Abstract
Study design
Narrative review.
Objectives
To provide an overview of contemporary therapies for the James Lind Alliance priority setting partnership for degenerative cervical myelopathy (DCM) question: ‘Can novel therapies, including stem-cell, gene, pharmacological and neuroprotective therapies, be identified to improve the health and wellbeing of people living with DCM and slow down disease progression?’
Methods
A review of the literature was conducted to outline the pathophysiology of DCM and present contemporary therapies that may hold therapeutic value in 3 broad categories of neuroprotection, neuroregeneration, and neuromodulation.
Results
Chronic spinal cord compression leads to ischaemia, neuroinflammation, demyelination, and neuronal loss. Surgical intervention may halt progression and improve symptoms, though the majority do not make a full recovery leading to lifelong disability. Neuroprotective agents disrupt deleterious secondary injury pathways, and one agent, Riluzole, has undergone Phase-III investigation in DCM. Although it did not show efficacy on the primary outcome modified Japanese Orthopaedic Association scale, it showed promising results in pain reduction. Regenerative approaches are in the early stage, with one agent, Ibudilast, currently in a phase-III investigation. Neuromodulation approaches aim to therapeutically alter the state of spinal cord excitation by electrical stimulation with a variety of approaches. Case studies using electrical neuromuscular and spinal cord stimulation have shown positive therapeutic utility.
Conclusion
There is limited research into interventions in the 3 broad areas of neuroprotection, neuroregeneration, and neuromodulation for DCM. Contemporary and novel therapies for DCM are now a top 10 priority, and whilst research in these areas is limited in DCM, it is hoped that this review will encourage research into this priority.
Keywords
Introduction
Individuals diagnosed with Degenerative Cervical Myelopathy (DCM) may require surgical and/or non-surgical interventions depending on the severity of the disease and clinical manifestations. Surgical intervention in the form of decompressive surgery with or without fusion is the mainstay of treatment for moderate to severe disease as assessed by modified Japanese Orthopaedic Association (mJOA) scale.1,2 The optimal management of mild DCM, early in its course, is unknown, as studies have not pointed towards a significant benefit for prophylactic surgical intervention.3,4 However, this opens a window for the possible application of non-surgical interventions such as disease modifying therapeutics physical therapy, cervical traction, collars and spinal injection.4,5
Despite the success of surgical decompression in halting neurological decline and providing some recovery, myelopathy symptoms may reoccur, 6 and full recovery is rarely seen, with estimates suggesting less than 5% make a full recovery,7,8 with lower limb and sphincter function demonstrating the slowest recovery responses. 9 Ongoing myelopathy may affect domains such as mobility, weakness, manual dexterity, pain and bladder/bowel dysfunction resulting in a considerable impact and decline in quality of life.10,11 Therefore, halting neurological decline and improving rates of functional recovery remain major priorities of patients and surgeons dealing with DCM. 12 The importance of this unmet need is further compounded by the fact that DCM is one of the commonest causes of spinal cord dysfunction worldwide, and the incidence is expected to increase with an ageing population.1,13
This uncertainty was highlighted in the recent James Lind Alliance 12 (JLA) priority setting partnership, with one of the top priorities and questions agreed upon being, ‘Can novel therapies, including stem-cell, gene, pharmacological and neuroprotective therapies, be identified to improve the health and wellbeing of people living with DCM and slow down disease progression?’. The objective of this narrative review is to provide an overview of potential contemporary therapies that may enhance recovery in DCM. We will briefly outline the pathophysiology of DCM and then present contemporary therapies in 3 main areas, namely, neuroprotection, neuroregeneration and neuromodulation, which may in the future have a therapeutic role in DCM.
Pathophysiology of DCM
Degenerative cervical myelopathy is an umbrella term encompassing a number of degenerative processes and responses in the cervical spine, which leads to progressive chronic spinal cord compression.14,15 Osseous, ligamentous, and intervertebral disc tissue undergo degenerative changes as part of normal ageing and/or repetitive stress leading to structural changes such as spondyloses, hypertrophy/ossification/calcification of ligaments (e.g Ossification of the posterior longitudinal ligament) and disc herniation/prolapse.1,16 Whilst these changes lead to static compression and direct injury to the spinal cord, they are it compounded by the mobility of the cervical spine, causing further compression due to physiological or pathological (ie spondylolisthesis) movements.1,16 Mechanical compression leads to direct primary injury but also initiates a complex sequence of secondary injury processes in the spinal cord. 17 These include macro- and microvascular compromise causing hypoxia and ischaemia18,19 and neuroinflammation.17,20 The consequence of these processes include loss of neurons, oligodendrocytes, 20 demyelination 21 and axonal degeneration. 17
Demyelination can act as a functional conduction block on axons contributing to the sensorimotor deficits seen in DCM. Post-mortem studies have found extensive thin myelinated fibres in the spinal cord white matter suggestive of focal demyelination and remyelination. 22 Studies have also found an association between reduced myelin content on myelin water imaging on MRI and impaired somatosensory evoked potential in DCM, which demonstrates the impact of demyelination seen on long tract axonal function in DCM. 23 Therefore, strategies to mitigate demyelination and stimulate endogenous mechanisms of myelin repair may provide a viable therapeutic strategy.
The neuroinflammatory cascade resulting from chronic compression is of interest as it can be both protective and damaging. An increase in activated microglia and macrophages has been observed at the site of chronic spinal cord compression and are a source of pro-inflammatory cytokines and can lead to further cell death by necrosis and apoptosis. 24 Alternatively, Immune cells can also exhibit neuroprotective effects such as releasing neuroprotective cytokines and growth factors. 25 This has created interest in immunomodulatory strategies to alter this balance as a therapeutic strategy.
Neuroprotection
Neuroprotective agents prevent further cell death and dysfunction by altering secondary injury pathways. A notable example is Riluzole, a benzothiazole sodium channel-blocker developed in the mid-20th century as a muscle relaxant, 26 with later indications as an anticonvulsant 27 and neuroprotective agent to prolong survival in ALS. 28 In the context of DCM, animal studies have shown RIluzole can mitigate secondary injury mechanisms related to sodium channels and glutamate excitotoxicity thus resulting in functional improvements.29-31 These promising findings have led to a phase III randomized placebo-controlled trial (CSM-PROTECT) to assess the efficacy of Riluzole as an adjunct to surgical decompression in chronic cervical myelopathy to promote neurologic recovery. 32 The study was recently published and reported no significant difference in the primary endpoint of mJOA at 6-month follow-up. However, Riluzole was associated with potentially promising reduction in neck pain at 6- and 12-month follow-up compared to placebo. 33 As pain is a major patient priority, further investigations may be warranted into which subgroups would benefit most. Inflammatory pathways are implicated in the deleterious effects of chronic cord compression and recent work in humans and animal models has implicated microRNA21 (miR21) as a key mediator of the inflammatory/ischemic cell injury in DCM. A prospective cohort study of patients with DCM has identified a positive correlation between miR21, initial symptom severity and poor treatment outcomes, the findings of which were further corroborated in mouse models of DCM. This work suggests the possibility of miR21 as a possible biomarker of disease risk in DCM and a potential therapeutic target for intervention. 34
Anti-inflammatory agents such as corticosteroids are also candidates for potential clinical translation as a neuroprotective adjunct to surgery and have been extensively studied in traumatic SCI.35-37 In a rodent study of DCM, Methylprednisolone, an agent used in acute-onset traumatic spinal cord injury, 38 was used as a perioperative adjunct to surgical decompression. 39 Significant improvement was found in the group treated by methylprednisolone when compared to the control 2-weeks post decompression in forepaw function. However, by 5-weeks, no significant difference was apparent between the groups. 39 Few human clinical studies have been conducted, though one retrospective study of thoracic myelopathy used intraoperative methylprednisolone found better neurological recovery at 2 weeks, though no significant difference in neurological outcomes between methylprednisolone and control group at long-term follow-up. 40 The use of corticosteroids perioperatively in anterior cervical spine surgery has however shown benefit in reducing post-operative complications with significant reduction in airway oedema, pain, hospital stay and improved swallowing in Phase-III randomized controlled trials.41,42 This surgical approach is used in the surgical management of DCM and future studies should look into the potential functional benefits of corticosteroids in these patients.
The perioperative period is a key time point in which neuroprotective agents should be considered due to ischaemia-reperfusion injury, but also due to axonal plasticity, which is thought to emerge after decompression. 43 Whilst no neuroprotective agent has shown efficacy in Phase 3 clinical trials, improved understanding of the pathophysiology of DCM may provide an avenue for the development of more targeted neuroprotective therapies. Of note, neck and arm pain represent potentially important targets if intervention in DCM, as suggested by the recent CSM-Protect trial. 33 However, this will require the design of trials which use more sensitive outcomes to detect changes in neck and arm pain.
Regenerative Medicine
People with DCM often develop and then suffer from lifelong disability, with less than 5% making a full recovery even despite surgical decompression.7,8 This occurs due to failure to recognize the clinical deterioration, delayed medical intervention, the limited regenerative capacity of the spinal cord, loss of local cellular structures and disruption of the spinal cord architecture. Replacing lost tissue and enhancing intrinsic recovery capacity is an area of interest in spinal cord injuries as a means to enhance recovery. Attempts are being made to achieve this through a number of different approaches, including pharmacological and cell-based therapies.44-48
One potential beneficial pharmacological agent is Ibudilast, which is currently licensed in Japan for the treatment of asthma and post-stroke dizziness. 49 The mechanisms by which it exerts its effects for the aforementioned indications have been attributed to its anti-inflammatory, bronchodilatory and vasodilatory effects.50-55 More recently, it has been found to exhibit central anti-inflammatory, neuroprotective and neurotrophic/regenerative effects by its inhibition of phosphodiesterase-4 (PDE-4 and -10) and macrophage migration inhibitory factor, leading to attenuation of activated glial cells and enhancement of neurotrophic factors.49,50,56,57 This has generated interest in its application in a number of neurological conditions. These include early-stage clinical trials in progressive Multiple Sclerosis,58,59 ALS, 60 alcoholism,61,62 drug addiction63,64 and pain.65,66 The combination of anti-inflammatory, neuroprotective and neuro-regenerative properties has led to interest for its use in DCM and is the basis for RECEDE-Myelopathy (NCT04631471), a phase 3, double-blind, randomized controlled trial assessing the efficacy of Ibudilast as an adjuvant treatment to decompressive surgery for DCM on mJOA score and neck pain.
Cell-based therapies have also gained considerable attention over the last few decades. Stem cells possess the ability to differentiate into a variety of cell types creating the possibility of a potential therapeutic tool to repair and/or replace damaged tissue. 67 Challenges for cell-based therapies include the cell type first surviving the transplantation, then migrating appropriately to the site of therapeutic action, differentiating into the correct lineage and finally to behave physiologically in the manner intended. 68 Currently, the most widely applied clinical use of this therapy is haematopoietic stem cell transplantation with research dating back to the 1950s and indicated for conditions such as lymphoma, leukaemia and anaplastic anaemia with curative potential.69,70 Whilst there has been little research for its application in DCM; specifically, there is ongoing research to develop stem cell therapies for traumatic spinal cord injury sharing pathophysiological features.67,71 Several mechanisms by which stem cells can promote recovery in spinal cord injuries can be applicable to DCM. These include replacement of lost tissue (ie neurones 72 /glial precursors/oligodendrocytes 73 ), integration into host neuronal circuits,74,75 release of neurotrophic factors(e.g BDNF, GDNF, NGF and VEGF),76-78 anti-apoptotic,79,80 anti-inflammatory81,82 and immunomodulatory effects. 83 Trials in SCI are typically underpowered for efficacy and lack controls as they are early-stage open-label trials.84,85 In addition, there is considerable heterogeneity between cell-based interventions such as type of cell utilized, source of stem cell (i.e embryonic stem cells, adult stem cells and induced pluripotent stem cells; autologous vs allogeneic), processing (i.e lab purification, amplification, good manufacturing practice adherence and quality control) and delivery of stem cells (i.e Intravenous, intrathecal, direct intraspinal and impregnated tissue engineered materials) highlighting uncertainty in optimal intervention parameters.44,45,85 Further, adjuncts to enhance cell-based therapies are undergoing investigation to overcome some of the challenges faced such as poor cell survival, migration and integration. These include co-administration of growth factors, cell delivery and structural support with biomaterials/scaffolds, guidance of migration and differentiation by electric fields (galvanotaxis), degradation of glial scar and self-assembling peptides to improve extracellular matrix environment.45,71,86
One of the inciting factors for DCM is cervical spondylosis and disc degeneration. Cell-based, growth-factor based and small molecule-based therapies aiming to repair or regenerate the degenerate intervertebral disc may offer an opportunity to halt DCM progression or even reverse its symptoms. This may be particularly important in patients with mild DCM symptoms and in asymptomatic patients with imaging evidence of cord compression. Several human clinical trials using stem cells (autologous or allogenic; bone marrow and adipose-derived mesenchymal, notochordal and chondrocyte-like nucleus pulposus cells) for intervertebral disc degeneration have been or are currently being undertaken. 87 While most studies report significant reduction in pain, increase in disc height, improved patient mobility and quality of life,88-91 concerns have been raised due to the poor design of some studies and their low number of patients and lack appropriate controls.
Due to the nature of DCM, the majority of patients are in an older age category (>55 years), which presents a unique challenge. The central nervous system undergoes structural and functional changes as part of the normal ageing process. 92 This includes reduced neuroplasticity, which could contribute to post-decompression recovery, 43 and strategies to enhance endogenous regenerative processes may be confounded by aging. Regenerative therapies provide prospects for new treatments in DCM; however, it is currently a very young field of research. Research in this field aiming to overcome some of the current challenges and specific to its role in DCM are warranted.
Gene therapy offers the prospect for sustained and localized production of therapeutics that is particularly attractive for ‘biologics’ that are otherwise complex to deliver. One therapy has been studied extensively for possible direct tissue application in SCI with a lead strategy aimed at neuroplasticity using an enzyme, chondroitinase, that remodels the basal lamina of neuronal nets to enhance neuroplasticity.93,94 In spinal muscular atrophy, a gene therapy (Zolgensma) that restores a critical functional protein (SMN protein, important in motor neurone survival) to motor neurons has been approved for clinical use. 95 Gene therapies such as myostatin inhibition are also under development for muscular dystrophy, 96 which aims to improve muscle function. Whilst gene therapy is in its early stages of development for many conditions, it offers the prospects for sustained production of therapeutic molecules which remains to be explored in DCM.
Neuromodulation
Neuromodulation is broadly defined by the International Neuromodulation Society as ‘the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation or chemical agents, to specific neurological sites in the body’. 97 Electrical neuromodulation is used in the treatment of conditions and symptoms including but not limited to chronic pain,98,99 movement disorders,100-102 epilepsy,102-104 psychiatric disorders,105,106 stroke,107,108 traumatic brain injury109,110 and sensory deficits.111,112 A variety of devices have been developed to deliver stimulation to the target of interest, with some being invasive such as deep brain stimulation and spinal cord stimulation (SCS) and others noninvasive such as transcranial Direct Current Stimulation (tcDCS), repetitive Transcranial Magnetic Stimulation (rTMS) and peripheral surface electrode Neuromuscular Electrical Stimulation (NMES) or Functional Electrical Stimulation (FES). The mechanism of action of these devices in modulating the nervous system and pathways and is not fully understood, but here we will briefly explore their application in related conditions.
Spinal cord stimulation has become an established therapeutic tool in the management of chronic neuropathic and ischaemic pain syndromes, including Failed Back Surgery Syndrome (FBSS),113,114 Complex Regional Pain Syndrome Type 1115,116 and chronic leg ischaemia. 117 Though the exact mechanism of its analgesic effects is elusive, early proposals included the gate control theory of pain, but more recent biochemical hypotheses propose SCS works by enhancing GABAergic systems of dorsal horn cells by stimulating their dendrites.118,119 There is limited evidence for the use of SCS for pain in DCM but one case study of a cervical spinal cord stimulator placed (C3-C6) for significant post-operative pain following posterior decompression (C5-C7), resulting in a significant reduction in pain. 120 Of note, the efficacy of SCS for SCI-induced pain appears to be more limited when compared to the aforementioned indications, such as FBSS, which may be due to the significant damage to underlying neural circuits required for the analgesic effects of SCS.121,122
Although the use in spinal cord–mediated pain has been limited, epidural SCS has shown to be capable in restoring motor and autonomic function in a small number of chronic complete SCI patients.123-125 It is thought that epidural SCS increases the excitability of the spared spinal cord circuitry within the injury site, leading to enhancement of transmission and volitional control. 126 Whether this strategy could be used for paralysis due to severe DCM remains to be investigated.
Transcranial Magnetic Stimulation (TMS) allows painless, noninvasive, cortical stimulation by means of electromagnetic induction from a coil positioned over the scalp. 127 It serves as a useful electrophysiological diagnostic tool by measuring parameters such as central motor conduction time (CMCT), a sensitive measure to detect myelopathy.128-130 Repeated stimulation in the form of rTMS has gained interest as a method of neuromodulation to induce changes in brain activity lasting beyond the duration of stimulation suggestive of neuroplastic changes. 131 In the clinical sphere, it has gained FDA approval as a treatment for major depressive disorder, obsessive-compulsive disorder and migraine,132-135 with ongoing research in a number of other psychiatric and neurological conditions. 136 Early clinical studies in SCI have found potential application of rTMS in the management of SCI-related pain,137-139 spasticity,140,141 motor function141-143 and autonomic function,144-146 albeit the outcomes across studies were not consistent. This may be attributed to the heterogeneity in rTMS protocols used, and ongoing research is warranted to further optimize existing protocols and to investigate their potential application in DCM. 130
Transcranial direct current stimulation modulates neural activity non-invasively by surface electrodes, which are placed over the scalp with low-intensity currents are passed across them. In contrast to TMS, tcDCS does not induce action potentials but modulates the resting membrane potentials to alter excitability. There is ongoing research investigating its role in a number of psychiatric and neurological conditions. 147 A recent meta-analysis found tcDCS improves upper-limb motor performance in healthy adults. 148 Its utility in chronic incomplete cervical SCI has been investigated in early-stage studies and noted significant improvement in hand grasp149,150 which were synergistic when combined with physical training. 151
Neuromuscular electrical stimulation, also known as FES, is a well-established tool in which surface electrodes are placed over muscles and peripheral nerves to deliver electrical stimulation and achieve muscle contraction. It is used routinely for motor retraining in conditions such as SCI and stroke to restore motor functions such as standing or grasping.152,153 Whilst the artificial stimulus leading to muscle contraction can lead to muscle strengthening, effects have also been demonstrated in the central nervous system with modulation of spinal reflex, corticospinal excitability and neurophysiological changes in the cortex. 154 It is proposed that the underlying mechanism stems not only from the propagation of action potentials leading to muscle contraction but also antidromic propagation of action potential across motor axons and sensory afferents, which traverse to the central nervous system.154,155 Despite extensive research for its use for injuries to the CNS such as SCI, only case reports have been published for its use in DCM, which have demonstrated significant improvement in upper limb function 156 and gait. 157 Whilst these case reports are encouraging, it presents low level of evidence and further research is necessary, especially as NMES is a safe and less inexpensive intervention.
An exciting new clinical trial in SCI is the Up-LIFT study (NCT04697472) that employs transcutaneous spinal stimulation over the injury region. In an open-label study, this method of stimulation was been shown to increase arm and hand function in subjects with chronic cervical SCI. 158 Given the similarities between chronic DCM and SCI, an effect of this methodology might be observed.
Implementation Strategies
A broad overview of contemporary interventions which may have therapeutic utility in DCM is presented in the 3 broad categories of neuroprotection, neuromodulation and neuroregeneration. Whilst specific interventions within these groups may overlap between these broad categories (i.e interventions with regenerative and neuroprotective properties), these categories also provide useful reference for when interventions could be given in the natural course of the disease. Mild DCM may benefit from neuroprotective agents to slow neurological deterioration. In moderate to severe cases, patients may have received surgical decompression and restorative therapies with neuromodulation or neuroregenrative strategies could be beneficial. The effects of these categories of intervention on the natural history of DCM are presented in Figures 1 and 2. Combinations of interventions may also enhance recovery as they can work on different aspects of the pathological and regenerative process’ to reduce neurological injury and enhance recovery. Combinatory strategies have been gaining traction in SCI research and are useful to consider in DCM.
159
Graph illustrating simplified natural history of degenerative cervical myelopathy with progressive deterioration of neurological function including slow change phase and rapid functional decline phase with red line. Timepoints in natural history which neuroprotective and neurorestorative intervention can be of therapeutic value in slowing neurological decline and regaining neurological function are highlighted. Paler lines indicate differing natural history which may be experienced by people with degenerative cervical myelopathy, including continuous slow decline with no rapid phase, rapid decline with no slow phase, and after intervention those with no significant improvement in neurology or deterioration. Timeline of potential application of contemporary interventions (neuroprotection, neuromodulation and neuroregeneration) according to natural history and severity of degenerative cervical myelopathy. In mild-moderate degenerative cervical myelopathy, neuroprotective strategies may prevent/slow down progression by interfering in pathological process. In severe degenerative cervical myelopathy, it is likely that the spinal cord is too damaged for there to be significant improvement gained from neuroprotective strategies. In moderate-severe cases, surgery will remove the focus of compression. At this stage, neuromodulatory strategies may enhance plasticity implicated in the recovery process and neuroregenerative strategies can be considered after the spine has been decompressed if significant neurological damage is present. Graphics produced with support of Myelopathy.org.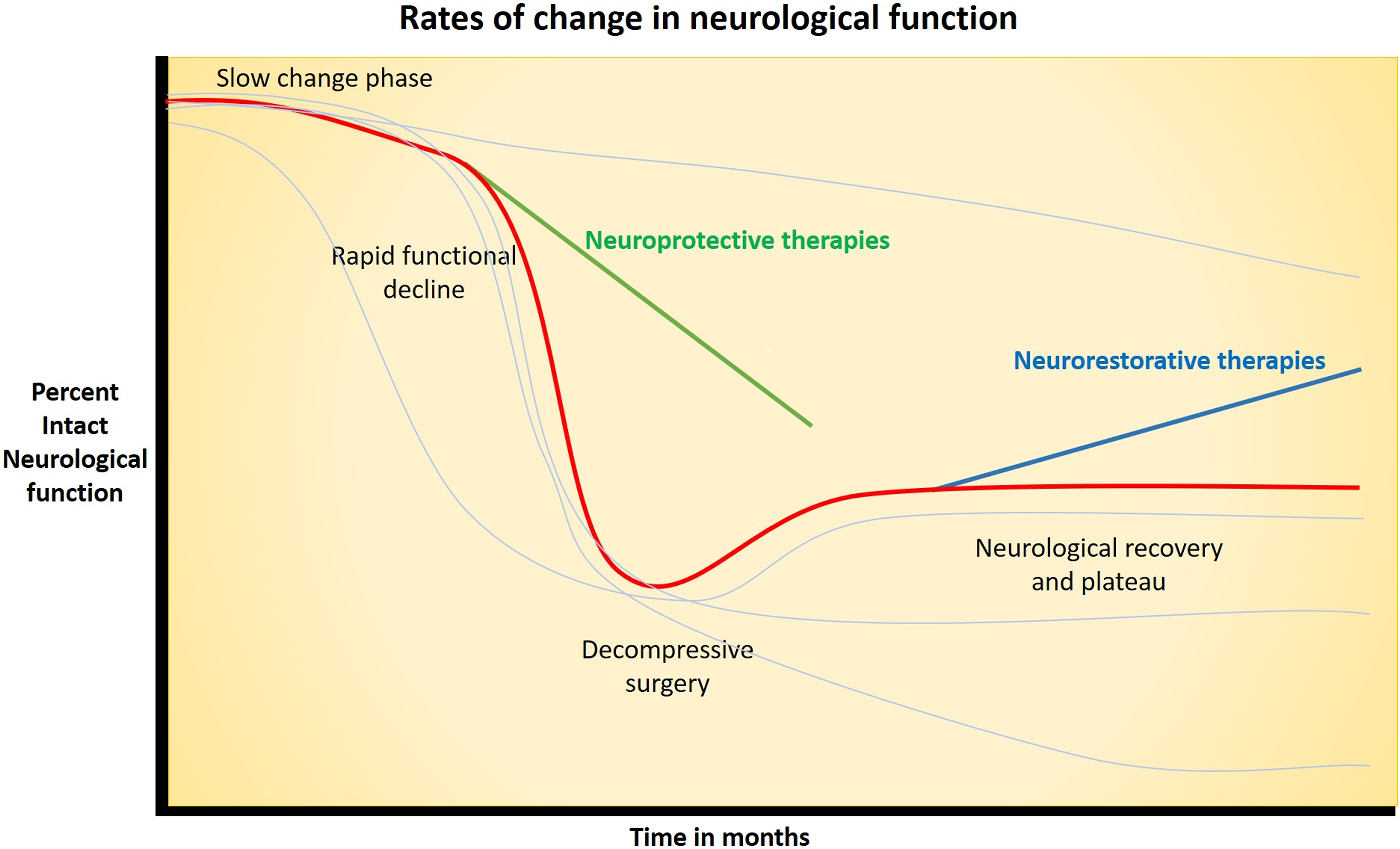
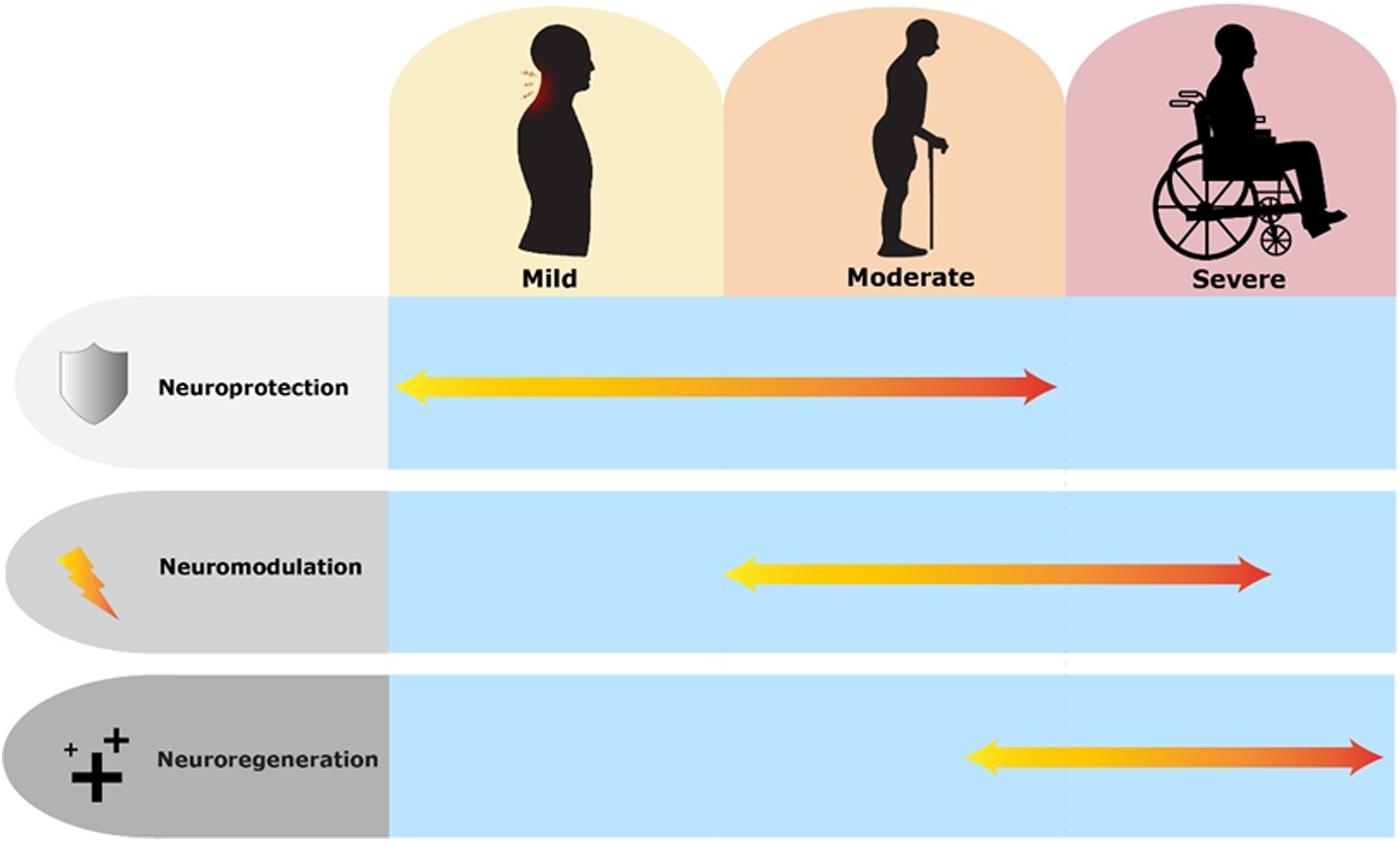
The interventions presented are at different stages of development and readiness for clinical testing in adequately powered efficacy studies. Some interventions are quite mature and are currently undergoing phase-III trials (e.g Ibudilast) or completed efficacy trials (e.g Riluzole), whilst other are undergoing early stage pre-clinical investigation (e.g miR21 regulators). Neuroprotective and neuroregenerative trials typically follow the more typical drug development process, whilst neuromodulatory interventions are devices, and are technologically mature and developed, though ongoing research into optimal intervention protocols and utilization in DCM are required. DCM and chronic cervical SCI have considerable pathophysiological overlap, and potential benefit of an intervention in one can be used to gauge potential efficacy in the other, though careful consideration of severity of cord injury and pathophysiological/restorative targets of interventions are required. An example is acute intermittent hypoxia, which has demonstrated efficacy as an adjunct to rehabilitation in improving gait function in subacute incomplete SCI, which may show similar benefit in DCM.
160
Figure 3 demonstrates technological development and readiness of potential interventions for DCM. Technological readiness of a number of contemporary therapies in the 3 broad categories of neuroprotection, neuromodulation and neuroregeneration. Stage 1 indicates therapies that are predominantly in preclinical developmental stage and will require further research at this stage. Stage 2 shows technologies that are developed, though require further early stage testing and exploration of protocols in degenerative cervical myelopathy prior to phase-III efficacy trials. These may have been investigated in chronic cervical SCI population. Stage 3 shows therapies which are mature in development and may be considered for investigation in degenerative cervical myelopathy at an earlier timeframe. Graphics developed with the support of Myelopathy.org. *These therapies have undergone or are currently undergoing phase-III trials.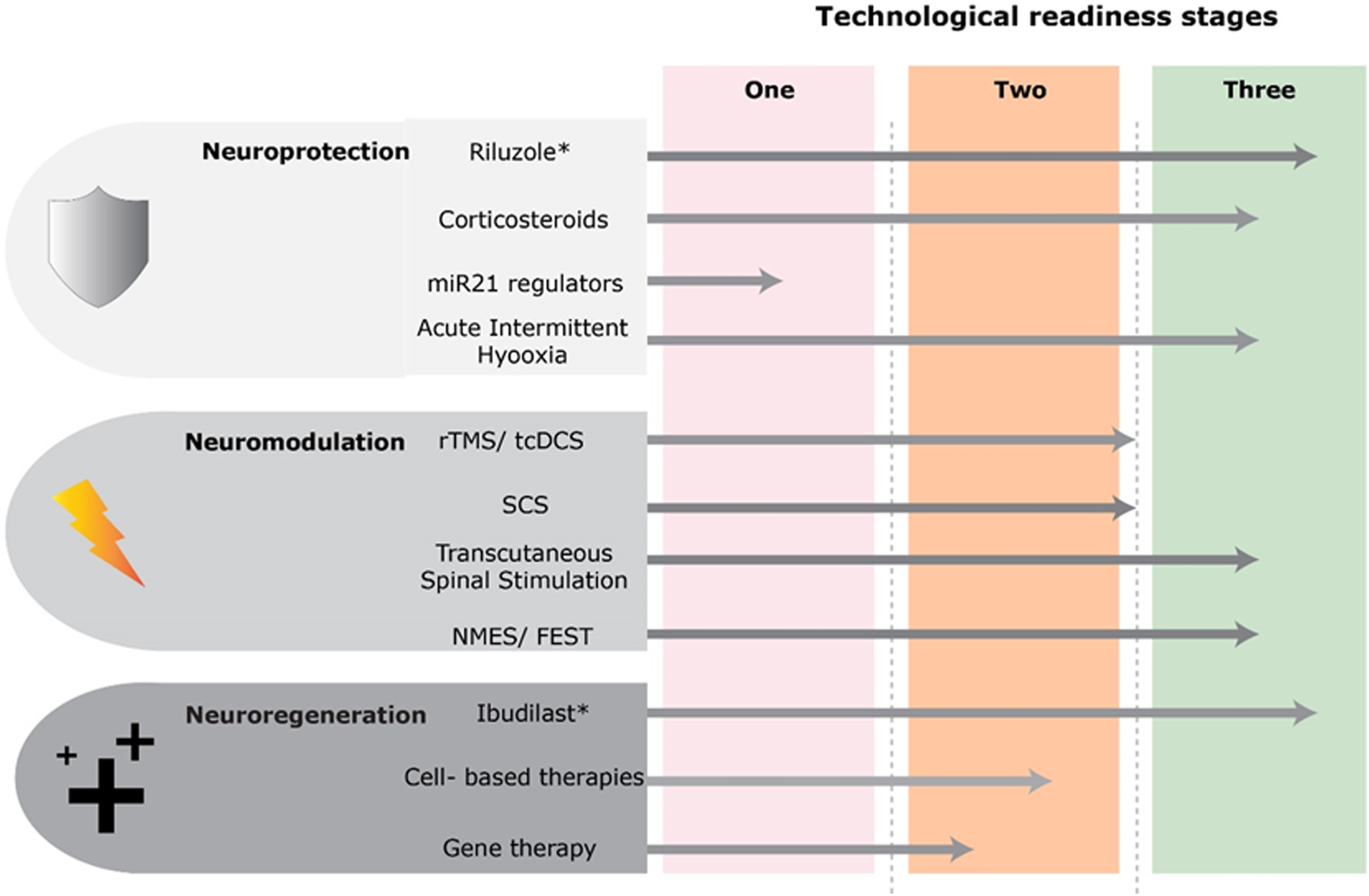
Conclusion
Therapeutic research in DCM over the last few decades has predominantly focused on the role and timing of surgical decompression. Surgery has shown improved outcomes for DCM patients; however, the majority do not make a full recovery and have a subsequent lifelong disability. The impetus for this article was the newly formed JLA priority setting partnership for DCM, which has determined the research priority and question of whether novel therapies can improve health and wellbeing in people with DCM. We present contemporary therapies in the broad domains of neuroprotection, neuroregeneration and neuromodulation, which may have potential therapeutic utility in DCM. As research in this area has been limited, it is hoped that this review will encourage research into this priority.
Footnotes
Acknowledgements
Further details on this priority, including how it was prioritized, why it was prioritized, and on-going research activity can be found at aospine.org/recode/novel-therapies We would like to thank ![]() for their time and support in the development of graphics utilized within this article.
for their time and support in the development of graphics utilized within this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research priorities were organized and funded by AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international Spinal Cord Injury experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically guided not-for-profit organization. Study support was provided directly through the AO Spine Research Department. MRNK is supported by the National Institute for Health Research (NIHR) Brain Injury MedTech Co-operative based at Cambridge University Hospitals NHS Foundation Trust and University of Cambridge, and BMD a NIHR Clinical Doctoral Research Fellowship. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care.
