The only treatment difference between the BMMC infusion group and the standard medication therapy group was cell transplantation.
Therefore, we postulate that the secretion of apelin is induced by grafted BMMCs. We also postulate that this potent endogenous positive inotropic substance, apelin, is secreted by BMMCs and acts as a paracrine mediator. Our data suggest that apelin could play an important role in early improvement of cardiac function in patients with ischemic heart failure. We conclude that the beneficial action of grafted BMMCs may result in part from the paracrine activity of secreted peptides such as apelin.
Footnotes
Acknowledgments
This study was supported by a grant of the National Advanced Technology Development the Plan of China (863 plan) (2006AA02Z469). We thank Dr. Joel S. Karliner, Professor of Medicine, University of California San Francisco, and Associate Chief of Medicine for Research, Department of Veterans Affairs Medical Center, University of California San Francisco, USA for valuable comments and helpful suggestions in writing and revising this manuscript.
