Abstract
Our previous study demonstrated that apelin level increased significantly after the treatment of intracoronary implantation of bone marrow mononuclear cells (BMMCs), followed by the improvement of cardiac function in patients with severe ischemic heart failure. The present studies both in vivo and in vitro explored whether mesenchymal stem cells derived from bone marrow (BMSCs) activate the apelin-APJ pathway when differentiating into cardiomyogenic cells. Isolated BMSCs from rat femurs and tibias were cultured and expanded for three passages, labeled with DAPI, and treated with 5-azacytidine (5-AZ). BMSCs labeled with ad-EGFP were injected intramyocardially into the peri-infarct area of rat models with acute myocardial infarction. Immunofluorescence staining exposed that CMGs expressed apelin together with myogenic-specific proteins such as α-actin, troponin T, GATA-4, and connexin-43 at 7 days after 5-AZ treatment or EGFP-BMSC injection. RT-PCR revealed that mRNA in CMGs started to express apelin and APJ from day 7 and progressively increased until day 28. Cardiac function, as measured by echocardiography in vivo, was significantly improved in parallel with the extent of apelin expression after BMSC transplantation. Our finding indicated that the expression of the apelin-APJ pathway during differentiation of BMSCs into CMGs may be an important mechanism in regulation of myocardial regeneration and functional recovery after BMSC transplantation.
Keywords
Introduction
Despite various therapeutic approaches that have been developed, ischemia heart disease is still a leading cause of death throughout the world. As necrotic cardiomyocytes are replaced inevitably by fibrous tissues after myocardial infarction, dysfunctional myocardial remodeling is a fatal common outcome (29). Therefore, cell-based therapies for cardiac regeneration are inducing great excitement. Recent studies indicate that bone marrow stem cells harbor a multilineage differentiation potential and are able to differentiate into smooth muscle, endothelial, and cardiac cell phenotypes, and contribute to improvement of cardiac function in diseased myocardium (10,16,22,23,25). More recently, some randomized clinical trials have also suggested that intramyocardial or intracoronary delivery of autologous bone marrow-derived cells improve regional perfusion and cardiac function in patients with myocardial infarction (1,6,11,21,28). However, the mechanisms of their cellular reparative capacity have not yet been determined (1,6,11,21,28).
Our previous study (8) revealed that intracoronary infusion of bone marrow-derived mononuclear cells (BMMCs) in patients with refractory chronic heart failure secondary to ischemic cardiomyopathy significantly increased urine output and improved symptoms of heart failure within 72 h after treatment. However, this early remarkable effect cannot be readily attributed to myocardial regeneration from the donor cells. Thus, we hypothesized that infused cells might stimulate or release soluble biological factors by paracrine mechanisms that account for their contributions to cardiac tissue repair and functional recovery (8).
Current studies from several investigators suggest that bone marrow-derived mesenchymal stem cells (BMSCs) secrete a wide array of cytokines that exert their protective benefits on surrounding tissue and stimulate vasculogenesis (4,30). However, there is a lack of sufficient evidence to confirm that BMSCs are capable of secreting active cardiovascular peptides and thereby protect against cardiac injury and modulate positive inotropy (4,30).
Apelin, a newly discovered inotropic peptide, is the endogenous ligand for the orphaned G protein-coupled receptor (APJ) (31,32). The apelin-APJ signaling pathway is widely represented in the heart and vasculature, and is emerging as an important regulator of cardiovascular homeostasis (13,14,20). In preclinical models, apelin-APJ signaling also attenuates ischemic myocardial injury and maintains cardiac performance in infarction and chronic pressure overload (2,3,17). The plasma levels of apelin significantly decrease in patients with heart failure (7). Moreover, recently the apelin-APJ pathway has been identified as an important regulator of myocardial cell specification and heart development. Zebrafish developmental models show that ectopic expression of apelin and APJ result in the complete absence of cardiomyocytes development. Apelin-APJ signaling has been proven to serve as a critical gradient for migration of mesodermal cells fated to contribute to the myocardial lineage, which plays a cell-autonomous role in myocardial specification and the formation of myocardial progenitors (26,35).
Previously, our study found that apelin increased significantly after intracoronary implantation of BMMCs, followed by the improvement of cardiac function in patients with severe ischemic heart failure (9). We considered that increased apelin levels may act as a paracrine mediator following BMMC therapy. Furthermore, we found that no one has reported whether stem cells implicate the expression of the apelin and APJ system in cardiomyogenic cells during the course of its differentiation.
In the present study, we designed experiments in both in vitro and in vivo models to explore: 1) whether the apelin-APJ pathway is expressed during BMSC differentiation into cardiomyogenic cells, and 2) the potential connection between the expression of apelin-APJ signaling and the improvement of heart function in acute myocardial infarction models.
Materials and Methods
BMSC Isolation, Culture, and Labeling
The procedures were performed in accordance with the guidelines for animal experimentation of Navy General Hospital Committee. Sprague-Dawley (SD) rats were sacrificed, and femurs and tibia were aseptically harvested. The adherent muscles and soft tissues were removed. The proximal and distal ends of the bone were excised just at the beginning of the marrow cavity. Following a rinse in phosphate-buffered saline (PBS), whole marrow plugs were obtained by flushing the bone marrow cavity with an 18-gauge needle connected to a syringe filled with culture medium composed of low-glucose Dulbecco's modified Eagle's medium (DMEM, Gibco BRL, USA) supplemented with 10% fetal bovine serum (FBS, Hangzhou Sijiqing Biological Engineering Materials Co. Ltd., China). Mononuclear cells were collected at the Percoll interface, rinsed twice in low-glucose DMEM, and plated at a density of 2 × 105/cm2 in low-glucose DMEM and incubated at 37°C in a humidified atmosphere of 95% air 5% CO2. Nonadherent cells were removed after 24 h and culture media were replaced every 2 or 3 days. When these primary BMSCs reached 90% confluence, they were trypsinized (trypsin-0.02% EDTA, Hyclone, China), counted, and passaged at a density of 6 × 105/cm2 and the cells of passage 3 were used for study. These cells were trypsinized and counted after they were rinsed in PBS (pH 7.2). Aliquots of 2 × 105 cells were incubated for 30 min with phycoerythrin (PE)/fluorescein (FITC)-labeled monoclonal antibodies against rat CD44, CD90, CD105, CD73, CD45, and CD34 (Beijing Boisynthesis Biotechnology Co, BJ). After a PBS wash, the stained cells were run on a flow cytometer (FACS Calibur, Becton-Dickinson, USA) to confirm BMSC purity.
Before induction of differentiation or transplantation, BMSCs were labeled with 4′6-diamidino-2′-phenylindole (DAPI) in vitro. For transplantation in vivo, BMSCs were transduced with a retroviral vector encoding the enhanced green fluorescent protein (EGFP-BMSCs). Transduction efficiency was assessed by fluorescence-activated cell sorting (FACS) analysis.
Cardiomyogenic (CMG) Cell Differentiation of BMSCs In Vitro
The differentiation of CMG cells from BMSCs was performed as previously described (25). The third passage of rat BMSCs were seeded into 35-mm dishes at a density of 20,000 cells/cm2 in DMEM medium containing 10% FBS and 1% antibiotics (100 U/ml penicillin and 250 ng/ml streptomycin). On the second day, the medium was changed, and the cells were exposed to the medium with 20% FBS or treated for 24 h with the medium consisting of 20% FBS and 5-azacytidine (5-AZ; 3 μmol/L). Thereafter, the cells were cultured in medium consisting of 20% FBS and 1% antibiotics, and the medium was changed twice a week for 4 weeks until the experiment was terminated. Control cells were cultured in DMEM supplemented with 5% FBS and antibiotics. CMG cells showed spontaneous beating.
Myocardial Infarction Models and BMSC Injection
Ligation of the left anterior descending coronary artery (LAD) was performed in 6-week-old SD female rats. Animals were anesthetized using a combination of xylazine (10 mg/kg) and ketamine (80 mg/kg) injected intramuscularly and a left thoracotomy was performed under artificial ventilation with air. The heart was accessed through the fourth intercostal space. The pericardial sac was cut and the heart exteriorized through the resulting space. The LAD was ligated with a 7–0 silk suture approximately midway between the left atrium and the apex of the heart. Successful performance of coronary occlusion was verified by observing the development of a pale color in the distal myocardium after ligation and by recording EKG evidence of infarction (ST segment elevation >1 mm in leads V1-V6). In the sham animals the ligature was left loose. Before BMSC transplantation, animals were randomized into three groups: (A) sham-operated animals with no injection performed; (B) control animals that received only PBS injection; (C) animals that received EGFP-BMSCs injection. Accordingly, 1 h later a total of 1 × 106 third passage EGFP-BMSCs suspended in PBS or an equivalent volume of PBS alone was injected at five different sites located in the infarct border zone. The investigators responsible for surgery and cell injection were blinded to the treatment groups. All procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act in China and approved by the Navy General Hospital Standing Committee.
Immunofluorescence Staining
DAPI-positive CMG cells were stained with mouse monoclonal antibodies against sarcomeric α-actin, troponin T (TnT), connexin-43, and GATA-4 at 1:200 dilution (Beijing Boisynthesis Biotechnology Co, LTD) and FITC-conjugated secondary antibody (Beijing ZGB, Biotechnology CO, LTD, China, 1:100) as a control. CMG cells were also stained with a rabbit monoclonal anti-apelin-12 antibody (1:200; Phoenix Pharmaceuticals, Belmont, CA, USA). Confocal images were obtained using a 40x objective on a Leica Microsystems TCS SP laser scanning confocal microscope. Digitized confocal images were processed by Leica confocal software and Adobe Photoshop (version 6.0).
The hearts of rats sacrificed at day 7, 14, and 28 were subject to histological analysis by hematoxylineosin and immunohistochemical staining. Assessment of the cell grafting was performed as follows. The apical myocardium below the coronary artery ligation site was sectioned. Cryostat sections (10 mm) were cut from frozen GFP-positive tissue samples; EGFP was detected and quantified with an anti-EGFP antibody (1:200; United States Biologica). Serial sections were immunolabeled with antibodies against sarcomeric α-actin, connexin-43, and GATA-4 and then were stained with a rabbit monoclonal anti-apelin-12 antibody followed by incubation with FITC-conjugated secondary antisera (Phoenix Pharmaceuticals, Belmont, CA, USA). The area of GFP-positive BMSCs in each tissue section was measured. Confocal images were obtained and processed as described above.
Apelin and APJ Gene Expression
To confirm messenger RNA (mRNA) expression of the apelin and APJ genes, reverse transcription PCR (RT-PCR) was performed. Total RNA from the CMG cells (baseline and 3, 7, 14, 21, and 28 days after 5-AZ treatment) were prepared by the acid guanidinium thiocyanate-phenol-chloroform extraction method. One microgram of total RNA with a combination of oligo(dT) and random hexamer primers was subject to first-strand complementary DNA (cDNA) synthesis in a 20-μl reaction for 60 min at 37°C. After completion of the cDNA synthesis, 20 μl was used for each PCR reaction according to the manufacturer's instruction (Promega, USA). The primer sequences used for amplification of the rat apelin were 5′-TGCTCTGGCTCTCCTTGACT-3′ (sense strand) and 5′-ATGGGTCCCTTATGGGAGAG-3′ (antisense strand). The primer sequences used for amplification of rat APJ were 5′-CTGCTGAGCATCATCGT GGT-3′ (sense strand) and 5′-AGGGCCAGTGCAG CAAATT-3′ (antisense strand); The primer sequences used for amplification of internal control GAPDH were AGTATGACTC- CACTCACGGCAA (sense strand) and TCTCGCTCCTGGAAGATGGT (antisense strand). The PCR products were separated on a 1% agarose gel, and the amplified cDNA bands were visualized by ethidium bromide staining. The bands imaged by Chemi-Genius Bio Imaging System were analyzed via Gene Tools software.
Echocardiography
Rat heart function was assessed by transthoracic echocardiography, which was performed at 1, 7, and 28 days after MI and cell transplantation, using 12-MHz transducer system designed for cardiac ultrasound (HP Sonos 5500). Two-dimensional images were obtained at midpapillary and apical levels. End-diastolic (EDV) and end-systolic (ESV) left ventricular volumes were obtained by biplane area length method. Percent left ventricular ejection fraction (LVEF) and the percent fractional shortening (FS) were calculated. All measurements were averaged on three consecutive cardiac cycles and were analyzed by two independent observers who were blinded to the treatment status of the animals.
Statistical Analysis
All results are presented as mean ± SE. Data were analyzed by one-way or two-way ANOVA with repeated measures followed by Bonferroni's post hoc test. Statistical significance was assumed at a value of p < 0.05.
Results
BMSC-Derived Cardiomyocytes Express Apelin and APJ In Vitro
BMSCs displayed a fibroblast-like morphology under phase-contrast microscopy and expressed CD44, CD90, CD105, and CD73, but did not express markers CD45 and CD34 and cardiac cell lineages before 5-AZ treatment (Fig. 1). DAPI staining was used to identify BMSCs (blue nucleus with multiple nucleoi) from differentiated myocytes. To confirm that BMSC-derived CMGs exhibited molecular characteristics of cardiac cells after 3, 7, 14, 21, and 28 days of 5-AZ treatment, we examined expression of several cardiac myocyte markers. These cells stained positive for the cardiac-specific transcription factor GATA-4, the cardiac-specific proteins sarcomeric α-actin, and TnT, and the intercellular connection and electrical coupling factor connexin-43 (Fig. 2). Apelin expression following 5-AZ treatment was also demonstrated with confocal laser scanning microscopy images. BMSC-derived CMGs coexpressed apelin with the cardiac sarcomeric α-actin, TnT, connexin-43, and the transcriptional factor GATA-4 at 7 days after 5-AZ treatment (Fig. 2).
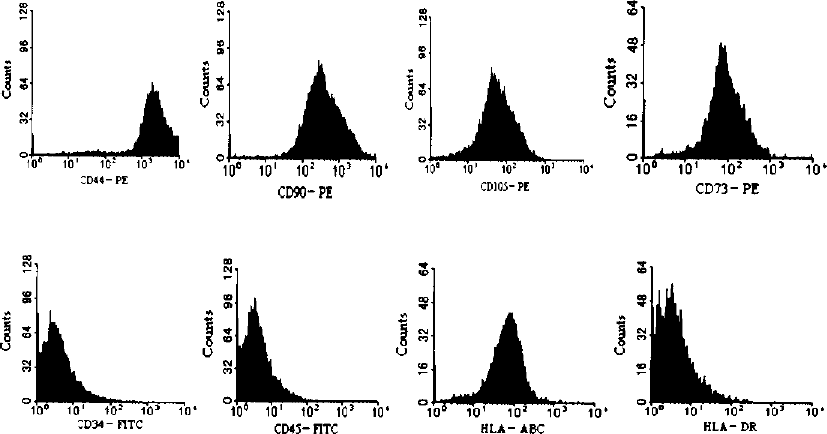
The surface antigens of bone marrow-derived MSCs (BMSC). BMSCs were positive for CD44, CD90, CD105, CD73, HLA-ABC, and negative for CD34, CD45, and HLA-DR. The results confirmed that the cells were a kind of nonhematopoietic MSC.

Confocal microscopy of fluorescent immunohistochemical staining of DAPI-labeled BMSCs induced by 5-AZ after 7 days. (A) (a) CMGs from DAPI-labeled BMSCs induced by 5-AZ (Dil-M-CMG) display blue nuclei. (b) CMGs stained with antibody to α-actin appear green. (c) CMGs stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows Dil-M-CMG coexpresses with α-actin and apelin (yellow-green, white arrows). Scale bar: 50 μm. (B) (a) Dil-M-CMG cells display blue nuclei. (b) Cells stained with antibody to troponin T (TnT) appear green. (c) Cells stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows Dil-M-CMG cells coexpress TnT and apelin (purple-blue, white arrows). Scale bar: 50 μm. (C) (a) Dil-M-CMG cells exhibit blue nuclei. (b) Cells stained with antibody to GATA-4 appear green. (c) Antibody to apelin stains red. (d) Merged image of (a), (b), and (c) shows Dil-M-CMG cells coexpress GATA-4 and apelin (orange-green, white arrows). Scale bar: 50 μm. (D) (a) Dil-M-CMG cells display blue nuclei. (b) Cells stained with antibody to connexin-43 appear green. (c) Cells stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows Dil-M-CMG cells coexpress connexin-43 and apelin (yellow-green, white arrows). Scale bar: 50 μm. (E) The apelin expression rate of CMGs from DAPI-labeled BMSCs induced by 5-AZ (Dil-M-CMG) at 7 days was defined as the percentage of α-actin-, TnT-, GATA-4-, connexin-43-positive cells in the DAPI-positive BMSCs. Measured data were averaged.
The induced expression of apelin after 5-AZ treatment was further confirmed by RT-PCR analysis. Apelin expression was not detected in BMSCs before 5-AZ treatment but it was detected afterwards. The expression level gradually increased in a time-dependent manner after 5-AZ treatment (Fig. 3A). The expression pattern of APJ paralleled apelin expression in BMSCs before and after 5-AZ treatment (Fig. 3B). Quantitative analyses of apelin expression and APJ were significantly higher in cells at 7, 14, 21, and 28 days in comparison to the untreated BMSC group.

(A) Expression of apelin mRNA. Expression of apelin mRNA in CMG cells derived from BMSCs after 5-AZ induced at 3, 7, 14, 21, and 28 days, respectively. Lane 1: DNA ladder marker; lane 2: rat cardiomyocytes; lane 3: isolated BMSCs not induced by 5-AZ; lane 4: CMG cells after 5-AZ exposure for 3 days; lane 5: CMGs 7 days; lane 6: CMGs 14 days; lane 7: CMGs 21 days; lane 8: CMGs 28 days. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal loading control.(a) Densitometric analysis was performed; ratio of RT-PCR product of apelin to that of GAPDH is shown. Data were obtained from five separate experiments and were presented as arbitrary units over control BMSCs (before 5-AZ treatment). Values are mean ± SEM. ∗p < 0.01 versus control BMSCs (before 5-AZ treatment). (B) Expression of APJ mRNA. Expression of APJ mRNA in CMG cells derived from BMSCs after 5-AZ induced at 3, 7, 14, 21, and 28 days, respectively. Lane 1: DNA ladder marker; lane 2: rat cardiomyocytes; lane 3: isolated MSCs not induced by 5-AZ; lane 4: CMGs 3 days; lane 5: CMGs 7 days; lane 6: CMGs 14 days; lane 7: CMGs 21 days; lane 8: CMGs 28 days. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal loading control. (b) Densitometric analysis was performed; ratio of RT-PCR product of APJ to that of GAPDH is shown. Data were obtained from five separate experiments and were presented as arbitrary units over control BMSCs (before 5-AZ treatment). Values are mean ± SEM. ∗p < 0.01 versus control BMSCs (before 5-AZ treatment).
CMG Cells Express Apelin In Vivo
We used enhanced green fluorescent protein (EGFP) as a marker to identify the engrafted cells in heart. Animals were sacrificed at days 7, 14, and 28 (n = 8 each) after the injection of male rat BMSCs-EGFP in the peri-infarct area of female rats. EGFP-positive cells were observed in the heart of the BMSC transplantation group. They were mostly localized in the border area. The frequency of EGFP-positive cardiomyocytes was very low in remote myocardium. Sarcomeric α-actin, TnT, GATA-4, and connexin-43 were used as markers for CMG cells, which were colocalized with apelin as shown in Figure 4. No EGFP-positive cells were detected in the PBS control group.
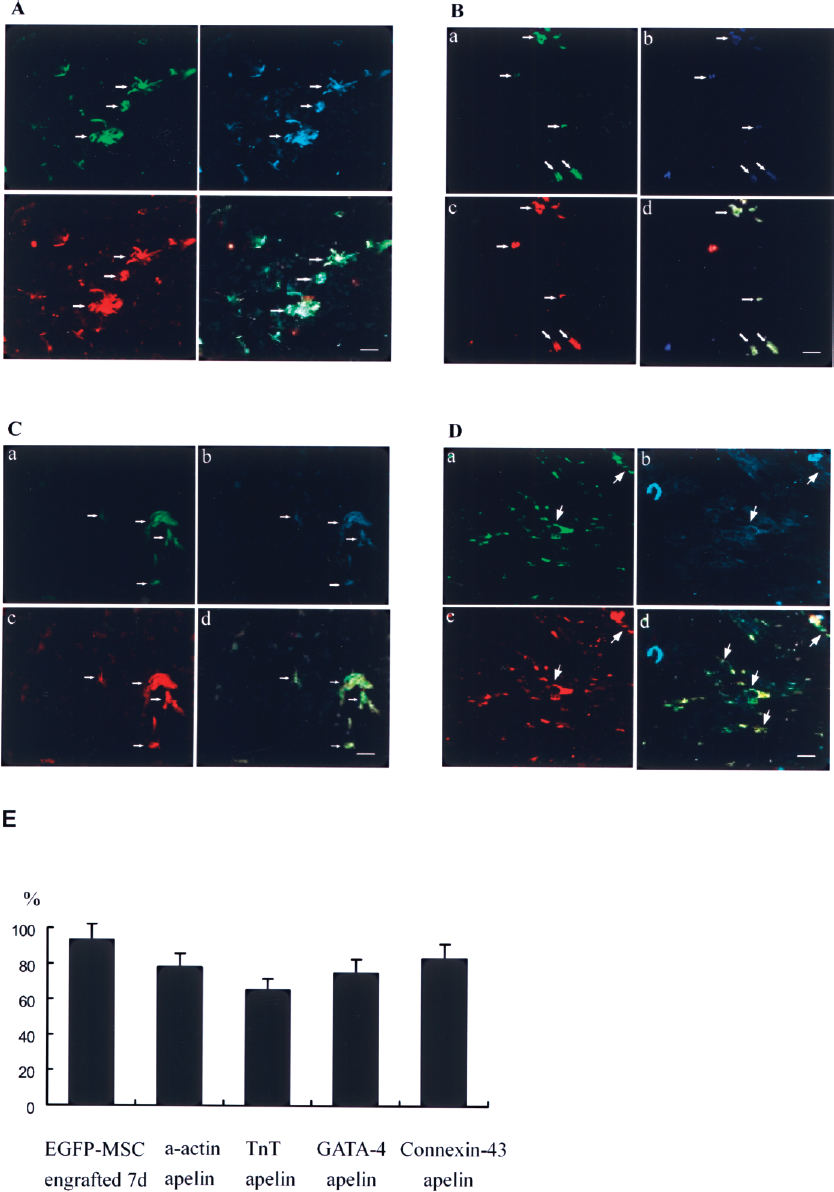
(A) Confocal microscopy of fluorescent immunohistochemical staining of CMG cells derived from EGFP-labeled BMSCs engrafted in infarcted myocardium for α-actin and apelin at 7 days. (a) EGFP-labeled BMSCs appear green (white arrows). (b) Sections stained with antibody to α-actin appear blue. White arrows point to transplanted BMSCs. (c) Sections stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows CMG from EGFP-BMSCs coexpress α-actin and apelin (yellow-green cells; white arrows). Scale bar: 50 μm. (B) Confocal microscopy of fluorescent immunohistochemical staining of CMG cells derived from EGFP-labeled BMSCs engrafted in infarcted myocardium for troponin T (TnT0 and apelin at 7 days. (a) EGFP-labeled BMSCs appear green (white arrows). (b) Sections stained with antibody to TnT appear blue. White arrows point to transplanted BMSCs. (c) Sections stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows CMGs from EGFP-labeled BMSCs coexpress TnT and apelin (yellow cells; white arrows). Scale bar: 50 μm. (C) Confocal microscopy of fluorescent immunohistochemical staining of CMG cells derived from EGFP-labeled BMSCs engrafted in infarcted myocardium for connexin-43 and apelin at 7 days. (a) EGFP-labeled BMSCs appear green (white arrows). (b) Sections stained with antibody to GATA-4 appear blue. White arrows point to transplanted BMSCs. (c) Sections stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows CMG from EGFP-labeled BMSCs coexpress GATA-4 and apelin (yellow cells; white arrows). Scale bar: 50 μm. (D) Confocal microscopy of fluorescent immunohistochemical staining of CMG cells derived from EGFP-labeled BMSCs engrafted in infarcted myocardium for connexin-43 and apelin at 7 days. (a) EGFP-labeled BMSCs appear green (white arrows). (b) Sections stained with antibody to connexin-43 appear blue. White arrows point to transplanted BMSCs. (c) Sections stained with antibody to apelin appear red. (d) Merged image of (a), (b), and (c) shows CMG from EGFP-labeled BMSCs coexpress connexin-43 and apelin (yellow cells; white arrows). Scale bar: 50 μm. (E) The apelin expression rate of engrafted EGFP-labeled BMSCs in infarcted myocardium at 7 days was defined as the percentage of α-actin-, TnT-, GATA-4-, connexin-43-positive cells in the EGFP-positive cells. Measured data were averaged.
Improvement of Cardiac Contract Function Was Accompanied by Apelin Expression
Eight rats in each group were examined for cardiac function by echocardiography at 1, 7, and 28 days after EGFP-BMSC engraftment. As shown in Table 1, left ventricular ejection fraction (LVEF) and fractional shortening (FS) progressively and significantly increased from 7 days to 28 days after BMSC transplantation (p < 0.05–0.01). Conversely, in the control group, LVEF and FS gradually decreased to 28 days (p < 0.01). However, no significant changes were seen for end-diastolic volumes after BMSC transplantation.
Parameters of LV Function by Echocardiography

LV function assessed by 2D echocardiography. EF, ejection fraction; LVEDV, left ventricular end-diastolic volume; LVESV, LV end-systolic volume; FS, fractional shortening. Baseline 1 day compared with 7 days, 28 days, respectively, after MSCs engrafted or control.
p < 0.05.
p < 0.01.
Discussion
Different types of BMSCs have been proposed as potential sources of cell therapy for ischemic heart disease (22). BMSCs are regarded as multipotent stem cells and have been used experimentally as treatment for ischemic heart disease in preclinical and clinical studies (1,8,11,16,21,23,25,35). Wollert and colleagues (21) and others (1,8,11) reported that intracoronary administration of one such cell type, bone marrow mononuclear cells (BMMCs), attenuated the progress of postinfarction ventricular remodeling and improved left ventricular function. The utility of these cells was ascribed to their ability to differentiate into cell types with reparative capacity (12,18,19,27,34). In our studies, we found that BMSC transplantation generated similar beneficial effects in injured myocardium, suggesting that engrafted BMSCs can either differentiate into functional reparative cells or that they generate one or more factors that enhance regeneration and increase function. Our data favor the latter explanation. Thus, we found that the expression of apelin in cardiomyogenic cells, which were derived from BMSCs, was significantly increased and paralleled the regeneration of cardiac cells in impaired cardiac tissue. These results suggest that apelin was involved in the mechanisms underlying the tissue repair of ischemic myocardial infarct.
APJ receptors have been found in developing heart tubes and vasculature, and in adult cardiac endothelium, atria, and endocardium. The locations correlate with the expression patterns of apelin and those of cognate components of the cardiovascular system (13,15,31). Although recent investigations concluded that apelin and G protein-coupled receptor Agtrl1b participated in modulation of early development of myocardial progenitors (26,35), these data require confirmation. However, depletion of the Agtrl1b receptor results in a reduced number of myocardial progenitor cells and the absence of an organized cardiac structure (26). Our in vitro experiments in CMG cells showed that the expression of apelin was significantly increased. The levels of apelin and APJ mRNA expression progressively intensified between day 7 and 28 after 5-AZ treatment. Experiments performed in vivo on the rat models with myocardial infarction demonstrated that the appearance of CMG cells was correlated with the presence of EGFP and other myogenic-specific peptide markers, such as α-actin, TnT, GATA-4, and connexin-43 after EGFP-BMSCs were injected into the infarct area of ischemic myocardium. These data indicate that BMSC transplantation is capable of enhancing the expression of apelin and APJ in the same situation.
Apelin has been reported to induce potent endogenous positive inotropic benefits and to reduce ventricular preload and afterload through triggering nitric oxide-dependent vasodilatation in normal and failing hearts (3,5,33). Ronkainen et al. found hypoxia inducible factor regulates the cardiac expression and secretion of apelin (24). In our study, depressed cardiac function, as determined by LVEF and FS in the rat model of cardiac infarction, was significantly improved, in parallel with the apelin expression in CMG cells. These findings concur with Szokodi et al., who reported that administration of apelin-16 produced a dose dependent increase in myocardial contractility in isolated rat heart preparations (31). Our study therefore suggests that in the process of BMSC differentiation into CMG cells, activated apelin-APJ signaling plays a critical role for both tissue repair and functional recovery of cardiac ischemic injury through paracrine and autocrine mechanisms.
In summary, ur findings for first time provide direct evidence that apelin-APJ signaling is expressed during the early development of the CMGs derived from MSC differentiation in vitro and in vivo. Apelin might play an important role in myocardial regeneration and functional recovery of the infarct heart through paracrine and autocrine mechanisms.
Footnotes
Acknowledgments
This study was supported by a grant of the National Advanced Technology Development the Plan of China (863 plan) (2006AA02Z469). We thank Dr. Joel S. Karliner, Professor of Medicine, University of California San Francisco, and Associate Chief of Medicine for Research, Department of Veterans Affairs Medical Center, University of California San Francisco, and Mark Sussman, Ph.D., Distinguished Professor of Biology, SDSU Heart Institute, San Diego State University, Department of Biology, San Diego, CA, for valuable comments and helpful suggestions in writing and revising this manuscript.
