Abstract
Bone marrow mononuclear cells (BMMC) effects have been investigated in small series of nonischemic dilated cardiomyopathy (NIDC). Left ventricular myocardial contractility improvements occur, but doubt remains about their mechanism of action. We compared contractility changes in areas treated (free wall) and nontreated (septal wall) with BMMC, in selected patients who have showed significant ventricular improvement after free wall-only intramyocardial stem cells injection. From 15 patients with functional class III/IV (NYHA) and LVEF inferior to 35%, who received 9.6 ± 2.6 × 107 BMMC divided into 10 points over the left ventricular free wall, 7 (46.7%) showed LVEF relative improvement greater than 15%. Those patients were selected for further contractility study. BMMC were collected from iliac bone and isolated with Ficoll-Hypaque. Magnetic resonance imaging was used to measure the systolic thickening of the septal (nontreated) and free wall (treated) before injection and 3 months postoperatively. Mean systolic septal wall thickening increased from 0.46 to 1.23 mm (an absolute 0.77 ± 1.3 mm and relative 167.4% increase) and in the free wall from 1.13 to 1.87 mm (an absolute 0.74 ± 1.5 mm and relative increase of 65.5%). There was no difference in the rate of absolute or relative systolic thickening between the two walls (p = 0.866 and 1.0, respectively), when cells were injected only in the left ventricular free wall. BMMC transplantation in nonischemic dilated cardiomyopathy can improve ventricular function by an overall effect, even in areas that are not directly injected. This finding favors the existence of a diffuse mechanism of action, rather than a local effect, and should be reminded when the pathophysiology of stem cells is considered.
Introduction
Eight years after the first successful applications of CD34+ cells in animal (18,29) and pilot clinical studies with bone marrow mononuclear cells (BMMC) (23,25), key issues in cell therapy for heart disease have not yet been fully understood. First, what mechanisms are responsible for functional gain observed in the treated areas of the myocardium? (14,16). Second, what is the clinical relevance of functional gain observed in experimental studies? There also remain issues on which type of disease would benefit, what kinds of cell (and dose) are most appropriate, and what are the better forms of delivery for each clinical scenario (10,13,21).
Direct intramyocardial application seems to be more effective than systemic or percutaneous intracoronary injection, especially in diseases or models of chronic disease, when there is a microenvironment conducive to cell migration (30). In dilated cardiomyopathy of nonischemic etiology (NIDC) this question is even more relevant, because the application in a restricted area of the myocardium could not improve global left ventricular function, even if effective.
Cardiac magnetic resonance imaging (CMRI) is the most accurate method to estimate left ventricular function, both segmental and global (3). It also allows accurate measurement of chamber diameters and wall thickness (22). Measurement of the systolic thickening of a myocardial segment can quantify regional contractility (8,26).
Patients from two previous studies, a finished pilot trial (7) and a randomized clinical trial under way, who received transplantation of autologous BMMC were assessed for segmental and global left ventricular function by CMRI. The objective was to determine whether, in a selected group of patients who showed improvement in cardiac function, this effect was only observed in the treated myocardial wall (i.e., the left ventricular free wall), or if it spread throughout the left ventricle, by comparing the systolic increase of thickness of the septal and the free ventricular wall regions by CMRI.
Materials and Methods
Patient Selection
In this cross-sectional study, patients were selected among those participating in a previous pilot study and in an ongoing randomized controlled clinical trial in which they underwent autologous transplantation of BMMC by direct transthoracic intramyocardial injection in the ventricular wall. Patients showing improvement on left ventricular ejection fraction (LVEF) greater than 15% in the CMRI examination performed 3 months after the procedure were selected. In this selected group, preoperative LVEF was a mean ± SD of 19.5 ± 12.3%, median 16.4%, and had increased 3 months postoperatively to 28.4 ± 15.2%, median 26.0%, p = 0.018.
Of a total of 15 patients, 7 (46.7%) were included according to these criteria. All patients had the disease diagnosed at least 1 year earlier, and were in NYHA functional class III/IV despite full pharmacological treatment, had previous LVEF below 35, and were aged less than 65 years. Patients with previous history of ventricular arrhythmia or significant comorbidities were excluded.
The protocols were approved by the local and the national ethics committees and have received grants from FAPERGS (State of Rio Grande do Sul Research Foundation) and CNPq (National Council of Technological and Scientific Development). All patients provided written informed consent.
BMMC Isolation and Transplantation
Approximately 4 h before the operation and with the patient under sedation, 80 ml of bone marrow was aspirated from the anterior iliac crest in medium containing anticoagulant preservative. BMMCs were isolated by density centrifugation over Ficoll-Hypaque-1077 (Sigma Diagnostics, St. Louis, MO), and washed in a heparinized saline solution containing 5% autologous serum. The cells were counted in a Neubauer chamber. Viability, determined by trypan blue exclusion test, was always higher than 90%. The cells were suspended in 5 ml saline solution with 5% autologous serum for intramyocardial injection. A small fraction was utilized for sterility and viability tests.
Surgical Technique
Surgical approach was through a left minithoracotomy, consisting of an incision of approximately 5 cm in length, anterolaterally in the fifth left intercostal space, in order to expose the pericardium. A T-shaped pericardial incision was made for accessing the free wall of the left ventricle. The position of the coronary arteries was defined and the cell suspension was directly injected, through a 21F butterfly needle that was positioned about 5 mm intramyocardially and connected to an extension managed by the surgical assistant. Twenty 0.25-ml injections were made in the myocardium, in the anterior, lateral, posterior, and apical faces of the left ventricle. After reviewing the hemostasis, the pericardium was closed, the thoracic cavity was drained, and the chest wall was closed.
Cardiac Magnetic Resonance Imaging
Left ventriculograms by nuclear magnetic resonance were obtained in patients before and 3 months after treatment. The following criteria were evaluated: global contractility of the left ventricle, and systolic thickening of the ventricular wall, divided by segments, as described below.
Analysis of global function was carried out quantitatively by the Simpson's method; ventriculography was used to measure end diastolic volume, end systolic volume, and ejection fraction of left ventricle.
Ventricular wall thickness was measured in both the septal and free walls of the left ventricle, in systole and diastole, in the following manner. Each wall had its thickness measured in five points: anterobasal, posterobasal, anteromedial, posteromedial, and apical. The arithmetic mean of these five measurements was considered as the mean thickness of the septal or the free wall, respectively. The difference between mean diastolic and mean systolic thickness represented the systolic thickening and this parameter was taken as an expression of the increase in contractility.
Statistical Analysis
Data were expressed as the mean ± SD and median for continuous variables and percentages for dichotomous variables. Wilcoxon signed ranks tests were used to compare medians of wall thickening between the free and septal walls. Statistical significance was defined at p < 0.05. Statistical analyses were performed using SPSS version 15.0.
Results
Patients and Cells
The sample consisted of seven patients, with mean age (±SD) of 43.9±13.6 years; five (71.4%) were males. All were in functional class III, despite full medical treatment. BMMC preparations contained a mean total of 9.6 ± 2.6 × 107 cells. Table 1 describes baseline characteristics of the patients under study.
Characteristics of Included Patients (n = 7)

Analysis of Segmental Ventricular Function by RNMC
Preoperative mean septal wall thickness was 10.5 ± 2.74 mm in diastole and 10.96 ± 3.56 mm in systole, representing an absolute systolic thickening of 0.46 mm over the mean diastolic thickness. Postoperative mean septal wall thickness was 9.32 ± 2.19 mm in diastole and 10.55 ± 2.62 mm in systole, representing an absolute systolic thickening of 1.23 mm over the mean diastolic thickness. Systolic wall thickening increased from 0.46 to 1.23 mm (an absolute 0.77± 1.3 mm and relative 167.4% increase) (Table 2).
Analysis of Segmental Wall Thickening by RNMC
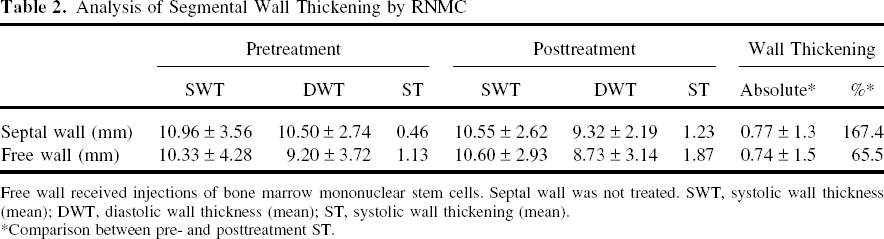
Free wall received injections of bone marrow mononuclear stem cells. Septal wall was not treated. SWT, systolic wall thickness (mean); DWT, diastolic wall thickness (mean); ST, systolic wall thickening (mean).
Comparison between pre- and posttreatment ST.
In the free wall, preoperative mean free wall thickness was 9.2 ± 3.72 mm in diastole and 10.33 ± 3.72 mm in systole, representing an absolute systolic thickening of 1.13 mm over the mean diastolic thickness. Postoperative mean septal wall thickness was 8.73 ± 3.14 mm in diastole and 10.6 ± 2.93 mm in systole, representing an absolute systolic thickening of 1.87 mm over the mean diastolic thickness. Systolic wall thickening increased from 1.13 to 1.87 mm (an absolute 0.74± 1.5 mm and relative 65.5% increase) (Table 2). There was no difference in systolic thickening between both walls (Table 3, Fig. 1), which can be interpreted as an overall, diffuse effect occurring after cells injection.
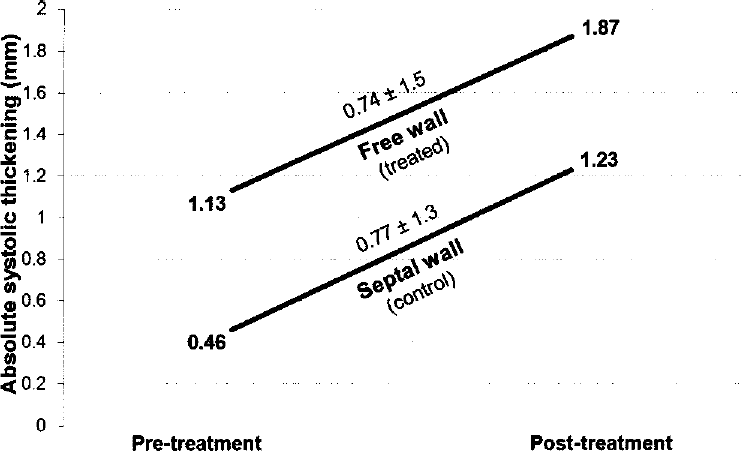
Comparison of the free (treated) and septal (control area) absolute wall thickness mean increase: pre- and posttreatment.
Statistical Analysis of Segmental Wall Thickening by RNMC

Values are means and medians of the systolic thickening increases. There was no difference in systolic thickening between both walls. ST, systolic wall thickening.
Discussion
Studies in small animals using experimental models of NIDC demonstrated functional gain by implantation of mesenchymal stem cells (MSC) in the basal, medial, and apical left ventricular wall (15) and with implantation of BMMC in the left ventricular free wall (6). In the study by Nagaya et al. (15), some of the MSC expressed connexin-43, a gap junction protein, at contact points between native cardiac myocytes and MSC, as well as cardiac markers such as troponin T and desmin. The analysis of isolated cardiac cells showed that 8.1% of MSC were double-positive for PKH26 and troponin T, suggesting that they had transdifferentiated in cardiomyocytes. The authors also demonstrated that the MSC are able to produce angiogenic cytokines and attenuate the formation of myocardial fibrosis, suggesting that MSC improve myocardial function through a mechanism of both cell transdiferentation and paracrine regulation of cytokine production. Li et al. (9), comparing the functional improvement achieved by cell therapy and cytokine therapy in both ischemic and nonischemic heart failure experimental models, had observed that both cell therapy and cytokine therapy were alternative treatments for ischemic heart failure, but cell therapy was more effective for the treatment of nonischemic heart failure than cytokine therapy, although cytokine therapy inhibited the fibrosis and apoptosis of the cardiomyocytes.
The first clinical report of transient improvement of left ventricular function after stem cell transplantation in nonischemic heart disease was a case report by Ogawa et al., in 2004 (17).
Clinical studies with bone marrow cells in nonischemic heart disease are still limited. A randomized clinical trial with NIDC patients showed that intracoronary injection of BMMC was associated with an increase of 5.4% in EF and a non significant decrease in final systolic volume (20). Endomyocardial biopsy performed 3 months after the procedure showed no significant difference in the number of capillaries or myocytes.
In acute myocardial infarction, their use is associated with improvement of regional contractility (1) in the same way as when applied by intramyocardial injection in chronic ischemia (19), but not by intracoronary route. On the other hand, Moreno-Gonzales et al. (12), in a experimental study, had shown a global effect using cardiomyocytes transplanted into adult rat hearts after infarction, observing that cardiomyocytes grafts isolated in the infarct zone increased Ca2+ sensitivity of remote uninjured myocardium.
BMMC were used in a trial of patients with dilated cardiomyopathy due to Chagas disease (27); the authors found a short-term tendency of improvement in myocardial function and relief of ventricular arrhythmias in treated patients. In NIDC, case reports and small clinical trials bring evidences of a persistent inotropic action when cells are injected intramyocardially (2,5). The effect, however, seems smaller when the intracoronary route is employed (11). Although a 6-month improvement has been achieved with this route of administration (20), others have failed to demonstrate a similar effect (28).
The intracoronary route could more evenly distribute cells over the entire myocardium, which would be appropriate in a diffuse disease, as nonischemic dilated cardiomyopathy. It has been demonstrated, however, that unlike the ischemic disease where homing factors are increased as expressed by upregulated circulating cytokines, “homing” is impaired in dilated cardiomyopathy, cytokine levels are reduced, and there are more CD34+ circulating cells (24). An alternative has been tried, injecting granulocyte colony-stimulating factor associated to intracoronary cell injection (4).
In the intent to obviate the low homing effect, the direct intramyocardial route is used, either through a percutaneous transendocardial way or, as in our series, transepicardially through a minithoracothomy. Directly delivered intramyocardial stem cells could have facilitated seeding, instead of finding their way to homing and differentiation, leading to a local and improved action.
In this study, we selected NIDC patients who had been injected with BMMC only in the free wall of the left ventricle and assessed left ventricular myocardial function, comparing differences in segments directly treated (free wall) with segments that were not directly treated (septal wall). MRI is an attractive method because it allows measurement of LVEF by the Simpson's method and segmental contractility by mapping the endocardial border both in systole and in diastole. CMRI obviates the need for any geometric assumptions regarding the shape of the left ventricle for determination of the ejection fraction value, as is necessary with 2D echocardiography.
Our main findings were: 1) the use of BMMC in patients with NIDC by direct injection in the left ventricular free wall can improve global left ventricular function; 2) this increased improvement in ventricular function was associated with an improvement in regional function of both treated and untreated areas of the left ventricle; 3) there was no difference between the regional myocardial function of treated and untreated areas.
The exact mechanisms responsible for these findings are unclear. As opposed to the experimental environment, histological examinations are not feasible in living patients, except by endomyocardial biopsy, which would increase invasiveness and risks. Very accurate means to assess cardiac function are available, however, in the clinical setting and we believe that once clinical trials with bone marrow cells for treatment of heart disease are justified, clinical and experimental studies should be complementary. Findings observed in our study could have complementary explanations. 1) As set out experimentally, the use of progenitor cells from bone marrow is associated with a beneficial effect on ventricular function through a mechanism possibly involving cell transdifferentiation into useful cell types for that scenario in small scale, and a paracrine effect mediated by cytokines in the adjacent myocardium. It is possible that this effect occurs in treated areas of the myocardium and extends to adjacent regions. The form of application we used, although aimed to achieve the greatest possible area, was limited to around 60% of all the left ventricle. 2) The improvement of regional function could lead to a positive ventricular remodeling and thereby mitigate ventricular dysfunction in areas not directly addressed. But unlike in cardiomyopathy of ischemic origin, where only ischemic areas are affected, in NIDC ventricular function is affected as a whole, so we believe that this should not be the predominant mechanism.
In conclusion, there is evidence, expressed by improved contractility evaluated by myocardial wall systolic thickening with nuclear magnetic resonance imaging, that BMMC intramyocardial transplantation in nonischemic dilated cardiomyopathy can improve ventricular function by an overall effect, even in areas that are not directly injected with them. This finding favors the understanding of a more diffuse effect than expected by local cell transdifferentiation of stem cells into cardiomyocytes and should be considered when the mechanisms of action of stem cells over the myocardium is considered.
Footnotes
Acknowledgments
The authors declare that they had full control of the design of the study, methods used, outcome parameters, analysis of data, and production of the written report. The study was funded by grants from government agencies, as FAPERGS, from the State of Rio Grande do Sul and CNPq, from the Federal Government of Brazil.
