Abstract
Postoperative liver failure is one of the most critical complications following extensive hepatectomy. Although transplantation of allogeneic hepatocytes is an attractive therapy for posthepatectomy liver failure, transplanting cells via the portal veins typically causes portal vein embolization. The embolization by transplanted cells would be lethal in patients who have undergone massive hepatectomy. Thus, transplant surgeons need to select extrahepatic sites as transplant sites to prevent portal vein embolization. We aimed to investigate the mechanism of how liver cells transplanted into the mesentery protect recipient rats from acute liver failure after massive hepatectomy. We induced posthepatectomy liver failure by 90% hepatectomy in rats. Liver cells harvested from rat livers were transplanted into the mesenteries of hepatectomized rats. Twenty percent of the harvested cells, which consisted of hepatocytes and nonparenchymal cells, were transplanted into each recipient. The survival rate improved significantly in the liver cell transplantation group compared to the control group 7 days after hepatectomy (69 vs. 7%). Histological findings of the transplantation site, in vivo imaging system study findings, quantitative polymerase chain reaction assays of the transplanted cells, and serum albumin measurements of transplanted Nagase analbuminemic rats showed rapid deterioration of viable transplanted cells. Although viable transplanted cells deteriorated in the transplanted site, histological findings and an adenosine-5′-triphosphate (ATP) assay showed that the transplanted cells had a protective effect on the remaining livers. These results indicated that the paracrine effects of transplanted liver cells had therapeutic effects. The same protective effects were observed in the hepatocyte transplantation group, but not in the liver nonparenchymal cell transplantation group. Therefore, this effect on the remnant liver was mainly due to the hepatocytes among the transplanted liver cells. We demonstrated that transplanted liver cells protect the remnant liver from severe damage after massive hepatectomy.
Keywords
Introduction
Although orthotopic liver transplantation has been one of the most effective therapies for lethal liver failure, a chronic donor shortage requires alternative therapies, including hepatocyte transplantation 1 . Some clinical trials for congenital enzyme deficiency have demonstrated that hepatocyte transplantation is useful for improving liver function, and it can be a temporal bridge for orthotopic liver transplantation2–4. Postoperative liver failure is one of the most critical complications following extensive hepatectomy for treating hepatobiliary cancer 5 . Patients typically present with massive ascites, progressive hyperbilirubinemia, coagulopathy, encephalopathy, and eventually death 6 .
Several previous studies have investigated the possibility of hepatocyte transplantation for resolving acute liver failure after massive hepatectomy, which requires a higher quantity of functional hepatocytes for transplantation7–10. A large amount of cells transplanted via portal veins, including the spleen, typically result in portal vein embolization11–15, which leads to functional deterioration 16 .
However, several extrahepatic sites, including the intraperitoneal cavity, pancreas, subcutaneous cavity, kidney capsule, and mesentery, were evaluated as suitable liver cell transplantation sites to avoid portal vein embolization17–20. Some researchers have reported on the recovery of a 90% hepatectomized rat model by hepatocyte transplantation in a peritoneal cavity, which allows a large amount of functional hepatocytes to be transplanted21–25. Indeed, extrahepatic sites that allow a large amount of transplanted cells with poor vascularity should lead to the poor maintenance of transplanted cells. Therefore, it should be elucidated how ectopic liver cell transplantation works to protect from acute liver failure after massive hepatectomy, as this remains unclear.
In the present study, we aimed to investigate the mechanism of how liver cells transplanted into the mesentery prevent acute liver failure after massive hepatectomy.
Materials and Methods
Animals
F344 rats, Sprague–Dawley (SD) rats, and Nagase analbuminemic (NA) rats were purchased from Japan SLC Inc. (Shizuoka, Japan). Transgenic green fluorescent protein (GFP)/F344 rats 26 were provided by Dr. Takahiro Ochiya (Section for Studies on Metastasis, National Cancer Center Research Institute, Tokyo, Japan). Rats used in the experiments weighed between 170 and 300 g, with the exception of the NA rats. Animals were housed at Kyoto University. Rats were fed a standard diet and tap water ad libitum. All of the animal experimental procedures were performed according to the Animal Protection Guidelines of Kyoto University and were approved by the Animal Research Committee of Kyoto University.
Cell Isolation
Grafted liver cells were obtained from male or female F344 wild-type rats or transgenic rats carrying the GFP gene, which helped distinguish grafted hepatocytes from original residual cells. Adult liver cells were isolated using a modified two-step collagenase perfusion technique, as described previously by our laboratory 27 .
Briefly, under general anesthesia with isoflurane (Wako Pure Chemical Industries, Osaka, Japan), donor rats underwent laparotomy, and their portal veins were cannulated. The livers were then preperfused with a Ca2+-free Hank's balanced salt solution (HBSS) containing 0.5 mM ethyleneglycol-bis-(b-aminoethylether)-N, N, N, N-tetraacetic acid (HBSS-EGTA) (HBSS; Wako Pure Chemical Industries) (EGTA; Sigma-Aldrich, St. Louis, MO, USA), followed by perfusion with a collagenase solution containing 0.3% dispase II (Sanko Junyaku Co. Ltd., Tokyo, Japan), 0.3% collagenase type II (Gibco, Grand Island, NY, USA), 150 mmol/L NaCl, 5.4 mmol/L KCl, 0.34 mmol/L NaHPO4, 0.1 mmol/L MgSO4, 5.0 mmol/L CaCl2, 4.2 mmol/L NaHCO3, 5.6 mmol/L glucose, 10 mmol/L HEPES, 0.01% deoxyribonuclease, and 0.005% trypsin inhibitor (all of the chemical reagents, unless otherwise specified, were purchased from Wako Pure Chemical Industries). The livers were excised, minced, and immediately filtered through 100-N polypropylene mesh (Kyoshin Rikoh Inc., Tokyo, Japan). In the isolated liver cells, the suspension including hepatocytes and nonparenchymal cells (NPCs) was observed using a light microscope (Keyence Co. Ltd., Osaka, Japan), showing that 20% of all the isolated liver cells contained approximately 6 × 107 hepatocytes and 1 × 107 NPCs. Liver cell homogenates were prepared by sonication for 10 min on ice (Handy Sonic UR-20P; Tomy Seiko Co., Ltd., Tokyo, Japan), and the sonicated samples were examined by light microscopy to confirm the loss of cell integrity. Cell homogenates from 20% of all the isolated liver cells were transplanted into each recipient rat in the homogenate group. The suspension was mixed with collagen gel (Nitta Gelatin Inc., Osaka, Japan) just before transplantation.
In addition, hepatocytes and NPCs were divided as follows. The suspension was centrifuged three times at 50 × g for 4 min, the hepatocytes were pelleted, and the NPCs were separated into the supernatant. The hepatocytes were isolated from the pellets. To isolate the NPCs, the supernatant was collected and centrifuged at 150 × g for 4 min 28 . After quantifying the hepatocytes or NPCs, they were suspended in the HBSS-EGTA solution and mixed with collagen gel just before transplantation.
Cell Transplantation
Male F344 wild-type rats and NA rats were used as recipients. First, posthepatectomy liver failure was induced by 90% hepatectomy, which was modified from the Higgins–Anderson operation 29 . Rats received isoflurane (Wako Pure Chemical Industries) anesthesia to begin the procedure. After laparotomy, the left and medial lobes (70% partial hepatectomy) and right lobes (20%) were removed, with the remaining caudate lobes (10%).
Just after hepatectomy, transplanted cells were suspended in a collagen gel mixture (HBSS-EGTA) and directly injected into each recipient's mesenteric fat. Equivalent amounts of the collagen gel mixture without liver cells were injected into the mesentery of the control rats. We evaluated the transplanted cells immediately posttransplantation using a transilluminator.
Survival Study
Thirty-nine male F344 wild-type rats were divided into three groups. The liver cell transplantation group (n = 13) received 20% of all the isolated liver cells from the donor GFP rat liver mixed with the collagen gel. The liver cell homogenate group (n = 13) received cell homogenates from 20% of all the isolated liver cells mixed with the collagen gel. The control group (n = 13) was injected with collagen without liver cells.
To evaluate differences between receiving hepatocytes (HEPs) alone and NPCs alone, 16 male F344 wild-type rats were divided into two groups (each group, n = 8). In the HEP group, 1 × 107 hepatocytes were transplanted, and in the NPC group, 1 × 107 NPCs were transplanted. Survival probability was calculated using the Kaplan–Meier method, and it was analyzed using the log-rank test.
Biochemical Analysis of the Liver Cell Transplantation Model
Peripheral blood samples were obtained by cutting the live animal's tail under general anesthesia. Serum ammonia levels were determined using an ammonia test kit (Wako Pure Chemical Industries) [transplantation (Tx): n = 4, control (Cont): n = 4], and the total bilirubin levels were determined using the QuantiChrom Bilirubin Assay Kit (Bioassay Systems LLC, Hayward, CA, USA) (Tx: n = 3, Cont: n = 3) according to the manufacturer's protocol. Statistical significance between these groups was evaluated by repeated-measures analysis of variance (ANOVA) followed by the Holm–Sidak test.
Histological, Periodic Acid-Schiff Stain, and Immunohistological Analyses of the Liver Cell Transplantation Model
After the rats were deeply anesthetized with isoflurane, the mesenteries or remnant livers were harvested followed by cervical dislocation. The samples were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E; Merck, Darmstadt, Germany). Engraftment of the transplanted liver cells in the mesentery was analyzed at 8, 24, and 72 h posttransplantation, and the remnant liver was evaluated at 8 and 24 h posttransplantation.
For immunohistochemical analysis, a Target Retrieval Solution (Dako, Glostrup, Denmark) was used for antigen retrieval, and then a nonspecific binding was blocked with 0.1% Triton X-100 (Sigma-Aldrich) dissolved in phosphate-buffered saline (PBS). The sections were incubated overnight at 4°C with primary antibodies as follows: rabbit polyclonal anti-GFP antibody (1:200; Molecular Probes Inc., Eugene, OR, USA) and sheep polyclonal anti-human albumin antibody (1:100; Abcam, Cambridge, UK). After washing, the stained sections were incubated with Alexa 488-conjugated donkey anti-rabbit immunoglobulin (Ig) G and Alexa 555-conjugated donkey anti-sheep IgG for 2 h at room temperature (RT). After washing, the stained sections were covered with Vectashield mounting medium with 4,6-diamidino-2-phenylindole (DAPI; Vector Laboratories, Burlingame, CA, USA).
Periodic acid-Schiff (PAS) stain was performed using the PAS Staining Kit (Muto Pure Chemicals, Tokyo, Japan) according to the manufacturer's protocol.
Qualitative and Quantitative Analysis of the Transplanted Cells' Existence
The expression of GFP in live rats of the liver cell transplantation group was measured using an in vivo imaging system (IVIS) (PerkinElmer Inc., Waltham, MA, USA) at 0, 8, and 16 h posttransplantation (n = 3).
Additionally, we compared the GFP expression at the transplantation sites between the hepatocyte transplantation group (HEP group) and the liver cell transplantation group (liver cell group), which consisted of hepatocytes and liver NPCs. In the liver cell group (n = 3), rats received 6 × 107 hepatocytes from GFP rats with 1 × 107 NPC liver cells from wild-type rats that were GFP negative. In the HEP group (n = 3), rats received 6 × 107 hepatocytes from GFP rats. We compared the GFP expression between the groups at 0, 4, and 8 h posttransplantation and used repeated-measures ANOVA followed by the Holm–Sidak test.
Quantitative Polymerase Chain Reaction
Total RNA was extracted from the liver cells just before transplantation and from the transplanted liver cells in the mesentery at 8, 24, and 72 h after transplantation using the PureLink RNA Mini Kit (Invitrogen, Carlsbad, CA, USA) (n = 3/each time point). The ReverTra Ace (Toyobo, Osaka, Japan) was used according to the manufacturer's protocol to reverse transcribe 1 μg of total RNA into complement DNA. We performed quantitative polymerase chain reaction (qPCR) assays with the SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA) using the ABI 7500 system (Applied Biosystems). The following primers were used for albumin: 5′-CGTTCTGGTTCGATACACC-3′ and 5′-GAAGTCACCCATCACCGTC-3′; for glucose-6-phosphatase (G6Pase): 5′-GACCTCCTGTGGACTTTGGA-3′ and 5′-AGGCTTGGGTTGCTCCTAGT-3′; for cytochrome 3A1 (CYP3A1): 5′-GGAAATTCGATGTGGAGTGC-3′ and 5′-AGGTTTGCCTTTCTCTTGCC-3′; for cytochrome 2E1 (CYP2E1): 5′-ACTTCTACCTGCTGAGCAC-3′ and 5′-TTCAGGTCTCATGAACGGG-3′; and for glyceraldehyde-3-phosphate dehydrogenase (GAPDH): 5′-GGCACAGTCAAGGCTGAGAATG-3′ and 5′-ATGGTGGTGAAGACGCCAGTA-3′ 30 . The quantification of the given genes was expressed as the relative messenger RNA (mRNA) level compared after normalization to GAPDH. Statistical significance between these groups was evaluated using Student's t-test.
Functional Analysis of the Transplanted Hepatocytes in the Liver Cell Transplantation Model
To evaluate the function of transplanted hepatocytes in the liver cell transplantation model, 20% of all the liver cells isolated from male SD rats were transplanted into the mesenteries of 6- or 7-week-old male NA rats (weight, 150–210 g), the original strain of which is an SD rat, and the serum albumin level of peripheral blood was subsequently measured. In the control group, collagen gel was injected into the mesentery fats. To measure albumin, blood samples were harvested every 4 h until 24 h posttransplantation, and they were evaluated using the rat albumin enzyme-linked immunosorbent assay quantitation kit (Bethyl Laboratories Inc., Montgomery, TX, USA) according to the manufacturer's protocol. Statistical significance between these groups (Tx: n = 3, Cont: n = 4) was evaluated by repeated-measures ANOVA followed by the Holm–Sidak test.
Adenosine-5′-Triphosphate Assay of the Remnant Liver
The remnant livers were collected 24 h after 90% hepatectomy and were stored at −80°C. The levels of adenosine-5′-triphosphate (ATP) in the remnant livers were measured using a firefly bioluminescence assay kit (AMERIC-ATP Kit; Wako Pure Chemical Industries) according to the manufacturer's instructions. The luciferase activity was measured using a plate reader (ARVO X5; PerkinElmer Inc.). Statistical significance between these groups was evaluated using Student's t-test (Tx: n = 3, Cont: n = 3) (HEP: n = 3, NPC: n = 3).
Proliferation Analysis of the Hepatocytes in the Remnant Livers
To detect cells synthesizing deoxyribonucleic acid, proliferating cell nuclear antigen (PCNA) expression in the liver sections was determined immunohistochemically. The deparaffinized sections were incubated in 3% hydrogen peroxide in methanol for 10 min to block endogenous peroxidase activity. Sections were incubated at 4°C for 16 h with mouse anti-PCNA antibody (1:100; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). After the primary antibody had been washed off, the samples were incubated for 60 min with horseradish peroxidase-conjugated anti-mouse IgG (EnVision+ Kit; Dako) and for 1 min with 3,3′-diaminobenzidine (DAB) substrates (Dako). Counterstaining was performed using hematoxylin. After staining, positive cells from 10 randomly selected fields were counted using a light microscope with a 20× objective lens. Statistical significance between these groups was evaluated by Student's t-test (Tx: n = 3, Cont: n = 3).
Removal Study of the Transplanted Cells
Briefly, transplanted cells were surgically removed from the transplant sites 8 h after transplantation (remove group). Twenty-four hours after transplantation, the serum ammonia levels and histological findings were analyzed and compared between the remove group (n = 3) and the nonremove group (n = 4).
Cytokine Array
To detect factors that were produced from transplanted cells, we comprehensively analyzed cytokines, growth factors, and other proteins in the portal vein blood, which connected the remnant liver with the transplanted site. It is often assumed that cytokines or growth factors produced from transplanted cells are distributed from mesenteric fats to the portal vein. Blood samples in the portal vein were obtained from each group (Tx: n = 3, Cont: n = 3) of rats at 4 h after hepatectomy, and they were centrifuged for 20 min at 2,400 × g. The serum was collected and stored at −20°C. Then the serum was diluted at approximately 25-fold and analyzed for specific proteins using a Rat Cytokine Antibody Array Membrane (Abcam) according to the manufacturer's instructions. Quantitation of the detected spots was performed using Quantity One Software 4.4.1 (Bio-Rad Laboratories, Hercules, CA, USA).
Statistical Analysis
Data are expressed as mean ± standard deviation. We performed statistical analyses using GraphPad Prism for Windows, version 6.0 (GraphPad Software, San Diego, CA, USA). Biochemical analysis, quantitative analysis of the transplanted cells, and functional analysis of the transplanted hepatocytes were performed by repeated-measures ANOVA followed by the Holm–Sidak test. Datasets consisting of two groups were evaluated using Student's t-test. Survival probability was calculated using the Kaplan–Meier method, and it was analyzed using the log-rank test. Statistical significance was defined as p < 0.05.
Results
Liver Cell Transplantation Recovery in Acute Liver Failure Models
The transplanted cells were identified as GFP-positive cell clusters by a transilluminator (Fig. 1A). The probability of survival was significantly higher in the transplantation group than in the control group (69.2 vs. 7.7%, p = 0.0004), or the homogenate group (69.2 vs. 23.1%, p = 0.0086). However, the probability of survival was not significantly higher in the homogenate group than in the control group (Fig. 1B). These results indicated that survival required liver cell transplantation, not the liver cell homogenate. All animals that survived longer than 7 days survived for the duration of the experiments (>100 days).

Green fluorescent protein-positive liver cell transplantation into the mesentery after massive hepatectomy. (A) Under an ultraviolet transilluminator, the transplanted green fluorescent protein-positive cells (encircled by red arrow heads) were confirmed in the mesentery. (B) Kaplan–Meier curves for survival in the control (dotted line), homogenate (broken line), and liver cell transplantation (Tx) (solid line) groups after 90% hepatectomy (n = 13/group). (C) The serum total bilirubin level (n = 3/group) and (D) serum ammonia level (n = 4/group) after 90% hepatectomy with [transplantation (Tx), solid line] and without (control, dotted line) liver cell transplantation. The p values represent the result of repeated-measures analysis of variance (ANOVA). **p < 0.01, ***p < 0.001, post hoc tests comparing the Tx and control groups at each time point (C, D).
Serum total bilirubin levels increased in the control group after 90% hepatectomy, which was significantly different from the relative lack of increase in the transplantation group (p < 0.005) (Fig. 1C). Serum ammonia levels were also lower in the transplantation group than in the control group (p < 0.005) after 90% hepatectomy (Fig. 1D). These results indicated that liver cell transplantation improved the posthepatectomy survival rate and prevented acute liver failure.
Assessment of the Transplanted Cells
The transplanted cells were successfully engrafted into the recipients' mesenteries, which was confirmed by histological and immunohistological analyses at 8, 24, and 72 h posttransplantation. The living cells with nuclei gradually decreased over time. To monitor donor-derived cells, the mesentery tissue sections were analyzed for the presence of GFP and albumin (Fig. 2A–I). The morphological characteristics of the GFP-positive cells were a cuboidal shape, which was compatible with hepatocytes. GFP-positive cells were immunohistochemically positive for albumin and were palely stained for PAS. The GFP and albumin expression deteriorated in a time-dependent manner until 72 h after transplantation. Furthermore, the number of PAS-positive cells and the strength of PAS staining gradually decreased over time.

Viability of the transplanted liver cells after intramesenteric transplantation. Histological (A–C), immunohistological (D–F), and periodic acid-Schiff (PAS) analyses (G–I) of the transplanted cells in the mesentery. Specimens were obtained at 8 (A, D, G), 24 (B, E, H), and 72 h (C, F, I) posttransplantation. Green fluorescence represents green fluorescent protein (GFP), and red fluorescence indicates rat albumin. Blue fluorescence indicates DAPI staining. Scale bars: 50 μm. (J–L) GFP expression at the transplanted site of live rats. (J, K) Viable transplanted cells, represented by the GFP signal intensity, are detected by the in vivo imaging system as high (red) to low (blue). (L) Signal counts of GFP after transplantation (n = 3). (M–P) Quantitative polymerase chain reaction (qPCR) analysis for hepatocyte-specific messenger RNA (mRNA) of transplanted cells in the mesentery. The mRNA of the transplanted cells in the mesentery at 8, 24, and 72 h after transplantation (n = 3/each time point) compared to that before transplantation (pre), using a set at a value of 1 (n = 3). (Q) Functional analysis of transplanted hepatocytes in the liver cell transplantation model. Serum albumin levels of the Nagase analbuminemic rats in the control (control, dotted line) and transplantation (Tx, solid line) groups after liver cell transplantation (Tx: n = 3, control: n = 4). The p values in the figure represent the results of the repeated-measures ANOVA. *p < 0.05, **p < 0.01, ****p < 0.0001, post hoc tests comparing the Tx and control groups at each time point. DAPI, 4′,6-diamidino-2-phenylindole; H&E, hematoxylin and eosin.
To assess any time-dependent changes in the transplanted cells across the entire transplanted site, we measured the GFP expression across the entire transplantation site of live rats with IVIS (Fig. 2J–L). Within 8 h after transplantation, the GFP expression decreased to 10% of the initially measured activity. During the next 8 h, the expression reduced to 10%. By 24 h after transplantation, the GFP expression was indistinguishable from the intestinal autofluorescence around the transplanted sites. Reverse transcription PCR showed that the mRNA levels of CYP2E1 and CYP3A1 at 24 and 72 h after transplantation were significantly lower than those before transplantation (Fig. 2M–P). The mRNA levels of albumin at 8, 24, and 72 h after transplantation were significantly lower than those before transplantation. Although there was no significant difference at 8, 24, and 72 h after transplantation, mRNA levels of G6Pase rapidly decreased over time. These results indicated that the hepatic function of transplanted liver cells had also rapidly deteriorated.
Serum Albumin Measurement of NA Rats
To evaluate the function of transplanted hepatocytes, we transplanted normal hepatocytes into the NA rats. Just after transplantation, the serum albumin level markedly increased in the transplanted NA rats and peaked at 16 h after transplantation (Fig. 2Q). Thus, the transplanted cells produced albumin in a time-dependent manner until 16 h after transplantation. However, the serum albumin levels of NA rats subsequently decreased (p < 0.05). This result also indicated the rapid deterioration of viable transplanted liver cells.
Remnant Liver Evaluation
H&E staining showed severe damage to the remnant liver structure. In the control group with 90% hepatectomy, diffuse hepatocyte vacuolar degeneration was observed in the remnant liver at 24 h after hepatectomy, whereas the hepatocytes and liver structure of the remnant liver were maintained in the transplantation group (Fig. 3A and B). The ATP levels of the remnant liver tissues were significantly lower in the control group than in the transplanted group (p < 0.05) (Fig. 3C).

Damage to the remnant liver tissue after massive hepatectomy. (A, B) H&E staining of the remnant liver sections (n = 5/group). (C) Adenosine-5′-triphosphate (ATP) assay (n = 3/group) of the remnant liver at 24 h after 90% hepatectomy with (Tx) and without (control) liver cell transplantation. (D, E) Representative photographs of the rat livers showing the proliferating cell nuclear antigen (PCNA) expression and (F) the PCNA index (n = 3/group) at 8 h after 90% hepatectomy with (Tx) and without (control) liver cell transplantation. (G) Experimental protocol for the removal study of transplanted cells. At 8 h after transplantation, the transplanted cells were removed. At 24 h after transplantation, the serum ammonia level and remnant liver tissues were evaluated with (removed) and without (Tx) removing the transplanted liver cells. (H) In vivo image showing GFP of the transplanted liver cells before and after removing the transplanted cells. It was confirmed that the transplanted cells were removed. (I) Serum ammonia measurement (remove: n = 3, dotted line; Tx: n = 4, solid line). (J, K) H&E staining of the remnant liver tissues in the remove and Tx groups. *p < 0.05. Scale bars: 50 μm (A, B, D, E, J, and K).
The number of PCNA-positive cells in the remnant liver 8 h after 90% hepatectomy was much higher in the transplanted group (p < 0.05) (Fig. 3D–F) than in the control group.
Removal Study of the Transplanted Cells
The serum ammonia level and histological findings of remnant livers were compared with and without removal of the transplanted cells (Fig. 3G) identified on IVIS (Fig. 3H). There was no significant difference in the serum ammonia level between groups, indicating that the remnant livers are responsible for maintaining low ammonia levels >8 h after transplantation (p = 0.5659) (Fig. 3I). In addition, histological findings showed a protective effect of the remnant liver, which occurred due to cell transplantation and was maintained despite the removal of transplanted cells 8 h after transplantation (Fig. 3J and K). These results indicated that the existence of transplanted cells within 8 h after transplantation was enough to protect the remnant liver from severe damage due to massive hepatectomy. Furthermore, these findings suggested that intramesenteric transplanted liver cells play essential roles during the early period after massive hepatectomy by protecting the remnant liver from liver damage via a remote effect such as secreting cytokines.
Detection of the Paracrine Factor for Protecting the Remnant Liver
To investigate factors working to protect the remnant liver, we performed a comprehensive analysis of cytokines and growth factors in the portal vein that flowed from the transplanted site to the remnant liver. No specific factor was significantly elevated in the transplantation group (Fig. 4).
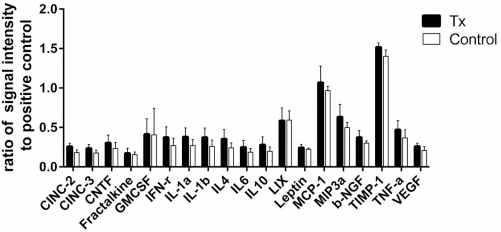
Proteome levels of the cytokines, growth factors, and other proteins in the serum of portal vein blood. Data at 4 h after transplantation in the transplantation (Tx) and control groups (n = 3/group). CINC, cytokine-induced neutrophil chemoattractant; CNTF, ciliary neurotrophic factor; GMCSF, granulocyte-macrophage colony-stimulating factor; IFN, interferon; IL, interleukin; LIX, lipopolysaccharide-induced CXC chemokine; MCP, monocyte chemoattractant protein; MIP, macrophage inflammatory protein; NFG, nerve growth factor; TIMP, tissue inhibitor of metalloproteinases; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.
The Role of Hepatocytes and NPCs in Liver Cell Transplantation
To investigate whether hepatocytes or NPCs played a more significant role in protecting remnant livers and promoting hepatic regeneration, liver cells harvested from the donor liver tissue were divided into hepatocytes and NPCs, and each was transplanted.
H&E staining showed that severe liver damage occurred in the remnant liver of the NPC transplanted group, which was the same as in the control group (nontransplanted group). However, the remnant liver of the HEP group was protected from severe liver damage due to massive hepatectomy (n = 3/group) (Fig. 5A and B). ATP levels of the remnant liver tissue 24 h after 90% hepatectomy were significantly higher in the HEP group than in the NPC group (p < 0.05) (Fig. 5C).

Intramesenteric cell transplantation of isolated hepatocytes and nonparenchymal cells (NPCs) for massive hepatectomy. (A, B) H&E staining of the remnant liver sections at 24 h after 90% hepatectomy in the hepatocyte (HEP) (A) and NPC (B) groups. Scale bars: 50 μm (n = 3/group). (C) The ATP measurement (n = 3/group). *p < 0.05. (D) Kaplan–Meier curves for 7-day survival in the HEP group (dashed line) and the NPC group (dotted line) (n = 8). (E) Green fluorescent protein expressions of the transplanted site in the liver cell group (n = 4) and the HEP group (n = 3). Tx, transplantation.
To assess the influence of transplanted hepatocytes and NPCs on improving survival, the Kaplan–Meier curves for 7-day survival after 90% hepatectomy were calculated and compared between the HEP and NPC groups (Fig. 5D). The HEP group had a higher probability of survival than the NPC group, but the difference was not significant (62.5 vs. 25.0%, p = 0.2811).
In addition, we compared the GFP expression at the transplantation sites between the liver cell and HEP groups to determine whether the NPCs supported transplanted hepatocytes. Although GFP expression decreased in both groups, the rate of decline of GFP expression was slightly lower in the total liver cell group than in the HEP group, with no significant difference (p = 0.1128) (Fig. 5E).
Discussion
In the present study, we demonstrated that liver cell transplantation into the mesentery recovered and prevented acute liver failure after massive hepatectomy.
Lee et al. reported that the small intestinal mesentery was a better transplantation site than the subcutaneous space in terms of engraftment of the donor hepatocytes with delivery devices 31 . However, in the present study, histological findings of the transplantation site, quantitative PCR assays of the transplanted cells, and IVIS study findings showed the rapid deterioration of viable transplanted liver cells, which would be caused by the inappropriate oxygen delivery of minimal vascularization32,33, or the attack of native granulocytes 34 , or the transplantation in the present study without the delivery devices as used by Lee et al. 31 . Furthermore, albumin is a protein with an extremely long half-life in the body (about 3 weeks). As long as the hepatocytes survive, the serum albumin levels do not decrease. The decreasing serum albumin levels of the NA rats indicated the rapid deterioration of viable transplanted liver cells.
Nevertheless, the overall mortality due to acute liver failure significantly improved in the transplantation group. Thus, it is reasonable to suppose that remnant liver function was maintained and improved by transplantation. H&E staining of the remnant liver showed swelling and vacuolar degeneration of the hepatocytes in the control group, which was consistent with the results of previous studies5,35, whereas in the transplantation group, there were almost normal findings. In addition, the ATP levels in the remnant liver tissue were significantly improved in the transplantation group. Energy supply is critical for cell survival and proliferation. In particular, ATP is required to supply energy for maintaining cell function36–39. Reportedly, ATP decreases markedly after partial hepatectomy37,38,40,41, and decreased ATP production in small-for-size liver grafts has been associated with increased hepatocyte injury and a higher mortality38,42,43.
Acute liver failure after massive hepatectomy is also characterized by suppressed hepatocyte proliferation44,45. We observed a significantly higher PCNA index at 8 h after 90% hepatectomy in the transplantation group than in the control. These findings suggested that the liver cells transplanted in the mesentery prevent the remnant liver from liver damage due to massive hepatectomy.
Furthermore, the findings of the removal study on the transplanted cells indicated that the existence of liver cells in the mesentery until 8 h after transplantation is sufficient for protecting the remnant liver. Therefore, intramesenteric transplanted liver cells play essential roles in the early period after massive hepatectomy by indirectly supporting the remnant liver.
The liver cells transplanted as donor graft in the present study contained hepatocytes and NPCs. Hepatocytes and NPCs secrete multiple cytokines, growth factors, and other molecules such as hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), transforming growth factor-β1 (TGF-β1), interleukin-6 (IL-6), and some chemokines46,47, and they promote liver regeneration in the injured liver48–50. We hypothesized that any of the several cytokines produced by transplanted hepatocytes and the NPCs either protected the remnant livers or promoted remnant liver regeneration. In addition, it is supposed that these paracrine factors secreted by transplanted cells were absorbed through mesenteric microvessels into the portal vein 17 . Concerning the portal blood flow draining into the liver, we speculated that the mesentery was a better site than the other extrahepatic sites in terms of the effectiveness of the paracrine effect of donor liver cells. Although we comprehensively analyzed cytokines in portal venous blood including HGF and TGF-β1 (data not shown), no specific factors significantly elevated in the transplantation group could be detected. However, the same protective effects represented by histological findings and an elevation in the ATP levels were observed in the hepatocyte transplantation group, as in the liver cell transplantation group previously described. Nevertheless, damage to the remnant liver was not prevented by isolated NPC transplantation in the same manner as in the nontransplanted groups. Therefore, the protective effect on the remnant liver was mainly attributable to hepatocytes among the liver cells transplanted in the mesentery. Although it would be assumed that several nonsignificant factors have multiplier effects on the protection of the remnant liver, further experiments that investigate factors released by transplanted hepatocytes are necessary. Additionally, the survival rate was slightly higher in the homogenate group than in the control group, without a significant difference between these two groups. This result indicated that the liver cell homogenate, which may contain many factors, including albumin, glucose cytokines, growth factors, etc., could not prevent liver failure in the liver cell transplantation group. Although the reason of this result has not yet been determined, several possibilities would be raised. For example, those liver protective factors would be chemically degraded in the process of preparing cell homogenate.
Some reports have stated that the liver NPCs support hepatocyte survival and maintain hepatic function51–53. We investigated whether NPCs support hepatocytes at transplanted sites by monitoring GFP fluorescence, which represents viable transplanted cells (Fig. 5E). Improvement in the GFP deterioration in the liver cell group, which contained hepatocytes and NPCs, compared to the hepatocyte group indicates that NPCs tend to support engraftment of the transplanted cells.
In conclusion, the present study described the effect of the intramesenteric transplantation of liver cells for acute liver failure after massive hepatectomy, the protective effects of transplanting liver cells, and improved survival. Namely, although transplanted liver cells hardly worked as functional liver cells in the very early period after transplantation, they protected the remnant liver from severe damage after massive hepatectomy, which would have a major effect on survival. Although these findings may provide the opportunity to utilize liver cells harvested from the resected liver tissue to prevent liver failure after massive hepatectomy, several issues need to be resolved. For example, the contamination of cancer cells must be prevented. In addition, further experiments should be planned for diseased recipients with chronic liver failure, metabolic liver failure, and liver cirrhosis to utilize this technique in the clinical setting in the foreseeable future.
Footnotes
Acknowledgments
We thank Dr. Takahiro Ochiya for providing the green fluorescent protein rats. This work was supported by grants from the Scientific Research Fund of Japan Science and Technology Agency (Research Project No. 24591999). The authors declare no conflicts of interest.
