Abstract
BACKGROUND:
Spinal cord injury (SCI) leads to spinal nerve fiber tract damage resulting in functional impairments. Volumetric muscle loss (VML), a skeletal muscle volume abnormal reduction, is represented by atrophy below the injury level. The strategies for VML management included personalized approaches, and no definite indications are available.
OBJECTIVE:
To identify the rehabilitation effects of VML in subjects with SCI (humans and animals).
METHODS:
PubMed, Scopus, and Web of Science databases were systematically searched to identify longitudinal observational studies with individuals affected by traumatic SCI as participants; rehabilitation treatment as intervention; no control, sham treatment, and electrical stimulation programs as control; total lean body and lower limb lean mass, cross-sectional area, functional gait recovery, muscle thickness, and ultrasound intensity, as outcome.
RESULTS:
Twenty-four longitudinal observational studies were included, evaluating different rehabilitation approaches’ effects on the VML reduction in subjects affected by SCI. The data showed that electrical stimulation and treadmill training are effective in reducing the VML in this population.
CONCLUSION:
This systematic review underlines the need to treat subjects with traumatic SCI (humans and animals) with different rehabilitation approaches to prevent VML in the subacute and chronic phases. Further clinical observations are needed to overcome the bias and to define the intervention’s timing and modalities.
Keywords
Introduction
Spinal cord injury (SCI) is a devastating condition that leads to spinal nerve fiber tracts damage resulting in motor, sensory, and autonomous functions impairments below the injury level (Rupp, 2020), with significant personal and societal loss (Hachem et al.., 2017). Worldwide, the incidence of SCI is 0.9 (95% UI, 0.7–1.2) million cases for both sexes, with an age-standardized incidence rate of 12 (95% UI, 9–15) per 100,000 (Ding et al.., 2022). SCI may be due to traumatic (e.g., contusion, compression, laceration) or non-traumatic events (e.g., degenerative cervical myelopathy, cancer, infection, intervertebral disc disease, vertebral injury, and spinal cord vascular disease) (Ge et al.., 2018), and the cervical spine is the most involved tract (Singh et al.., 2014). Particularly, the traumatic spinal cord injury (TSCI)’s documented incidence ranges between 12.1 and 57.8 cases per million inhabitants in high-income countries and between 12.7 and 29.7 in low-income countries (Barbiellini Amidei et al.., 2022). Even if no significant changes in the incidence of SCI were observed over the last 25 years, it is necessary to underline relevant differences concerning its demographic characteristics: the average age of onset increased from 29 to 42 years, reflecting a higher number of people who get this injury as a result of accidental falls, which, more frequently, resulting in incomplete tetraplegia (Algahtany et al.., 2021; Bárbara-Bataller et al.., 2018; Devivo, 2012).
The International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI), endorsed by the American Spinal Injury Association (ASIA) and the International Spinal Cord Society (ISCoS), is the most widely used classification (ASIA and ISCoS International Standards Committee, 2019). This classification allows to examine of sensory and motor functions and define the lesion completeness or incompleteness according to the ASIA Impairment Scale (AIS), with a huge prognostic role if applied within the first 72 hours after the traumatic event (Kirshblum, 2020; Kirshblum, 2020). Furthermore, van Middendorp et al.. and the European Multicenter Study about Spinal Cord Injury (EMSCI), in a longitudinal cohort study, stated that sensitivity and muscle strength assessed on L3 and S1 within 2-weeks of injury might predict with 95% confidence the probability of independent walking one year after injury (van Middendorp et al.., 2011).
From a pathophysiological point of view, SCI is characterized by a primary and a second phase (McDonald & Sadowsky, 2002; Tator, 1995). Primary SCI results directly from the physical forces due to the traumatic event and is often the injury severity principal determinant (Ackery et al., 2004; Yip & Malaspina, 2012). The second phase is a delayed and progressive tissue lesion during which macrophages, microglia, T-cells, and neutrophils in the marrow stimulate the inflammatory cytokines release, such as tumor necrosis factor (TNF)α, interleukin (IL)-1α, IL-1β, and IL-6 (Nakamura et al., 2003). Moreover, the mitochondrial dysfunction and the excitatory amino acid receptors’ hyperactivation result in excitotoxicity with neuronal loss due to necrosis and apoptosis (H. Liu et al., 2015). These mechanical and inflammatory damages induce central and peripheral nervous structures significant alterations, resulting in motor, sensory, and autonomous functions impairments below the injury level (Invernizzi, 2020); furthermore, considering the functional, embryological, and biochemical linkages between nerves and muscles, this condition may lead to musculoskeletal sequelae both in the acute and chronic phases (Clark & Findlay, 2017).
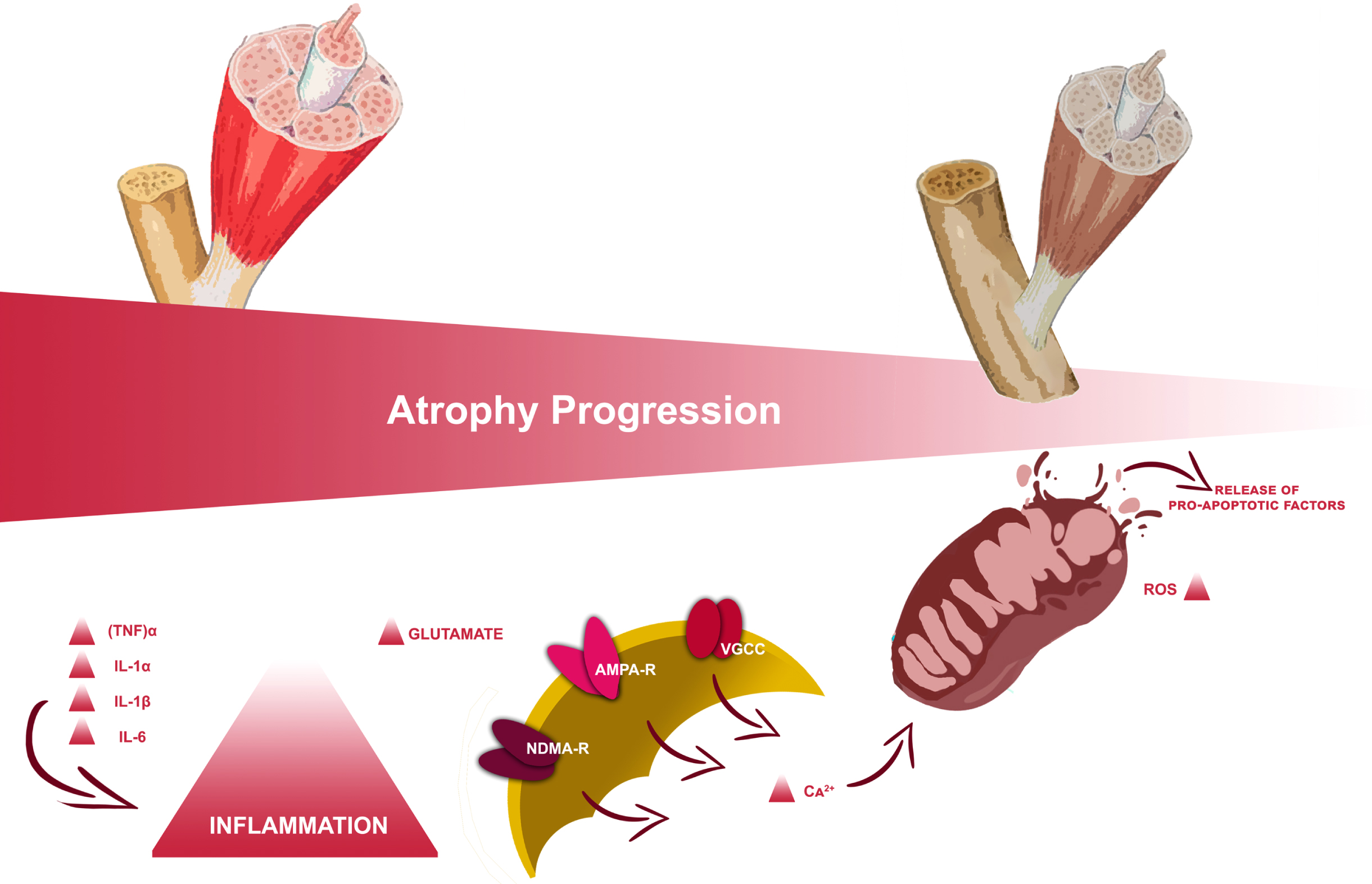
In particular, volumetric muscle loss (VML), usually defined as the abnormal decrease of skeletal muscle volume, is associated with variable degrees of functional deficits (Corona et al., 2018). Its most frequent clinical manifestation is represented by sublesional muscle atrophy according to the spine lesion level and severity (Carda et al., 2013; Invernizzi et al., 2015). During the acute phase following SCI, the pro-inflammatory state established results in a rapid series of muscle modifications with consequential muscle catabolism through autophagy supported by mitochondrial mechanisms (Gorgey & Dudley, 2007a; Invernizzi et al., 2021), as depicted in Fig. 1. This could result in a decrease of up to 45% in the thigh muscle cross-section approximately six weeks after the injury (Panisset et al., 2016); this process slowly stabilizes towards the chronic phase, a few months after the injury, with a huge impact on delayed recovery from trauma and functional outcome (Invernizzi, 2020).
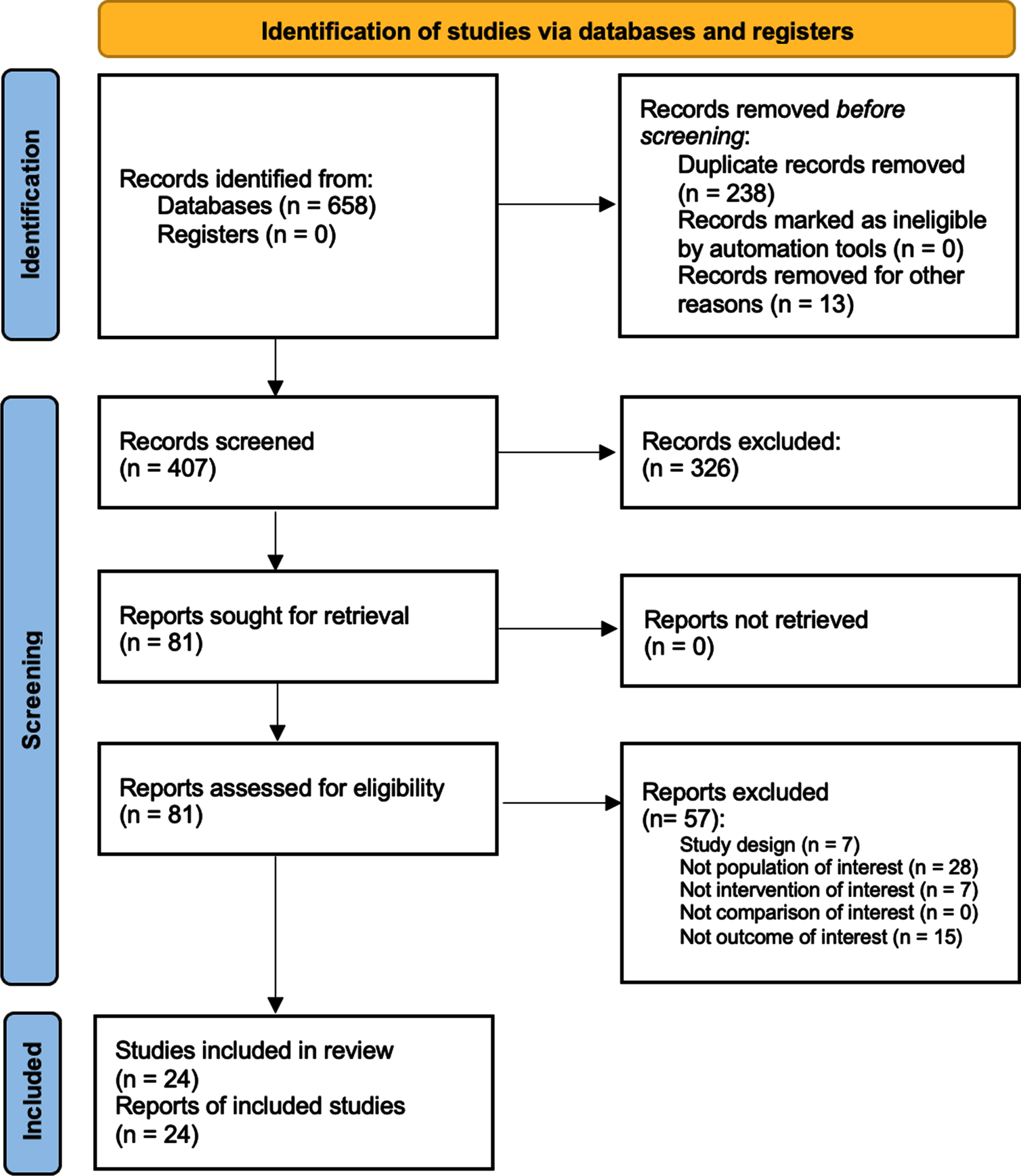
Study flowchart.
In this context, the early assessment and identification of this condition are essential to improve the overall outcome of patients affected by SCI who develop VML (Downing et al., 2021; Kojouharov et al., 2021). The main strategies proposed in the literature include personalized approaches consisting of dietary supplements (Farkas et al., 2019; Iolascon et al., 2017), drugs (Bauman & Cardozo, 2015; Schopp et al., 2006), and exercise (Beckwée et al., 2019; Durán et al., 2001; Maher et al., 2017; Sandrow-Feinberg & Houlé, 2015; Spangenburg et al., 2008).
A previous study by our group (Invernizzi et al., 2021) provided a portrayal of the state-of-the-art treatment of muscle modifications after SCI, focusing on the connections between the different types of approaches with the main pathophysiological mechanisms underpinning the VLM in subjects affected by SCI. However, the review did not provide clear data on the duration, timing, and effects of the different types of interventions.
Resistance training (RT) might increase muscle mass, however, in person with SCI it might be unfeasible, or need a partial weight lift. Nutritional supplementation is mandatory both to prevent VML and to avoid SCI systemic consequences (Farkas et al., 2019). In particular, essential and branched-chain amino acids, such as leucine (An et al., 2020), creatine, a non-protein nitrogenous tripeptide (Jacobs et al., 2002), vitamin D (Mailhot et al., 2018), irisin, and L-β-aminoisobutyric acid (L-BAIBA) (Colaianni et al., 2017) have been demonstrated to reduce muscle atrophy in population with chronic SCI. In this context, rehabilitation might play a key role in the clinical management of these subjects to improve muscle mass and function (Beckwée et al., 2019).
The inactivity-related VML, as in subjects suffering from SCI, might take advantage of physical exercise, isometric or cycle-ergometry functional electrical stimulation (FES), neuromuscular electrical stimulation (NMES), treadmill step-trained (Stp-T) or stand-trained (Std-T), body weight supported treadmill training (BWSTT), spinal magnetic stimulation, acrobatic exercises, testosterone replacement therapy, exoskeletal body-powered gait orthosis (EBPGO), and epidural spinal cord stimulation (eSCS) (Bustamante et al., 2016; Choi et al., 2020; Durán et al., 2001; Gao et al., 2017; Maher et al., 2017; Marquez-Chin & Popovic, 2020; McHugh et al., 2021; Mehrholz et al., 2017; Nightingale et al., 2018). The physical exercise efficacy seems to be related to its action on skeletal muscle tissue structure and neuroplasticity by the motor cortex activation (Fu et al., 2016; Jurkiewicz et al., 2007), and may result in resistance and cardiopulmonary conditioning improvement due to its neuroprotective and regenerative properties in people with SCI (Durán et al., 2001). FES and NMES are clinically applied as both assistive devices and therapeutic interventions to facilitate functioning restoration through short electrical pulses to generate muscle contractions, resulting in physical health benefits (Bochkezanian et al., 2018; Kapadia et al., 2014). Treadmill training, typically assisted with sensory stimulation, with or without body weight support, may recover some locomotor functions (R. V. Ung et al., 2010; R.-V. Ung et al., 2012). rTMS central nervous system stimulation occurs according to Faraday’s law of electromagnetic induction and seems to improve muscle strength and locomotor function in mice affected by SCI (Ahmed & Wieraszko, 2008). Testosterone Replacement Therapy (TRT) has been identified as an effective strategy for restoring lean body mass and decreasing fat mass in different clinical populations (Bassil et al., 2009), and may be a potential approach to manage body composition changes and improve metabolism in persons with SCI (Nightingale et al., 2018). In individuals with SCI, long-term sitting may result in secondary complications including pressure ulcers, muscle-tendon retractions, and osteoporosis (Karimi, 2011). Thus, the EBPGO training program was effective in preventing VML and in reducing body fat (Choi et al., 2020). eSCS has been identified as able to elicit motor function and to reestablish volitional movement in patients affected by SCI (Megía García et al., 2020) due to its action on some supraspinal control over motor function below the level of injury (Darrow et al., 2019).
To date, there are no clear indications in the literature concerning the type of intervention, timing, and duration to reduce muscle loss in subjects suffering from traumatic spinal cord injury outcomes. Indeed, studies on VML on SCI are often based on animal models, for ethical reasons. Including these papers in the analysis might aid to understand from a different point of view the more effective techniques to restore muscle mass.
Therefore, the present systematic review aims to investigate the role of rehabilitation in the reduction of volume muscle loss, to provide useful data for clinical practice concerning the timing, duration, and treatment parameters in subjects (humans and animals) with traumatic SCI.
Search strategy
PubMed, Scopus, and Web of Science databases were systematically searched for articles published from the inception until September 26th, 2022, according to each specific thesaurus, following the strategy described in Table 1. Furthermore, a manual search of the references of previous systematic reviews on a similar topic was conducted as well.
Search strategy
Search strategy
This systematic review was conducted according to the guidance of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The systematic review protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) (number: CRD42022313117).
All observational longitudinal studies were assessed for eligibility according to the following population, intervention, comparison, and outcomes (PICO) model:
P) Participants consisted of humans and animals with a diagnosis of traumatic SCI.
I) Intervention consisted of rehabilitative approaches aimed to reduce volumetric muscle loss (e.g., physical therapy, FES, NMES, body weight supported or not supported treadmill training, spinal magnetic stimulation, weight lift exercises, testosterone replacement therapy, EBPGO, and eSCS).
C) Comparison: no control group, healthy controls, SCI subjects that did not undergo any treatment, individually tailored exercise program, sham electrical stimulation.
O) Outcome measures consisted of total body lean body mass, lower limb lean body mass, cross-sectional area (CSA), fiber type composition, isometric muscle contraction strength, functional gait recovery, such as Basso-Beattie-Bresnahan locomotor rating scale or Irvine, Beattie and Bresnahan Forelimb recovery scale, and muscle thickness and echo intensity.
Two reviewers independently screened all potential articles for eligibility after duplication removal. Any disagreement has been resolved through discussion or, if necessary, by a consultation with a third reviewer.
Only prospective studies were included. Exclusion criteria were: 1) studies involving non traumatic SCI subjects; 2) studies without assessment of muscle atrophy by specific evaluation tools; 2) studies on children (age < 18 years); 3) studies on patients with previous psychological disorders; 4) studies written in a language different from English; 5) full-text unavailability (i.e. posters and conference abstracts).
Data extraction and data synthesis
Data from included studies were extracted by two reviewers independently through a customized data extraction on a Microsoft Excel sheet. In case of disagreement, a third reviewer was asked for a consensus. The following data were extracted: 1) First author; 2) Journal; 3) Publication year; 4) Nationality; 5) Study design; 4) Age of study participants; 5) Sex of study participants; 6) Body mass index (BMI) of study participants; 6) Anamnestic characteristics of study participants; 7) Comorbidities of study participants; 8) Time from the acute event; 9) Neurological level according to ASIA Scale; 10) Extent of SCI defined by ASIA Impairments Scale (AIS); 11) Rehabilitative intervention; 12) Muscle atrophy assessment at the baseline and at the end of treatment (T1); 13) Other clinical functional assessments.
The data have been synthesized by two authors from full-text articles included. Any disagreement between the two reviewers has been solved by collegial discussion among the authors. In case of disagreement, a third author was asked. Text and tables were used to provide a narrative synthesis and explanation of both study characteristics and findings.
Due to the heterogeneity of study participants and intervention assessed, a meta-analysis was not possible in accordance with the Cochrane Handbook for Systematic Reviews of Interventions (version 6.2; Higgins J.P. et al., 2021).
Quality assessment
We evaluated the risk of bias by Joanna Briggs Institute Critical Appraisal Checklist for Quasi-Experimental Studies (non-randomized experimental studies) checklist to estimate the included studies’ methodological quality. Each article was evaluated by two authors separately; a third author was involved to solve any disagreements. The JBI-QES tool consists of nine domains that allow for finding any study bias. Options for each judgment are: 1) low risk of bias, 2) moderate risk of bias/some concerns, 3) serious risk of bias, 4) critical risk of bias, and 5) no information. Domain-level reports providing the basis for an overall risk of-bias judgment.
Results
Study characteristics
Altogether, 658 studies were identified at the end of the database search. After duplication removal, 407 were considered suitable for the title and abstract screening. As a result, 328 were excluded and 81 articles were screened in full text. Lastly, 24 studies (Adams et al., 2011; Ahmed & Wieraszko, 2008; Baldi et al., 1998; Battistuzzo et al., 2017; Choi et al., 2020; Crameri et al., 2002; de Freitas et al., 2018; Demchak et al., 2005; Ditterline et al., 2020; Dolbow & Credeur, 2018; Everaert et al., 2021; Galea et al., 2017; L. Giangregorio et al., 2012; L. M. Giangregorio et al., 2006; Gorgey & Dudley, 2007a; Holman & Gorgey, 2019; Karelis et al., 2017; M. Liu et al., 2008; Roy et al., 1998; Singh et al., 2011; Skiba et al., 2021; Sutor et al., 2022; R. V. Ung et al., 2010) were included in this systematic review, as illustrated by the PRISMA flowchart in Fig. 2. The included studies (Adams et al., 2011; Ahmed & Wieraszko, 2008; Baldi et al., 1998; Battistuzzo et al., 2017; Choi et al., 2020; Crameri et al., 2002; de Freitas et al., 2018; Demchak et al., 2005; Ditterline et al., 2020; Dolbow & Credeur, 2018; Everaert et al., 2021; Galea et al., 2017; L. Giangregorio et al., 2012; L. M. Giangregorio et al., 2006; Gorgey & Dudley, 2007a; Holman & Gorgey, 2019; Karelis et al., 2017; M. Liu et al., 2008; Roy et al., 1998; Singh et al., 2011; Skiba et al., 2021; Sutor et al., 2022; R. V. Ung et al., 2010) have been published from 1998 to 2022. Twelve (50%) were conducted in the USA (Adams et al., 2011; Ahmed & Wieraszko, 2008; Baldi et al., 1998; Demchak et al., 2005; Ditterline et al., 2020; Dolbow & Credeur, 2018; Gorgey & Shepherd, 2010; Holman & Gorgey, 2019; M. Liu et al., 2008; Roy et al., 1998; Singh et al., 2011; Sutor et al., 2022), two in Brazil (8,3%) (de Freitas et al., 2018; Skiba et al., 2021), six in Canada (25%) (Everaert et al., 2021; L. Giangregorio et al., 2012; L. M. Giangregorio et al., 2006; Karelis et al., 2017; R. V. Ung et al., 2010), two in Australia (8.3%) (Battistuzzo et al., 2017; Galea et al., 2017), one in Denmark (4.16%) (Crameri et al., 2002), and one in Korea (4.16%) (Choi et al., 2020).
Volume muscle loss’ progression in subjects affected by spinal cord injury.
A total of 370 subjects were analyzed, of whom 203 were adult humans (Adams et al., 2011; Baldi et al., 1998; Choi et al., 2020; Crameri et al., 2002; Demchak et al., 2005; Ditterline et al., 2020; Dolbow & Credeur, 2018; Everaert et al., 2021; Galea et al., 2017; L. Giangregorio et al., 2012; L. M. Giangregorio et al., 2006; Gorgey & Shepherd, 2010; Holman & Gorgey, 2019; Karelis et al., 2017; Skiba et al., 2021; Sutor et al., 2022), 20 Mongrel cats (Roy et al., 1998), and 177 mice (13 CD1 female rats (Ahmed & Wieraszko, 2008), 22 CD1 male rats (R. V. Ung et al., 2010), 72 Sprague-Dawley female rats (M. Liu et al., 2008; Singh et al., 2011), 49 C57Bl/6 male rats (Battistuzzo et al., 2017), and 21 Wistar female rats (de Freitas et al., 2018)). Study cohorts ranged from 1 (Dolbow & Credeur, 2018; Gorgey & Shepherd, 2010) to 49 subjects (human and animals) (Battistuzzo et al., 2017). About intervention, twelve (Adams et al., 2011; Baldi et al., 1998; Crameri et al., 2002; de Freitas et al., 2018; Demchak et al., 2005; Dolbow & Credeur, 2018; Everaert et al., 2021; Galea et al., 2017; L. Giangregorio et al., 2012; Gorgey & Shepherd, 2010; Holman & Gorgey, 2019; Skiba et al., 2021) studies provided several types of electrical stimulation, seven administered body weight supported or not supported treadmill training (Battistuzzo et al., 2017; L. M. Giangregorio et al., 2006; M. Liu et al., 2008; Roy et al., 1998; Singh et al., 2011; R. V. Ung et al., 2010), and five performed other approaches (Ahmed & Wieraszko, 2008; Choi et al., 2020; Ditterline et al., 2020; Karelis et al., 2017; Sutor et al., 2022). Concerning the outcome measures, most of the studies evaluated the lower-limb muscles’ cross-sectional area (CSA) (Adams et al., 2011; Ahmed & Wieraszko, 2008; Battistuzzo et al., 2017; Crameri et al., 2002; Demchak et al., 2005; Everaert et al., 2021; Galea et al., 2017; L. Giangregorio et al., 2012; L. M. Giangregorio et al., 2006; Gorgey & Shepherd, 2010; Holman & Gorgey, 2019; M. Liu et al., 2008; R. V. Ung et al., 2010) and the muscle function (Ahmed & Wieraszko, 2008; Crameri et al., 2002; Demchak et al., 2005; Ditterline et al., 2020; Everaert et al., 2021; Holman & Gorgey, 2019; R. V. Ung et al., 2010), while nine studies (Ahmed & Wieraszko, 2008; Baldi et al., 1998; Choi et al., 2020; Ditterline et al., 2020; Galea et al., 2017; L. Giangregorio et al., 2012; Karelis et al., 2017; Singh et al., 2011; Sutor et al., 2022) analyzed the muscle features. The main characteristics of the studies included in this systematic review are summarized in Table 2.
Main characteristics and findings of the included studies
Legend: AIS = ASIA impairments scale; BMS = Basso mouse scale; BWSTT = body weight supported treadmill training; CSA = cross-sectional area; EBPGO = exoskeletal body-powered gait orthosis; FES = functional electrical stimulation; FI = fatigue index; G-LBM = gluteal lean body mass; LL-LBM = lower limb lean body mass; MHC = major histocompatibility complex; NMES = neuromuscular electrical stimulation; scES = spinal cord epidural stimulation; SCI = spinal cord injury; TB-LBM = Total body lean body mass.
Electrical stimulation
de Freitas and colleagues (de Freitas et al., 2018) analyzed the effects of biceps brachii NMES on muscle function, tropism, and the Akt pathway signaling involved in muscular plasticity in rats after C5-C7 level surgical induced hemisection, observing an increase in bicep muscle strength in the NMES group when compared with the untreated SCI group.
Treadmill on sub-acute phase SCI
In 1998, Roy et al. (1998) trained twenty adult cats to determine the role of short periods of daily stepping on a treadmill (rhythmic activity) or standing (continuous activity) in ameliorating the adaptations in the muscle properties, demonstrating that training ameliorated soleus atrophy and enhanced maximum force capability.
Liu et al. (2008) concluded that both cycling and treadmill training on rats paused the atrophic process and improved the recovery rate, with a positive correlation between locomotor functional scores and hindlimb muscle size following SCI.
Ung et al. (2012), through experiments in a mouse model of complete paraplegia which underwent non-assisted treadmill training, stated that body weight, adipose tissue, whole muscle, and individual fiber CSA values were significantly lower in trained compared with untrained animals.
Singh and colleagues (2011) found that BWSTT-contused animals showed accelerated locomotor recovery and reduced muscle atrophy.
Battistuzzo et al. (2017) also demonstrated that 9 weeks of training after incomplete SCI was effective in preventing fast-twitch muscles atrophy, but there were limited effects on slow-twitch muscles and muscle fiber type composition.
Spinal magnetic stimulation on sub-acute phase SCI)
Ahmed and Wierasko (2008) provided preliminary data about the efficacy in the maintenance of CSA in epidural spinal cord magnetic stimulation and acrobatic exercise on exercising/exposed animals.
Studies on humans
Electrical stimulation
3.3.1.1. Sub-acute phase SCI. Baldi et al. (1998) and Demchak et al. (2005) determined that FES cycle ergometry (FES-CE) or non-loaded FES isometric contractions (FES-IC) could prevent disuse muscle atrophy after SCI, with a less loss of lean body mass and CSA than controls.
In 2017, the Switch-On study (Galea et al., 2017), examined the efficacy, safety, feasibility, and tolerability of FES-assisted cycling (FESC) and passive cycling 4-week post-traumatic complete or incomplete SCI, stating that no significant between-group difference in post-intervention muscle CSA was found. Furthermore, Everaert et al. (2021) reported that muscle CSA decreased and recovered after a variable period of FES cycling, concluding that changes in muscle CSA did not always correspond with the dose of FES cycling.
3.3.1.2. Humans (chronic phase SCI). In Crameri and colleagues’ study (2002), the paralyzed vastus lateralis muscle showed significant alterations in skeletal muscle characteristics after FES cycle ergometry leg training.
Four studies (Adams et al., 2011; Dolbow & Credeur, 2018; L. Giangregorio et al., 2012; Gorgey & Shepherd, 2010) demonstrated that, after the electrical stimulation training, the average skeletal muscle CSA increased and torque, fatigue index, contractile speed, and cross-sectional area were preserved.
In 2019, Holman and Gorgey (2019) concluded that 16 weeks of combined TRT and NMES-RT improved muscle quality in men with motor-complete SCI.
Skiba and colleagues (2021), identified a chronic increase in muscle thickness in the subjects treated with FES + blood flow restriction (BFR) in thirty-two adult humans affected by complete SCI.
Treadmill training
3.3.2.1 Sub-acute phase SCI. In 2005, Giangregorio and colleagues (2005) demonstrated an increased muscle CSAs and a small reduction in lower limb bone mineral density (BMD), after BWSTT training in the sub-acute phase of SCI, and, in 2006 (L. M. Giangregorio et al., 2006), the same results were obtained in subjects at least 12 months after injury.
Exoskeletal body-powered gait orthosis or exoskeleton-assisted walking
3.3.3.1. Humans (chronic phase SCI). Karelis et al. (2017), after a personalized locomotor training program using a robotic exoskeleton, found a significant increase in CSA, leg and appendicular lean body mass, while Choi et al. (2020) did not identify significant changes in weight, fat-free mass, lean body mass, and percent fat mass in the exercise group.
Sutor and colleagues (Sutor et al., 2022) performed an exploratory study to investigate the efficacy of exoskeleton training and trans-spinal stimulation in 8 subjects after spinal cord injury (EXTra-SCI), concluding that percent fat was reduced.
Spinal cord epidural stimulation
3.3.4.1. Humans (chronic phase SCI). Ditterline and colleagues (Ditterline et al., 2020) stated that implanted eSCS significantly increased LL-LBM, muscle strength, and fatigability.
Study quality
To evaluate the quality of evidence of this systematic review, we adopted the Joanna Briggs Institute Critical Appraisal Checklist for Quasi-Experimental Studies (non-randomized experimental studies). As depicted in Table 3, we assessed the 9-question risk-of-bias domains. All the included articles presented full-text availability. Most of the studies (n = 19; 79.16%) were judged with at least one serious risk of bias, which translated into an overall serious risk of bias for that study. Fourteen (58.33%) included studies reported a lack of data on follow-up outcomes.
Joanna briggs institute critical appraisal checklist for quasi-experimental studies (non-randomized experimental studies).
Joanna briggs institute critical appraisal checklist for quasi-experimental studies (non-randomized experimental studies).
Legend: Q1 = Is it clear in the study what is the ‘cause’ and what is the ‘effect’ (i.e. there is no confusion about which variable comes first)?; Q2 = Were the participants included in any comparisons similar?; Q3 = Were the participants included in any comparisons receiving similar treatment/care, other than the exposure or intervention of interest?; Q4 = Was there a control group?; Q5 = Were there multiple measurements of the outcome both pre and post the intervention/exposure?; Q6 = Was follow up complete and if not, were differences between groups in terms of their follow up adequately described and analyzed?; Q7 = Were the outcomes of participants included in any comparisons measured in the same way?; Q8 = Were outcomes measured in a reliable way?; Q9 = Was appropriate statistical analysis used?; N = no, Y = yes; N/A = not applicable.
The present systematic review describes the state-of-art of rehabilitation effects on volume muscle loss in subjects (humans and animals) with SCI. Our results underlined that electrical stimulation, whether functional or neuromuscular and exercise on a treadmill with or without load relief seem to be effective in the VML reduction in subjects suffering from SCI. Moreover, we highlighted that most of the studies in the literature presented a relevant risk of bias, as more rigorous trials are needed to conclude on best practices. As previously stated, VML in individuals with SCI may significantly impair the patient’s functional independence. Due to this, it is mandatory to monitor changes in body composition over time, detecting early muscle volume reduction and the most effective type of intervention, also considering the social and health implications of this condition. Several authors evaluated the effects of different rehabilitative approaches in VML management, without providing reliable data about the optimal treatment parameters and the intervention timing. The mechanisms of VML, which occurs for 40–60% in the first two weeks after SCI, are not fully understood but seem primarily supported by an upregulation of the proteolytic degradation of muscle proteins, such as muscle-restricted E3 ubiquitin ligases muscle atrophy F-box (MAFBx, atrogin-1) and muscle ring finger-1 (MuRF1, Trim63) (Gorgey et al., 2019; Qin et al., 2010). Both functional and neuromuscular electrical stimulation is the most commonly used approaches in addition to conventional rehabilitation treatment. In 2020, Zhao et al. (2021) evaluated the efficacy of surface or implantable ES to elicit muscle contraction and prevent muscle atrophy. In this study, the authors found a significant reduction of the mass of soleus, plantaris and extensor digitorum longus muscles in a rat model 6 weeks post-SCI treated with prolonged electrical stimulation and /or testosterone enanthate compared to sham-transected animals. Zhang and colleagues (Zhang et al., 2017) examined the effects of tail nerve electrical stimulation (TANES) 5 times/week and electroacupuncture (EA) 1 time/week on lumbar motor neurons and hindlimb muscle in spinal transected rats. Both TANES and EA showed a significant impact in ameliorating the atrophy of hindlimb muscle after SCI four weeks after the beginning of treatment, suggesting that these approaches seem to protect the motor neurons as well as alleviate muscle atrophy. An experimental prospective study with four parallel treatment groups (Butezloff et al., 2015) analyzed the skeletal muscle changes after a complete SCI in rats and aimed to compare the ES and whole-body vibration’s effects on the prevention of muscle hypotrophy in these animals. After 30 days of treatment with ES (3 days/week, 20 mins per day) the Authors obtained a significant partial muscle hypotrophy prevention. As stated before, insightful changes in skeletal muscle size and strength might occur after injury, often in conjunction with increased intramuscular fat (Elder et al., 2004; Gorgey & Dudley, 2007b). This condition is also associated with increased muscle fatigue in response to the ES, as well as the reduced oxidative capacity of the skeletal muscle (Mahoney et al., 2007; Olive et al., 2003). Erickson et al. (2017) demonstrated that the endurance training elicited with NMES could reverse these changes and result in muscle metabolic health improvement. They recruited 14 subjects affected by SCI undergoing a 16-week home-based endurance NMES training of knee extensors muscles; after this rehabilitation protocol, an increased number of contractions (performed throughout training) was observed, with a significant improvement in skeletal muscle oxidative capacity of 119% (p = 0.019).
As underlined by the results of our systematic review, treadmill training with or without weight relief also appears to significantly reduce VML. More in detail, incomplete SCI animal models, and quadrupedal bodyweight-supported treadmill training could reduce muscle atrophy and improve muscle force production and recovery of voluntary locomotion (M. Liu et al., 2010; Stevens et al., 2006); these results might be obtained through better afferent neuromodulation, the prevention of axonal degradation, and the normalization of the spinal reflex pathways that regulate spasticity and motoneuron excitability (Hou et al., 2014). Moreover, several systemic hormonal irregularities occur secondary to SCI and may worsen musculoskeletal deficits, such as low serum testosterone (Qin et al., 2010). Yarrow et al. (2020) concluded that short-term bodyweight-supported treadmill training alone did not improve bone, muscle, or locomotor recovery in adult rats after severe SCI. In contrast, longer-term bodyweight-supported treadmill training associated with adjuvant testosterone-enanthate more comprehensive musculoskeletal benefit than adjuvant testosterone-enanthate alone, suggesting that adjuvant testosterone-enanthate influenced locomotor recovery. Marques et al. (2018) randomly divided 95 Wistar rats into four groups: control, SCI (rats with spinal cord contusion), and SCI groups exposed to locomotor training starting 7, 14 or 28 days after the injury (SCI-T7, SCI-T14 and SCI-T28), performed on a treadmill, five days a week, 20 minutes per day, for ten weeks. Interestingly, the authors reported that late training groups had preservation of the motoneurons in the spinal cord and larger muscle fibers area in tibialis anterior muscle; in contrast, the SCI-T7 group had higher lesion volume after locomotor training in comparison with the SCI group, demonstrating that identify a locomotor training starting time after the injury is essential to define the best therapeutic window for rehabilitation. Santo et al. (2018) investigated the effects of locomotor treadmill training on muscle tropism mediated by protein kinase B (Akt)/mammalian target of rapamycin (mTOR)/p70 ribosomal protein S6 kinase (p70S6K) in paraplegic adult female Wistar rats underwent an incomplete thoracic SCI induced by compression using an aneurysm clip, and, after 7 days, a 3-week locomotor treadmill training with body weight-support. In particular, trained animals did not show locomotor improvement, but presented an increase in muscle weight and myofiber CSA, confirming the locomotor treadmill training’s role in the prevention of soleus muscle hypotrophy in rats with SCI. To date, we found only a single recent systematic review in the literature investigating the strategies to increase skeletal muscle mass in individuals with SCI (Santos et al., 2022); however, the Authors focused on the chronic phases (after at least one year from the event) and considered only human studies.
Concerning the risk of bias in the studies included in the present systematic review, we performed an evaluation through the Joanna Briggs Institute Critical Appraisal Checklist for Quasi-Experimental Studies (non-randomized experimental studies). It was interesting to be noted that only 5 papers (20.83%) did not present any risk of bias. Furthermore, 10 studies (41.66%) showed a lack of data on follow-up outcomes. Finally, 13 (54.16%) of the 24 papers included in our systematic review did not provide data from a control group.
To the best of our knowledge, this is the first systematic review providing a broad overview of the currently available rehabilitation options to prevent VML in patients with SCI to promote an early therapeutic intervention targeting the multilevel mechanisms underpinning this disabling condition. However, we are aware that this paper is not free from limitations. First, the studies included in this systematic review administered the treatments to both humans and animals (i.e., cats and mice), resulting in complicated comparability of the obtained data. However, we believe that the inclusion of animal studies might be beneficial in the SCI field, to better understand the mechanisms underpinning muscle mass restoration. In addition, the subjects presented a very different AIS classification, with consequent different findings; lastly, we should take into consideration the high heterogeneity of the rehabilitation approaches administered, and the outcome measures assessed.
Conclusion
Taking together the findings of the present systematic review showed that the electrical stimulation and treadmill exercises might be effective to counteract the VML in both the sub-acute and the chronic individuals with SCI. In the recent scientific literature, several rehabilitative treatments have been investigated, albeit there is no agreement on the duration and the modality of the intervention. The data obtained in the present systematic review confirm the role of approaches widely used in clinical practice as effective for the prevention of VML; this allows rehabilitators to customize the treatment for each patient while maintaining the methods investigated as a gold standard.
In this context, considering the key role that a personalized medicine might have in these subjects, further observational studies are still needed to overcome the limitations of a standardize rehabilitation in patients with SCI affected by VML. Moreover, future studies should focus on the definition of clear protocols, with timing, duration, and parameters that might be a benchmark for the treatment of a such disabling condition.
Author contributions
Conceptualization: AdS; Methodology: AdS, LM, and KM; Investigation: LM, NM, and CC; Formal analysis: AdS, LM, and NM; Data curation: AdS, KM, and AB; Writing—original draft preparation, AdS and LM; Writing—review and editing: KM and AA; Visualization: NM, CC, LL, and MI; Supervision: AdS, KM, and AA. All authors read and approved the final version of the manuscript.
Conflict of interest
None of the authors declare any conflict of interests, funding sources or consultant relationships with any organizations involved in this research.
Footnotes
Acknowledgments
The authors would like to thank Martina Ragona for her contribution to the work.
